Abstract
The triterpenoid, bauerenol, from Suregada angustifolia (Baill. ex Muell.-Arg.) Airy Shaw (Euphorbiaceae) was screened for anti-cancer property using hepatocellular carcinoma cell line, HepG2. Bauerenol exhibited growth inhibitory and apoptosis inducing potential against HepG2 cancer cells. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide cytotoxic assay revealed that bauerenol treatment significantly reduced the growth of HepG2 cells in a time- and dose-dependent manner with 50% growth inhibitory concentration doses of 45 and 25 µg/mL at 24 and 48 h treatments, respectively. Bauerenol-induced cell death reflected apoptotic morphological features, that is, cell membrane blebbing, vacuolization, chromatin condensation, and nuclear fragmentation. In addition, bauerenol treatment diminished the mitochondrial membrane potential, by inducing the efflux of cytochrome c, downregulating the levels of anti-apoptotic Bcl-2 as well as upregulating the levels of pro-apoptotic Bax, and inducing caspase activation and poly (ADP-ribose) polymerase cleavage. Moreover, bauerenol treatment activates p38MAPK and inactivates the anti-apoptotic kinases Akt and ERK1/2 through the induction of reactive oxygen species. Furthermore, bauerenol-mediated S-phase arrest was associated with downregulation of cell cycle-rate-limiting factor (cyclin D1) and upregulation of cyclin-dependent kinase inhibitor p21 and tumor suppressor p53. Interestingly, pre-treatment of cells with reactive oxygen species inhibitor and p38 inhibitor significantly decreases bauerenol-induced cytotoxicity, Bax upregulation, and p38 activation. This study clearly states that bauerenol induces cell cycle arrest and apoptosis through the reactive oxygen species–dependent p38MAPK activation in HepG2 cancer cells.
Introduction
Hepatocellular carcinoma (HCC) is a common and aggressive malignant tumor worldwide. It is the third most frequent cause of cancer-related deaths and the sixth most prevalent cancer. Currently, it is mainly treated by surgery and chemotherapy. The curative effect of several existing chemotherapeutic drugs is associated with numerous side effects, including drug resistance.1,2 Yet natural products that play an important role in chemotherapy still remain as one of the best approaches for the discovery of new molecules 3 and have contributed approximately 60% cancer chemotherapeutic drugs. 4 Recently, the search for novel chemotherapeutic drugs from traditional medicinal plants to combat liver carcinogenesis has been increasing in order to reduce the adverse effect of the existing drugs. Therefore, an Indian Euphorbiaceous species, S. angustifolia (Baill. ex Muell.-Arg.) Airy Shaw, was chosen for investigation. This species is a small tree distributed in the deciduous and semi-evergreen forests of the tropical region. The Kanis in India use this plant medicine to treat skin infections and toothache. 5 Moreover, anti-fungal and anti-bacterial properties of this medicinal plant have been reported.6,7 Bioactive triterpenoid, bauerenol, isolated from this species was screened against hepatocellular carcinoma (HepG2) cells and the results are reported here to support the anti-cancerous property of the compound.
Materials and methods
Collection of plant material
The plant material was collected from Tirunelveli district, Tamil Nadu, India, and authenticated by one of the authors (Prof. M.B. Viswanathan) and a voucher specimen (MBV, CR & PSK 1374) has been deposited in the herbarium of the Centre for Research and Development of Siddha-Ayurveda Medicines (CRDSAM), Department of Plant Science, Bharathidasan University, Tiruchirappalli, Tamil Nadu, India.
Instrumentation
Melting point was determined using open capillary method on a heating block apparatus. Fourier transform infrared spectroscopy (FT-IR) was recorded on a PerkinElmer FT-IR grating spectrophotometer (PerkinElmer Singapore Pte Limited, Singapore). 1H NMR (nuclear magnetic resonance) and 13C NMR were recorded in CDCl3 on a Bruker instrument at 400 and 100 MHz, respectively, with tetramethylsilane (TMS) as the internal standard (Bruker Corporation, Billerica, Massachusetts, USA).
Electron ionization mass spectrometry
Mass spectrum was recorded on a Shimadzu instrument by direct inlet method at 70 eV. All chemicals and solvents used were of analytical grade. Fractionation of the active hexane extract was carried out by column chromatography over silica gel (Acmes’ 100–200 mesh ; Acme Synthetic Chemicals, Mumbai, India). Elution was carried out with hexane, benzene, chloroform, and their mixtures. The fraction, each 100 mL, was collected and combined based on their thin-layer chromatography (TLC) profiles. The active fraction was located and the active principle was isolated and identified by physical and spectroscopic data (infrared (IR), 1H NMR, 13C NMR, and electron ionization mass spectrometry (EI-MS)).
Extraction and isolation of the active principle
A volume of 8 kg of the shade-dried and coarsely powdered stem bark of the plant was extracted successively with n-hexane, chloroform, and methanol in a Soxhlet apparatus (S.N. Scientific Company, Tiruchirappalli, Tamil Nadu, India). The extracts were filtered through Whatman No. 1 filter paper and concentrated in a rotary evaporator and finally dried in a vacuum desiccator. The n-hexane extract which showed activity was taken up for further studies.
Cell culture reagents and antibodies
RPMI-1640 cell culture medium, fetal bovine serum (FBS), phosphate-buffered saline (PBS), and trypsin-ethylenediaminetetraacetic acid (EDTA) were purchased from Gibco and Invitrogen (ThermoFisher Scientific, Waltham, Massachusetts, USA). Acridine orange (AO), ethidium bromide (EtBr), Hoechst 33258, JC-1 (5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimi-dazolylcarbocyanine iodide), 2,7-diacetyl dichlorofluorescein diacetate (DCFH-DA), N-acetyl-L-cysteine (NAC), and other chemicals were supplied by Sigma-Aldrich (MO, USA). Mouse- or rabbit-monoclonal antibodies against human Bax, Bcl-2, p53, cyclin D1, cytochrome c (cyt-c), p21, caspase-3 and -9, cleaved-poly (ADP-ribose) polymerase (PARP), proliferating cell nuclear antigen (PCNA), and β-actin were procured from Santa Cruz Biotechnology (TX, USA). The secondary antibody and alkaline phosphatase (ALP)-conjugated anti-mouse and anti-rabbit immunoglobulin (IgG) were obtained from GeNei (Bengaluru, India). Small-molecular inhibitor SB202190 (p38MAPK inhibitor) was purchased from Cayman Chemical (MI, USA).
Cell culture
The hepatocellular carcinoma cell line HepG2 was obtained from the National Centre for Cell Science (NCCS, Pune, India). The cells were cultured in T25 culture flasks containing RPMI 1640 supplemented with 10% (v/v) FBS and 2% antibiotics (100 U/mL penicillin and 100 µg/mL streptomycin) at 37°C and 5% CO2 in a CO2 incubator. Upon reaching 80%–90% confluency, the cells were trypsinized and passaged.
Cytotoxicity assay
Growth inhibitory effect of bauerenol was screened by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method. Briefly, cells were seeded onto 96-well plates at a density of 5000 cells/well in 200 µL of medium. After 24 h, the cells were incubated with different concentrations of bauerenol (0–100 µg/mL) for 24 and 48 h. After incubation period, medium containing bauerenol was removed and 100 µL of MTT solution (5 mg/mL in PBS) was added to each well. After 3–4 h incubation in dark, MTT was discarded and 100 µL of dimethyl sulfoxide (DMSO) was added to each well. Then, the absorbance was measured using ELISA (enzyme-linked immunosorbent assay) Microplate Reader (Bio-Rad Laboratories, CA, USA) at 570 nm. The percentage of growth inhibition was calculated as follows: growth inhibition = optical density (OD) of control − OD of test/OD of control × 100.
AO/EtBr staining
The morphological changes in control and treated cells were assessed by AO/EtBr staining. Briefly, HepG2 cells (106 cells/well) were seeded onto six-well plates and treated with the 50% growth inhibitory concentration (IC50) doses of bauerenol for 24 and 48 h. After treatment, the cells were harvested, washed once with PBS, and then stained with AO/EtBr for 5 min. Then, the morphological changes were observed under an Axioskop 2 Plus fluorescent microscope (Carl Zeiss, Jena, Germany).
Analysis of nuclear damage by Hoechst 33258 staining
Cells were seeded at a density of 106 cells/well onto six-well plates and treated with bauerenol (45 µg/mL and 25 µg/mL) for 24 and 48 h. After treatment, cells were harvested and washed with ice-cold PBS twice and then, stained with Hoechst 33258 (100 µg/mL) for 5 min at 37°C. Then, the nuclear morphological changes were visualized under the Axioskop 2 Plus fluorescent microscope.
Determination of mitochondrial membrane potential
The alteration in mitochondrial membrane potential (ΔΨm) was assessed using 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethyl benzimidazolyl carbocyanine iodide (JC-1 dye). It is commonly used to detect mitochondrial depolarization, an early indication of apoptosis. HepG2 cells were grown on six-well plates and treated with IC50 doses of bauerenol for 24 and 48 h. After treatment, the HepG2 cells were incubated with JC-1 fluorescent dye for 30 min at 37°C in dark. After washing twice with PBS, the ΔΨm was analyzed under the fluorescent microscope (Model CH40, Olympus, Tokyo, Japan).
Cell cycle analysis by flow cytometry
HepG2 cells were cultured in T25 flask and incubated with respective IC50 doses of bauerenol for 24 and 48 h. After incubation period, cells were harvested and fixed in 70% ice-cold ethanol. The next day, cells were washed with PBS, incubated with 1 µg/mL RNase (1 mg/mL) for 30 min before they were stained with propidium iodide (PI; Sigma-Aldrich). Finally, cell cycle distributions were analyzed by a BD FACSCalibur™ flow cytometer (BD BioSciences, NJ, USA) and the percentage of cells in each phase was determined.
Terminal deoxynucleotidyl transferase dUTP nick end labeling apoptosis assay
Bauerenol-induced apoptosis in HepG2 cells was detected by TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) in situ Apoptosis Detection Kit (GenScript, NJ, USA) in accordance with the manufacturer’s protocol. In brief, cells were treated similarly as described above in cell cycle analysis, fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and stained with 3,3′-diaminobenzidine (DAB) and observed under the light microscope.
Measurement of intracellular reactive oxygen species
HepG2 cells were cultured in six-well plates at a density of 106 cells/well and incubated with the IC50 doses of bauerenol for 24 and 48 h. After the treatment, cells were harvested and washed with PBS, incubated with 1 mL of 10 mM DCFH-DA in PBS at 0 and 30 min under dark. Then, the fluorescence intensity was measured at 480 nm excitation and 530 nm emissions at 0 and 30 min using multimode reader (BioTek, VT, USA) and the values were expressed as percentage of fluorescence and calculated as follows
where Ft0 is the fluorescence at t = 0 min and Ft30 is the fluorescence at t = 30 min.
N-acetyl cysteine and SB202190 treatments
To study the role of reactive oxygen species (ROS) generation, chemical inhibitors of ROS (n-acetyl cysteine) and p38MAP kinase (SB202190) were employed to substantiate the involvement of ROS generation and p38MAP kinase activation in bauerenol-induced cell death. In brief, HepG2 cells were pre-treated with 6.5 mM N-acetyl cysteine (NAC) for 1 h and then incubated with IC50 doses of bauerenol for 24 and 48 h. Then, the changes in growth inhibition, ΔΨm, intracellular ROS levels, and expression of apoptosis-related proteins were studied. To study the involvement of p38MAP kinase activation in bauerenol-induced cell death, HepG2 cells were pre-treated with 10 µM SB202190 for 1 h and then incubated with IC50 doses of bauerenol for 24 and 48 h and protein expressions were studied by Western Blotting analysis.
Western Blotting analysis
After the treatment period, cells were lysed with radioimmunoprecipitation assay (RIPA) buffer and protein concentration was determined by Bradford method. 8 After lysis, 50 µg of cell lysates were electrophoresed in 10%–12% SDS-PAGE (sodium dodecyl sulfate–polyacrylamide gel electrophoresis) and then transferred to polyvinylidene difluoride (PVDF) membrane. The membranes were incubated with primary antibodies against p53, Bax, Bcl-2, cyt-c, PCNA, cyclin D1, caspase-3 and -9, PARP, p21, and β-actin (Santa Cruz Biotechnology). After washing, the membranes were incubated with secondary antibodies (anti-mouse or anti-rabbit IgG conjugated with ALP was used for the detection of primary antibody binding). Protein bands were detected using 5-bromo-4-chloro-3-indolyl phosphate (BCIP)/nitro blue tetrazolium chloride (NBT). The density of blots was quantified by Quanti-one software (Bio-Rad Laboratories).
Statistical analysis
All the experiments were performed at least three times and the data were expressed as mean ± standard deviation 7D). Statistical significance was denoted by parametric one-way analysis of variance (ANOVA). *p < 0.05 shows significant difference between control and treatment groups.
Results
Isolation, purification, and identification
The crude hexane extract (88.0 g) was chromatographed over silica gel and elution with hexane:benzene (1:1) gave the active constituent (yield 8.2 g) which was identified as the triterpenoid bauerenol (Figure 1). It was obtained as a colorless solid (mp 208–209°), (α)p <20° (CHCl3). It was analyzed for C30H50O (m+, m/z 426) IR

Chemical structure of bauerenol.
Bauerenol induces time- and dose-dependent growth inhibition in HepG2 cells
The growth inhibitory potential of bauerenol was determined by MTT Assay. The results showed that bauerenol-treated HepG2 cells had a significant decrease in growth in time- and dose-dependent manner (Figure 2). The IC50 was found at 45 and 25 µg/mL after 24 and 48 h treatments, respectively. We therefore treated the cells with IC50 doses of bauerenol in the following experiments unless stated otherwise.

Cytotoxicity of bauerenol in HepG2 cancer cell line by MTT assay in different concentrations (0–100 µg/mL) for 24 and 48 h. Mean ± SD of three independent experiments (*p < 0.05 and **p < 0.01 indicate the significance compared to control).
Bauerenol-treated cells reflecting morphological features of apoptosis
The morphological changes in bauerenol-treated HepG2 cells were analyzed by AO/EtBr dual fluorescent staining. AO is a vital dye that stains both live and dead cells, whereas EtBr stains only those cells that have lost their membrane integrity. As shown in Figure 3, control cells show uniform green fluorescence indicating that the cells are viable, whereas bauerenol-treated cells show yellow, orange, and red fluorescence that represent early and late apoptotic features (cell membrane blebbing, chromatin condensation, and apoptotic bodies formation). These morphological alterations in bauerenol-treated HepG2 cells clearly indicate its apoptotic potential.

Morphological changes in HepG2 cancer cells, reflecting apoptosis after exposure to bauerenol for 24 and 48 h. (a) Cells were treated with IC50 values of bauerenol showing morphological changes under fluorescent microscope after staining with AO/EB (a: upper panel) and Hoechst 33258 (a: lower panel). Bauerenol-treated cells showing shrinkage of cells, fragmentation, condensed chromatin, and fragmented nuclei (indicated by arrows). (b and c) The ratio of apoptotic and nuclear morphological changes at 24 and 48 h treatment periods are indicated. All the results were obtained from Mean ± SD of three independent experiments (a significant difference between treatment and control cells indicated by *p < 0.05; scale bar 50 µm).
Nuclear morphological changes in HepG2 cells
To analyze the nuclear morphological changes of bauerenol-treated HepG2 cells, Hoechst 33258 staining was performed. It is a membrane-permeable blue fluorescent dye that stains nuclei in the cells. The results showed that the control cells had light blue nuclei uniformly under the fluorescent microscope that demonstrated that the cells are healthy. Interestingly, bauerenol-treated cells exhibited a bright blue fluorescence confirming the death of cells which are marked by arrows indicating condensed and fragmented nuclei (Figure 3).
ΔΨm loss in HepG2 cells
To determine bauerenol-induced cell death by the loss of ΔΨm, JC-1 staining was performed. The JC-1 is a mitochondria-specific probe that can be used as an indicator of ΔΨm. It is a voltage-sensitive fluorescent dye that detects normal polarized mitochondria as red and depolarized mitochondrial membranes as green under fluorescence. As shown in Figure 4, the loss of ΔΨm induced by bauerenol was noticeably increased in HepG2 cells after 24 and 48 h treatments.

Bauerenol-induced mitochondrial depolarization in HepG2 cancer cells. (a) Cells were incubated with IC50 values of bauerenol with or without NAC and stained with JC-1 showing mitochondrial depolarization pattern of cells under fluorescent microscope at 400x magnification. Changes in red to green fluorescence indicate the mitochondrial depolarization. (b) Percentage of mitochondrial membrane potential between control and treated cells. All the results were obtained from mean ± SD of three independent experiments (a significant difference between treatment and control cells indicated by *p < 0.05; scale bar 25 µm).
Bauerenol treatment increases the ratio of Bax/Bcl-2 in HepG2 cells
To investigate changes in the apoptosis-related protein expression, Bax and Bcl-2 proteins expressions were detected by Western Blotting. The Bcl-2 family proteins play a key role in mitochondria-dependent apoptosis. Exposure of HepG2 cells to bauerenol, after being normalized to actin (internal control), decreased the expression of Bcl-2 significantly, while Bax protein level was increased markedly in a time- and dose-dependent manner. The results implied that apoptosis induced by bauerenol might be due to the changes in the ratio of Bcl-2/Bax (Figure 5). Bauerenol-induced Bax upregulation was considerably inhibited by the ROS scavenger and p38 inhibitor. It indicates the key role played by ROS in bauerenol-induced HepG2 cell death.

Bauerenol-induced apoptosis through intrinsic or mitochondria-dependent pathway. (a) Cell lysates were prepared from control and bauerenol-treated cells and proteins were separated in 10%–12% SDS-PAGE. Western Blotting performed with specific primary antibodies against Bcl-2, Bax, cytochrome c, caspase-3 and pro-caspase-9, and PARP showing the upregulation of Bax, downregulation of Bcl-2, cytochrome c release, and caspase and PARP cleavage at 24 and 48 h treatment periods. (b) Mean ± SD of three independent experiments (statistically significant changes at *p < 0.05 vs the control).
Caspase-cascade activation, PARP cleavage, and apoptosis in HepG2 cells
The activation of caspase is one of the key events in mitochondria-dependent apoptosis. The proteolysis of caspase-3 and -9 (cleavage of pro-caspase into cleaved-caspase) was detected by Western Blotting. The results showed that bauerenol treatment markedly increased caspase-3 and -9 cleavages in a concentration-dependent manner at 24 and 48 h in HepG2 cells and more so caspase cleavage reached its maximum level at 48 h (Figure 5). Bauerenol treatment caused the cleavage of PARP, a substrate of caspase-3, and increased the cleavage of PARP fragments over time (Figure 5). Bauerenol-induced apoptosis in HepG2 cells was detected by TUNEL assay. Most nuclei were stained as visible brown in cells treated with bauerenol but in contrast no visible brown was observed in control cells. These results proved that most of the bauerenol-treated cells underwent apoptosis (Figure 6).

TUNEL assay analyzing bauerenol-induced apoptosis in HepG2 cells. (a) TUNEL assay in control and treated cells under light microscope. (b) Ratio of apoptotic cells. All the results were obtained from three independent experiments (a significant difference between treatment and control cells indicated by *p < 0.05; scale bar 50 µm).
ROS levels in HepG2 cells
ROS acts as a growth inhibitory and apoptotic signaling agent. Perturbation in the balance of ROS in cancer cells may offer an opportunity for chemoprevention. To find out the importance of ROS in bauerenol-induced cytotoxicity in HepG2 cells, intracellular ROS levels were measured using a ROS-sensitive fluorescent probe, DCFH-DA. The basal level of dichlorofluorescein (DCF)-sensitive ROS in HepG2 cells was not readily detectable. Following the treatment with bauerenol, a significant generation of ROS appeared at 24 h (Figure 7) when compared to control. The cells showed a sharp increase in intracellular ROS generation at 48-h treatment with bauerenol. Thus, it implies that bauerenol-induced ROS production has a certain relationship with mitochondrial dysfunction and apoptosis mechanism.

Bauerenol stimulated intracellular ROS levels in HepG2 cells. Cells were incubated with IC50 values of bauerenol followed by incubation with DCFH-DA for 30 min. Relative fluorescence of DCF measured by using fluorescence microplate reader. Mean ± SD of three independent experiments (*p < 0.05 vs treatment with control).
Bauerenol induces the activation of stress-related protein kinase
To elucidate the putative mechanism of bauerenol-induced apoptosis, the effects of bauerenol on MAPK pathway were examined by Western Blotting to assess the protein expression and phosphorylation of p38MAPK and ERK1/2. In HepG2 cells, bauerenol treatment significantly increased the phosphorylation level of p38MAPK while ERK1/2 phosphorylation level was significantly reduced after 24 and 48 h (Figure 8).

Bauerenol-induced phosphorylation of p38MAPK and dephosphorylation of ERK1/2 and Akt in HepG2 cells. (a) Cells were treated with IC50 values of bauerenol, and proteins were separated by SDS-PAGE. The levels of total ERK1/2, p38MAPK, Akt, and their phosphorylated forms were detected using specific antibodies. β-actin served as internal control. (b) Relative protein expression of ERK1/2, p38MAPK, and Akt. Mean ± SD of three independent experiments (*p < 0.05 vs treatment with control).
Bauerenol treatment downregulates the activation of Akt in HepG2 cells
To determine the effect of bauerenol on Akt signaling, the total protein expression and phosphorylation of Akt were detected by the Western Blotting in cells treated with bauerenol. Phosphorylation of Akt (p-Akt) significantly decreased in a time- and dose-dependent manner and no changes in the total Akt protein expression were detected. Therefore, the results indicate the involvement of Akt inactivation in bauerenol-induced growth inhibition and apoptosis (Figure 8).
Inhibitor treatment
Small molecule inhibitor NAC (n-acetyl cysteine) was used to substantiate the role of ROS in bauerenol-induced p38MAPK activation and apoptotic cell death. Bauerenol-induced growth inhibition was abrogated significantly by the co-treatment of NAC at 24 and 48 h (Figure 9(a)). Blocking of ROS generation by NAC prevented bauerenol-induced ΔΨm loss at 24 and 48 h (Figure 9(b)). Pre-treatment of HepG2 cells with NAC strongly suppressed the bauerenol-induced intracellular ROS accumulation at 24 and 48 h (Figure 9(c)). Bauerenol-induced p38MAPK phosphorylation and apoptosis was potentially prevented by p38 inhibitor and ROS inhibitor at 24 and 48 h, respectively (Figure 9(d)).
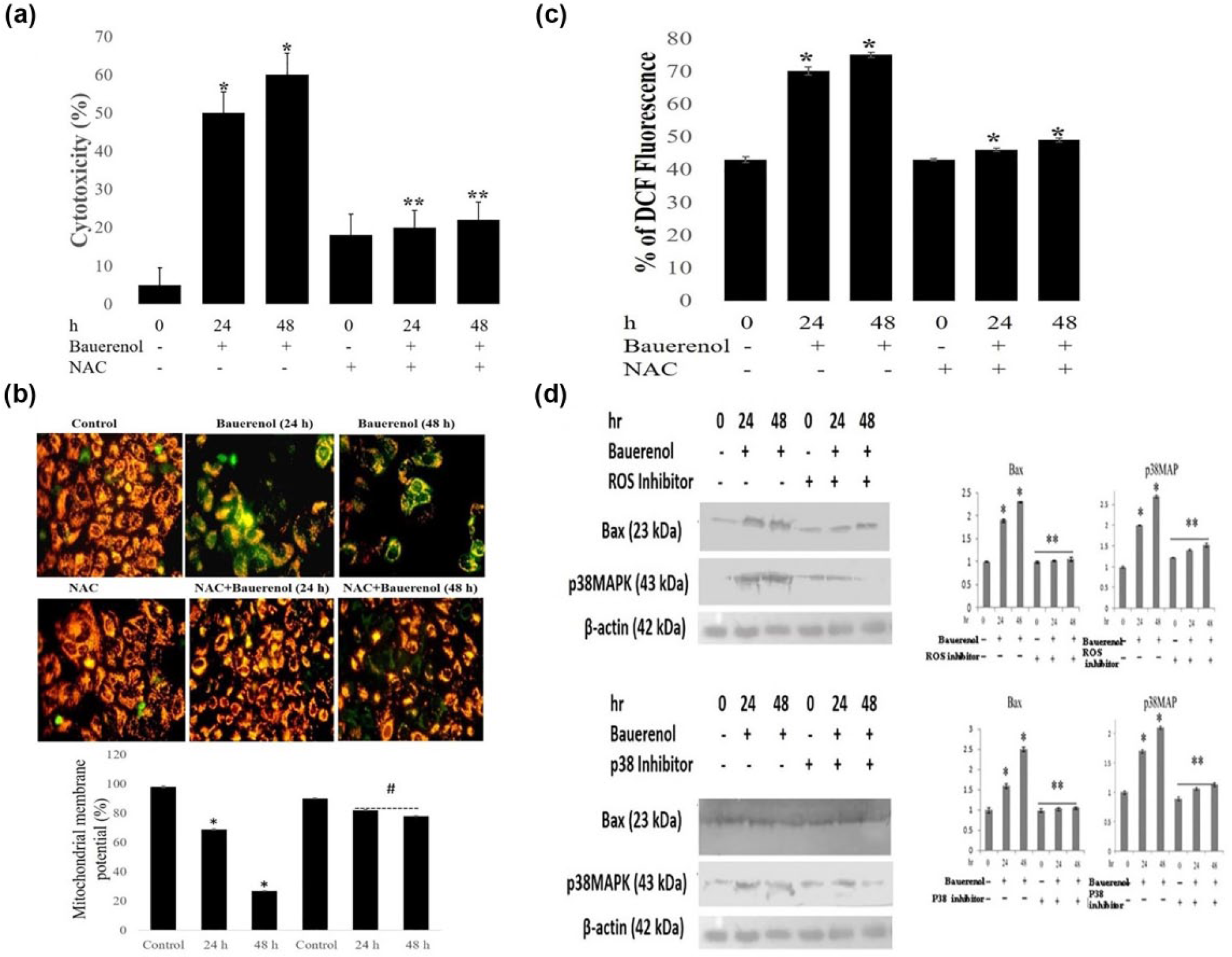
Bauerenol-induced apoptosis through ROS-dependent p38MAPK activation in HepG2 cells. (a) Impact of NAC pre-treatment on bauerenol-induced cytotoxic effect on HepG2 cells, analyzed by MTT assay. Results show cytotoxic effect of bauerenol was abolished by NAC pre-treatment at 24 and 48 h. (b) Cells were incubated with IC50 values of bauerenol with or without NAC and stained with JC-1 showing mitochondrial depolarization pattern of cells under fluorescent microscope at 400× magnification. Changes in red to green fluorescence indicate the mitochondrial depolarization. The graph shows the percentage of mitochondrial membrane potential between control and treated cells. (c) To analyze the ROS accumulation levels, cells were treated with bauerenol (with or without NAC) for 24 and 48 h and incubated with DCFH-DA for 30 min. Relative fluorescence of DCF measured using fluorescence microplate reader. (d) Cells were exposed with or without ROS and p38 inhibitors for 1 h and then treated with bauerenol for 24 and 48 h. The protein expression levels of p38 and Bax were analyzed by Western Blotting. Mean ± SD of three independent experiments (statistically significant changes at *p < 0.05 vs control).
Bauerenol induces S-phase arrest by modulating the expression of cell cycle regulatory proteins in HepG2 cells
To elucidate the mechanism of bauerenol-induced alterations in cell cycle progression and cell cycle distribution, flow cytometry analysis was performed and the results were recorded. Bauerenol treatment caused a significant inhibition of cell cycle progression in HepG2 cancer cells at 24 and 48 h, and alteration in the percentage of cells in S phase compared to control cells was observed (Figure 10). To estimate the changes by the expression of cell cycle regulatory proteins (cyclin D1, p21, p53, and PCNA) in the bauerenol-mediated cell cycle arrest, the Western Blotting analysis was performed. After incubation with bauerenol, p21 and p53 protein levels were significantly increased in a dose-dependent manner. Simultaneously, a dose-dependent downregulation of cyclin D1 and PCNA was also observed (Figure 11).

Bauerenol-induced cell cycle perturbation in HepG2 cells. (a) Cells were exposed to bauerenol for 24 and 48 h. The cells were stained with propidium iodide (PI) and analyzed by the flow cytometry. (b) The ratio of cells at G0/G1, S, and G2–M phase in the control and bauerenol-treated cells. Data are represented as mean ± SD of three independent experiments (*p < 0.05 vs treatment with control).

Bauerenol altered the expression of cell cycle regulatory proteins in HepG2 cells. (a) Cells were treated with bauerenol for 24 and 48 h. The protein expression of cyclin D1, p53, p21, and PCNA was detected by Western Blotting. Blots show downregulation of cyclin D1 and PCNA, whereas p21 and p53 expressions were upregulated in bauerenol-treated cells. (b) Mean ± SD of three independent experiments (statistically significant changes at *p < 0.05 vs the control).
Discussion
The bioactive compound eluted from hexane:benzene (1:1) answered for Noller’s test. 9 Mass spectrum showed molecular ion peak of m/z 426 having molecular formula C30H50O. The FT-IR spectrum showed band for the presence of hydroxyl (3369 cm−1), trisubstituted double bond (1628 cm−1), gem dimethyl (1383 cm−1), and C=O bonding of alcohol (1037 cm−1). 1H NMR spectrum showed eight methyl groups in the region of 1.10 to 1.14 ppm and nine methylene and five methine protons in the region of 1.29 to 2.13 ppm. The multiplet appeared at 3.10 to 3.24 ppm suggests the presence of proton adjacent to oxygen. Furthermore, vinylic proton appeared at 5.50 ppm strongly suggests that the compound having trisubstituted double bond. The structure of bauerenol also corresponds to 1H NMR shifts. 10 13 C NMR spectrum showed 30 carbon signals as expected for a triterpenoid. 13C NMR spectrum showed one secondary hydroxyl group, a trisubstituted double bond, two secondary methyls, and six tertiary methyls, and the remaining peaks being methylene groups. The olefinic carbon signals appear at 147 and 124 ppm and carbon nearer to hydroxyl group at 79.2 ppm. The 13C NMR values were assignable to bauerenol. 11 Finally, the structure was confirmed by mass spectral fragmentation. 12 The above data is comparable with those of bauerenol reported in literature.13–15 Bauerenol was reported from S. bifaria. 16 This is the first report of bauerenol from S. angustifolia.
Medicinal plants research in the family of Euphorbiaceae has revealed chemopreventive as well as anti-cancer efficacy in pre-clinical animal models.17,18 Alcoholic extract of Euphorbia hirta, E. lunulata, and Phyllanthus urinaria showed cytotoxicity against HepG2 cells.19,20 Jolkinolide B (50 µg/mL), a diterpene, derived from E. fischeriana, exhibited greater cytotoxicity potential against HepG2 cell line. 21 It is presumed that bauerenol might have cytotoxicity against cancer cells. Hence, it was investigated and the results are presented and interpreted here. As expected, bauerenol exerted significant cytotoxic potential against HepG2 cells with an IC50 value of 45 and 25 µg/mL at 24 and 48 h, respectively (Figure 2).
Targeting programmed cell death (apoptosis) is a promising candidate for anti-cancer drug discovery. Apoptosis is a cellular suicide or a programmed cell death which is mediated by the activation of an evolutionary conserved intracellular pathway. The relationship between apoptosis and cancer showed that the process of neoplastic transformation, progression, and metastasis involves alteration of normal apoptotic pathway. 22 In our study, bauerenol-treated cells showed significant morphological changes. AO, a membrane-permeable fluorescent dye, is specific for apoptotic cell death and does not significantly stain necrotic cells. It is used to visualize the number of cells which have undergone apoptosis but it cannot distinguish viable from non-viable cells. To achieve this, a mixture of AO and EtBr stain was used to visualize cells with aberrant chromatin organization and differential uptake property as the dye finally allows the identification of viable and non-viable cells. 23 Exposure to the bauerenol against HepG2 cells showed early and late apoptotic morphological changes that indicate the anti-tumor activity of bauerenol. In this study, bauerenol-treated cells exhibited condensed and fragmented nuclei, cell membrane blebbing, vacuolation, and apoptotic bodies. When stained with a Hoechst 33258 nuclear fluorochrome, the chromatin of the bauerenol-treated HepG2 cells showed lumps by condensation that exhibits the punctuated morphology of typical apoptotic cells. The ability of bauerenol in the inhibition of cancer cells proliferation and induction of apoptosis purports that it is a potential anti-cancer agent against liver carcinoma (Figure 3).
Mitochondria play an important role in the propagation of apoptosis and are responsible for 90% of energy needed for cell functioning. The disruption of mitochondrial integrity is one of the early events leading to apoptosis.24,25 In this study, JC-1 staining of bauerenol-treated HepG2 cells showed the loss of ΔΨm (Figure 4). Furthermore, mitochondrial intermembrane space (IMS) proteins (e.g. Cyt-c) upon cellular fractionation proved the disruption of ΔΨm. It is proved that bauerenol induced the dysfunction of mitochondrial membrane that has contributed to the apoptosis in HepG2 cells.
Mitochondria-mediated apoptosis includes downstream of ROS production and upstream of caspase activation as regulated by Bcl-2 family proteins. The Bcl-2 family proteins consist of an anti-apoptotic protein (Bcl-2) as well as a number of pro-apoptotic molecules (Bax), whereas overexpression of the anti-apoptotic protein Bcl-2 blocks mitochondrial outer membrane permeabilization and inhibits apoptosis.26,27 An agent that could enhance the expression of pro-apoptotic Bax protein and/or inhibit the expression of anti-apoptotic Bcl-2 protein might induce apoptosis in cancer cells. Targeting Bcl-2 family proteins and tumor suppressor proteins is crucial in molecular target that ultimately leads to anti-cancer drug discovery. To analyze the mechanism underlying the bauerenol-induced apoptosis, we tested the expression of Bcl-2 and Bax. Our data showed that the expression of anti-apoptotic Bcl-2 was decreased significantly while death promoting pro-apoptotic Bax protein level was increased significantly in a dose-dependent manner (Figure 5). Genetic mutations of p53 gene are the most common alteration detected in HCC. 28 The p53 protein functions as a transcriptional activator, associated with cell metabolism, cell survival, and apoptosis. 29 In particular, Bax protein is a transcriptional target of p53 that facilitates apoptosis upon its activation. In this study, the expression levels of p53 were increased following bauerenol treatment. The results are consistent with Bax upregulation in bauerenol-treated HepG2 cells and thus indicate that the apoptosis of HepG2 cells after bauerenol treatment is p53 dependent.
Intracellular ROS production is the by-product of normal cellular oxidative processes, and it is suggested to be involved in the regulation of apoptotic signaling. Increased levels of ROS have demonstrated induction of mitochondrial membrane depolarization that eventually produces an increase in the level of other cell pro-apoptotic molecules.30,31 In this study, the production of intracellular ROS was significantly increased at 24 and 48 h of bauerenol treatments. It clearly demonstrated the accumulation of intracellular ROS in bauerenol-treated cells. Bauerenol-induced apoptosis was initiated by the generation of ROS, followed by the disruption of ΔΨm, and cyt-c was released into the cytosol, altogether that led to the activation of caspase-3 and -9 (Figures 5–7).
The MAPK pathways are critical for converting diverse extracellular signals, including ROS, to apoptotic responses for the treatment of chemotherapeutic agents. Of these, MAPK subfamilies are considered as important mediators of apoptotic signals in many biological systems. In MAPK signal transduction, p38 and ERK1/2 play an important role in apoptosis/pro-survival. The p38MAPK is more commonly activated in response to stress and cellular damage, whereas ERK1/2 is activated through mitogenic stimuli. The p38 signaling pathway is an important regulator of stress responses, and there is also an evidence that p38 can function as tumor suppressor by inducing cell cycle arrest, differentiation, and apoptosis. 32 Hence, the balance between ERK and p38 phosphorylation is responsible for cells’ fate, that is, whether the cells undergo differentiation, proliferation, or apoptosis. 33 Therefore, we studied the ERK1/2 and p38MAPK to elucidate the signal transduction leading to apoptosis in bauerenol-treated HepG2 cells. Our results demonstrated that bauerenol treatment enhanced p38 activation (phosphorylation) in HepG2 cells and the apoptotic effect of bauerenol in association with stress-induced p38MAPK activation (Figure 8).
Anti-apoptotic protein kinase (AKT), a serine/threonine-protein kinase also known as protein kinase B, is involved in the regulation of various signaling pathways of cell metabolism, cell proliferation, survival, growth, and angiogenesis. The mechanism by which Akt protects cells from death is likely to be multifactorial because Akt directly phosphorylates several components of the cell death machinery. It is a member of the most frequently activated proliferation and survival pathway in cancer. 34 This study also indicates the apoptotic effect of bauerenol in association with phosphorylation of p38MAPK and Akt dephosphorylation. These data indicate the involvement of MAPK signaling in bauerenol-induced apoptosis.
NAC is a potent anti-oxidant that prevents chemotherapeutic agent induced ROS accumulation. 35 In this study, it is revealed that NAC significantly reversed bauerenol-induced apoptosis in HepG2 cells and confirmed the role of ROS in bauerenol-induced apoptosis. The cells pre-treated with NAC and incubated with bauerenol at 24 and 48 h showed activation of p38MAPK which was effectively inhibited by NAC and clearly revealed the role of ROS in bauerenol-induced p38MAPK activation (Figure 9). Furthermore, the cells pre-treated with SB202190 (a p38MAPK inhibitor) at 1 h and incubated with bauerenol at 24 and 48 h showed that SB202190 effectively diminished bauerenol-increased Bax expression and activated p38MAPK in bauerenol-induced apoptosis (Figure 9).
Cell cycle is an essential process in the development, differentiation, and proliferation of mammalian cells. 36 Cell cycle progression is regulated by the activity of cyclins, a family of proteins that activates the cell cycle–dependent kinases (cyclin-dependent kinase (CDKs)).
Altogether, overexpressions of cyclins lead to either tumorogenesis or cell cycle arrest. 37 Since deregulation of the cell cycle machinery has been associated with cancer initiation and progression, suppression of the cell cycle has become an appreciated target for management and treatment of tumor cells with cytotoxic agent.38,39 In this study, the data clearly showed the significant increase of cells in S phase, decrease in G0/G1 and G2/M phase, and perturb of the cell cycle in bauerenol-treated HepG2 cells (Figure 10).
PCNA is an essential auxiliary protein centrally involved in the regulation and coordination of cell cycle progression and DNA replication. 40 In this study, bauerenol suppressed the PCNA expression in HepG2 cells. The regulation of p21 is largely dependent on the presence of functional p53, a transcriptional regulator that mediated cell cycle arrest following DNA damage. 41 Mammalian cell cycle is known to be controlled by anti-mitogenic signals where it involves p21 that binds and inhibits CDKs required for the initiation of S phase. The protein p21 also binds to the DNA polymerase δ processivity factor, PCNA and inhibits PCNA-dependent DNA replication. 42 The suppression of PCNA expression with the simultaneous p21 upregulation and cell cycle arrest in this study explains inhibitory action of bauerenol by the execution of cell cycle arrest through p21 induction (Figure 11). It is reported that terpenoids of plant origin were found to induce S-phase arrest. 43 In this study, it is very clear that bauerenol induces S-phase arrest in HepG2 cells at 24 and 48 h post-treatment and there was no evidence of apoptotic population at that time. Hence, it is revealed that bauerenol induces cell cycle arrest primarily through p53 overexpression and later apoptosis follows.
Conclusion
Bauerenol possesses cytotoxic and apoptotic potential against human HepG2 cancer cells. Furthermore, the cytotoxic effect of bauerenol substantiated using human primary lymphocytes and did not show significant cell death at each IC50 dose used in this study. The apoptosis induced by bauerenol is associated with ROS elevation, mitochondrial dysfunction, cyt-c release, increase of Bax/Bcl-2 ratio, caspase activation, and PARP cleavage. As there has been no report on the apoptotic property of bauerenol, an active component of S. angustifolia, this study clearly addresses the mechanism present behind the growth inhibition of cancer cells. However, further in-depth studies to explain the molecular mechanisms on the cytotoxic effect of bauerenol are to be carried out for using it as a potential therapeutic agent in future.
Footnotes
Acknowledgements
This research was awarded second prize in the International Conference on Translational Medicine and Imaging (ICTMI 2016) held on 4 March 2016 and 5 March 2016 at Rajiv Gandhi Centre for Biotechnology, Tiruvananthapuram, India, organized by Rajiv Gandhi Centre for Biotechnology, Tiruvananthapuram and Mar Ephraem College of Engineering and Technology, Marthandam.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

