Abstract
A complex network of chemokines can influence cancer progression with the recruitment and activation of hematopoietic cells, including macrophages to the supporting tumor stroma promoting carcinogenesis and metastasis. The aim of this study was to investigate the relation between tissue and plasma chemokine levels involved in macrophage recruitment with tumor-associated macrophage profile markers and clinicopathological features such as tumor–node–metastases stage, desmoplasia, tumor necrosis factor-α, and vascular endothelial growth factor plasma content. Plasma and tumor/healthy mucosa were obtained from Chilean patients undergoing colon cancer surgery. Chemokines were evaluated from tissue lysates (CCL2, CCL3, CCL4, CCL5, and CX3CL1) by Luminex. Statistical analysis was performed using Wilcoxon match-paired test (p < 0.05). Macrophage markers (CD68, CD163, and iNOS) were evaluated by immunohistochemistry samples derived from colorectal cancer patients. Correlation analysis between chemokines and macrophage markers and clinicopathological features were performed using Spearman’s test. Plasmatic levels of chemokines and inflammatory mediators’ vascular endothelial growth factor and tumor necrosis factor-α were evaluated by Luminex. Tumor levels of CCL2 (mean ± standard deviation = 530.1 ± 613.9 pg/mg), CCL3 (102.7 ± 106.0 pg/mg), and CCL4 (64.98 ± 48.09 pg/mg) were higher than those found in healthy tissue (182.1 ± 116.5, 26.79 ± 22.40, and 27.06 ± 23.69 pg/mg, respectively p < 0.05). The tumor characterization allowed us to identify a positive correlation between CCL4 and the pro-tumor macrophages marker CD163 (p = 0.0443), and a negative correlation of iNOS with desmoplastic reaction (p = 0.0467). Moreover, we identified that tumors with immature desmoplasia have a higher CD163 density compared to those with a mature/intermediated stromal tissue (p = 0.0288). Plasmatic CCL4 has shown a positive correlation with inflammatory mediators (tumor necrosis factor-α and vascular endothelial growth factor) that have previously been associated with poor prognosis in patients. In conclusion High expression of CCL4 in colon cancer could induce the infiltration of tumor-associated macrophages and specifically a pro-tumor macrophage profile (CD163+ cells). Moreover, plasmatic chemokines could be considered inflammatory mediators associated to CRC progression as well as tumor necrosis factor-α and vascular endothelial growth factor. These data reinforce the idea of chemokines as potential therapeutic targets or biomarker in CRC.
Introduction
Colorectal cancer (CRC) is one of the most frequently diagnosed cancers worldwide, being the second most common cancer in women and the third most in men. 1 In Chile, the crude mortality rate has sharply risen between 1983 and 2008. 2
In most cases, the disease occurs sporadically through a transition from dysplastic adenomatous polyps to carcinoma, which is caused by genetic alterations that promote proliferation and inhibit apoptosis of colonic epithelial cells.3,4
In addition to the alterations in the tumor cells, a microenvironment that promotes the progression of cancer is essential with the interaction between the tumor mass and different cell populations such as fibroblasts, endothelial cells, and immune system cells. 5 Thus, tumors can be classified according to the inflammatory grade, infiltration, and the presence of fibrosis that characterizes desmoplasia.6,7 Desmoplasia is the rearrangement of an organized, anisotropic extracellular matrix (ECM) fiber in a pathological microenvironment associated to metastasis or poor patient survival.8,9
The immune cells involve macrophages, neutrophils, mast cells, and T and B cells, which release inflammatory mediators such as cytokines and chemokines (CKs); all these can participate in the tumor progression. 10
Macrophages are usually the most abundant cell group in tumors, 11 and they belong to the innate immune system where they participate in inflammation, phagocytosis, and cytotoxicity, among others. 12 They are called tumor-associated macrophages (TAMs) when they infiltrate the tumor mass, and are highly heterogenic cell populations with differing participations in carcinogenesis. 13 Usually, two extreme states of differentiation in macrophages are recognized: the classic phenotype or M1, associated to antitumor and proinflammatory activity (mediated by the secretion of cytokines interleukin (IL)-1β, tumor necrosis factor (TNF)-α, and IL-6), and the alternative phenotype or M2, with protumor and anti-inflammatory activity. 14 M2 acts directly on the tumor cells and indirectly on the tumor microenvironment 15 by the production of growth factors (e.g. fibroblast growth factor (FGF), vascular endothelial growth factor (VEGF), and IL-6), matrix degrading enzymes and CKs that induce the neo-angiogenesis switch, tumor progression, tissue invasion and repair.5,11,16–18 CKs are small size proteins, belonging to the cytokine family with pleiotropic functions (cellular chemotaxis, cytokine secretion, reactive oxygen species production, cell proliferation, and survival). 19 These functions can participate in the pathogenesis of cancer, promoting growth of tumor cells, metastasis, and angiogenesis. 20
In relation to the TAMs, studies in CRC showed that greater macrophage infiltration was found in patients with better prognosis, such as less recurrence or complications and favorable clinical and histological tumor characteristics.21–24 However, with regard to M2 type macrophages, evidence demonstrates that a greater amount of this phenotype is associated with a worse prognosis, less survival, and late stages of the disease.25–27
Studies performed in CRC have shown an increase in several CKs in tumor mucosa compared to the normal tissue.28–32 In 2007, Bailey et al. 29 analyzed the expression of different CKs in CRC, highlighting an increase in CCL2, which correlated with an accumulation of TAMs. Later, Erreni et al. 30 reported an increase in CCL3 and CCL4 independent of the tumor stage, together with the overexpression of CCR1 and CCR5 (CCL3, CCL4, and CCL5 receptors). Subsequently, Cambien et al. 31 identified CCL5 overexpression and its receptors CCR1 and CCR3 to a murine CRC model and when applying an anti-CCL5 neutralizing antibody, tumor growth reduction was observed.
In addition, CKs can be secreted and enter into the blood stream. Moreover, cancer patients have shown a systemic inflammatory response that has been associated to a poorer outcome, in both early- and advanced-stage CRC. 33 In this regard, circulating high levels of TNF-α and VEGF are soluble mediators that have been associated to increased risk of disease recurrence and poor survival and therefore could have some prognostic value.34–36
With this knowledge, we investigated tissue and plasma levels of CKs (involved in macrophage recruitment) and their association with colon cancer progression, such as macrophage profile markers, clinicopathological, and histological features (e.g. grade of desmoplasia and inflammatory infiltration) and plasmatic cytokines.
Methods
Patients
Adult patients with CRC (n = 48; 33–91 years), who underwent surgical resection, were enrolled by Clínica Las Condes, Hospital Clínico Universidad de Chile (HCUCH) and Biobanco de Tejidos (HCUCH) between 2015 and 2017 (Table 1). Patients receiving radiation or chemotherapy prior to surgery were excluded. Tumor staging was categorized according to the tumor–node–metastases (TNM) classification (The Union for International Cancer Control (UICC)). 37
Clinical–demographic features of patients.

CRC: colorectal cancer; TNM: tumor–node–metastases.
Healthy and tumor tissue samples (fresh-frozen) from 25 patients were used to determine levels of intestinal CKs. Tumor and healthy tissue (paraformaldehyde (PFA) fixed) from 31 patients were used to characterize the macrophages in the intestinal mucosa. The selected areas correspond to tumor core, neither the infiltration/expansion border, nor were the areas of the tumor surface considered. According to the regulations of the Biobank, a mirror image of the tissue that was digested (cytokine quantification) was obtained in order to guarantee the exact position from which the sample was taken.
Plasma samples from 32 patients were used to evaluate the cytokine and CK content. As a control group, plasma from 15 healthy volunteers was obtained (subjects with inflammatory diseases such as arthritis or inflammatory bowel diseases were excluded).
This study was approved by the ethics committee from Clínica Las Condes (certified 15/12/2014) and Hospital Clínico Universidad de Chile (certified acta no. 044 July 2015 and No. OAIC 844/16), and informed consent was obtained from all participants in research for analysis of medical history, plasma and tissue biopsies.
Determination of tissue CK levels
Tissues were lysed in radioimmunoprecipitation assay (RIPA) buffer with a tissue disruptor and the protein concentration was measured using the bicinchoninic acid (BCA) method (ThermoScientificTM, catalog no. 23227).
CKs levels (CCL2, CCL3, CCL4, CCL5, and CX3CL1) were assessed using MilliplexTM MAP (Millipore, Billerica, MA, USA) multiplex magnetic bead–based antibody detection kits according to the manufacturer’s protocols. Our analysis was performed with a 5-analyte (5-plex) kit in two tissue sample dilutions.
VEGF and TNF-α levels were assessed with DuoSet® enzyme-linked immunosorbent assay (ELISA) kit (DuoSet; R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s protocols. The CK and cytokine levels were expressed in picogram/milligram total protein.
Determination of plasma cytokine and CK levels
Plasma from controls and CRC patients were obtained from whole blood collected in ethylenediaminetetraacetic acid (EDTA) tubes (vacutainers). CCL2, CCL3, CCL4, TNF-α, and VEGF levels were quantified using MilliplexTM MAP multiplex magnetic bead–based antibody detection kits according to the manufacturer’s protocols and expressed in picogram/milliliter of plasma.
Tissue microarray (TMA) generation
TMAs were assembled from formalin-fixed paraffin-embedded tissues using a 0.6-mm-diameter punch (Beecher Instruments, Silver Spring, MD, USA). The arrays encompass 24 tissue cores from colonic tumors and healthy tissue derived from 31 patients. Also, two cores from kidney tissue were used (for orientation purposes).
Using a tape-transfer system (Instrumedics, Hackensack, NJ, USA), 2 μm sections were transferred to glass slides and analyzed by immunohistochemistry (IHC).
Histological characteristics
Hematoxylin-/eosin-stained sections from tumor and normal tissue derived from 21 CRC patients were evaluated by a pathologist who was blind to the patients’ information. Histological characteristics such as tumor grade, inflammatory and desmoplastic reaction were identified. A score was assigned to each patient according to tumor grade, which indicates how tumor cells resemble to normal epithelia in well-differentiated (1), moderately differentiated (2), and poorly differentiated (3) tumor. 38 Regarding inflammatory infiltrate, scores were considered by density of inflammatory infiltrate in the stromal compartment of normal and tumoral tissues as follows: none or scattered inflammatory cells (0), <20% (1), 20%–50% (2), and more than 50% (3). Desmoplastic reaction (a fibrotic reaction to the tumor) was scored also with a three-point scale according to tumor density, in low (1), moderate (2), and abundant (3). Also, we analyzed the type of desmoplasia, according to criteria described by Ueno et al.,39,40 categorizing each patient’s type of desmoplasia as mature, intermediate, or immature based on cellular and fibrous stromal organization.
IHC
Conventional IHC on TMA sections was performed on 2 μm sections using antibodies against CD68 (clone PG-M1; Dako, Carpinteria, CA, USA), iNOS (ab178945; Abcam, Cambridge, MA, USA), and CD163 (ab87099; Abcam, Cambridge, MA, USA).
IHC was performed using a polymer-based detection system (Envision; Dako, Carpinteria, CA, USA), according to the manufacturer’s instructions. Sections were deparaffinized and rehydrated with deionized water. Then, they were heated in citrate buffer, pH 6.0, using an electric pressure cooker for 3 min at 12–15 pounds per square inch at approximately 120°C and cooled for 10 min before immunostaining. All samples were blocked by exposure to 3% hydrogen peroxide for 10 min, incubated first with primary antibody for 30 min, then second with labeled polymer (Envision Flex HRP) for 30 min, next in 3′3-diaminobenzidine as a chromogen for 5 min and finally counterstained with hematoxylin for 5 min. The above was carried out at room temperature and, between incubations, sections were washed with Tris-buffered saline. Coverslips were placed with the Tissue-Tek SCA (Sakura Finetek USA, Inc, Torrance, CA, USA). Positive and negative controls were tested with each batch of patient/study slides.
The images were captured with the Aperio ScanScope equipment. The analysis of the images was made by the Aperio ImageScope Software and the algorithm to evaluate the images was Positive Pixel Count 9. The proportion of positive pixels with respect to the total pixels (positive and negative) per area was considered for the association with the clinical and histological features of the patients (positivity/area).
Double IHC was performed by a sequential double labeling technique using two different detection systems. Briefly, sections were first stained for CD163 or iNOS, using the streptavidin–biotin peroxidase detection system, and visualization with DAB (brown). Then, sections were washed and stained for CD68 using the streptavidin–biotin alkaline phosphatase detection system and visualization with fast red (red).
Statistical analysis
To compare levels of CKs in paired samples (healthy and tumor tissue from patients), statistical analysis of the data was performed using the Wilcoxon match-paired test, and p values <0.05 were considered significant.
To compare plasma levels of CKs and cytokines from controls versus patients, statistical analysis was performed using the Mann–Whitney test, and p values <0.05 were considered significant.
Spearman’s test was used to analyze correlation between soluble and histological markers in tumor.
Statistical analysis of density of macrophage markers in tumor (CD68, CD163, and iNOS; according to the type of desmoplasia; mature/intermediate vs immature) was performed using Mann–Whitney test, in which two-tailed p values <0.05 were considered significant.
Results
Clinicopathological features
Table 1 includes clinical information of the 48 CRC patients studied; 22 patients were diagnosed with local metastasis to lymph nodes and 1 with distant metastasis that corresponds to more advanced disease stages (TNM II and III). Patients were diagnosed mainly with left colon cancer (32/48, 66.6%).
CK and inflammatory mediator levels in tumor versus healthy tissue
From analysis of five CKs in tumor and paired healthy samples taken from 25 patients with colon cancer, it was observed that three (CCL2, CCL3, and CCL4) demonstrated higher levels in tumor compared to healthy tissue, using the Wilcoxon match-paired test (Figure 1).

Chemokine levels in healthy colon tissue and tumor samples. The graph represents the levels of chemokine in each sample and the mean ± SD. Levels of CCL2, CCL3, and CCL4 were higher in tumors compared to healthy tissue (non-parametric Wilcoxon matched pair test, p < 0.05). Differences in CCL5 and CX3CL1 were not observed among tumor and healthy tissues.
The levels of CCL2, CCL3, and CCL4 (mean ± SD) found in the malignant tissue were as follows 530.1 ± 613.9, 102.7 ± 106.0, and 64.98 ± 48.09 pg/mg protein, whereas in the normal mucosa they were 182.1 ± 116.5, 26.79 ± 22.40, and 27.06 ± 23.69 pg/mg protein.
However, CCL5 (p = 0.2473) and CX3CL1 (p = 0.3746) did not show a statistical difference, identifying 3.785 ± 2.307 pg/µg and 197.2 ± 152.1 pg/mg protein in healthy tissue, whereas 3.057 ± 2.614 pg/µg and 172.7 ± 204.0 pg/mg protein in tumor tissue.
The levels of inflammatory mediators, TNF-α and VEGF, in tumor tissue were 8.471 ± 5.456 and 203.6 ± 142.8 pg/mg, respectively, whereas in normal tissue they were 10.81 ± 18.53 and 74.91 ± 75.08, demonstrating higher VEGF levels in tumor samples compared to paired normal tissue (p < 0.0001; Supplementary Figure 1).
TAMs’ markers identification in tumor samples
To characterize the TAMs population in CRC, the presence of a lineage monocyte-macrophage marker (CD68) together with M1/M2 subpopulation markers (iNOS/CD163) were evaluated in tissue sections. Double IHC analysis demonstrated expression of CD163 or iNOS in CD68 positive cells in stroma from tumor and healthy tissue from CRC patients (Supplementary Figure 2). 31 tumor samples and their healthy counterpart were analyzed for stromal protein density of CD68, CD163, and iNOS through single-labeling IHC (Figure 2). Spearman’s test was used to establish the relationship between macrophage marker quantification, levels of CKs and histopathological characteristics (TNM, grade of differentiation, desmoplasia, inflammation score, and lymphatic metastasis).

Immunohistochemistry of macrophage markers (CD68, CD163, and iNOS) in tumor samples. Representative image of (a–c) CD68, (d–f) CD163, and (g–i) iNOS macrophage markers determined by single labeling immunohistochemistry in mucosa samples of healthy colon and tumor from CRC patients. Tumor tissue with high or low density of the different macrophage markers is shown. Magnification 20× (bar 200 µm).
Due to the importance of CKs in the chemotaxis and cell infiltration to the tumor, correlation analysis between CCL2, CCL3, and CCL4 with the macrophage markers in the samples demonstrated a positive correlation between CCL4 and CD163 (Spearman correlation coefficient: 0.4660, p = 0.0443; Table 2). No correlation was identified between CCL2 and CCL3 with macrophage markers.
Correlation analysis between macrophage markers and chemokine levels in tumor tissue through Spearman’s test.

r: Spearman correlation coefficient.
Bold numbers indicate p values <0.05.
Similarly, correlation analysis between CK levels and macrophage markers with histopathological characteristics showed a negative correlation of iNOS and desmoplastic reaction (Spearman correlation coefficient: −0.3724, p = 0.0467; Table 3). Alternatively, when comparing the density of macrophage markers according to the type of desmoplasia (Figure 3), we identified that tumors with immature desmoplasia have a higher CD163 density compared to mature/intermediate desmoplasia. No correlation was identified between CKs tumor levels or macrophage markers with the TNM stage (Table 3).
Correlation analysis of macrophage markers and tumor CKs levels with tumoral stage (TNM), differentiation and desmoplasia grade, leukocytic infiltration and lymphatic metastasis, through Spearman’s test.

r: Spearman correlation coefficient.

Tumor density of macrophage markers according to type of desmoplasia. The macrophage markers CD68, CD163, and iNOS were plotted according to the type of desmoplasia (mature/intermediate or immature). CD163 levels were higher in tumor with immature desmoplasia (Mann–Whitney test, two-tailed, p < 0.05).
These results suggest that CCL4 could participate in the profile of TAMs; moreover, the subpopulation of these macrophages in the tumor microenvironment can be associated to the characteristics of the tumor.
In relation to the inflammatory mediators, no correlation of tumor VEGF content with macrophage markers was determined. However, although no differences were found in TNF-α content in healthy and tumor tissues, direct correlation between tumor TNF-α levels with CD163 was observed (Supplementary Table 1).
CK and cytokine levels in plasma samples from CCR patients and controls
The content of CKs in plasma was studied to relate to the clinical–demographic characteristics of the patients and to determine whether changes at the tumor tissue are reflected in peripheral blood. For this, plasma levels of the CCL2, CCL3, CCL4, and the cytokines TNF-α and VEGF were examined in 32 patients with colon cancer and 15 controls (Figure 4).

Plasma levels of chemokines and cytokines in healthy controls and CRC patients. The graphs represent the CCL2, CCL3, CCL4, TNF-α, and VEGF levels in plasma samples from controls and CRC patients and the mean ± SD. CCL3 levels were higher in patients compared to controls (Mann–Whitney test, two-tailed, p < 0.05).
CCL3 was the only one that showed higher levels in CRC patient’s plasma (with a mean ± SD 5.186 ± 3.016 pg/mL) compared to control group (3.489 ± 1.446 pg/mL) (p = 0.0154).
A correlation analysis applied between the content of CCL2, CCL3, and CCL4 in tumor tissue with plasma levels of 20 CRC patients demonstrated that only CCL2 showed a negative correlation (p = 0.0420; Figure 5). TNF-α and VEGF did not show correlation between plasma and tumor levels in CRC patients (Supplementary Figure 3).
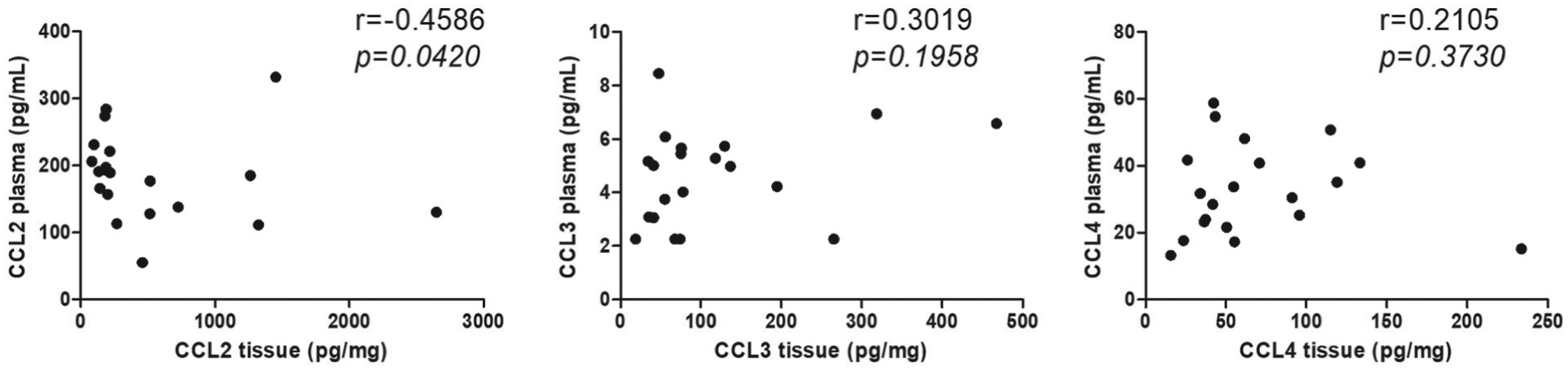
Correlation between chemokines in tumor and plasma samples. The graph represents the levels of chemokine in plasma samples and tissue from CRC patients. Moreover, correlation analysis was made through Spearman’s test (r = Spearman correlation coefficient and p value).
Because plasma cytokine levels are age related, 41 we confirmed this correlation with CCL2, CCL3, and VEGF, but not with TNF-α (p = 0.0528) or CCL4 (p = 0.1090) in control subjects. However, in CRC patients, no correlations were found between age and CKs (Table 4). These data suggest that plasma CK/cytokine levels can be tumor influenced, but not by age in colon cancer patients. Changes in CK plasma levels can be a sign of a systemic inflammatory condition in cancer patients, which may prove to be a very interesting study.
Correlation analysis of the plasma cytokines and CK levels with the characteristics of patients (n = 32) and controls (n = 15) through Spearman’s test.

r: Spearman correlation coefficient.
Bold numbers indicate p values <0.05.
Due to the fact that VEGF and TNF-α are associated with increased risk of disease recurrence and poor survival of cancer patients, we further evaluated whether the content of CKs in plasma are related to these soluble mediators. Analysis showed that plasma levels of CCL3 and CCL4 had a positive correlation with VEGF (Table 5). In the same way, the plasma levels of CCL2, CCL3, and CCL4 showed positive correlation with TNF-α (Table 5). These data may suggest that the plasma CK levels can be biomarkers of colon cancer prognosis as well as TNF-α or VEGF.
Correlation analysis of the plasma cytokine levels versus plasma CKs content in patients with colon cancer (n = 32), through Spearman’s test.

r: Spearman correlation coefficient.
Bold numbers indicate p values <0.05.
Discussion
In this study, we identified elevated protein levels of CKs involved in migration of macrophages in tumor samples from patients with colon cancer.
Moreover, we observed correlations between CKs levels, macrophage molecular markers and clinical–pathological characteristics, suggesting that CKs act as a key factor in the cell types that can infiltrate the tumor and enhance disease progression.
Our analysis indicated that CCL2, CCL3, and CCL4 are overexpressed in colon cancer, but show no changes in the levels of CCL5 and CX3CL1. Other groups have also described these changes in CK levels in colon cancer using techniques such as ELISA, 32 ribonuclease protection assay, 29 and RT-PCR. 30 Unlike our work, Erreni et al. 42 identified a higher content of CX3CL1 in an analysis of 100 colorectal tumors by IHC, predominantly in the tumors of patients in stage TNM I and II, although not in III. It is probable that in our study, the characteristics of the cohort of patients enrolled or the technique used to detect CK content could explain the differences observed with this report.
TNF-α and VEGF are considered to reflect inflammatory response and participate in both the initiation and progression of cancer. 5 Our results have shown a high VEGF content in tumors compared to paired normal tissue, although no differences were found in TNF-α content, confirming other reported studies.43,44 In addition, TNF-α content in tumor tissue directly correlated with CD163, which is not consistent with the literature describing TNF-α repression of macrophage polarization to an M2 profile.45–47 A deeper characterization of the microenvironment and the cytokines that predominate in CRC is necessary to define the participation of TNF-α in macrophage activation. In addition, high levels in CKs and cytokines have been identified in plasma and serum from CRC patients, suggesting a systemic inflammatory condition in patients with cancer, thus resulting in a worse outcome.36,48 An inherent factor is TNF-α, a cytokine with a diverse role in immunological and metabolic responses, related to promotion of either the death or survival of tumor cells.49,50 Moreover, the high levels of plasma TNF-α in patients under surgery have been associated with increased CRC-specific mortality. 36 However, VEGF is involved in vascularization processes, which may be key in tumorigenesis and metastasis. Many tumors produce large amounts of VEGF, with potential in the diagnosis and prognosis of cancer, 51 specifically, serum and plasma VEGF levels have been associated to reduced survival in colon cancer. 34
Our study identified that the CKs and cytokines at the tumor are not systemically reflected to the plasma content. This confirms current literature, in which poor prognosis in cancer patients is associated with systemic inflammation, while a good prognosis is associated with a proinflammatory response in the tumor, thus the relationship between the two types of inflammation remains unclear. 52 This suggests that systemic inflammatory mediators derive from processes associated with the pathological state, but not directly from the tumor.
Although the levels of plasma cytokines and CKs in patients do not show significant differences with the control group, it seems interesting that in the study, these inflammatory mediators have a positive correlation with age in the control group but not in patients. This correlation between plasma cytokine levels and age in the population has been previously described and could be explained by pathological factors associated with age such as atherosclerosis, arthritis and hypertension. 41 However, this correlation does not replicate in patients with cancer which strongly suggests that the pathology is a determinant factor in the plasma levels of cytokines and CKs regardless of age. The levels of plasma VEGF and TNF-α directly correlate with CCL4 and CCL3, thus alluding that the plasma CKs levels can be a biomarker of poor prognosis.
The cellular source of CKs in cancer appears to be stromal cells (fibroblasts and leukocytes), as well as tumor cells, with CCL2 produced by tumor cells in CRC. 29 In murine models of colon cancer, it has been demonstrated that cancer-associated fibroblast (CAF) can be an important source of CCL3. 53
In CRC, the protein levels of CCL2 have been correlated with the degree of infiltration of macrophages CD68+. 29 In this study, we demonstrate that CCL4 tumor levels have a positive correlation with CD163, usually related to poor clinical outcomes and lower cumulative survival than patients with low expression of the M2 marker. 27 This poor prognosis related to TAMs with M2 profile can produce enzymes and inhibitors that regulate the digestion of the ECM, metastasis, and angiogenesis.54,55
The relationship between CCL4 and CD163 could explain a participation of this CK in monocyte and macrophage infiltration into the tissue. However, CKs in tumors are more than just leukocyte attractants because they can activate a distinct differentiation program in monocytes. Roca et al. 56 demonstrated that CCL2, which is highly expressed in prostate cancer, polarizes human monocytes toward a type-M2 macrophage profile, suggesting a pro-tumorigenic role of this CK in this cancer. This explanation has not been explored for CCL4, which could favor a M2 macrophage profile in colon cancer. Recent data support that CCR5 blockade (one of the receptors of CCL4, CCL3, and CCL5) in in vitro organotypic CRC cultures induces a repolarization of TAMs. This is achieved through STAT3 expression (a transcription factor associated with M1 macrophage profile), favoring an anti-tumor microenvironment. 57 Similarly, CCR5 or CCR3 deficient murine colon cancer model (induced by azoxymethane (AOM) and sodium dextran sulfate (DSS)) contained less tumor formation than wild-type mice. 53 These combined results suggest that the CKs studied favor a pro-tumoral microenvironment predominantly with M2 macrophages.
The M1 profile marker (iNOS) showed negative correlation with desmoplastic reaction score, whereas the M2 marker (CD163) has higher expression in tumors with immature desmoplasia. Furthermore, a well-recognized ECM alteration in cancer is collagen deposition in tumor tissue. 58 Immature desmoplasia correspond to tumors with keloid-like collagen or myxoid stroma and this has been associated to poor survival in CRC patients. 7 Different works have shown that TAMs control the composition of the ECM directly or through the activation of fibroblasts. Recently, in this field of study, it was described that TAMs promote tumor progression by contributing to ECM remodeling in a CRC murine model. 59 At the same time, Ly6Chi monocytes were infiltrated into tumors through a CCR2-dependent process and then matured into TAMs. Also, many ECM-related proteins differentially expressed in TAMs from CCR2−/− and WT colorectal tumors (including collagens, TGFβ-induced protein, some proteoglycans, and others) were detected in a proteome analysis.
Moreover, TAMs located preferentially at the invasive margins of colorectal tumors significantly contributed to organization of collagenous matrix through regulation of CAFs producing type-XIV and I collagen. In addition, an increase in TAMs could define ECM proteins (type I, VI, and XIV collagen) in CRC patients, which associate with a clinical application for TAMs-induced ECM remodeling. 59
In other studies, macrophages differentiated within a three-dimensional (3D) organotypic model (based on decellularization of normal and tumor tissues from CRC patients), upregulated anti-inflammatory markers and downregulated proinflammatory markers, therefore ECM composition and organization contributes to macrophage activation. 60
Finally, a decrease in iNOS could be a consequence of the combined effect of upregulated CKs in tumors with increased desmoplastic reaction, favoring the M1 < M2 balance.
In conclusion, high levels in tumor or plasma samples of CKs associated to macrophages infiltration in CCR patients could promote a pro-tumor microenvironment. We believe that CCL4 could be a good candidate as a prognosis biomarker (such as survival) in CCR and a potential therapeutic target.
Supplemental Material
Fig_2_Suppl – Supplemental material for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer
Supplemental material, Fig_2_Suppl for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer by Marjorie De la Fuente López, Glauben Landskron, Daniela Parada, Karen Dubois-Camacho, Daniela Simian, Maripaz Martinez, Diego Romero, Juan Carlos Roa, Isidora Chahuán, Rocío Gutiérrez, Francisco Lopez-K, Karin Alvarez, Udo Kronberg, Sebastian López, Antonella Sanguinetti, Natalia Moreno, Mario Abedrapo, María-Julieta González, Rodrigo Quera and Marcela A Hermoso-R in Tumor Biology
Supplemental Material
supplementary_Figure_3 – Supplemental material for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer
Supplemental material, supplementary_Figure_3 for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer by Marjorie De la Fuente López, Glauben Landskron, Daniela Parada, Karen Dubois-Camacho, Daniela Simian, Maripaz Martinez, Diego Romero, Juan Carlos Roa, Isidora Chahuán, Rocío Gutiérrez, Francisco Lopez-K, Karin Alvarez, Udo Kronberg, Sebastian López, Antonella Sanguinetti, Natalia Moreno, Mario Abedrapo, María-Julieta González, Rodrigo Quera and Marcela A Hermoso-R in Tumor Biology
Supplemental Material
supplementary_Table_1_corr_MH – Supplemental material for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer
Supplemental material, supplementary_Table_1_corr_MH for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer by Marjorie De la Fuente López, Glauben Landskron, Daniela Parada, Karen Dubois-Camacho, Daniela Simian, Maripaz Martinez, Diego Romero, Juan Carlos Roa, Isidora Chahuán, Rocío Gutiérrez, Francisco Lopez-K, Karin Alvarez, Udo Kronberg, Sebastian López, Antonella Sanguinetti, Natalia Moreno, Mario Abedrapo, María-Julieta González, Rodrigo Quera and Marcela A Hermoso-R in Tumor Biology
Supplemental Material
Suppl_Fig_1 – Supplemental material for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer
Supplemental material, Suppl_Fig_1 for The relationship between chemokines CCL2, CCL3, and CCL4 with the tumor microenvironment and tumor-associated macrophage markers in colorectal cancer by Marjorie De la Fuente López, Glauben Landskron, Daniela Parada, Karen Dubois-Camacho, Daniela Simian, Maripaz Martinez, Diego Romero, Juan Carlos Roa, Isidora Chahuán, Rocío Gutiérrez, Francisco Lopez-K, Karin Alvarez, Udo Kronberg, Sebastian López, Antonella Sanguinetti, Natalia Moreno, Mario Abedrapo, María-Julieta González, Rodrigo Quera and Marcela A Hermoso-R in Tumor Biology
Footnotes
Acknowledgements
The authors thank Biobanco de Tejidos de la Universidad de Chile. They would also like to thank David Cox for his editing contribution and Santiago Sepulveda for his technical support.
Marjorie De la Fuente López and Marcela A Hermoso-R contributed to design concept; Marjorie De la Fuente López, Glauben Landskron, Daniela Parada, Karen Dubois-Camacho, and Diego Romero contributed to data acquisition; Marjorie De la Fuente López, Daniela Parada, Karen Dubois-Camacho, Isidora Chahuán, and Diego Romero contributed to data analysis and interpretation; María-Julieta González, Rodrigo Quera, and Marcela A Hermoso-R contributed to results interpretation; Marjorie De la Fuente López, Isidora Chahuán, and Rocío Gutiérrez drafted the manuscript; Daniela Simian, Maripaz Martinez, Francisco Lopez-K, Udo Kronberg, Sebastian López, Antonella Sanguinetti, and Natalia Moreno involved in patient recruitment; Daniela Simian, Maripaz Martinez, Sebastian López, Antonella Sanguinetti, and Natalia Moreno contributed to clinical data base generation; Marjorie De la Fuente López, Glauben Landskron, Diego Romero, and Juan Carlos Roa contributed to histological classification, acquisition, analysis, and interpretation of immunohistochemistry assay; Marjorie De la Fuente López, Glauben Landskron, Daniela Parada, Karen Dubois-Camacho, Daniela Simian, María-Julieta González, Rodrigo Quera, and Marcela A Hermoso-R reviewed the manuscript critically. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the ethics committee from Clínica Las Condes and Hospital Clínico Universidad de Chile, Santiago, Chile. All patients gave their written informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grant nos 3150328 and 1170648 from the National Fund for Scientific and Technological Development (FONDECYT; OAIC No. 844/16), Santiago, Chile.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
