Abstract
Tumor-associated macrophages are key regulators of the complex interplay between tumor and tumor microenvironment. M2 Macrophages, one type of tumor-associated macrophages, are involved in prostate cancer growth and progression. Protein kinase C zeta has been shown to suppress prostate cancer cell growth, invasion, and metastasis as a tumor suppressor; however, its role in chemotaxis and activation of tumor-associated macrophages remains unclear. Here, we investigated the role of protein kinase C zeta of prostate cancer cells in regulation of macrophage chemotaxis and M2 phenotype activation. Immunohistochemistry was performed to analyze the expression of protein kinase C zeta and the number of CD206+ M2 macrophages in human prostate tissue. Macrophage chemotaxis and polarization were examined using Transwell migration assays and a co-culture system. Quantitative real-time polymerase chain reaction, western blotting, and enzyme-linked immunosorbent assay were used to detect M2 markers, protein kinase C zeta, interleukin-4, and interleukin-10 expression. We found the expression of protein kinase C zeta increased in prostate cancer tissues, especially in the early stage, and was negatively associated with tumor grade and the number of CD206+ macrophages. Inhibition of protein kinase C zeta expression in prostate cancer cells promoted chemotaxis of peripheral macrophages and acquisition of M2 phenotypic features. These results were further supported by the finding that silencing of endogenous protein kinase C zeta promoted the expression of prostate cancer cell–derived interleukin-4 and interleukin-10. These results suggest that protein kinase C zeta plays an important role in reducing infiltration of tumor-associated macrophages and activation of a pro-tumor M2 phenotype, which may constitute an important mechanism by which protein kinase C zeta represses cancer progression.
Introduction
Prostate cancer (PCa) has become the second most commonly diagnosed cancer among men worldwide. 1 While surgery and radiation are potential therapeutic choices for men, the high rate of secondary metastasis results in a poor prognosis. It is well established that the tumor microenvironment plays a critical role in the malignant progression and metastasis of PCa. 2
Many different cell types are contained within the tumor microenvironment, including fibroblasts, adipocytes, endothelial cells, smooth muscle cells, and a number of inflammatory cells that can influence tumor progression. 3 Among these cells, infiltrating macrophages called tumor-associated macrophages (TAMs) are most abundant and play a dual role in tumor progression: M1-like or classically activated macrophages promote anti-tumor immunity; M2-like or alternatively activated macrophages promote tumor progression,4,5 including promotion of angiogenesis, matrix remodeling, and suppression of adaptive immunity. 6 Many observations indicate that M2 macrophages have pro-tumor functions in different neoplastic tissues7–9 as well as in PCa.10,11 However, little is known with respect to how macrophages are recruited to the PCa microenvironment and differentiate into an M2 phenotype, and the underlying mechanisms controlling polarization of macrophages to an M1 or M2 phenotype remain unclear.
Protein kinase C (PKC) isozymes comprise three classes: 12 conventional PKC (cPKC: α, β, and γ), novel PKC (nPKC: δ, ε, η, and θ), and atypical PKC (aPKC: ζ and ι). cPKC isozymes are activated by binding to diacylglycerol (DAG) and Ca2+, whereas nPKC isozymes are activated solely by DAG. Protein kinase C zeta (PKCζ) is one subtype of aPKC which requires neither DAG nor calcium for optimal activity. 13 Studies have demonstrated that PKCζ is a key regulator of cellular processes operating in cancer, and the different functions of PKCζ correlate with its tissue specificity and intracellular distribution. 14 In vitro experiments have shown that PKCζ exerts both pro-tumoral effects15,16 and anti-tumoral effects, 17 but the role of PKCζ in human PCa has been controversial in previous studies.18,19 However, a recent elegant study performed in vivo by Kim et al. 20 showed that PKCζ plays a critical role in inhibition of prostate tumorigenesis as it represses c-Myc activity by inducing phosphorylation of c-Myc on Ser-373, confirming that PKCζ is a tumor suppressor in the prostate. Another study in the mouse PKCζ-knockout phenotype demonstrated that PKCζ acts as a critical metabolic tumor suppressor in mouse and human cancer by repressing expression of two key enzymes in the serine biosynthetic pathway in the absence of glucose. 21 In addition, a growing body of evidence indicates that PKCζ is involved in inflammatory responses in the microenvironment, 22 and loss of PKCζ in vivo leads to increased tumorigenicity linked to the overproduction of interleukin (IL)-6. 23 IL-6, transforming growth factor (TGF)-β, IL-1β, tumor necrosis factor (TNF)-α, IL-10, and other cytokines are important signaling molecules in the tumor microenvironment24,25 produced by inflammatory cells like macrophages. Recent evidence suggests that PKCδ is involved in the change of interferon (IFN)-γ-induced human monocyte-derived macrophages to an M1-like phenotype. 26 However, the role of PKCζ on the conditioning of inflammatory cells in the tumor microenvironment is still unknown. Therefore, we tested whether PKCζ expression in PCa produced an effect on TAM infiltration or polarization to restrain tumor progression and metastasis.
In this study, we observed that the expression of PKCζ increased in PCa tissues, especially in the early stage, and was negatively associated with tumor grade and the number of CD206+ macrophages. Transwell migration assays showed that PKCζ in PCa cells regulates the recruitment of the macrophage cell line RAW264.7. In addition, silencing of PKCζ skewed macrophage polarization to the M2 phenotype as shown by increased expression of M2 markers mediated by IL-4 and IL-10.
Materials and methods
Chemicals and reagents
Lipofectamine® 3000 Transfection Reagent was obtained from Invitrogen (L3000015; Thermo Fisher Scientific, USA). HilyMax transfection reagent and Cell Counting Kit-8 were purchased from Dojindo (Japan). Other purchased reagents were recombinant murine IL-4 (Cat. No. 214-14; PeproTech, USA), lipopolysaccharide (LPS; L2880; Sigma, USA), anti-PKCζ (Cat. No. EnT37365; Elabscience, China), anti–glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibodies (Ray Antibody, China), goat anti-rabbit and anti-mouse secondary antibodies (Bioss, China), IL-4 and IL-10 enzyme-linked immunosorbent assay (ELISA) kits (Elabscience), and anti-human IL-4 and anti-human IL-10 (eBioscience Cat. No. 16-7048 and Cat. No. 16-7108; Affymetrix, USA).
Cell culture, small interfering RNA, and plasmid transfections
All cell lines were obtained from the American Type Culture Collection (Manassas, VA, USA). Cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, USA) supplemented with 10% fetal bovine serum (FBS) and penicillin–streptomycin (Invitrogen, USA), and were maintained at 37°C in a 5% CO2 humidified atmosphere. The medium for the murine macrophage cell line RAW264.7 was changed every 48 h. The small interfering RNA (siRNA) to silence PKCζ (sequence 5′-GAGGAAGTGAGAGACATGTGT-3′; Gene-Pharma, China) and CA-PKCζ plasmid (carried by pcDNA3.1 vector, a type of constitutive active) to overexpress PKCζ was transfected into PCa cell lines PC3 and DU145 using Lipofectamine 3000 and HilyMax, respectively, according to the manufacturers’ instructions. After 6 h, cells were washed with phosphate buffered solution (PBS) and cultured in DMEM supplemented with 1% FBS for 48 h. The culture supernatants (conditioned media (CM)) were collected and named PC3-CM and DU145-CM.
Co-culture experiments with RAW264.7
Co-culture inserts (0.4 µm pores; Corning, USA) were placed in six-well culture plates. RAW264.7 cells were added to the lower culture wells (2 × 105 cells per well) and cancer cells (1 × 105 cells per well) were placed in the inserts, and both were cultured in DMEM supplemented with 5% FBS for 3 days before harvesting the macrophages for gene expression analysis.
RNA extraction and quantitative real-time polymerase chain reaction
Total RNA from each cell line was extracted using TRIzol reagent (TaKaRa, China) according to the manufacturer’s protocol. Reverse transcription was carried out using a PrimeScript RT Reagent Kit (TaKaRa). Quantitative real-time polymerase chain reaction (qRT-PCR) was performed on a 7500 Fast Real-Time PCR system (Applied Biosystems, USA) using a program: 95°C, 30 s; 95°C, 3 s; and 60°C, 30 s for 40 cycles. Target gene expression was normalized to GAPDH as endogenous controls. All results are presented as the mean ± standard deviation (SD) of at least three independent experiments. Comparative quantification was determined using the 2−ΔΔCt method. The primers used are presented in Supplementary Table S1.
Western blotting
Total protein from each cell line was obtained by complete cell lysis (Keygen Biotech, China) with protease and phosphatase inhibitors. Equivalent quantities of proteins (calibrated by Coomassie Brilliant Blue staining) were resolved by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gels and transferred onto polyvinylidene difluoride membranes. The membranes were blocked with 5% skimmed milk for 3 h and incubated with primary antibodies anti-PKCζ and anti-GAPDH overnight at 4°C. Immunolabeling of primary antibodies was detected with goat anti-rabbit or anti-mouse secondary antibodies conjugated to horseradish peroxidase (HRP). Blotting results were visualized with a chemiluminescence HRP substrate kit (Cat. No. WBKL S0500; Millipore, USA).
Immunohistochemistry and tissue microarrays
Immunohistochemistry (IHC) was performed on high-density tissue microarrays (Cat. No. PR242b, T195c; Alenabio Biotechnology, China). Normal prostate tissues were obtained from men excluding PCa pathologically and median age is 26 years. Primary antibodies against PKCζ and CD206 (Cat. No. ab64693; Abcam, UK) were diluted 1:1000 in antibody diluent, respectively. Immunolabeling was visualized using anti-mouse/rabbit-HRP (K5007; Dako, Denmark). The expression of PKCζ was evaluated according to the percentage of positive cells per specimen and the staining intensity as described previously. 27 Staining was assessed as negative, weakly positive or only focally positive, or strongly positive. Counts of M2 macrophages were determined by the number of CD206+ cells per field.
Transwell migration and chemotaxis assays
All cell migration assays in vitro were performed using Transwell chambers (8 µm pore size; Corning, USA) in 24-well plates. RAW264.7 cells (0.5 × 105) in 200 µL of serum-free medium were seeded in the upper chamber, and RAW264.7 cells in 450 µL of CM collected from PCa cells were placed in the lower chamber. After 24 h incubation, the chamber was fixed with methanol, followed by staining with 0.1% crystal violet. Cells on the upper side of the Transwell membrane were wiped off with a cotton swab and microphotographs of the cells attached to the underside of the membranes were taken (at least five random fields; 200×), from which cells were counted.
ELISA cytokine assays
IL-4 and IL-10 protein levels were determined using ELISA kits. PCa cells were seeded in six-well culture plates and cultured for 48 h with serum-free medium. Cell supernatants were then collected and stored at −80°C before cytokine concentrations were measured by ELISA following the manufacturer’s instructions. The actual concentration was calculated from a standard curve prepared for each assay.
Statistical analysis
All values were expressed as means ± SD from at least three replicates. All statistical analyses were conducted using SPSS 17.0 software and GraphPad Prism V software. Statistical significance between groups was determined using unpaired two-tailed Student’s
Results
Negative correlation between expression of PKCζ and CD206+ M2 macrophages in human PCa
PKCζ was widely distributed in cells of mammalian tissues 21 and stroma.28,29 In order to determine whether the expression of PKCζ was related to the number of infiltrating macrophages of M2 phenotype in the development of PCa, we analyzed the expression of PKCζ and CD206 in human prostate tissue arrays using IHC. CD206 is a biomarker for macrophages with a M2 phenotype. 30 More CD206-positive macrophages were observed in 19 cases of prostate adenocarcinoma than in 4 cases of normal prostate tissue (Figure S1). Representative micrographs of the distribution of cytoplasmic PKCζ and membrane CD206 expression in different tumor grades of PCa are shown in Figure 1(a). We found that PKCζ expression levels increased in PCa, especially in the early stage (Figure 1(b)), and were inversely correlated with Gleason scores (Supplementary Table S2). However, contrasting findings were observed for the number of M2 macrophages, which increased in advanced tumor grades (Figure 1(c)). We conclude from these analyses that PKCζ expression was inversely correlated with the presence of CD206+ macrophages in the stroma of PCa tissues. This inverse correlation may be a useful prognostic indicator associated with PCa.
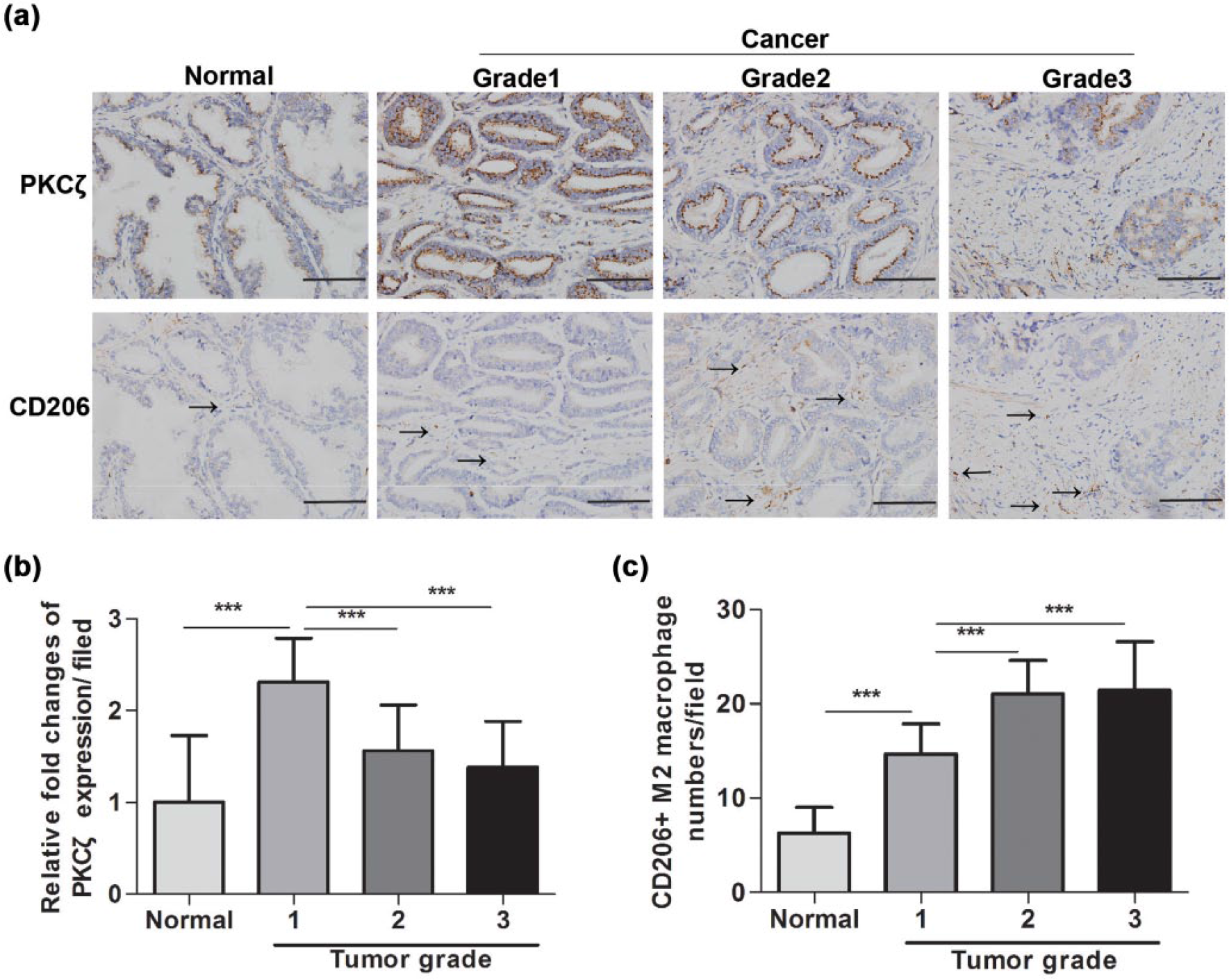
PKCζ expression is reversely correlated with M2 macrophage infiltration and tumor grade in human prostate cancer. (a) Immunohistochemical images of PKCζ and CD206 expression in normal prostate tissue and prostate cancer of different tumor grades. (b) Statistical results of PKCζ-relative expression. (c) Quantification of the number of CD206+ macrophage. (b) and (c) were counted in five independent fields under a 40× objective (***p < 0.001).
Expression of PKCζ in PCa cells inhibits the recruitment of macrophages in vitro
The clinical relationship between PKCζ expression and M2 macrophages suggested that macrophage chemotaxis was correlated with the expression of PKCζ. To test this hypothesis, the effect of siPKCζ and CA-PKCζ on PCa cells was examined by qRT-PCR, which showed corresponding changes in PKCζ messenger RNA (mRNA) expression (Figure S2). Subsequently, Transwell migration assays of the macrophage cell line RAW264.7 were performed with CM from PCa cells obtained after PKCζ silencing (Figure 2(a)). Results showed that PKCζ inhibition in PCa cells increased the migration of RAW264.7 cells (Figure 2(b)), while overexpression decreased migration (Figure 2(c) and (d)). In addition, western blotting was performed to confirm the effect of PKCζ gene silencing and overexpression in PCa cells (Figure 2(b) and (c)). Data suggest that lower expression of PKCζ in PCa was a key driver of macrophage recruitment that may ultimately increase the potential of tumor progression.

Expression of PKCζ in prostate cancer cells inhibits the chemotaxis migration of RAW264.7 macrophages. (a) Condition media from PCa cells with PKCζ silencing was used to induce the migration of RAW264.7 cells. (c) Condition media from PCa cells with PKCζ overexpression was used to induce the migration of RAW264.7 cells. The effect of siRNA (siPKCζ) and overexpression of plasmid (CA-PKCζ) on PCa cell lines were confirmed. (b and d) Quantification of recruited macrophages in each image field. Representative micrographs of the cells attached to lower side of the membranes were taken under a 20× objective and counted in five independent fields (*p < 0.05, **p < 0.01).
Expression of PKCζ in PCa cells suppresses the polarization of innate RAW264.7 macrophages to a M2 phenotype via inhibition of M2 marker expression
At the tumor site, various physical and chemical interactions with the tumor and surrounding microenvironment induce the differentiation of M1 or M2 macrophage. 31 A recent study showed that REG4 secreted by pancreatic cancer cells promoted the polarization of macrophages to an M2 phenotype. 32 Accordingly, we hypothesized that PKCζ might not only recruit macrophages but also polarize them to the M1 or M2 phenotype.
The expression of representative M1 markers such as IL-6, TNF-α, and IL-1β 33 and M2 markers such as CD163, arginase-1 (Arg-1), mannose receptor C type 1 (Mrc-1/CD206), and mannose receptor C type 2 (Mrc-2) have often been used to identify subtypes of TAMs.30,31,34,35 To determine whether expression of PKCζ in PCa cells affected innate RAW264.7 differentiation into M2 macrophages, RAW264.7 cells were co-cultured with PCa cells in which PKCζ was either silenced or overexpressed, and the expression of M1 and M2 macrophage markers was examined by qRT-PCR. The relative mRNA expression of M2 or M1 cells marker genes treated with IL-4 (40 ng/mL) or LPS (2.5 μg/mL), respectively, were given as positive control. As shown in Figure 3, mRNA expression of M2 macrophage markers increased markedly after 72 h co-culture with DU145 cells in which PKCζ was knocked down compared to nontargeting siCTL control RNA (Figure 3(a)). In contrast, expression of M1 macrophage markers (IL-6, TNF-α, and IL-1β) failed to increase after similar treatment (Figure 3(b)). Moreover, there was no change in the expression of M2 and M1 markers in macrophages co-cultured with DU145 cells overexpressing PKCζ (Figure 3(c) and (d)). Similar results were obtained in the PC3 cell line (Figure S3). These results suggested that PKCζ not only restrained the recruitment of macrophages but also controlled macrophage differentiation to the M2 phenotype.

Expression of PKCζ in DU145 cells restrains the polarization of RAW264.7 macrophages into an M2 phenotype via inhibition of M2 marker expression. (a) RAW264.7 macrophages were co-cultured with siPKCζ-DU145 cells in co-culture inserts, siCTL as the control treatment, IL-4 (40 ng/mL) as a positive control of M2, and LPS (2.5 g/mL) as a positive control of M1. M2 marker of CD206, CD163, Arg-1, and Mrc-2 mRNA expression was detected by qRT-PCR. (b) M1 marker of IL-6, TNF-α, and IL-1β was detected by qRT-PCR. (c and d) M2 and M1 marker mRNA expression was detected after RAW264.7 cells were co-cultured with CA-PKCζ-DU145 cells. (*p < 0.05, **p < 0.01, and ***p < 0.001; *compared with the control group).
Expression of PKCζ in PCa cells inhibits the M2 phenotype by regulating the secretion of IL-4 and IL-10
Since PCa cells can also release a number of cytokines and chemokines into the tumor microenvironment,36,37 we hypothesized that PKCζ expression could control the conversion of macrophages to the pro-tumor M2 phenotype by regulating cytokine expression. Macrophage colony-stimulating factor (M-CSF) and TNF-α are stimulating factors of M1 polarization; IL-4, IL-6, IL-10, IL-13, and TGF-β are polarization factors for M2.38,39 These cytokines were detected by qRT-PCR in siPKCζ and its control siCTL; IL-4 and IL-10 changed significantly and similar trends were obtained in PCa cells (Figure S4). We further examined IL-4 and IL-10 expression in PCa cells with PKCζ silenced and observed increased levels of mRNA and protein (Figure 4(a) and (b)). Moreover, the enhancement of M2 marker expression by PKCζ silencing was abolished in the presence of neutralizing antibodies against IL-4 and IL-10 in our co-culture system (Figure 4(c)). Therefore, PKCζ in PCa cells suppresses conversion to the M2 phenotype by regulating the secretion of IL-4 and IL-10.

PKCζ inhibits the M2 phenotype by regulating the secretion of IL-4 and IL-10 in prostate cancer cells. (a) The mRNA expression level of IL-4 and IL-10 in siPKCζ-PCa cells. (b) The protein expression level of IL-4 and IL-10 in siPKCζ-PCa cells. (c) M2 markers were detected after RAW264.7 cells were co-cultured with siPKCζ-DU145 cell in the presence of neutralizing antibody against IL-4 and IL-10 (*p < 0.05 and **p < 0.01; *compared with the control treatment; ##p < 0.01, #compared to treatment without neutralizing antibody against IL-4 or IL-10 (Group b)).
Collectively, the above results suggest that loss of PKCζ in PCa cells could promote macrophage recruitment and M2 polarization by upregulating IL-4 and IL-10 production, which may constitute an important mechanism by which PKCζ represses cancer progression.
Discussion
While many researchers have focused on immunotherapy for PCa and melanoma, a significant obstacle is the presence of immunosuppressive cells including myeloid-derived suppressor cells and TAMs which induce immune tolerance and promote tumor development. 40 Here, we report that downregulation of PKCζ in PCa promotes the recruitment of macrophages to the tumor microenvironment and induces their polarization to the M2 phenotype. First, we observed that expression of PKCζ in PCa cells was inversely correlated with CD206+ TAMs by IHC. Furthermore, the number of migrating macrophages was reduced by CM collected from PCa cells overexpressing PKCζ. Second, we demonstrated that PKCζ inhibited the polarization of RAW264.7 macrophages to the M2 phenotype using a co-culture system. Finally, we found that PKCζ regulated the expression of IL-4 and IL-10, which contributes to M2 polarization.
Macrophages have a high rate of infiltration into tumors 41 and may develop a pro-tumoral phenotype that has been demonstrated both in vivo and in vitro experiments.7,9 Our migration assay data confirmed that lower expression of PKCζ in PCa was essential for migration of macrophages. We also screened chemokines in PCa cells that could stimulate macrophage chemotaxis, including CCL2, CCL3, CCL5, CCL7, fractalkine, and SDF-1, but found only undetectable or low-level expression (Figure S4). We did not perform further analysis of the causes of macrophage migration, but it has been reported that IL-4 contributes to the migration of THP-1 monocyte cells. 42 Downregulation of PKCζ in PCa might therefore stimulate macrophage migration through direct effects on tumor-derived IL-4 levels. Previous studies demonstrated that IL-4 enhances androgen receptor (AR) activation and overexpression of IL-4 enhances the growth of androgen-sensitive LNCaP cells; 43 IL-4 has been associated with PCa progression. 44 Analysis of benign and malignant prostate tissue demonstrates that the source of IL-4 is epithelial cells rather than infiltrating leukocytes. 45
It is generally recognized that TAMs may polarize to the M2 macrophage phenotype stimulated by IL-4, IL-10, and IL-13. 5 In our study, co-culture experiments were performed with cell co-culture inserts, which allowed no direct contact between PCa cells and macrophages, so that only soluble mediators were able to affect cellular activity. Our results indicated that PKCζ expression in PCa cells prevented macrophage chemotaxis and M2-like polarization via negative regulation of tumor-secreted IL-4 and IL-10 levels. To our knowledge, this study is the first to report that silencing of PKCζ stimulates production of tumor-derived IL-4 and IL-10 in PCa cells. Interestingly, some investigators have found that PKCζ knockdown in obese mice leads to a hyperinflammatory state and IL-6 accumulation. 22 Another study demonstrated that IL-6 from cervical carcinoma induced the differentiation of M2 macrophages. 38
Strong evidence has been provided showing a negative relationship between TAM cell density and clinical prognosis in tumors of the lung, cervix, ovary, and prostate.46,47 This finding has revealed several processes that can potentially be exploited therapeutically to dampen cancer cell metastasis by reducing the number of M2 macrophages. With further validation, the restrictions imposed by PKCζ on M2 differentiation may exert an indirect inhibitory effect on metastasis in PCa.
In summary, our findings suggest that the PKCζ gene is a critical tumor suppressor in PCa acting through the inhibition of macrophage chemotaxis and the acquisition of M2 features. Modulation of the tumor microenvironment through inhibition of IL-4 and IL-10 production may be a potential target for prevention and therapy. To date, targeting TAMs instead of tumor cells has had clearly beneficial clinical outcomes in fibrosarcoma as well as ovarian and lung cancers. 48 Our study thus raises the possibility that a combination of agents that block both IL-4 and IL-10 may be an effective approach to inhibit TAMs in PCa.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81272852, 81472407, 81672540), Oversea Hong Kong & Macao Scholars Collaborative Research Fund of NSFC in China (81328020), Natural Science Foundation of Guangdong Province (S2013010014482), and Science and Technology Project of Guangzhou (201607010351).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
