Abstract
Urothelial cancer patients are prone to recurrence, and there is no marker to predict which cases become refractory to the immunotherapy given to these patients. Tumour behaviour is decided by the dynamics between the pro- and anti-tumorigenic cytokines. In this study, 27 cytokines were estimated in serum and urine of 72 urothelial cancer patients and 42 healthy volunteer controls. Serum cytokines IL-1RA, IL-4 and RANTES were in significantly higher concentration in serum of patients compared to controls, while IL-2 was significantly less in concentration. Patients were found to have significantly high concentrations of 12 urinary cytokines (IL-2, IL-4, IL-8, IL-10, GM-CSF, IFN-γ, IP-10, MIP-1a, PDGF, MIP-1b, RANTES and VEGF) in comparison to healthy controls. Serum VEGF and urinary IL-1ra, IL-4, IL-10, GM-CSF, IP-10, MIP-1a and MIP-1b concentrations were found significantly higher in concentration in high-grade tumours compared to low-grade tumours. There was no difference in either the serum or urinary cytokines between non-invasive and muscle-invasive cases. Serum IL-1ra, IL-6, IL-10, TNF-α and VEGF and urinary IL-1ra, IL-4, IL-8, IL-10, GM-CSF, IP-10, MIP-1a, PDGF, MIP-1b and VEGF were found to be significantly higher in recurrent patients compared to non-recurrent patients. Of these, high concentrations of urinary IL-1RA, IL-4, IL-10, IP-10, PDGF and VEGF and serum IL-1ra, IL-6, IL-10, VEGF and TNF-α were associated with poor recurrence-free survival. Poor recurrence-free survival was also seen with increasing number of cytokines showing high concentrations. The study shows that the estimation of a combination of these cytokines in minimally or non-invasive samples may act as a prognostic indicator.
Introduction
Tumours incite both systemic and local immune response in cancer patients. The systemic response can be modulated by extraneous factors such as smoking, the presence of chronic disorders or metabolic diseases, occupation and genetic factors. It is assessed by the estimation of serum and can differ significantly in individual patients accounting for differing behaviour of the tumour. The systemic immune response is reported to be Th2 dominant in urinary bladder carcinoma patients, especially when recurrent.1,2 The study of immune responses is especially important in urinary bladder cancer because of the frequent recurrences in spite of intravesical therapy given in these patients. 3 Local immune response in most tumours manifests as infiltration of CD4+/CD8+ T-cells, T-regulatory cells, dendritic cells, natural killer (NK) cells and macrophages. 4 These immune cells release cytokines, chemokines and growth factors which can promote the development of a rich tumour stroma and growth of new blood vessels. In urinary bladder cancer, the cytokines which are released by the tumour milieu may get secreted in the urine or blood. As multiple cytokines act in concert to cause a multitude of effects, interact with each other and activate pro- as well as anti-tumorigenic actions, it is understandable that the end result is due to a combination of all their effects. Identification of these secreted cytokines in urine or serum and correlation with clinical features will help understand the role of immune factors in bladder cancer.
Hence, multiplex cytokine analysis may give a cytokine signature for prognostic assessment in patients. In this study, a panel of cytokines, chemokines and growth factors were estimated in non-invasive or minimally invasive samples such as blood and urine to identify the cytokine signature predictive of recurrence.
Materials and methods
The study included 72 urothelial bladder cancer (UC) patients, 52 non-muscle-invasive urothelial bladder cancer (NMIUC) and 20 muscle-invasive urothelial bladder cancer. Institutional ethical approval and patient consent were obtained before start of the study. Of the 52 NMIUC, 28 had recurrences during follow-up. In the non-muscle invasive group, 34/52 (65.4%) were low grade. High-grade cases showed significantly more number of recurrences and were more invasive (Table 1).
Patient characteristics.

NMIUC: non-muscle-invasive urothelial bladder cancer; MIUC: muscle-invasive urothelial bladder cancer.
Boldface signifies statistical significance
Both serum and urine were collected from the patients prior to surgery and cytokine concentration was estimated by using 27-plex multiplex kits (Bio-Rad). The kits included the cytokines (IFN-γ (interferon-γ), IL-1b (interleukin-1 beta), IL-1RA (interleukin-1 receptor antagonist), IL-12, TNF-α (tumor necrosis factor-α), IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-13, IL-15 and IL-17), chemokines (IP-10 (interferon gamma-induced protein 10), Eotaxin, MCP-1 (monocyte chemoattractant protein-1), MIP (macrophage inflammatory protein)-1a, MIP-1b and RANTES (regulated on activation, normal T expressed and secreted)) and growth factors (FGF (fibroblast growth factor) basic, PDGF (platelet-derived growth factor)-bb, VEGF (vascular endothelial growth factor), G-CSF (granulocyte–colony stimulating factor) and GM-CSF (granulocyte-macrophage colony-stimulating factor)), and experiments were performed as per manufacturer’s instructions. Briefly, 50 µL of sample per well was used to perform the assay. Serum samples were diluted with the assay diluents provided (1:4). Urine samples were used as is without the assay diluents. The assays were performed in duplicate. Phycoerythrin (PE)-conjugated secondary antibody was used for detection. All washing steps were done thrice with washing buffer using a vacuum manifold. Data were acquired as median fluorescence intensity and concentration in picogram/millilitre and analysis of experimental data was done using five-parametric curve fitting. Concentrations which were lower or higher than the lower and higher limit of detection (LOD), respectively, were defined as non-measurable.
Enzyme-linked immunosorbent assay (ELISA) was performed for concentration of urinary IP-10 and VEGF in 22 patients with cystitis, 20 healthy controls and 20 cancer patients using kits (IP-10/CXCL10 (Sigma Aldrich, St Louis, MO, USA) and VEGF (R&D Systems, Minneapolis, MN, USA)). The assays were performed in duplicate with 50 µL of urine sample added to each well. The protocol given in the manufacturers’ instructions was followed. The readings were taken in the ELISA plate reader at 450 nm. Cytokine concentrations were normalised to the creatinine concentration in urine and expressed as amount of cytokine in picograms (pg) excreted per milligram of creatinine.
Statistical analysis
As each cytokine shows wide concentration ranges in both groups, non-parametric Mann–Whitney U test was performed for significance testing. To assess the ability of each system to identify patients at highest risk of recurrence (i.e. the discriminatory power), we calculated the area under the receiver-operating characteristic curve (AUROC). A cut-off serum or urinary cytokine concentration having highest sensitivity and specificity was taken for further analysis and the cytokines coded as low or high as per the values below or above the cut-off. The patients were followed up for a median period of 20 months (interquartile range: 6–28 months), and Kaplan–Meier (KM) analysis was performed to evaluate the recurrence-free survival (RFS) between patients with low and high cytokine concentrations, and significance was computed by log rank. A probability less than 0.05 was considered significant. All statistical analyses were performed using the statistical package for the social sciences (SPSS) software version 16 (SPSS, Chicago, IL, USA) and KM survival analysis with log-rank test.
Results
Proinflammatory response in patients’ serum
The estimation of 27 serum cytokines revealed a proinflammatory response in bladder cancer patients compared to controls. The cytokines IL-1RA, IL-4 and RANTES were in significantly higher concentration in serum of patients compared to controls, while IL-2 was significantly more in controls (Figure 1). There was no significant difference in the concentration of other cytokines between patients and controls. The concentration of serum cytokines showed no difference between patients with muscle-invasive tumours compared to those with non-invasive tumours. The concentrations of serum IL-1RA, IL-6, IL-10, TNF-α and VEGF were found to be significantly higher in recurrent patients compared to non-recurrent patients. Only VEGF was found significantly higher in concentration in high-grade tumours compared to low-grade tumours (Figure 2).

Box plots show that high concentrations of serum IL-1RA, IL-4 and RANTES in patients while IL-2 was significantly lower in bladder cancer patients (p < 0.05 by Mann–Whitney U test).

Box plots showing the distribution of serum cytokines (a) IL-1RA, (b) IL-6, (c) IL-10, (d) TNF-α and (e) VEGF in recurrent and non-recurrent patients, all of which were in significantly higher concentrations in the recurrent patients and (f) VEGF was found to be significantly increased in high-grade cases compared to low-grade cases.
Urinary cytokines
Of the 27 cytokines evaluated in urine of 72 patients and 42 controls, significantly high concentrations of 12 cytokines (IL-2, IL-4, IL-8, IL-10, GM-CSF, IFN-γ, IP-10, MIP-1a, PDGF, MIP-1b, RANTES and VEGF) were found in urine of patients compared to healthy controls (Figure 3). High urinary IL-1RA, IL-4, IL-10, GM-CSF, IP-10, MIP-1a and MIP-1b concentrations were associated significantly with high grade, while high IL-6 concentration was associated with low grade (Figure 4). There was no difference in the urinary cytokines between non-invasive and muscle-invasive cases. High concentrations of urinary IL-1RA, IL-4, IL-8, IL-10, GM-CSF, IP-10, MIP-1a, PDGF, MIP-1b and VEGF were found associated significantly with recurrence (Figure 5).
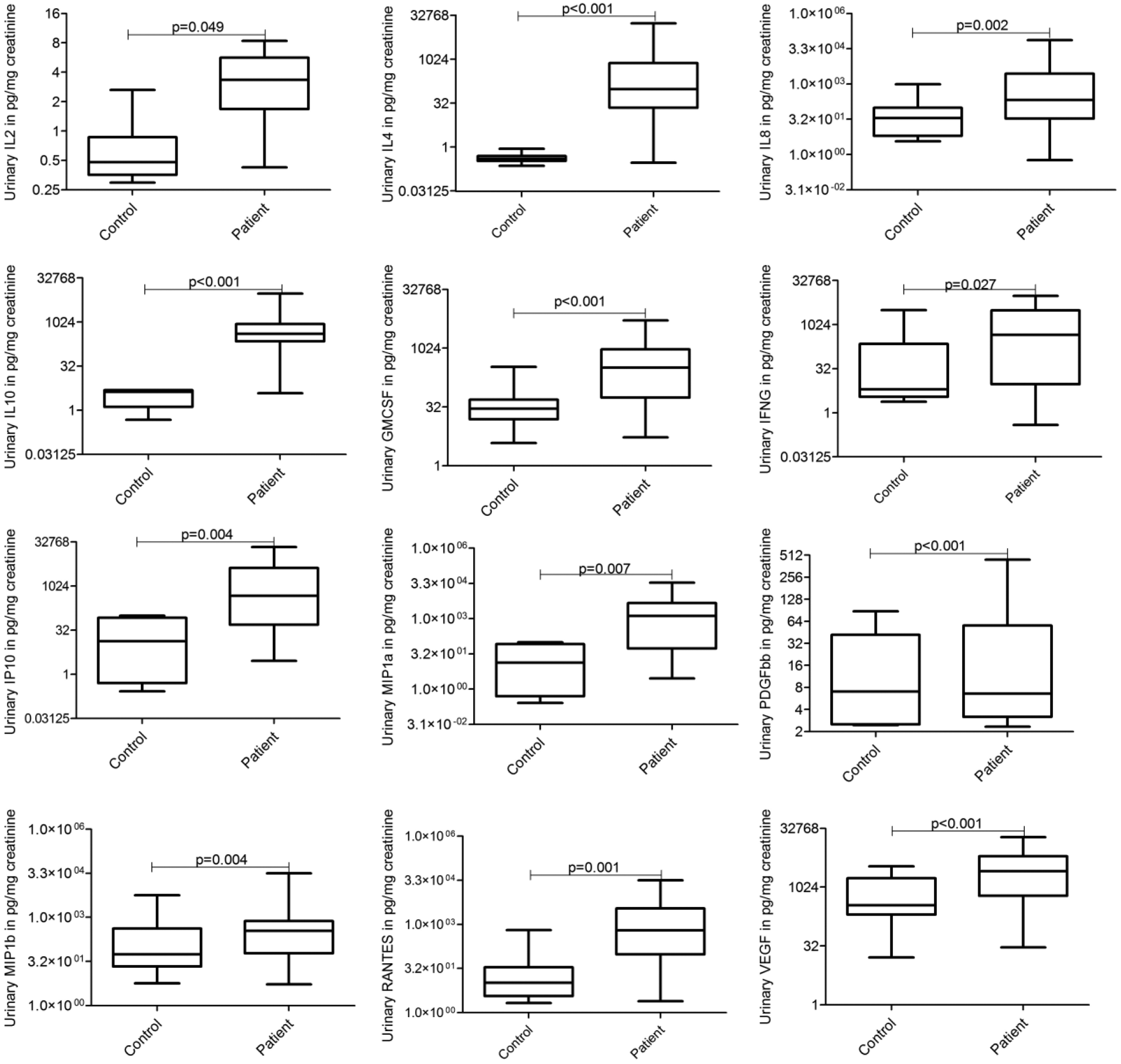
Box plots showing the concentration of urinary cytokines IL-2, IFN-γ, IP-10, IL-4, IL-10, IL-8, RANTES, MIP-1a, MIP-1b, PDGF-bb and VEGF in the patients and controls. All the 12 cytokines were found to be present in significantly higher concentration in patients’ urine compared to healthy controls.

Box plots showing concentration of urinary cytokines IL-1RA, IL-4, IL-6, IL-10, GM-CSF, IP-10, MIP-1a and MIP-1b in low- and high-grade cases.
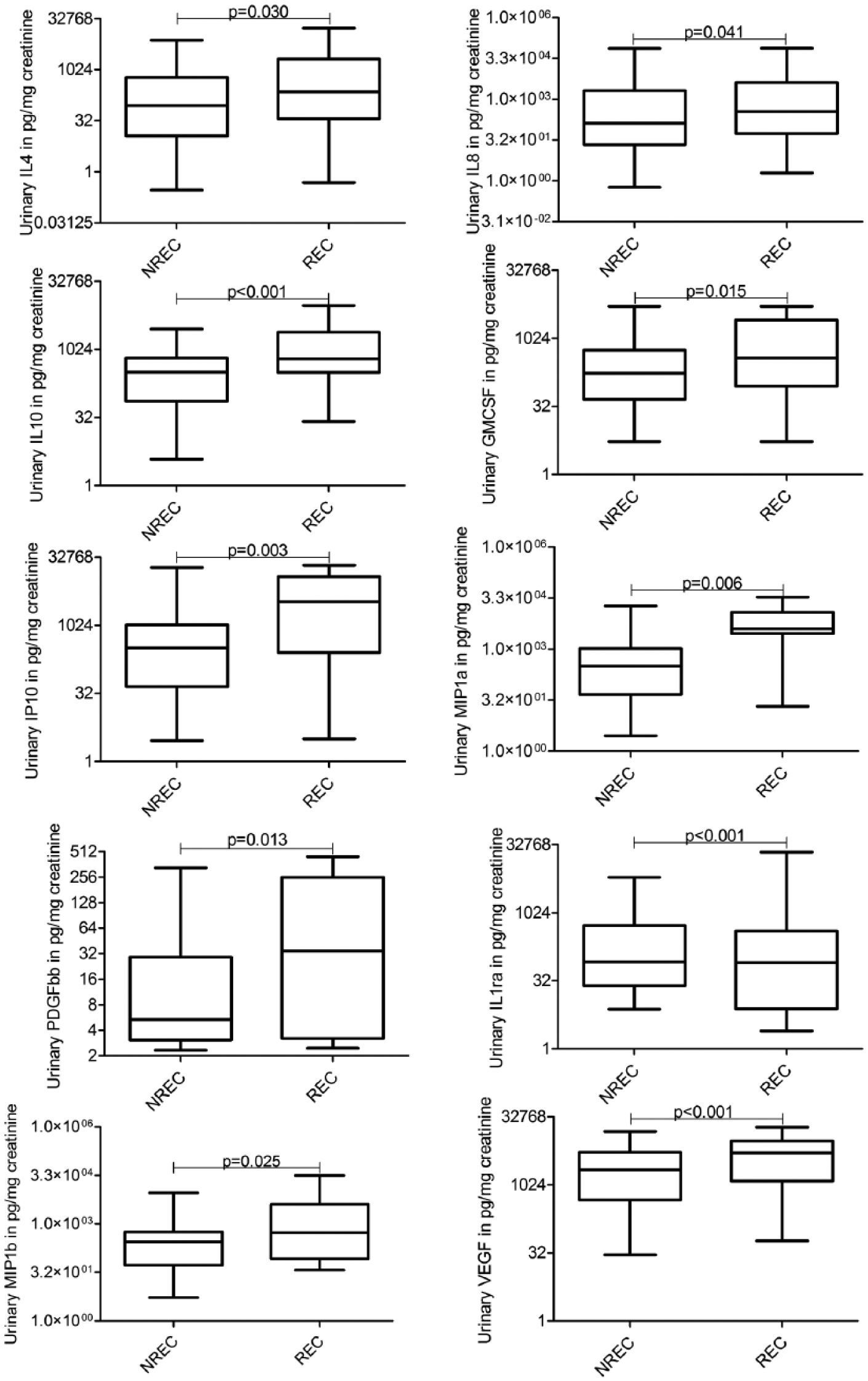
Box plots showing the concentration of urinary cytokines IL-4, IL-8, IL-10, IP-10, MIP-1a, PDGF, IL-1RA, MIP-1b and VEGF in recurrent and non-recurrent cases.
Survival analysis
High concentrations of the five serum cytokines (IL-1RA, IL-6, IL-10, TNF-α and VEGF) which were found to be associated with recurrence were also seen to be associated with poor RFS (Figure 6) significant by log-rank test. High concentrations of serum IL-1RA were associated with a poor RFS of 52.7% compared to 63.2% cumulative survival with high serum IL-1RA at 36 months of follow-up (p = 0.010). High serum IL-6 concentrations were compared to low concentration (36.5% vs 72.3%, p = 0.002) and found associated with poor RFS at 34 months follow-up. High concentrations of serum IL-10 were associated with significantly poorer RFS, 20.2% compared to cumulative survival of 79.6% with low concentrations of serum IL-10. Similarly, high serum TNF-α and VEGF showed a poorer RFS of 29.8% and 42.4% compared to low serum TNF-α and VEGF with survival of 64.2% and 62.0%, respectively, at 30 months of follow-up (p = 0.005 and 0.027, respectively; Table 2).

Kaplan–Meier survival analysis shows that elevated serum IL-1RA, IL-6, IL-10, TNF-α and VEGF concentrations are associated with poor recurrence-free survival. Kaplan–Meier survival analysis shows that (a) elevated sIL1ra. (b) sIL6. (c) sIL10. (d) sTNFα. (e) sVEGF concentrations are associated with poor recurrence-free survival as compared to low concentrations of sIL1ra, sIL6, sIL10, sTNFα and sVEGF respectively. hi and lo indicates cytokine concentrations above and below cut-off values given in Table 2 for the respective cytokine.
AUROC cut-off and significance of Kaplan–Meier analysis of serum (s) and urine (u) cytokines.

Low and high concentrations of urinary cytokines with cut-off based on AUROC analysis were used for KM survival analysis, and log-rank test was used for comparison of survival between these two groups. Of the 27 cytokines evaluated, high concentrations of urinary IL-1RA, IL-4, IL-10, IP-10, PDGF and VEGF were associated with poor RFS (Table 2, Figure 7). High concentrations of urine IL-10 were associated with significantly poorer RFS, 87.3% compared to 16.5% with low concentrations of urine IL-10 at 36 months follow-up. High IP-10 concentration in urine was associated with poorer RFS (33.0% compared to 51.6%, p = 0.032), and high PDGF concentration in urine was associated with poorer RFS (37.0% compared to 82.4%, p = 0.020). High urine IL-1RA, IL-4 and VEGF showed a poorer RFS of 17.2%, 14.9% and 12.0%, respectively, when compared to 100.0% RFS with low urinary concentrations of the same cytokine. The number of cytokines with high concentrations was computed for each case, and KM survival analysis showed that the more the number of cytokines which were high, the poorer the RFS was for both serum and urine cytokines (Figure 8).

KM survival analysis shows that higher concentrations of urinary IL-1RA, IL-4, IL-10, IP-10, PDGF and VEGF are associated with poor recurrence-free survival. Kaplan–Meier survival analysis shows that (a) elevated uIL1ra. (b) uIL4. (c) uIL10. (d) uIP10. (e) uPDGF. (f) uVEGF concentrations are associated with poor recurrence-free survival as compared to low concentrations of uIL1ra, uIL4, uIL10, uIP10, uPDGF, uVEGF respectively. hi and lo indicates cytokine concentrations above and below cut-off values given in Table 2 for the respective cytokine.
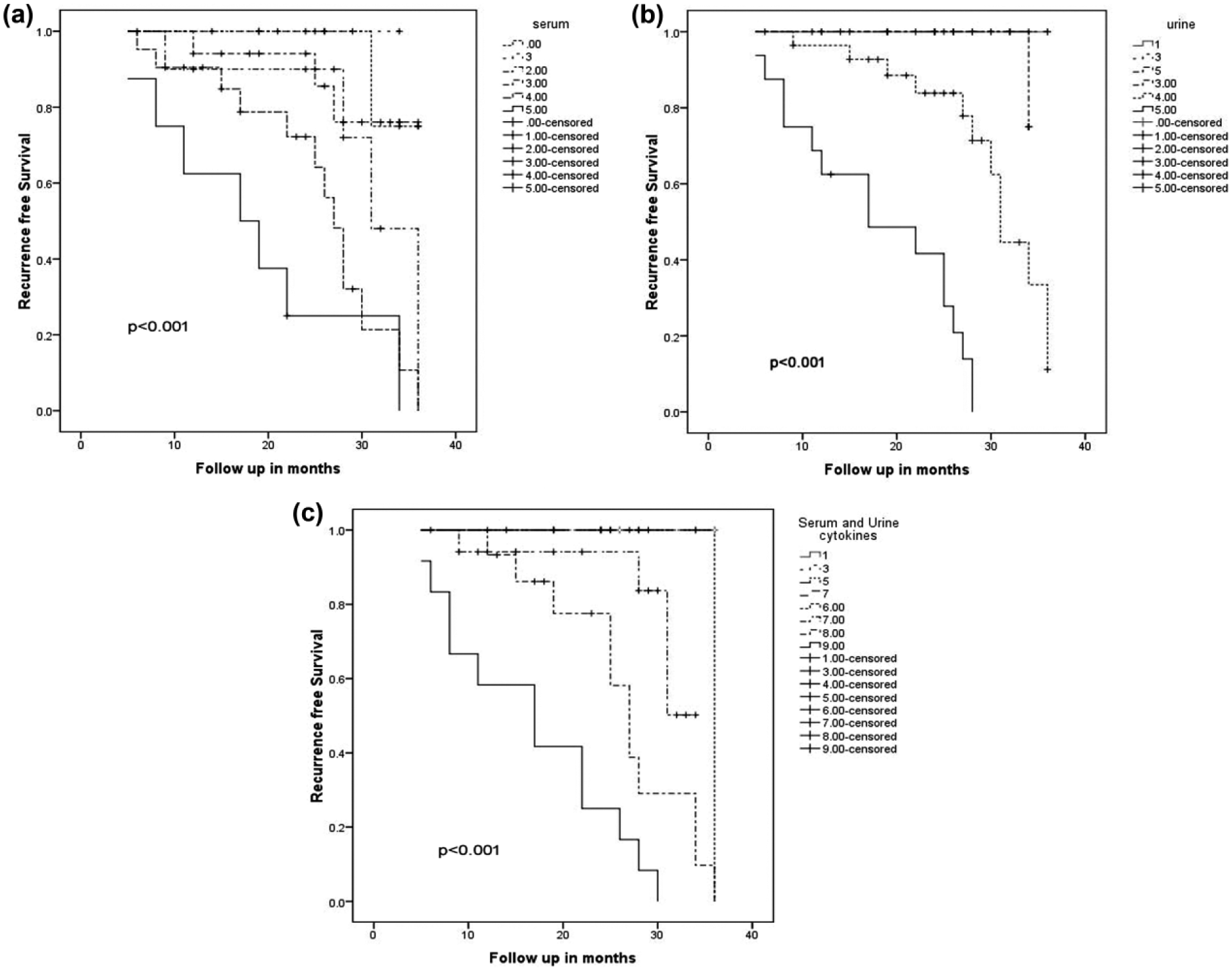
KM analysis: (a) combination of cytokines in serum shows poorer RFS when all five (IL-1RA, IL-6, IL-10, TNF-α and VEGF) cytokines are present in high concentration, (b) poorer RFS seen when five urinary cytokines are in high concentration and (c) poorer RFS seen in KM survival analysis when combination of serum and urine cytokines in each case was more than nine.
Correlation of serum and urine cytokines
Pearson’s correlation between those serum and urine cytokines found associated significantly with poor RFS showed significant positive correlation between the serum and urine cytokines (Table 3).
Correlations between serum and urine cytokines.
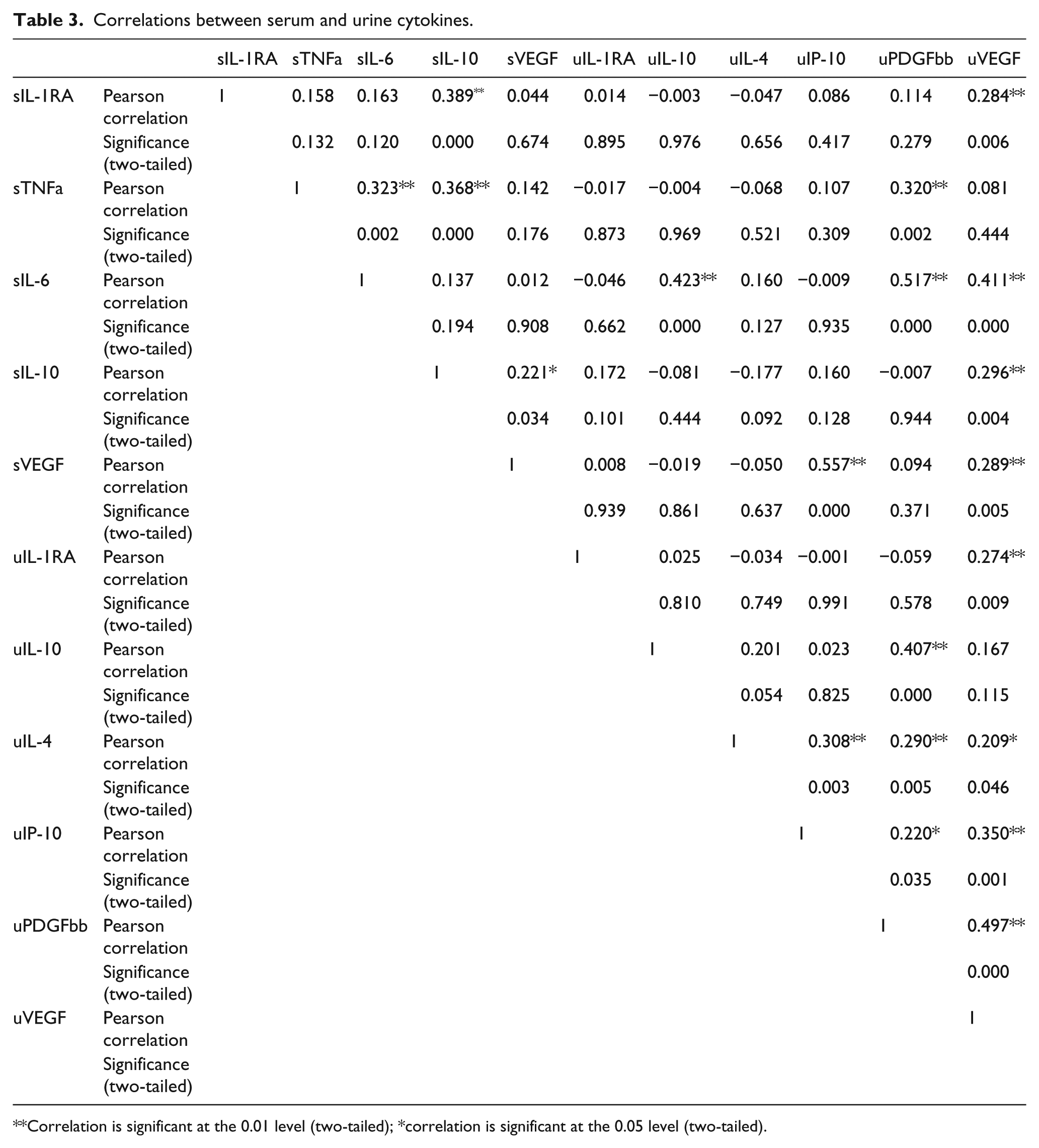
Correlation is significant at the 0.01 level (two-tailed); *correlation is significant at the 0.05 level (two-tailed).
Of the six urine cytokines found to have an association with poor RFS, concentrations of VEGF and IP-10 were compared by ELISA in cystitis and cancer cases with healthy controls. Both VEGF and IP-10 were found to be significantly higher in cancer cases compared to both cystitis cases and controls. There was no significant difference between concentration of VEGF and IP-10 in cystitis cases compared to controls (p > 0.05 by Mann–Whitney U test; Figure 9).

The concentrations of VEGF and IP-10 in urine of patients were compared in three groups of patients (cystitis, cancer and healthy controls). Both VEGF and IP-10 showed significant difference (p < 0.001) in concentration between control and cancer cases and also between cystitis and cancer cases. Though the concentration of both cytokines was high in cystitis cases, it was not significantly more than in controls.
Discussion
Cytokines have pleiotropic effects and also act in concert with sometimes a slight trigger resulting in a ‘cytokine cascade’. 5 Tumour cells themselves release certain chemokines and also express cytokine/chemokine receptors. 6 The recruitment of the inflammatory cells into the tumour milieu results in the further release of cytokines, and it is the balance of these cytokines in the environment which decides the fate of the tumour cells. 7 This may tip in favour of the immune response resulting in a cytotoxic effect on the tumour cells or it may end in the tumour cell immunoevasion with proliferation of the tumour cells. In this study, concentration of multiple cytokines was estimated in both serum and urine to understand the body’s response to cancer.
Multiplex assay for cytokines has previously been used for studying the immune response in patients and includes techniques such as ELISA, flow cytometry bead assays, and the Luminex/Bio-Plex platform. The advantage of multiplex arrays lies in the fact that multiple cytokines are estimated in a small amount of sample and the sensitivity of these kits is high and concentrations are measured in picograms. In a study of breast cancer patients and healthy controls, a cytokine profile of six differential cytokines including significantly elevated MCP-1, eotaxin, RANTES and GM-CSF and significantly decreased IL-1α and IL-4 was reported. 8 Similarly, in lung cancer, cytokine gene signatures associated with prognosis have been identified in different ethnic groups. These included IL-6, associated with worse prognosis in both African Americans and Caucasians; IL-10 and IL-12 associated with lung cancer survival in African Americans and TNF-α associated with survival in Caucasians. 9 In pancreatic ductal adenocarcinoma patients, a panel of IP-10, IL-6 and PDGF plus CA19-9 offered improved diagnostic performance over CA19-9 alone for differentiating from patients with benign disease. 10 These studies reveal that multiplex cytokine analysis may give cytokine signature for prognostic assessment in cancer patients. As cytokines interact with each other and activate or inhibit both pro- and anti-tumorigenic actions, it is more practical to find a combination of markers which have a role in prognostication.
The estimation of cytokines in this study showed that three cytokines in serum (IL-1RA, IL-4 and RANTES) were in significantly higher concentrations in bladder cancer patients compared to healthy control, while serum IL-2 was significantly higher in controls when compared to the patients. Of these four cytokines, IL-1RA was found significantly higher in recurrent bladder cancer patients. In addition, IL-6, IL-10, TNF-α and VEGF were found associated with recurrence, and high serum concentrations of these five cytokines were significantly associated with a poorer RFS.
Analysis of the concentration of urinary cytokines showed significantly high concentrations of 12 cytokines including IL-2, IFN-γ, IP-10, IL-4, IL-10, IL-8, RANTES, MIP-1a, MIP-1b, PDGF, GM-CSF and VEGF in urine of bladder cancer patients compared to healthy controls. At first glance, it appears that significantly elevated urinary IL-2 in bladder cancer patients is in contrast to the significantly decreased serum IL-2 in the same group of patients. However, a closer look at the results shows that while serum profile does not show elevation of any of the Th1 cytokines, the urinary profile shows elevation of IL-2, IFN-γ and IP-10, in addition to the Th2 cytokines IL-4 and IL-10. This profile indicates a proinflammatory response to the tumour with release of both Th1 and Th2 cytokines, chemokines and growth factors by the tumour infiltrating inflammatory cells which may be a mixture of T-cells (CD4+/CD8+), NK cells, dendritic cells and macrophages. 4 While there was no significant difference in cytokine profile between muscle-invasive and non-muscle invasive cases, high concentrations of IP-10, IL-1RA, IL-4, IL-6, IL-10, IL-8, MIP-1a, MIP-1b and GM-CSF were found associated with high grade. High concentrations of nine of these cytokines including IP-10, IL-1RA, IL-4, IL-10, IL-8, MIP-1a, MIP-1b, PDGF and VEGF were also found associated with recurrence. Of these, high concentrations of six cytokines (IP-10, IL-1RA, IL-4, IL-10, PDGF and VEGF) have been found associated with poorer RFS. However, all the cytokines and growth factors are also released in urine of cystitis patients,11–13 and hence, it is necessary to know the concentrations in urine of cancer patients compared to patients with cystitis if these markers have to be used as biomarkers of recurrence in future. To address this issue, a small cohort of patients with cystitis was recruited, and it was found that the concentrations of VEGF and IP-10 were significantly higher in cancer cases compared to both controls and patients with cystitis.
The initial proinflammatory response to the tumour cells triggers the release of the acute-phase cytokines IL-1, IL-6 and TNF-α. IL-1RA is released soon after to mitigate the injurious effects of IL-1b and is also considered an acute-phase protein. IL-1RA is a secretory protein produced by macrophages, monocytes and neutrophils and is reported to be present in high levels in circulation in inflammatory, infectious and neoplastic conditions. High concentrations of IL-1RA have been reported in certain cancers such as bronchogenic carcinoma, hepatocellular carcinoma, Hodgkin’s lymphoma and in pituitary tumours. However, in some cancers, such as thyroid cancer, reduced levels have been reported. 14 It is proposed that the high levels of IL-1RA may inhibit IL-1 and help in evading the immune system and thus potentiate cell proliferation. 15 In this study, IL-1RA was found raised in both serum and urine of patients and associated with poor RFS. The systemic response showed high serum concentrations of IL-6, TNF-α and IL-1RA, and the local cytokine secretion into the urine revealed a high concentration of IL-1RA in bladder cancer patients. The acute-phase proteins, IL-6 and TNF-α activate nuclear factor-kappaB (NF-κB), and the downstream signalling in this pathway plays a role in cell survival, carcinogenesis and poor survival. 16 High IL-1RA and IL-6 were reported in colorectal cancer and purported to result in hypoalbuminemia and cancer cachexia. 17
IL-4 and RANTES were both found increased in both serum and urine of bladder cancer patients. However, only high urinary IL-4 was found to be associated with poor RFS. IL-4, a Th2 cytokine, appears to have a direct anti-apoptotic effect and an indirect pro-tumorigenic effect. 18 A Th2 response against tumours is generally associated with a worse prognosis, and this study shows a similar trend with IL-4 associated with poor RFS.
Other factors found raised in both serum and urine in bladder cancer patients and associated with poor RFS include IL-10 and VEGF. IL-10 acts as an immunosuppressive and at the same time triggers tumour progression. 19 VEGF has been reported to play a significant role in carcinogenesis and is associated with poor survival, invasion and metastasis in prostate, breast and pancreatic cancer.20–22 It is also a pro-angiogenic factor and possibly mediates the cellular proliferation in cancer by this action. Other pro-angiogenic factors raised in urine included IL-8 and PDGF. IL-8 is a leukocyte chemoattractant and has previously been reported as an independent factor in urine for detection of bladder cancer. IL-8 has also been reported to be significantly high in high-grade, muscle-invasive and recurrent cases.23,24
The Th1 cytokines IL-2, IFNγ and IP-10 (CXCL10) were also found raised in urine of bladder cancer patients, but only IP-10 was found associated with poor RFS. The mixture of both Th1 and Th2 cytokines found in the urine of bladder cancer patients indicates the proinflammatory immune response. IP-10 is a C-X-C chemokine which has proinflammatory and anti-angiogenic properties. However, it binds to its receptor CXCR3 and promotes tumour proliferation, invasion and metastasis of cancer cells in several tumours.25,26 IP-10 is induced by Ras signalling by way of Raf and PI3 kinase signalling pathways. 26 IP-10 has been shown to be carcinogenic by its activity in recruiting T-regulatory lymphocytes which release IL-10, thus suppressing the immune response. 27
It is well known that the cytokine network acts in concert to achieve an end effect, and hence, the association of high concentrations of the number of cytokines with survival was evaluated and it was found that with the estimation of a panel of five cytokines (IL-1RA, IL-6, IL-10, TNF-α and VEGF) in serum and six cytokines (IP-10, IL-1RA, IL-4, IL-10, PDGF and VEGF) in urine, the predictive power for recurrence may be increased. This raises the possibility of using this combination of cytokines as a signature profile in non-invasive samples for identification of poor prognosis.
Conclusion
The study shows association of high concentration of cytokines, IL-1RA, IL-10, IL-4, IL-6, IP-10, TNF-α, PDGF and VEGF with poor RFS in bladder cancer patients. It is also evident that estimation of multiplex cytokine panel constituting these cytokines by ELISA would be a powerful predictive non-invasive biomarker in urothelial cancer patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors acknowledge the financial support given for this work by the Indian Council of Medical Research.
