Abstract
Partitioning defective 3–like protein is a novel cell polarity protein. Recently, partitioning defective 3–like protein has been demonstrated with tumor-promoting function by disrupting tight junction, inhibiting tumor suppressor liver kinase B1, and maintaining mammary stem cells. For the first time, we studied partitioning defective 3–like protein expression in malignant colorectal cancer. We used immunohistochemistry scoring system to evaluate partitioning defective 3–like protein expression in 196 colorectal cancer tissues and 33 adjacent normal tissues. We found that colorectal cancer tissues had much stronger partitioning defective 3–like protein immunoreactivity than normal tissues, and colorectal cancer patients with positive partitioning defective 3–like protein expression were characterized with higher cancer stages, metastasis, poor tumor differentiation, larger tumor size, as well as high levels of colorectal cancer markers carcinoembryonic antigen and cancer antigen 19-9. Besides, partitioning defective 3–like protein overexpression was independently predictive of lower survival rate in colorectal cancer patients, even after adjusting the influence of cofactors. Moreover, we also found that partitioning defective 3–like protein was associated with rapid growing colorectal cancer, while knockdown of partitioning defective 3–like protein expression largely inhibited cancer cell proliferation. Our study provided the first evidence that partitioning defective 3–like protein was overexpressed in colorectal cancer and associated with disease malignancy. Also, partitioning defective 3–like protein may serve as a promising prognostic marker and a potential therapeutic target for colorectal cancer treatment. Further study is necessary to understand the regulatory mechanism of partitioning defective 3–like protein in colorectal cancer and the feasibility of its application in clinic.
Keywords
Introduction
Partitioning defective 3 homolog (PAR3) is a key regulator of cell polarity and, is therefore, involved in many polarization events during embryo development, neural differentiation, epithelial morphology, and cell migration.1,2 Recently, abnormal cell polarity caused by PAR3 dysfunction has been found to modulate a series of oncogenic and tumor suppressor pathways and finally contribute to human cancers. For example, downregulated PAR3 decreases cell–cell cohesion and promotes breast cancer invasion and metastasis.3,4 Also, PAR3 controls tight junction assembly in pancreatic cells and absence of PAR3 is associated with poor prognosis of pancreatic cancer. 5 In addition, PAR3 reduction has also been frequently reported in esophageal squamous cell carcinoma, 6 head and neck squamous cell carcinoma, and primary glioblastoma tumors, 7 all of which suggest a tumor-suppressing function of PAR3. However, all the growing evidences implicated the oncogenic role of PAR3. Increased PAR3 expression is evident in the metastatic human liver cancer and renal clear cell carcinoma8–10 and correlated with the poor prognosis of ovarian cancer, 11 while knockdown of PAR3 delays the formation of papillomas by decreasing cell proliferation and increasing apoptosis. 12
Previously, a novel homolog of PAR3, PAR3-like protein (PAR3L), has been identified to colocalize with PAR3 at tight junctions. 13 Although the biological functions of PAR3L remain as largely unknown, N-terminal region of PAR3L has been revealed to disrupt tight junction formation.13,14 Besides, PAR3L expression inhibits tumor suppressor protein liver kinase B1 (LKB1) and is essential for mammary gland regeneration and mammary stem cell maintenance, while depletion of PAR3L suppresses self-renewal and promotes apoptosis. 15 These reports implicated a tumorigenic potential of PAR3L by sustaining the stemness of the cell and disrupting cell–cell adhesions. However, the role of PAR3L in the development and prognosis of colorectal cancer (CRC) has not been well characterized.
CRC originates from colon and rectum. It is the third most common cancer in the world with approximately 1.4 million new cases and 700,000 deaths each year. 16 Like many other cancer types, CRC is characterized with rapid cell growth and disrupted cell polarity,17,18 and loss of LKB1 function has also been observed in CRC from time to time.19–22 Our recent study has provided evidence that PAR3L may support CRC cell growth by inhibiting LKB1 and downstream adenosine monophosphate–activated protein kinase (AMPK) signaling pathway. 23 In this study, we evaluated the potential application of PAR3L in clinic by analyzing PAR3L expression in CRC cancer tissues and its association with CRC prognosis.
Material and methods
CRC specimens
196 CRC patients undergoing surgery at The First Affiliated Hospital of Nanchang University from 2000 to 2005 were recruited and followed up from 1 to 122 months, with a median of 51 months. Informed consent was obtained from all the patients prior to the study. All procedures were performed with the approval of Institutional Ethics Committee of The First Affiliated Hospital of Nanchang University in accordance with 1964 Helsinki declaration. The patients included 100 males (51.02%) and 96 females (48.97%), with an average age of 67.24 ± 13.42 years. Pathologic stage was determined according to the tumor–node–metastasis (TNM) classification revised in 1977. With this, 36 patients were classified at Stage I, 59 were at Stage II, 61 were Stage III, and the rest 40 were Stage at IV. In addition, 23 patients had well-differentiated tumor, 142 had moderately differentiated, and 31 had poorly differentiated. Blood carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA 19-9) were obtained from routine blood tests at the time of surgery (normal range: CEA < 5 ng/mL; CA 19-9 < 37 U/mL). Patient survival was measured from the date of surgery to the date of disease progression diagnosed or the date of death from any cause.
Immunohistochemistry
In total, 196 formalin-fixed and paraffin-embedded CRC tissues and 33 adjacent normal tissues were obtained. Tissue blocks were cut into 4 µm sections followed by deparaffinization, dehydration, and heat-induced epitope retrieval, and endogenous peroxidase activity was then blocked by incubation with hydrogen peroxide solution in methanol. Then, the sections were blocked with serum and incubated overnight with antibody targeting PAR3L (1:10 dilution; ab122264, Abcam) and Ki-67 (1:100 dilution; ab15580, Abcam, Cambridge, MA, USA) in a moist chamber. Then, EnVision reagent (Dako, Carpinteria, CA, USA) was added to the sections, which were further visualized with 3,3′-diaminobenzidine tetrahydrochloride and counterstained with hematoxylin. To confirm the staining specificity, sections without primary antibody treatment were immunostained and used as negative controls.
Immunohistochemistry scoring system
Immunohistochemistry (IHC) scoring system was then used to quantitatively evaluate the expression of PAR3L and Ki-67. 24 For a particular protein, its immunoreactive intensity in each cell was defined as weak (score: 1), moderate (score: 2), and strong (score: 3). In each field of vision at 400× magnification, IHC score was calculated as 3 × (percentage of strongly stained cells) + 2 × (percentage of moderately stained cells) + 1 × (percentage of weakly stained cells) and the mean IHC score of five random visual fields in each section was determined as the expression of corresponding protein.
Gene silencing
According to American Type Culture Collection (ATCC) protocol, human CRC cell line DLD-1 was cultured in RPMI-1640 medium (HyClone) containing 10% fetal bovine serum (FBS) and Caco-2 was cultured in Eagle’s Minimum Essential medium (HyClone, Logan, UT, USA) with 20% FBS. All cell cultures were incubated at 37°C in a 5% CO2 humidified atmosphere. Small interfering RNA (siRNA) targeting PAR3L (siPAR3L; 5′-CUGUUCUUGUCAUAUCACUCA-3′, Thermo Fisher Scientific, Waltham, MA, USA) was introduced to each cell with Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA, USA). Non-targeting control (siNC; 5′-AAGCUCGUCUCGUGCCAAUAA-3′, Thermo Fisher Scientific) was introduced into CRC cells as negative controls.
Cell proliferation assay
siPAR3L and siNC transfected CRC cells were seeded onto 96-well plates at a density of 1 × 103 cells per well and incubated for 6, 12, 24, 48, and 72 h. BrdU Cell Proliferation Kit (Abcam) was used to detect cell density at each time point for each cell line. Experiments were performed with five replicates.
Statistical analysis
All statistical analyses were performed using SPSS v.20.0 (IBM, Armonk, NY, USA). Chi-square test and Student’s t test were used to evaluate the association between patient clinical characteristics properly. Pearson correlation was used to assess the correlation between PAR3L and CEA, CA 19-9, and Ki-67. Overall survival and disease-free survival were analyzed using Kaplan–Meier method, and log-rank test was used to identify the differences between survival curves. Multivariate prognosis analysis was done with Cox proportional hazards regression model with forward stepwise procedure. A p value less than 0.05 was regarded as statistically significant for all analysis.
Results
PAR3L is overexpressed in CRC tissues and associated with the severity of the disease
We used IHC scoring system to analyze PAR3L protein expression in primary CRC tissues as well as in adjacent normal tissues (see Material and methods section). As shown in Figure 1(a) and (b), CRC tissues exhibited strong PAR3L immunoreactivity while the staining was absent in normal tissues (Student’s t test: p < 0.001), indicating elevated PAR3L expression in CRC. Besides, we also found that PAR3L expression is gradually increased with CRC progression (Figure 1(c), p < 0.001) and all CRC stages can be well discriminated from controls based on PAR3L expression (Figure 1(d), All: area under the curve (AUC) = 0.938, p < 0.001; Stage I: AUC = 0.873, p < 0.001; Stage II: AUC = 0.939, p < 0.001; Stage III: AUC = 0.951, p < 0.001; Stage IV: AUC = 0.975, p < 0.001).

(a) Representative IHC staining of PAR3L in CRC (right) and adjacent normal tissues (left). (b) Comparison of PAR3L expression between normal and CRC tissues based on IHC scoring system. (c) PAR3L expression in CRC tissues at different cancer stages. (d) ROC curves for the use of PAR3L expression in discriminating CRC at different stages from controls. (e) Pearson correlation between PAR3L expression and plasma CEA levels. (f) Pearson correlation between PAR3L expression and plasma CA 19-9 levels. (g) Pearson correlation between PAR3L expression and Ki-67 labeling index.
In clinic, CEA and CA 19-9 are widely used CRC biomarkers in blood (normal range: CEA < 5 ng/mL; CA 19-9 < 37 U/mL). As shown in Figure 1(e) and (f), PAR3L expression is positively correlated with plasma levels of CEA (Figure 1(e), Spearman correlation: rs = 0.669, p < 0.001) and CA 19-9 (Figure 1(f), Spearman correlation: rs = 0.697, p < 0.001) in CRC patients. All these results demonstrated that PAR3L overexpression is highly associated with the severity of CRC.
PAR3L IHC scores varied from 7 to 170 in CRC tissues (median: 49). Positive and negative PAR3L reactions were defined in a previously described way, 25 in which, CRC tissues with PAR3L IHC scores exceeding 41 (the maximal score within +3 standard deviation of the mean value in normal tissues) were classified as positive (n = 116, 59%) and the rest were negative (n = 80, 41%). We then compared patient clinicopathological features between the two groups. As listed in Table 1, positive PAR3L expression was correlated with advanced cancer stages (p = 0.003), lymph node metastasis (p < 0.001), distant metastasis (p < 0.001), poor differentiation (p = 0.037), larger tumor size (p < 0.001) as well as high plasma levels of CEA (p < 0.001) and CA 19-9 (p < 0.001).
Correlation between immunohistochemical analysis of PAR3L and clinicopathological features of CRC patients.

PAR3L: partitioning defective 3–like protein; CRC: colorectal cancer; CEA: carcinoembryonic antigen; CA 19-9: cancer antigen 19-9.
As mentioned above, PAR3L has been found to support self-renewal of mammary stem cells; 15 therefore, we also compared PAR3L expression with a well-known cancer proliferation marker Ki-67. 26 The expression of Ki-67 in CRC tissues was also evaluated by IHC scoring system. As shown in Figure 1(f) and Table 1, PAR3L was highly correlated Ki-67 expression (Spearman correlation: rs = 0.694, p < 0.001), indicating that PAR3L overexpression is associated with the high proliferation of cancer cells.
PAR3L overexpression is predictive of poor CRC prognosis
As reported, increased PAR3 expression was predictive of the good prognosis of pancreatic cancer 5 as well as the poor prognosis of hepatocellular carcinoma and renal clear cell carcinoma.8–10 We then used Kaplan–Meier analysis to investigate the association of its homolog PAR3L with the survival of CRC patients. As shown in Figure 2(a), CRC patients with positive PAR3L expression had significantly lower survival rate than those with negative PAR3L tumors (log-rank test: p = 0.047). The disease-free survival rate was also significantly lower in patients with positive PAR3L tumors (Figure 2(b), log-rank test: p = 0.008). Moreover, we also used Cox proportional hazards regression was used to adjust the influence of covariates, including age, gender, and the severity of the disease and identified PAR3L as an independent predictive factor for the overall survival in CRC (Table 2, PAR3L: p = 0.036, relative risk (RR): 1.008, 95% confidence interval (CI): 1, 1.015).
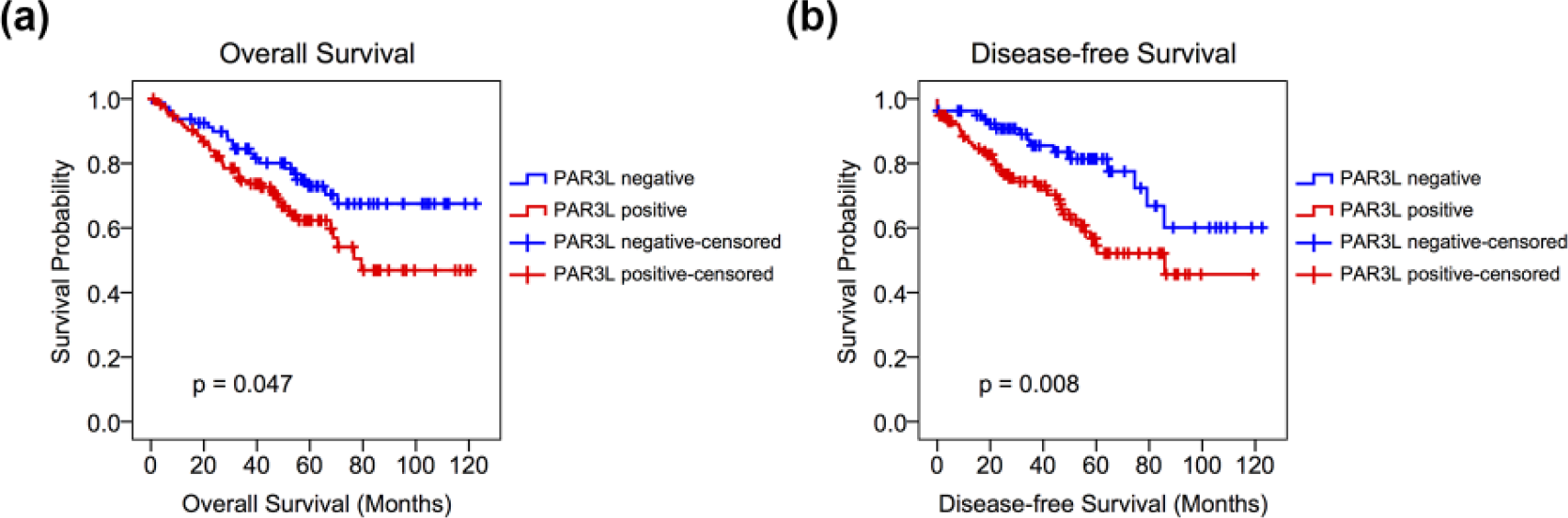
Kaplan–Meier curves for (a) overall survival and (b) disease-free survival with respect to the expression of PAR3L. Blue curves stand for CRC patients with negative PAR3L expression, while red curves are for patients with positive PAR3L expression.
Cox proportional hazards regression model analysis of CRC patients.

CRC: colorectal cancer; CI: confidence interval; RR: relative risk; PAR3L: partitioning defective 3–like protein; CEA: carcinoembryonic antigen; CA 19-9: cancer antigen 19-9.
Knockdown of PAR3L inhibits CRC cell proliferation
The above results demonstrated the high expression of PAR3L in CRC. We then examined whether suppressing PAR3L expression can inhibit the growth of CRC cells. To this end, we transfected siPAR3L into two different CRC cancer cell lines, DLD-1 and Caco-2 (Figure 3(a) and (c)), and we found that the proliferation of all CRC cells was largely reduced by PAR3L silencing (Figure 3(b) and (d)). This result indicated that PAR3L was essential for CRC cell viability, and knockdown of PAR3L might be a promising strategy for CRC treatment.

Knockdown of PAR3L expression in CRC cell lines (a) DLD-1 and (c) Caco-2. Cell proliferation was inhibited in both (b) DLD-1 and (d) Caco-2 cell lines (*p < 0.05; **p < 0.01; ***p < 0.001).
Discussion
For most of the cells, the intracellular organelles and plasma membrane are organized in an asymmetric way, which is known as cell polarity. 27 Correct cell polarity determines cell fate and behavior and plays a crucial role in regulating cell proliferation, differentiation, and morphogenesis, while disruption of cell polarity has been considered as a hallmark of advanced human cancer by decreasing cell–cell adhesion and assisting cancer cell to invade into surrounding normal tissues.1,2 Consistent with this, many polarity regulators are proposed as tumor suppressors, and their decreased expression is considered to support hyperproliferation and metastasis, and has been observed during the progression of many cancers. However, recent evidences have revealed that some members of cell polarization machinery also carry tumor-promoting property, and their aberrant expression and function occur at the early stages of cancer, which can therefore be used as a predictive marker. For example, PAR6 has been reported to induce breast cancer cell proliferation without affecting cell polarity, and its overexpression was observed early in precancerous breast lesions and sustained during breast cancer development, suggesting the regulatory role of PAR6 in the initiation and progression of breast cancer. 28 Besides, another cell polarity protein atypical protein kinase C (aPKC) has also been frequently identified to promote tumor growth. Overexpressed aPKC has been implicated in malignancies originating from lung, breast, liver, colon, and ovary and correlated with poor cancer prognosis.18,29–32 Moreover, polarity protein PAR3 possesses both anti-tumor and pro-tumor functions in a tumor type–dependent manner. In pancreatic tumor, glioblastoma, head and neck squamous cell carcinoma, and esophageal squamous cell carcinoma, PAR3 acts as tumor suppressor and is positively associated with overall survival rate of cancer patients.3–7 However, in some other tumors, such as liver cancer, ovarian cancer, and renal clear cell carcinoma, PAR3 overexpression supports cancer cell proliferation and is associated with poor cancer prognosis.8–11 All these evidences implicated the complex regulatory network of cell polarity and its interaction with oncogenic and tumor suppressor pathways.
Recently, PAR3L has been identified as a new member of polarity protein family and exhibits pro-oncogenic activity by maintaining cell stemness and disrupting cell tight junctions.13–15 In this study, we, for the first time, evaluated the expression of PAR3L in malignant CRC. Our results revealed that PAR3L was overexpressed in CRC tissues and is an indicative of cancer severity and poor prognosis. Moreover, we also provided evidence that knocking down PAR3L expression efficiently inhibited cancer cell proliferation and may serve as a novel therapeutic strategy for CRC. PAR3L is located at tight junctions between cells. The N-terminal region of PAR3L has been found to disrupt tight junctions by blocking the normal association of tight junction protein 1 (TJP1) to cell–cell boundaries,13,14 which may facilitate CRC cancer cells to disassociate from primary tissue and migrate into adjacent tissue. In addition, PAR3L can also bind to LKB1 and inhibit LKB1 activity. 15 LKB1 is a tumor suppressor, and absence of LKB1 has been frequently observed in CRC.19–22 Our recent study has demonstrated that PAR3L may support CRC cell growth by inhibiting LKB1/AMPK signaling pathway, while PAR3L depletion resulted in the activation of AMPK signaling cascade and, therefore, subjected CRC cell to death. 23 Besides, LKB1 has also been reported to physically interact with p53 to activate apoptotic cell death33,34 and suppress cell migration by activating anoikis. 35 Given these, PAR3L overexpression in CRC may also lead to uncontrolled cell proliferation and aggravate cancer cell metastasis and invasion by suppressing LKB1/p53 cascade.
Altogether, our study supported the oncogenic role of PAR3L in CRC development and progression and demonstrated the possibility of PAR3L as a prognostic marker and therapeutic target in clinic. However, several limitations in this study should be acknowledged. First, the patient cohort in the study was still relatively small and only one study center was involved. We are currently trying to recruit more patients at different disease stages and validate our result with more other centers. Second, although previous studies demonstrated that PAR3L may execute tumorigenic function through inhibiting LKB1 and affecting downstream signaling network, the regulatory mechanism of PAR3L remains largely unknown and needs to be investigated in the future. Third, in this study, we only detected a single biomarker PAR3L. Recently, a scoring system comprising four biomarkers (p53, p21, Ki-67, and CD133) has been proposed to predict therapy response in rectal cancer patients and achieve high sensitivity and sufficient accuracy. 36 This indicated that, in addition to PAR3L, it is necessary to explore more potential biomarkers and generate more reliable diagnosis. At last, besides CRC, abnormal cell polarity function and abnormal LKB1 activity have been implicated in the development and progression of many other cancers. It is possible that PAR3L expression is correlated with those abnormalities and more efforts should be placed to understand the association of PAR3L with more other cancer types.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by National Natural Science Foundation of China (Project No. 81402401).
