Abstract
This study was to evaluate the prognosis of peritonectomy following gastrectomy for gastric adenocarcinoma patients with intraoperatively proven single P1/P2 carcinomatosis and to define the best therapeutic strategy of the patient cohort. The patients with intraoperatively proven single P1/P2 carcinomatosis from a prospectively maintained database were divided into resection group and non-resection group based on complete gross resection of peritoneal carcinomatosis. From 2005 to 2012, there were 103 patients in the resection group and 122 patients in the non-resection group. There was no difference in morbidity and mortality between groups. The patients did not have improved median survival in P1 carcinomatosis compared to P2 carcinomatosis (15.53 vs 14.80 months,
Introduction
Gastric cancer is currently the third leading cause of cancer-related death worldwide. 1 The overall survival of patients with locally advanced gastric cancer has continually improved due to the implementation of standard D2 lymphadenectomy and the advancement of chemotherapy and targeted treatments.2–4 However, the prognosis of advanced gastric cancer remains poor and most patients have advanced stage diseases at the time of diagnosis in China. 5
Peritoneal dissemination is the most frequent pattern of metastasis and recurrence in patients with gastric cancer.6,7 After the cancer invades the serosa, the exfoliated cancer cells can be scattered into the peritoneal cavity. 8 It has been reported that 44% of patients with serosa invasion have free cancer cells in the peritoneal cavity. 9 In addition, some patients with gastric cancer present with peritoneal carcinomatosis. Peritoneal metastasis is not usually observed before laparotomy because of the poor sensitivity and specificity of imaging techniques. Previous studies reported 10%–20% of patients with intraoperatively proven peritoneal carcinomatosis were not diagnosed preoperatively.10,11 Thus, effective strategies that prevent or delay peritoneal dissemination are critical for gastric cancer.
The patients with peritoneal dissemination are not considered to be curable. Chemotherapy is standard of care for these patients. Although hyperthermic intraperitoneal chemotherapy (HIPEC) could eradicate microscopic residual diseases with 20 times higher concentrations of regional chemical drug combined with hyperthermia 12 and then improve outcomes of gastric cancer patients with peritoneal dissemination,13,14 the locoregional effects of HIPEC are limited with a penetration to superficial 3–6 mm into malignant nodules and an increased antimitotic effect. 15 Surgical treatment is generally palliative, and gastrectomy is considered to well relieve serious complications, such as bleeding, obstruction, and perforation,16,17 or reduces the tumor burden to extend survival of asymptomatic patients.18–20 Achieving a complete surgical resection of metastatic tumors is an aggressive treatment method with curative intent.11,21 However, the effectiveness of peritonectomy for peritoneal carcinomatosis is unclear. For gross peritoneal carcinomatosis, peritonectomy could remove macroscopic tumor burden, and several clinical studies reported the addition of peritonectomy to gastrectomy might prolong overall survival without increasing the mortality rate of gastric cancer patients with peritoneal carcinomatosis.11,21–24 However, these studies were retrospective and had small sample populations. There is also inherent selection bias that confounds the results. For example, patients with good acceptable Eastern Cooperative Oncology Group (ECOG) performance status and small tumor burden were more likely to undergo peritonectomy, which increases the positive outcomes.
The extent of peritoneal dissemination influences the surgical resection of metastatic tumors. Patients with P3 carcinomatosis have multiple obvious peritoneal disseminations to the distant peritoneum, and complete gross peritonectomy with standard gastrectomy is not considered complete removal by surgeons. However, patients with P1/P2 carcinomatosis have minimal and localized peritoneal metastasis. Therefore, these patients are able to undergo complete gross peritonectomy with standard gastrectomy without residual disease. 25 As a result, this study was restricted to patients with P1/P2 carcinomatosis.
There is currently no convincing evidence demonstrating the survival benefits of peritonectomy for gastric cancer patients with intraoperatively proven peritoneal carcinomatosis. This study was to evaluate the prognosis of peritonectomy following gastrectomy for gastric adenocarcinoma patients with intraoperatively proven single P1/P2 carcinomatosis and to define the therapeutic strategy of this cohort.
Methods
Patients
The medical data of all diagnosed gastric cancer patients with intraoperatively proven single P1/P2 carcinomatosis who had undergone gastrectomy were identified from a prospective database and retrospectively analyzed from February 2005 to December 2012. The preoperative diagnosis of gastric adenocarcinoma was confirmed using histological evidence. The diagnosis of peritoneal carcinomatosis was based on the operative findings. All patients were diagnosed with adenocarcinoma and were not treated with neoadjuvant chemotherapy. The patients diagnosed with other gastric malignances such as lymphoma and gastrointestinal stromal tumor were excluded from the study. In addition, patients diagnosed with other multisite distant metastases such as concurrent hepatic metastasis, lung metastasis, and concurrent extra-regional lymph node metastasis, among others, were also excluded. Patients with tumor histological types that were non-adenocarcinoma such as squamous carcinoma and neuroendocrine carcinoma were excluded. Furthermore, remnant stomach cancers were also excluded from the analysis. All patients presenting with residual disease from a primary tumor after surgical resection were also excluded to avoid the influence of residual disease on prognosis; the residual disease of the primary tumor made the role of additional peritonectomy in the treatment of peritoneal carcinomatosis unclear. There were 225 patients enrolled in the study. The following patients’ clinicopathologic and treatment-related characteristics were compared between the two groups: gender, age, performance status, The American Society of Anesthesiologists (ASA) score, body mass index (BMI), tumor location, tumor size, depth of infiltration, lymph node metastasis, Borrmann type, tumor differentiation, peritoneal dissemination, postoperative chemotherapy, morbidity, mortality, and survival outcomes. This retrospective study was approved by the ethics committee of Harbin Medical University Cancer Hospital, China.
Treatments
All patients receive gastrectomy in our institution. The primary tumor and greater omentum were removed and then total or subtotal gastrectomy was performed according to the location of the primary tumor. Either Roux-en-Y or Longmire esophagojejunostomy with a mechanical stapler was performed to reconstruct the digestive tract for total gastrectomy. Billroth I or Billroth II anastomosis was used for the distal gastrectomy, and esophagogastric anastomosis was used after proximal gastrectomy. D2 or D3 lymphadenectomy is our standard nodal dissection method performed for all patients. The decision to proceed with peritoneal carcinomatosis resection was determined mainly by surgeons according to the feasibility of resection of peritoneal carcinomatosis and general health status of the patient. Within 8 weeks of surgery, a chemotherapy regimen of fluoropyrimidine such as S-1 or capecitabine plus platinum was given to the patients who received postoperative chemotherapy. In this study, five patients receiving complete gross resection of the peritoneal carcinomatosis intraoperatively and with proven microscopic residual disease in the postoperative pathology report were classified in the non-resection group. The clinicopathologic terminology was based on the Japanese classification of gastric carcinoma (third English version). 26
Peritoneal carcinomatosis
It is common knowledge that the extent of tumor burden influences the survival of gastric cancer patients with synchronous peritoneal carcinomatosis after gastrectomy.
We classified peritoneal metastasis according to the first edition of the General Rules for Gastric Cancer Study published by the Japanese Research Society for Gastric Cancer using the following criteria: P0, no peritoneal dissemination; P1, disseminating metastasis to the region directly adjacent to the peritoneum of stomach (above the transverse colon), including the greater omentum; P2, several scattered metastases to the distant peritoneum and ovarian metastasis alone; and P3, numerous metastases to the distant peritoneum. The peritoneal carcinomatosis patients with P1 or P2 disease based on the Japanese classification of gastric carcinoma (first English edition) definition were considered completely removed (resected) by surgeons. 25
Follow-up
Patients underwent follow-up every 3 months. The survival data were obtained from telephone calls or outpatient visits after completion of postoperative chemotherapy and/or surgery. The patient follow-up lasted until death or the cutoff date of 31 March 2015. The overall follow-up rate was 91.09% (225/247). There were 9 patients in the resection group and 13 patients in the non-resection group lost to follow-up. The median follow-up duration was 10.73 months in the resection group and 7.63 months in the non-resection group.
Statistical analyses
SPSS 19.0 software (SPSS, Chicago, IL) and GraphPad prism 5.0 were used for all statistical analyses. The variables of normality were tested and the data were expressed as the mean ± standard deviation or mean with a range where appropriate. The quantitative data were compared by using Student’s
Results
Patient characteristics
There were 103 patients in the resection group and 122 patients in the non-resection group. The peritoneal dissemination was more extensive and severe in the non-resection group. The following clinicopathologic and treatment-related characteristics were comparable between groups: gender, age, performance status, ASA score, BMI, comorbidities, tumor location, tumor size, depth of infiltration, lymph node metastasis, Borrmann type, tumor differentiation, peritoneal dissemination, and postoperative chemotherapy. The clinicopathologic and treatment-related characteristics of the patients are summarized in Table 1. There were 96 patients who underwent distal gastrectomy, 11 patients received proximal gastrectomy, and 118 patients received total gastrectomy. A total of 36 patients had combined multiorgan resections including the following: 9 transverse colon resections, 19 transverse mesocolon resections, 11 tail of pancreas resections, 5 splenectomies, 2 cholecystectomies, 12 oophorectomies, and 5 hepatectomies.
The clinicopathologic and treatment-related characteristics of the patients.

ASA: The American Society of Anesthesiologists; BMI: body mass index; ECOG: Eastern Cooperative Group Performance Status; W/D: well-differentiated adenocarcinoma; M/D: moderately differentiated adenocarcinoma; P/D: poorly differentiated adenocarcinoma; SRC: signet ring cell carcinoma.
Morbidity and mortality
The postoperative morbidity rate was 20.39% (21/103) in the resection group and 13.93% (19/122) in the non-resection group, respectively (
The incidence of morbidity and mortality.

Survival
The median survival was 21.07 (95% confidence interval (CI), 15.22–26.91) months in the resection group and 13.37 (95% CI, 11.85–14.88) months in the non-resection group (Figure 1). The survival difference was statistically significant (

Overall survival of resection group and non-resection group for gastric cancer patients with intraoperatively proven single P1/P2 carcinomatosis (
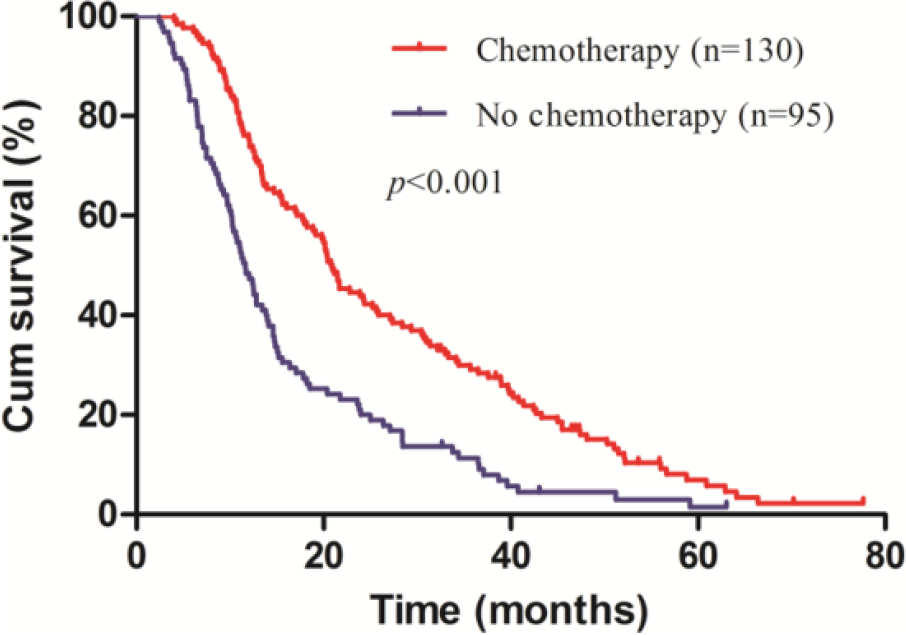
Overall survival of resection group and non-resection group for gastric cancer patients with intraoperatively proven single P1/P2 carcinomatosis stratified by postoperative chemotherapy (

Overall survival of resection group and non-resection group for gastric cancer patients with intraoperatively proven single P1/P2 carcinomatosis stratified by extent of peritoneal dissemination (
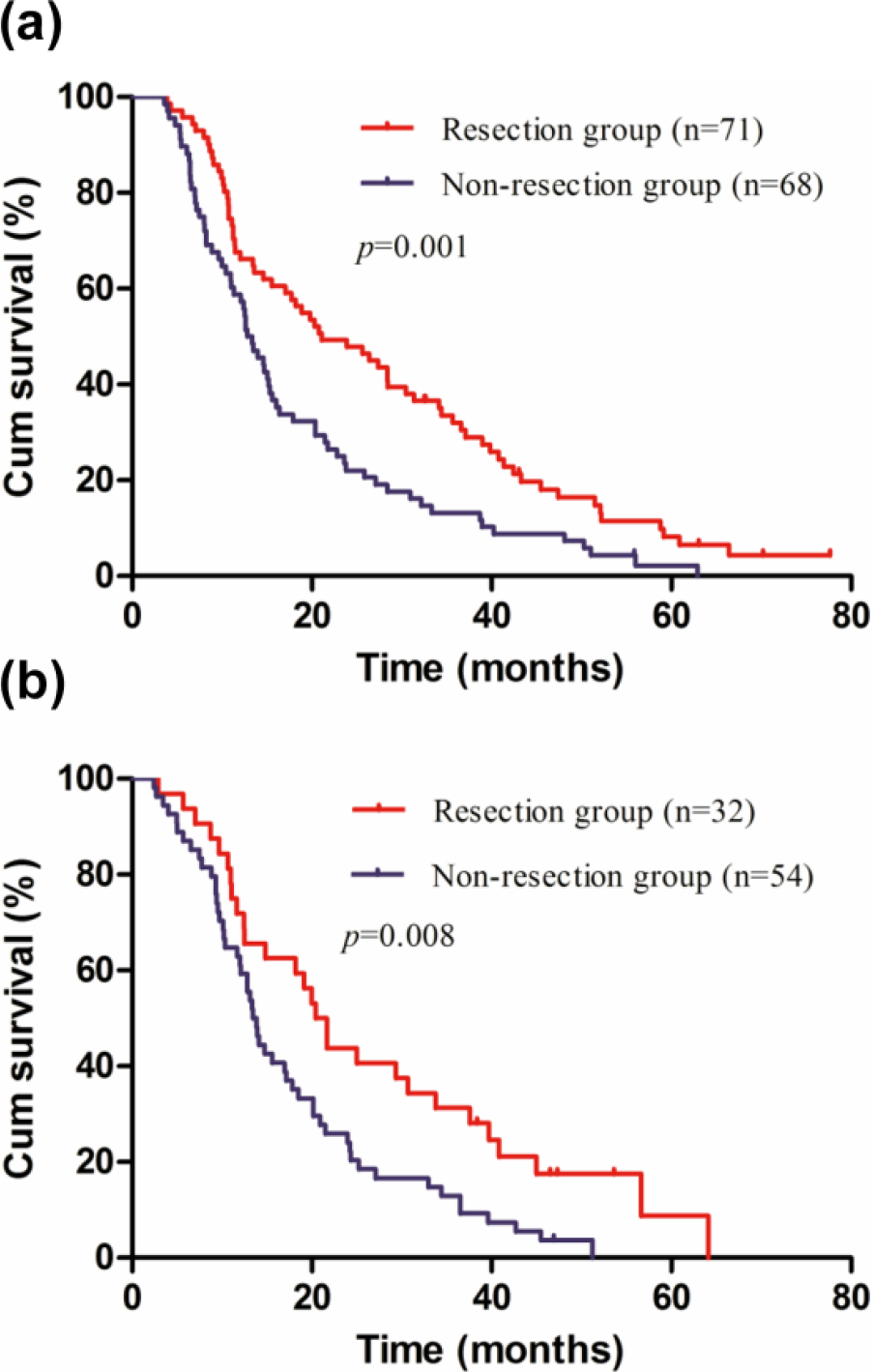
Overall survival of resection group and non-resection group for gastric cancer patients with intraoperatively proven single P1/P2 carcinomatosis stratified by extent of peritoneal dissemination. (a) P1 carcinomatosis (

Overall survival of gastric cancer patients with intraoperatively proven single P1/P2 carcinomatosis according to the treatment strategies received (
Multivariable analysis for overall survival
The univariate survival analysis revealed that gender, performance status, tumor size, Borrmann type, complete gross peritonectomy, and postoperative chemotherapy were associated with survival. The multivariate analysis identified poor performance status (
Univariate and multivariate analyses of prognostic factors for overall survival.
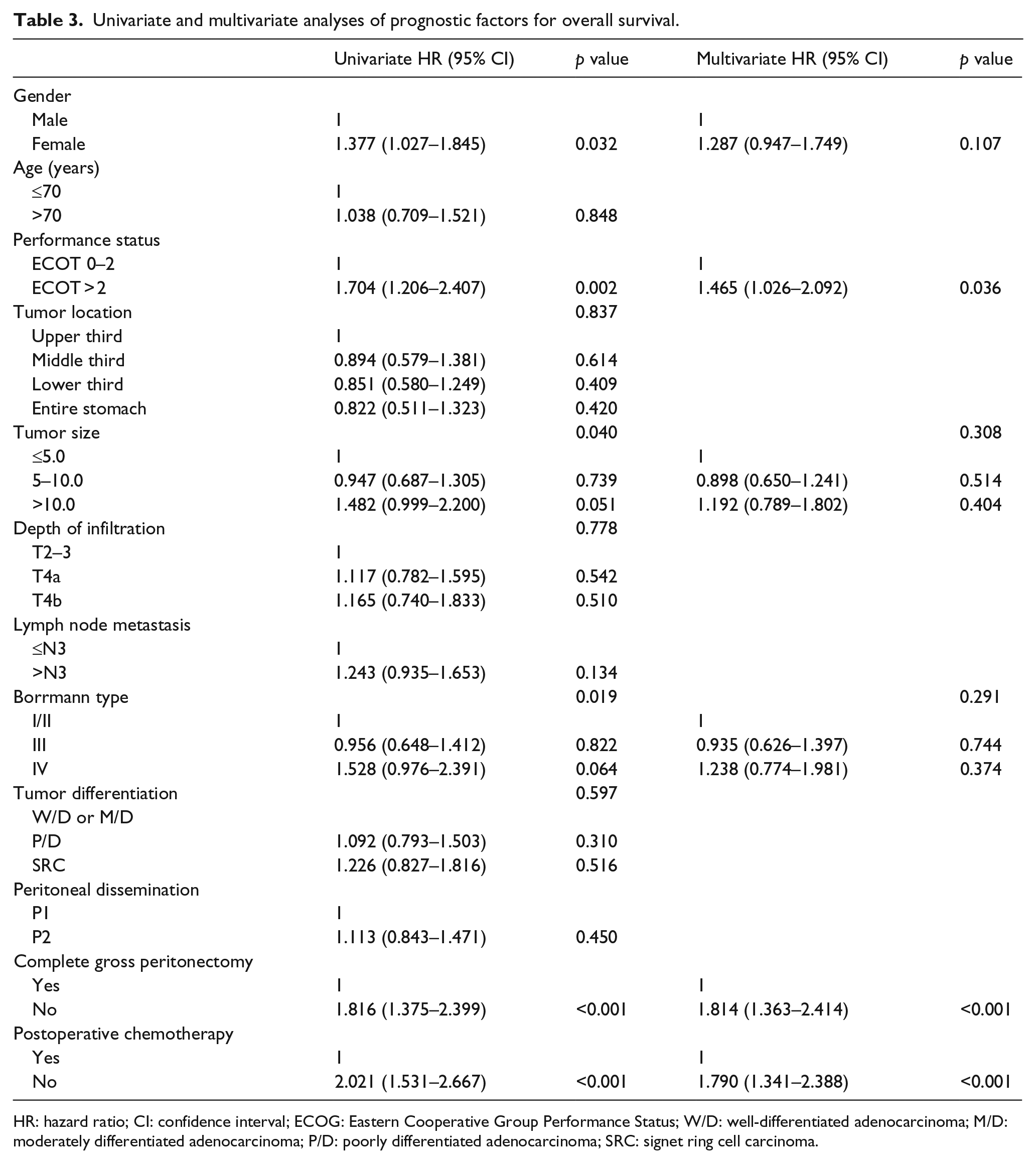
HR: hazard ratio; CI: confidence interval; ECOG: Eastern Cooperative Group Performance Status; W/D: well-differentiated adenocarcinoma; M/D: moderately differentiated adenocarcinoma; P/D: poorly differentiated adenocarcinoma; SRC: signet ring cell carcinoma.
Discussion
The presence of peritoneal dissemination is undetectable preoperatively by conventional radiology imaging. 28 Approximately 10%–20% of patients with gastric cancer investigated for potentially curative resection have peritoneal dissemination at the time of surgery,10,11 and 60% of the patients will develop peritoneal carcinomatosis after curative resection. 29 The prognosis of gastric cancer with peritoneal dissemination remains poor. Thus, the treatment of peritoneal dissemination is an important issue for gastric cancer worldwide.
The patients who develop peritoneal metastasis receive treatments such as systemic chemotherapy, HIPEC, and surgical resection to improve survival. Chemotherapy is currently the standard of care for these patients. HIPEC has a role in eradicating microscopic residual diseases in the peritoneal cavity after surgical resection. Surgical treatment is generally palliative, and gastrectomy is frequently considered to well relieve serious complications such as bleeding, obstruction, or perforation16,17 or reduces tumor burden to improve survival of asymptomatic patients18–20 (median overall survival of 7.78–14.0 months). Complete surgical resection of metastatic tumors as an aggressive treatment method is a way to aim for curative intent.11,21 However, it is unknown whether the addition of peritonectomy to gastrectomy could further improve patient prognosis.
The extent of peritoneal dissemination influences the surgical resection of metastatic tumors and the survival of gastric cancer patients. Patients with P3 carcinomatosis have extensive peritoneal dissemination and high tumor burden. Therefore, complete gross peritonectomy with standard gastrectomy is not considered complete removal by surgeons. Due to the presence of minimal and localized peritoneal metastasis, patients with P1/P2 carcinomatosis are eligible for complete gross peritonectomy with standard gastrectomy and do not have residual disease, and many patients will likely survive without recurrence. Previous study reported that P1/P2 carcinomatosis but not P3 carcinomatosis can be completely removed surgically.21,25 These findings suggest it is advisable for patients with P1/P2 carcinomatosis to be considered as candidates for peritonectomy plus gastrectomy.11,21,30 Complete gross peritonectomy reduces tumor burden and confers a survival benefit in patients with peritoneal carcinomatosis. The resection of the primary gastric tumors and peritoneal carcinomatosis reduces the large and potentially immunosuppressive tumor burden and removes the source of new metastases caused by the gastric tumor. These changes may facilitate durable chemotherapy responses. By contrast, peritonectomy plus gastrectomy might increase postoperative complications and delay the initiation of systemic chemotherapy. 31 Several studies of surgical resection for metastatic gastric cancer have demonstrated that the addition of peritonectomy plus gastrectomy improves patient survival for a small number of metastatic sites.11,21–24 These results suggest that reducing the tumor burden can lead to longer overall survival in patients with advanced gastric cancer. Our results showed that the median survival time was significantly longer in the resection group than in the non-resection group (20.07 vs 13.37 months). Therefore, peritonectomy reduces the tumor burden and confers a survival benefit in selected patients with P1/P2 carcinomatosis.
We also analyzed the usefulness of different treatment strategies received. We found that patients undergoing complete gross peritonectomy plus postoperative chemotherapy had a significantly longer median survival of 27.33 (95% CI, 18.36–36.31) months, compared with 12.00 (95% CI, 7.88–16.12) months for patients who had complete gross peritonectomy alone, 16.00 (95% CI, 10.49–21.51) months for patients receiving postoperative chemotherapy alone and 10.33 (95% CI, 6.59–14.08) months for patients who received neither peritonectomy nor postoperative chemotherapy. Some patients in the resection group had prolonged disease-free survival without any evidence of tumor recurrence which suggest a possible cure for some patients with distant metastasis. Although the value of metastasectomy is well established in colorectal cancer, it is still controversial surgery targeting metastatic lesions is beneficial for patients with peritoneal carcinomatosis in gastric cancer. Previous retrospective studies have shown positive results and median overall survival of 11.2–28.0 months.11,21 These analyses suggest there are survival benefits for aggressive surgical treatment in patients with peritoneal carcinomatosis. The results of our study are consistent with previous studies suggesting that gastrectomy plus peritonectomy is beneficial for patients with gastric cancer with peritoneal carcinomatosis. In addition, we found patients who underwent postoperative chemotherapy without peritonectomy had a better median survival than patients who underwent complete gross peritonectomy without postoperative chemotherapy. The univariate and multivariate analyses indicated that chemotherapy was associated with survival, and lack of chemotherapy was inversely related to survival. Our data suggest it is beneficial for patients to have reduction surgery, including primary tumor resection and peritonectomy. Chemotherapy is also recommended after reduction surgery.
Clinicians have expressed concerns regarding the morbidity and mortality of surgical resection. It is known that radical resection increases postoperative morbidity and prolongs the hospital stay in patients with distant metastasis. The overall postoperative morbidity rate was higher in the resection group than in the non-resection group. However, the difference was not statistically significant. This result is consistent with the data reported in previous studies.11,21 In this study, the postoperative morbidity rate in the resection group was not superior to the non-resection group. There were no significant differences in the postoperative hospital stay and mortality between the resection group and the non-resection group. Therefore, peritonectomy for gastric cancer patients with intraoperatively proven single peritoneal carcinomatosis should be considered safe and has an acceptable incidence of morbidity and mortality in experienced institutes.
There are some intrinsic limitations of this study. Our study may have a patient selection bias, because patients undergoing surgical resection with the intent of complete resection in the resection group had better performance status or a smaller tumor burden than patients in the non-resection group. This patient difference could influence the survival results. We tried to minimize these biases by adjusting for performance status, tumor burden, and other clinical parameters in the multivariate analysis. We also showed the consistent survival benefits of gastrectomy plus peritonectomy. However, we cannot help but admit that even this process has inherent biases associated with a retrospective study. Regardless, large-scale prospective studies are needed to explore the survival benefit and safety of gastrectomy plus peritonectomy for gastric cancer patients with intraoperatively proven peritoneal carcinomatosis.
Conclusion
The study data indicated complete gross peritonectomy following gastrectomy might be beneficial to the survival of gastric cancer patients with single P1/P2 carcinomatosis. In addition, postoperative chemotherapy improves survival regardless of resection of peritoneal carcinomatosis and should be recommended. Patients with good performance status and P1/P2 carcinomatosis should be considered appropriate surgical candidates.
Footnotes
Acknowledgements
S.L. edited the manuscript; X.L. conducted the statistical analyses; H.G. provided support for the registry website; W.W., L.C., and S.S. provided technical help and fruitful discussion.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Harbin Medical University Cancer Hospital.
Informed consent
Informed consent was obtained from all patients before they were included in the study.
