Abstract
Cancer immunotherapy using cytotoxic T cells demonstrates dramatic survival benefits in lymphomas, but its efficacy in solid tumors is limited. Here, we investigated the possibility of using cytotoxic T cells to treat malignant Schwannoma, a rare but aggressive nerve sheath tumor, by examining the native T-cell immunity in the host. We found that compared to CD8+ T cells from healthy controls or benign Schwannoma patients, the CD8+ T cells from malignant Schwannoma patients were present at normal frequencies but were substantially enriched with PD-1−TIM-3+ and PD-1+TIM-3+ cells. Compared to the PD-1−TIM-3− CD8+ T cells, the PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells presented significantly lower proliferation capacity, reduced interleukin 2 and interferon gamma expression, and/or dramatically decreased perforin and granzyme B secretion, indicating a whole-spectrum immunosuppression and reduced cytotoxicity. TIM-3 expression alone was associated with lower proliferation and less perforin and granzyme B secretion, whereas PD-1 expression alone was not associated with functional impairments, suggesting that TIM-3 expression was a better marker of exhausted CD8+ T cells. The expression of galectin 9, a TIM-3 ligand, in CD4+ Th cells was significantly elevated in malignant, but not benign, Schwannoma patients and were enriched in CD25+ Treg cells. Both the PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells responded to Treg-mediated and galectin 9–mediated suppression, whereas the PD-1+TIM-3− CD8+ T cells only responded to Treg-mediated suppression. In resected tumors, the malignant Schwannomas had more tumor-infiltrating CD4+ and CD8+ T cells than the benign Schwannomas, but a large fraction of these tumor-infiltrating CD4+ and CD8+ T cells expressed PD-1 and/or TIM-3, which indicated that their antitumor immunity was compromised. Together, our results suggested that PD-1 and TIM-3 blockade might be necessary in developing effective immunotherapeutic strategies in malignant Schwannoma, in which TIM-3 may play a more important role.
Introduction
Malignant Schwannoma is a rare but aggressive nerve sheath tumor, representing an estimated 0.5%–5% of all Schwannomas. 1 It is thought that malignant tumors can arise as a primary tumor generated de novo. 2 In other cases, benign primary Schwannoma can undergo malignant transformation, even after complete surgical resection.3,4 The underlying mechanism for the development of malignant Schwannoma is not completely understood. Treatment options are limited to conventional chemotherapy, radiotherapy, and surgical resections.5,6 Currently, the 5-year survival rate is between 39% and 85%.1,7 Further research is needed to develop better treatment strategies.
Robust and effective T-cell-mediated immunity is critical in the prevention of tumor formation and further development. 8 The level of CD4+ and/or CD8+ T-cell infiltration in the tumor is directly correlated with the prognosis of many cancers.9–12 The cytotoxic effects mediated by CD8+ T cells, and in some cases, CD4+ T cells, are considered the primary mechanism in tumor elimination and rejection.8,13–15 Immunotherapies utilizing tumor antigen–specific CD8+ T cells, such as chimeric antigen receptor–modified CD8+ T cells, demonstrate efficacy in treating mobile phase tumors. 16 In many malignant solid tumors, however, multiple mechanisms exist to suppress the normal antitumor response of the immune system.17,18 The anti-inflammatory antigen presentation by tumor-associated macrophages (TAMs), myeloid-derived suppressor cells (MDSCs), and regulatory dendritic cells (DCs), for example, preferentially primes regulatory T (Tregs) cells over tumor-reactive effector T cells through the secretion of anti-inflammatory interleukin (IL) 10 instead of proinflammatory IL-12.19,20 The Tregs can further downregulate inflammation by expressing CD25, which is an IL-2 receptor that functions to deplete local IL-2, and by releasing TGF-β, which is a inhibitory cytokine that suppresses CD8+ T-cell cytotoxicity.21–24 In addition, the expression of PD-L1 by tumor cells can directly mediate T-cell exhaustion and apoptosis by interacting with the PD-1 receptor expressed on the surface of activated T cells.25–27 Together, these mechanisms potently inhibit the antitumor immunity of T cells and prevent future developments of T-cell-based immunotherapies.
Since immunotherapy represents one promising new strategies to treat malignant Schwannoma, more research is needed to examine the current status of antitumor T-cell immunity in malignant Schwannoma patients. In this study, we found that although the peripheral blood from benign and malignant Schwannoma patients contained similar frequencies of CD4+ and CD8+ T cells, the CD8+ T cells from malignant patients expressed significantly elevated frequencies of PD-1 and/or TIM-3. These PD-1/TIM-3-expressing CD8+ T cells had reduced capacity to proliferate, to express IL-2 and interferon gamma (IFN-γ), and to mediate cytotoxicity through granzyme B and perforin. The TIM-3 ligand galectin 9 (Gal-9) expression was dysregulated in malignant, but not benign, Schwannoma patients. In malignant tumors, the percentage of PD-1/TIM-3-expressing CD4+ and CD8+ T cells were further increased compared to those in blood. Together, our results presented another challenge to the development of immunotherapies in malignant Schwannoma.
Materials and methods
Study subjects
The benign and malignant Schwannomas were diagnosed at the Third Affiliated Hospital of Hebei Medical University, with histological confirmations using previously established references.3,28 Briefly, benign tumors presented minimal cellular and nuclear pleomorphism, without evidence of mitosis or necrosis, whereas malignant tumors presented prominent pleomorphic cells and nuclei, with scattered giant cell forms, as well as mitotic and necrotic cells. Age- and sex-matched healthy controls were also recruited. Peripheral blood collection by venipuncture and tumor collection by resection were conducted according to the Declarations of Helsinki, under the approval of the Third Affiliated Hospital of Hebei Medical University. All participants provided written informed consent.
Cell culture
Peripheral blood mononuclear cells (PBMCs) were collected by standard Ficoll–Hypaque centrifugation. Tumors were dissociated by cutting and enzymatic digestion with collagenase and DNase (Sigma). Tumor mononuclear cells were collected by Ficoll–Hypaque centrifugation. Cells were grown under 37°C and 5% CO2, in RPMI 1640 supplemented with
Surface and intracellular staining
To measure proliferation, 1 µL CellTrace carboxyfluorescein succinimidyl ester (CFSE; Invitrogen) was added to 106 cells, which were incubated at 37°C for 10 min, washed five times in cold medium, rested on ice for another 10 min, washed twice in warm medium, and then placed in warm medium for further experiments. For T-cell receptor (TCR) stimulation, cells were treated with antibody OKT3 and antibody CD28.2 at 3 µg per 106 cells each. For trapping of intracellular cytokines, brefeldin A and monensin (BD Pharmingen) were added 5 h before harvest. Fluorescently labeled anti-human CD3, CD4, CD8, PD-1, and TIM-3 (BioLegend) were added to cells at 5 µg per 106 cells for 25 min at 4°C. Excess surface antibody was removed by two washings with 2% FBS–phosphate-buffered saline (PBS). For intracellular staining, cells were first fixed and permeabilized with CytoFix/CytoPerm (BD Pharmingen) and then treated with fluorescently labeled anti-human IL-2 and IFN-γ (BioLegend) at 5 µg per 106 cells for 25 min at 4°C. Excess surface antibody was removed by two washings with 1× Perm Wash (BD Pharmingen).
Fluorescence-activated cell sorting
Acquisition and examination of surface and intracellular molecule expressions were performed in BD LSR II cytometer. Sorting of CD4+ and CD8+ T cells according to their PD-1 and TIM-3 expressions was performed in BD FACSAria. Data analyses were performed using FlowJo software (Tree Star).
ELISA
The LEGEND MAX Human Granzyme B ELISA Kit (detection limit: 9.4–600 pg/mL) from BioLegend, the Perforin (PRF1) Human ELISA Kit (detection limit: 62.5–2000 pg/mL) from Abcam, and the Human Galectin-9 Quantikine ELISA Kit (sensitivity: 0.028 ng/mL) from R&D Systems were used according to manufacturer’s instructions. In some experiments, 1 µg/mL recombinant human Gal-9 protein (Abcam) was added to the cell culture.
Chromium-51 release assay
B cells were immortalized by Epstein–Barr virus (EBV) using a published protocol. 29 The EBV-transformed B cells were labeled with 100 µCi 51Cr (Sodium Chromate, Perkin Elmer) for 1 h at 37°C, pulsed with 3 µg/mL superantigen Staphylococcal enterotoxin B (SEB; Sigma Aldrich) for 1 h, and then cocultured with autologous sorted CD8+ T-cell subsets at indicated effector to target (E/T) ratios. Chromium-51 (51Cr) release was measured after 5-h incubation. No T-cell control and Triton-X conditions were included as minimum and maximum releases, respectively, for the calculation of specific lysis.
Statistics
Data were represented as mean ± SD (standard deviation) where applicable. Analyses were performed in Prism software (GraphPad). D’Agostino–Pearson test was first applied to determine data normality. Appropriate parametric or nonparametric tests were then chosen. Value of p < 0.05 was considered significant.
Results
Peripheral blood CD4+ and CD8+ T cells from malignant Schwannoma subjects had elevated the expression of exhaustion markers
Age- and sex-matched healthy controls (N = 10), patients with benign Schwannoma (N = 10), and patients with malignant Schwannoma (N = 6) were recruited for the study. We first compared the peripheral blood CD3+ T cells between these three groups. The peripheral blood frequencies of CD4+ T cells and CD8+ T cells and the CD4-to-CD8 ratio were not significantly different among the three groups (Figure 1). In chronic viral infection as well as tumor, the expression of PD-1 and TIM-3 is used to denote exhausted T cells.30–32 Using those two markers, we found that compared to those in controls, CD4+ T cells from malignant Schwannoma subjects did not present significantly different PD-1 or TIM-3 expression, whereas CD8+ T cells from malignant Schwannoma subjects contained significantly higher TIM-3 single-positive (PD-1−TIM-3+) and PD-1/TIM-3 double-positive (PD-1+TIM-3+) cells (Figure 2). The CD4+ and CD8+ T cells from benign subjects did not display significantly different PD-1 and TIM-3 expression compared to healthy controls (Figure 2). Together, these results demonstrated that although malignant Schwannoma subjects had similar peripheral blood frequencies of CD4+ and CD8+ T cells, their CD8+ T cells tended to express elevated exhaustion markers such as PD-1 and TIM-3.

Analyses of peripheral blood CD4+ and CD8+ T cells in Schwannoma patients. (a) Representative step-wise (left to right) gating strategy of circulating CD4+ and CD8+ T cells in one malignant Schwannoma patient. (b) The frequencies of circulating CD4+ T cells and CD8+ T cells and the CD4-to-CD8 ratio in healthy control subjects, benign Schwannoma patients, and malignant Schwannoma patients. One-way ANOVA followed by Tukey’s multiple comparisons test.
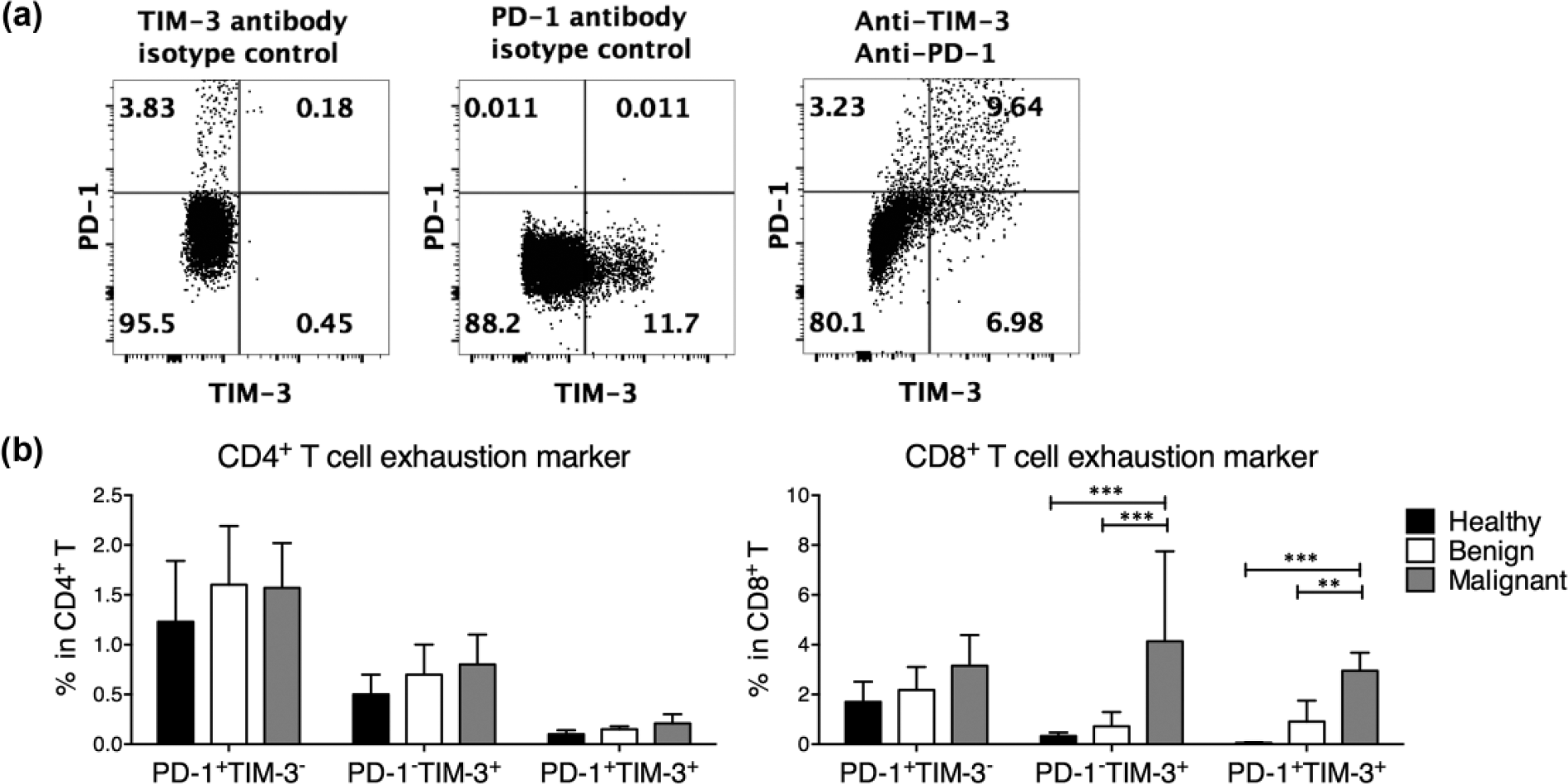
PD-1 and TIM-3 expression in circulating CD4+ and CD8+ T cells from Schwannoma patients. (a) Representative gating strategy of PD-1 and TIM-3 on circulating CD4+ and CD8+ T cells in one malignant Schwannoma patient. The boundaries of PD-1+ and TIM-3+ cells were determined using isotype control antibodies. (b) The frequencies of PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ cells in circulating CD4+ and CD8+ T cells from healthy control subjects, benign Schwannoma patients, and malignant Schwannoma patients. Two-way ANOVA followed by Tukey’s multiple comparisons test.
PD-1−TIM-3+, and PD-1+TIM-3+ CD8+ T cells presented significantly reduced proliferation and/or cytokine production
To evaluate the extent of CD8+ T-cell exhaustion in malignant Schwannoma patients, we analyzed the proliferation and cytokine production of CD8+ T cells, fractionated based on PD-1 and TIM-3 expressions. The CFSE-labeled PBMCs from all subjects were TCR-stimulated for 5 days. The proliferation and cytokine secretion were analyzed by flow cytometry. We found that PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells, but not PD-1+TIM-3− CD8+ T cells, presented significantly reduced proliferation compared to PD-1/TIM-3 double-negative PD-1−TIM-3− CD8+ T cells (Figure 3). IL-2, a cytokine that mediates CD8+ T-cell proliferation, was present in significantly lower frequencies of PD-1+TIM-3+ CD8+ T cells (Figure 3). IFN-γ, an effector T-cell cytokine, was also lower in PD-1+TIM-3+ CD8+ T cells (Figure 3). These results demonstrated that PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells presented inhibited effector functions.

Proliferation and cytokine production by PD-1−TIM-3−, PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ cells in circulating CD8+ T cells in malignant Schwannoma subjects. (a) Representative gating strategy of CFSE-low (proliferating) cells, IL-2-expressing cells, and IFN-γ-expressing cells in circulating CD8+ T cells. (b) Summary of CFSE-low (proliferating) cells, IL-2-expressing cells, and IFN-γ-expressing cells in PD-1−TIM-3−, PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ circulating CD8+ T cells from malignant subjects. One-way ANOVA followed by Tukey’s multiple comparisons test. Asterisks (*) denote the statistical difference between the labeled population and the PD-1−TIM-3− population.
PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells presented reduced cytotoxicity
CD8+ T cells can mediate cytolytic activity through the secretion of perforin and granzyme B, which penetrates the membrane of the target cell and mediates the apoptosis signaling cascade, respectively. Therefore, we sorted CD8+ T cells according to the surface PD-1 and TIM-3 expression, which were then incubated separately at 0.5–2 × 105 per 200 µL for 12 h in vitro, in the presence of TCR stimulation. The normalized granzyme B and perforin release in the supernatant was measured by enzyme-linked immunosorbent assay (ELISA). We found that both the PD-1−TIM-3+ and the PD-1+TIM-3+ CD8+ T cells were significantly suppressed in their ability to secrete granzyme B and perforin, compared to the PD-1−TIM-3− CD8+ T cells, which suggested a reduced capacity to eliminate target cells (Figure 4(a)). To examine the cytotoxicity of the CD8+ T-cell subsets, we performed a 51Cr release assay using autologous EBV-immortalized B cells as target cells. PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells presented significantly impaired cytoxicity compared to PD-1−TIM-3− CD8+ T cells at both 10/1 and 50/1 E/T ratios (Figure 4(b)). Although PD-1+TIM-3− CD8+ T cells also presented lower cytotoxicity than PD-1−TIM-3− CD8+ T cells at 50/1 E/T ratio, the extent was less pronounced.

Cytolytic molecule release by circulating PD-1−TIM-3−, PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ CD8+ T cells in malignant Schwannoma subjects. (a) The release of granzyme B and perforin was measured by ELISA of 12-h supernatant from pure subpopulations of circulating CD8+ T cells. Data were normalized for 2 × 105 cells in 200 µL media. (b) The cytotoxicity of pure subpopulations of circulating CD8+ T cells was examined using 51Cr-labeled EBV-transformed autologous B cells as targets, at 10/1 and 50/1 effector to target (E/T) ratios. One-way ANOVA followed by Tukey’s multiple comparisons test. Asterisks (*) denote the statistical difference between the labeled population and the PD-1−TIM-3− population.
PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells were more susceptible to Gal-9-mediated suppression than PD-1+TIM-3− CD8+ T cells
Previous investigations demonstrated that TIM-3-expressing CD8+ T cells in Schwannoma patients demonstrated enhanced suppression. We investigated the potential mechanisms that mediated the inhibition of TIM-3-expressing CD8+ T cells. TIM-3 has two known ligands: Gal-9 and phosphotidylserine. Gal-9 is expressed on epithelial cells, CD4+ T helper (Th) cells, and neutrophils and was shown to regulate Th responses.33,34 Here, we investigated whether Gal-9 expression in Schwannoma patients was dysregulated and whether it contributed to TIM-3-mediated CD8+ T-cell suppression. We found that CD4+ Th cells from malignant, but not benign, Schwannoma patients presented significantly elevated Gal-9 expression (Figure 5(a)). In malignant patients, the Gal-9 expression was primarily concentrated in the CD25+ Treg cells (Figure 5(b)). In addition, we stimulated PD-1+TIM-3−, PD-1−TIM-3+, or PD-1+TIM-3+ circulating CD8+ T cells in media only, with additional autologous CD25+ Treg cells or with exogenous Gal-9. PD-1+TIM-3− CD8+ T cells responded to Treg-mediated suppression but did not respond to exogenous Gal-9. The PD-1−TIM-3+ or PD-1+TIM-3+ CD8+ T cells responded to both Treg-mediated suppression and exogenous Gal-9 by downregulating IFN-γ secretion (Figure 5(c)). Together, these results demonstrated that the Gal-9 expression by Th cells in malignant Schwannoma patients was elevated, rendering PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells, but not PD-1+TIM-3− CD8+ T cells, susceptible to Gal-9-mediated suppression.
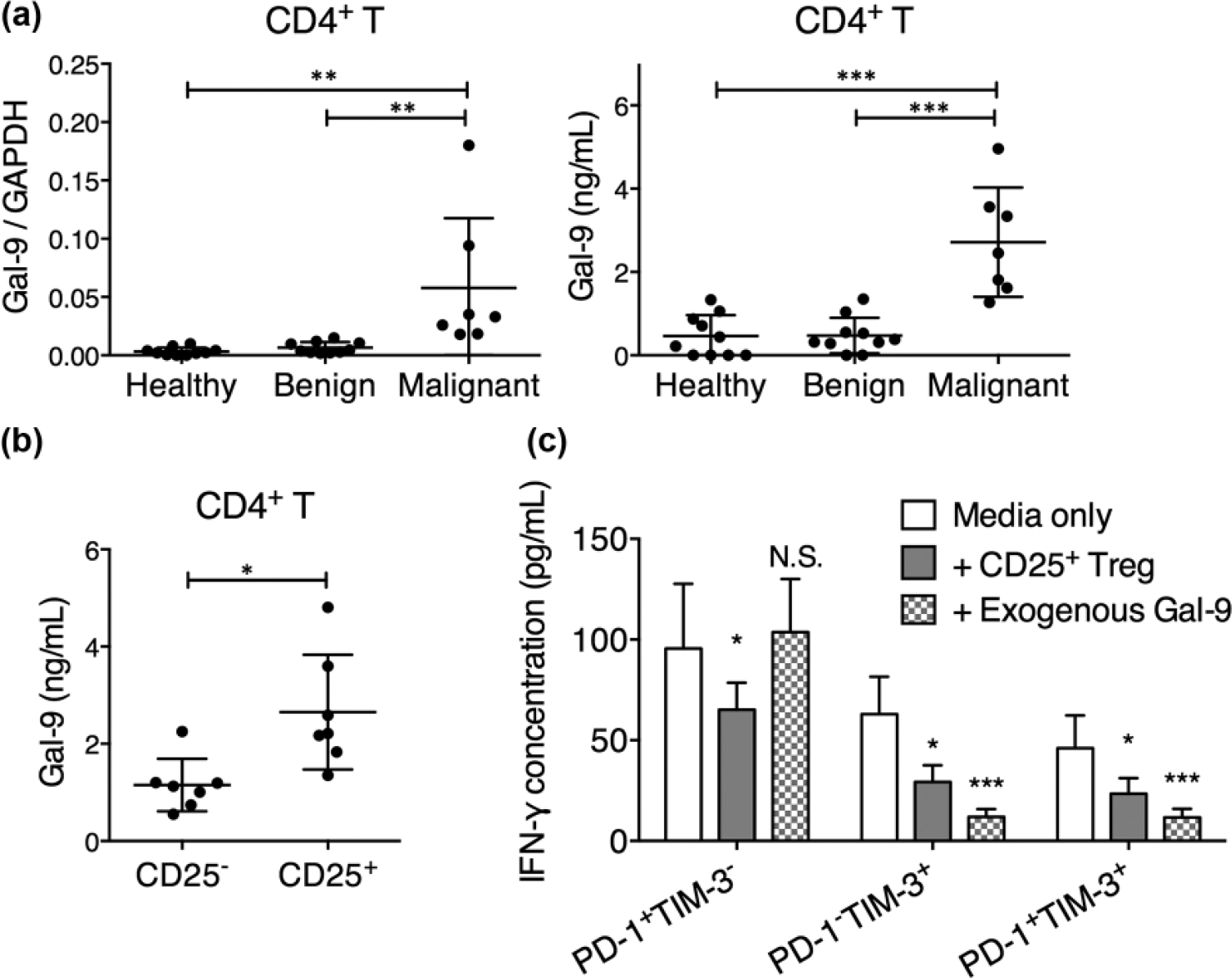
Susceptibility of PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ CD8+ T cells to Gal-9-mediated suppression. (a) The expression of Gal-9 by CD4+ T cells from healthy controls, benign Schwannoma patients, or malignant Schwannoma patients after 12-h TCR stimulation. Both mRNA expression (relative to GAPDH) and protein secretion were examined. (b) The expression of Gal-9 by sorted CD4+CD25− T cells or CD4+CD25+ T cells after 12-h TCR stimulation. The protein secretion in the supernatant was examined. One-way ANOVA followed by Tukey’s multiple comparisons test. *p < 0.05; **p < 0.01; ***p < 0.001. (c) The PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ CD8+ T cells were cocultured with irradiated feeder cells, in media only, with autologous CD25+ Tregs at 1: 1 ratio, or with 1 µg/mL Gal-9 for 12 h with anti-CD3/CD28 antibodies. The IFN-γ secretion was then examined in the supernatant. Two-way ANOVA followed by Dunnett’s multiple comparisons test. Asterisks (*) denote the statistical difference between the labeled data and the media-only data. *p < 0.05; **p < 0.01; ***p < 0.001.
PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T cells were present at high frequencies in the malignant Schwannoma tumors
Although previous results demonstrated a clear suppression of PD-1−TIM-3+ and PD-1+TIM-3+ CD8+ T-cell functions, the peripheral blood CD8+ T cells overall did not appear to be severely exhausted since the PD-1−TIM-3+ and the PD-1+TIM-3+ CD8+ T cells only made up a small percentage of all CD8+ T cells. We then examined whether this is the case in the tumor. Benign tumors contained few CD4+ or CD8+ T-cell infiltrations, while malignant tumors contained significantly elevated numbers of CD4+ and CD8+ T cells (Figure 6(a)). However, a significant proportion of the CD4+ and CD8+ T cells in the tumor expressed exhaustion markers. In our cohort, up to 53.7% of CD4+ T cells and up to 35.6% CD8+ of T cells were PD-1+TIM-3+, compared to less than 5% in the peripheral blood (Figure 6(b)).

Tumor-infiltrating CD4+ or CD8+ T-cell frequency and composition in benign or malignant Schwannoma tumors. (a) The number of CD4+ and CD8+ T cells per 2 g of freshly digested benign and malignant tumor samples. Mann–Whitney test; ***p < 0.001. (b) The frequency of PD-1+TIM-3−, PD-1−TIM-3+, and PD-1+TIM-3+ cells in tumor-infiltrating CD4+ and CD8+ T cells in benign and malignant Schwannomas. One-way ANOVA followed by Tukey’s multiple comparisons test.
Discussion
In recent years, multiple immunotherapy strategies have been developed as novel treatment options to cancer. Given that most tumors present weakly immunogenic self-molecules, the possibility to eliminate tumor cells using tumor-reactive cytotoxic T cells has been explored in multiple tumors. T cells that are engineered to express tumor antigens have demonstrated clinical benefits in the treatment of multiple cancers. 35 Most notably, genetically modified T cells that express chimeric receptors, which are fusions of the high-affinity CD19-specific immunoglobulin ectodomain and the CD3 intracellular signaling complex as well as the CD28 and 41BB molecules, have showed dramatic effects in B-cell lymphomas.36,37 Similar strategies have also been explored in solid tumors, but the results are not as promising as in mobile phase tumors, partly due to immunosuppressive mechanisms mediated by high PD-L1 expression in tumor cells. 26 In this study, we observed that the CD8+ T cells in malignant Schwannoma patients expressed significantly higher levels of PD-1 and TIM-3, a feature not observed in benign Schwannoma patients. The dual expression of both PD-1 and TIM-3 was associated with whole-spectrum functional impairment, as PD-1+TIM-3+ CD8+ T cells presented significantly low proliferation capacity, low IL-2 and IFN-γ expression, and low perforin and granzyme B secretion than PD-1−TIM-3− CD8+ T cells. TIM-3 expression alone also conferred low proliferation capacity, low perforin and granzyme B secretion, and reduced cytotoxicity. Furthermore, we discovered that malignant Schwannoma patients presented elevated Gal-9 expression by CD4+ Th cells, especially CD25+ Treg cells, which could exert suppression on TIM-3-expressing CD8+ T cells. Therefore, although malignant Schwannomas have significantly higher CD4+ and CD8+ T-cell infiltration than benign Schwannomas, it is unlikely that the tumor-infiltrating CD8+ T cells could exert effective antitumor immunity.
Our study indicated that TIM-3 presented a better marker for T-cell exhaustion than PD-1 since TIM-3 alone was associated with lower proliferation and cytolytic molecule secretion in PD-1−TIM-3+ CD8+ T cells, while PD-1 expression alone, on the contrary, was not associated with functional impairments. This observation is consistent with a previous report showing that TIM-3 expression on top of PD-1 expression, but not PD-1 expression by itself, indicated exhausted tumor antigen–specific CD8+ T cells in melanoma patients. 38 The upregulation mechanism of TIM-3 in malignant Schwannoma patients remains unclear. It has been reported that constitutive stimulation by common γ-chain cytokines, such as IL-2, IL-7, IL-15, IL-12, and IL-21, could induce TIM-3 expression on T cells in an antigen-independent manner.39,40 Whether these cytokines play a similar role in the upregulation of TIM-3 in malignant Schwannoma patients requires future studies. Interestingly, a recent report demonstrated that the blockade of PD-1 using nivolumab in the tumor-infiltrating T cells of human head and neck squamous cell carcinoma patients resulted in an upregulation of TIM-3 expression on these T cells. 41 Similarly, blockade of PD-1 in a murine tumor model resulted in significant TIM-3 upregulation. These results suggested that compensatory TIM-3 upregulation could be induced after PD-1 blockade. Indeed, blocking the PD-1/PD-L1 axis has yielded improved efficacy in a subset of, but not all, patients with renal cell carcinoma, melanoma, and non–small cell lung cancer,42–44 whereas in other cancers, the results are less promising. The upregulation of TIM-3 could provide another cellular target through which the tumor cells could suppress T-cell immunity. Together, these results demonstrated dual PD-1 and TIM-3 blockade is probably required for the suppression of tumors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
