Abstract
ABT-737, a B-cell lymphoma 2 homology 3 mimetic, not only induces cell apoptosis by inhibiting the interaction of B-cell lymphoma 2 and Bax but also induces cell autophagy by interrupting the interaction of B-cell lymphoma 2 and Beclin1. Several recent studies have reported that ABT-737 has antitumor efficacy in diverse cancers. However, another study showed that hepatocellular carcinoma cells with high B-cell lymphoma 2 expression were resistant to ABT-737 compared to hepatocellular carcinoma cells with low B-cell lymphoma 2 expression. It was also found that ABT-737-induced autophagy is crucial for drug resistance. Here, we observed that of B-cell lymphoma 2 expression in Adriamycin-resistant human hepatocellular carcinoma HepG2/ADM cells is higher than that in human hepatocellular carcinoma HepG2 cells. Therefore, we further confirmed the mechanism and effect of autophagy induced by ABT-737 on apoptosis in HepG2/ADM cells with high B-cell lymphoma 2 expression. Our results showed that ABT-737 induced apoptosis and autophagy in time- and dose-dependent manner in HepG2/ADM cells, and this ABT-737-induced autophagy was Beclin1-dependent. In addition, we demonstrated that ABT-737 induced reactive oxygen species-mediated autophagy, and the reactive oxygen species-inhibitor
Introduction
Human hepatocellular carcinoma (HCC), the third most common malignancy, has a very high mortality rate.1,2 In recent years, almost all patients with HCC have been treated with traditional chemotherapy, which often leads to unsatisfactory results, drug resistance, and poor prognosis. 3 This indicates the necessity to develop novel molecular target complexes to improve HCC treatment. Of these compounds, the B-cell lymphoma 2 (Bcl-2) inhibitors have better antitumor activity than other compounds, 4 and better clinical results.
The Bcl-2 family proteins, which play a key role in the process of apoptosis, contain the conservative Bcl-2 homology (BH) regions and are divided into two categories, based on their functions: pro-survival proteins and pro-apoptotic proteins.5,6 Pro-survival proteins contain four BH regions (BH1–BH4) and include Bcl-2, Bcl-xL, Bcl-w, Mcl-1, and A1. Pro-apoptotic proteins are further divided into two categories: BH3-only proteins (Bim, Puma, Bid, Bad, Bik, Bmf, Hrk, and Noxa) 7 and Bax and Bak, which contain three BH domains (BH1, BH2, and BH3).8–11 In recent years, some reports have indicated that overexpression of the Bcl-2 protein is associated with many tumors, such as prostate cancer, colon cancer, breast cancer, non-small cell lung cancer, and human HCC. 12 An increasing number of studies suggest that inhibition of Bcl-2 expression is an effective strategy for cancer therapy.13–15
ABT-737, a BH3 mimetic, is a small-molecule inhibitor of Bcl-2 family proteins and it specifically inhibits the interaction of Bcl-2/Bcl-xL and Bax/Bak through competitive binding with the BH3 domain and induces apoptosis. Several studies have reported that ABT-737 has a positive effect on cancer treatment by targeting the apoptosis machinery. ABT-737 is a mimetic of BH3 protein Bad and binds to Bcl-2, Bcl-xL, and Bcl-w, but not to Mcl-1 or Bfl-1.16–18 Several studies have demonstrated the antitumor activity of ABT-737 in different types of cancers, including several hematopoietic cancers and some solid tumors.19,20
In recent years, many studies have reported that autophagy plays an important role in tumorigenesis and is becoming a key regulator of cancer survival.14,15,21 It has been reported that ABT-737 induces apoptosis in diverse cancer cells such as those in acute lymphoblastic leukemia, small cell lung cancer,18,19 and liver cancers.
22
In recent years, some data demonstrated that the Bcl-2 inhibitor gossypolone induces reactive oxygen species (ROS)-dependent protective autophagy by the activation of c-Jun N-terminal kinase (JNK) in HCC cells,
23
and the research group also reported that ABT-737 induces protective autophagy in HCC cells with high level of Bcl-2.
24
In our previous study, we observed that the expression of Bcl-2 in Adriamycin-resistant human HCC HepG2/ADM cells is higher than that in human HCC HepG2 cells. Therefore, in this study, we further demonstrate that ABT-737 induced ROS- and Beclin1-mediated autophagy in Adriamycin-resistant human HCC HepG2/ADM cells. We also demonstrate that the inhibition of autophagy by the ROS inhibitor
Materials and methods
Reagents and antibodies
ABT-737, a BH3 mimetic, was purchased from Selleck Chemicals LLC (Houston and Texas, USA) and dissolved in dimethyl sulfoxide (DMSO). Antibodies against Bax (1:100, mouse) and β-actin were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA). The antibody against Beclin1 (1:100) was purchased from BD Bioscience (Franklin Lakes, NJ, USA). Antibodies against Bcl-2 (1:200, rabbit) and LC3 (1:200, rabbit) were purchased from Abcam (Cambridge, MA, USA). Antibody against caspase 3 (1:200, rabbit) was purchased from Cell Signaling Technology (Beverly, MA, USA). 3-MA, NAC, and DCFH-DA (2′,7′-dichlorodihydrofluorescein diacetate) were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Cell culture
Human HCC HepG2 cells and Adriamycin-resistant human HCC HepG2/ADM cells were purchased from BeNa Culture Collection (Beijing, China), cultured in Dulbecco’s modified Eagle’s media (Gibco, Gaithersburg, MD, USA), and supplemented with 10% (v/v) fetal bovine serum (Gibco) at 37°C with 5% CO2.
Cell viability assays
Adriamycin-resistant human HCC HepG2/ADM cells were plated at 1 × 104 cells/well in 96-well plates (Corning Incorporated, Tewksbury, MA, USA). After 24 h, cells were treated with different concentrations (0, 2.5, 5, 10, 20, 40, and 80 µmol/L) of ABT-737 for 24 h or treated with the autophagy inhibitor 3-MA (5 mmol/L) and the ROS inhibitor NAC (10 mmol/L) for 30 min, and then incubated with ABT-737 (10 µmol/L) for 24 h. 3-(4,5-Dimethylthiazol-2yl)-2,5-diphenyltetrazolium bromide (MTT) reagent (20 µL, 5 mg/mL in phosphate buffered saline (PBS)) was then added in each well, and plates were incubated for 4 h. Subsequently, 150 µL DMSO was added in each well to dissolve formazan crystals, and absorbance was recorded at a wavelength of 490 nm (Bio-Rad, Hercules, CA, USA).
Flow cytometry analysis
Adriamycin-resistant human HCC HepG2/ADM cells were plated in six-well plates (Corning Incorporated) (15 × 104 cells/well). After 24 h, cells were treated with different concentrations (0, 5, 10, 20, and 40 µmol/L) of ABT-737 for 24 h or treated with the autophagy inhibitor 3-MA (5 mmol/L) and ROS inhibitor NAC (10 mmol/L) for 30 min, and then incubated with ABT-737 (10 or 25 µmol/L) for 24 h. Cells were collected and washed three times with PBS. Cells were then incubated at 37°C for 20 min with propidium iodide (PI; 1 mg/mL; Invitrogen, Carlsbad, California, USA) and Annexin V-FITC (1 mg/mL; Invitrogen) to determine the number of apoptotic cells. Finally, the samples were analyzed on a FACScan flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA).
Measurement of ROS production
Adriamycin-resistant human HCC HepG2/ADM cells were plated in 24-well plates (Corning Incorporated) (5 × 104 cells/well). After 24 h, cells were treated with different concentrations (0, 2.5, 5, 10, 15, and 20 µmol/L) of ABT-737 for 24 h. The cells were then washed three times with PBS and incubated with DCFH-DA (10 µmol/L) at 37°C for 30 min. Cells were washed with PBS a further three times. DCF-dependent fluorescence was detected by immunofluorescence microscopy (Olympus).
Western blotting assays
Adriamycin-resistant human HCC HepG2/ADM cells were lyzed in radioimmunoprecipitation assay (RIPA) buffer by sonication. Proteins were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes (Millipore, Bedford, MA, USA). Then, 5% non-fat dry milk in phospate buffered saline Tween 20 (PBST) buffer (10 mM Tris–HCl pH 7.6, 100 mM NaCl, and 0.1% Tween 20) was added to block the membranes for 60 min at room temperature, and the membranes were incubated with the relevant primary antibody overnight at 4°C. The next day, the membranes were incubated with a horseradish peroxidase-conjugated secondary antibody at a 1:1000 dilution for 60 min at room temperature. The bands were measured with an ECL (enhanced chemiluminescence detecting agents; Thermo Fisher Scientific, Waltham, MA, USA) image detecting system (Gene Company Ltd, Hong Kong, China).
Immunofluorescent confocal laser microscopy
Adriamycin-resistant human HCC HepG2/ADM cells were cultured on coverslips at a density of 5 × 104 cells/well in 500 µL of complete medium. After 24 h, cells were treated with indicated reagents for indicated times. After treatment, HepG2/ADM cells were washed three times with cold PBS, fixed in 4% (w/v) paraformaldehyde/PBS at 37°C for 15 min, and washed with cold PBS three times. The fixed cells were digested by protein enzyme K for 1 min and washed three times with PBS. Cells were incubated with 0.1% (v/v) Triton X-100 for 8 min, washed three times with PBS, and then blocked with 5% (v/v) non-immune animal serum/PBS for 30 min at room temperature. Cells were incubated overnight with primary antibody at 4°C and washed three times with PBS. Cells were then incubated with fluorescent secondary antibody (rabbit and mouse, dilution 1:400; Thermo Fisher Scientific) for 30 min in a dark and humid box. Plates were washed three times in PBS, treated with Hoechst 33342/H2O (1 µg/mL) for 2 min, and washed a further three times with PBS. Cells were examined by Olympus FV1000 confocal laser microscope.
TUNEL assays
Adriamycin-resistant human HCC HepG2/ADM cells were cultured on coverslips at a density of 5 × 104 cells/well in 500 µL of complete medium. After 24 h, cells were treated with NAC (10 mmol/L) for 30 min and then incubated with ABT-737 (10 µmol/L) for 24 h. Apoptosis analysis was performed using an In Situ Cell Death Detection Kit (Roche, Indianapolis, IN, USA) to find DNA breaks according to the manufacturer’s instructions. HepG2/ADM cells were cultured on coverslips, treated with 4% (w/v) paraformaldehyde/PBS at 37°C for 15 min, and then washed three times with cold PBS. Cells were incubated in the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) reaction mix containing 10 U of terminal deoxyribosyltransferase, 10 mM dUTP-biotin, and 2.5 mM cobalt chloride in 1 × terminal transferase reaction buffer for 1 h at 37°C in a dark and humid box. Apoptotic cells with characteristic nuclear fragmentation (green staining) were counted in six randomly chosen fields. The experiment was repeated three times.
Transfection
The plasmids of shScramble and shBeclin1 were purchased from Shanghai GenePharma Co., Ltd. The short hairpin RNA (shRNA) plasmid target sequences were as follows: shScramble, GTATGACAACAGCCTCAAG; and shBeclin1-1056, GGAATGGAATGAGATTAATGC. HepG2/ADM cells were transfected with shRNA plasmids and shScramble in a six-well plate with lipofectamine 2000 according to the manufacturer’s protocol.
Statistical analysis
Statistical analysis of the data was performed by T-test. The data are presented as the mean ± standard deviation (SD). The p-value <0.05 was considered statistically significant. Data represent three independent results.
Results
ABT-737 inhibits Adriamycin-resistant human HCC HepG2/ADM cell growth and induces mitochondria-mediated apoptosis
Ni Zhenhong et al. have reported that ABT-73 induced protective autophagy partly through the production of ROS in HCC cells with high expression of Bcl-2. 24 To our surprise, we found here that the ratio of Bcl-2/Bax in HepG2/ADM cells is higher than that in HepG2 cells (Figure 1(a)). Therefore, we investigated whether ABT-737 can induce protective autophagy in HepG2/ADM cells. HepG2/ADM cells were treated with different concentrations (0, 2.5, 5, 10, 20, 40, and 80 µmol/L) of ABT-737 for 24 h, and cell growth rate was examined by MTT assay. The results showed that the viability of HepG2/ADM cells was decreased by the ABT-737 treatment (Figure 1(b)). According to the MTT assay results, we selected 10 µmol/L as the optimum concentration of ABT-737.

ABT-737 inhibits HepG2/ADM cell growth and induces apoptosis. (a) Western blot analysis of Bax and Bcl-2 expression in HepG2 and HepG2/ADM cells, (b) quantitative analysis of Bax and Bcl-2 protein levels from (a). Data are presented as mean ± SD, n = 3; *p < 0.05 and **p < 0.01 vs control group, (c) HepG2/ADM cells were treated with different concentrations (0, 2.5, 5, 10, 20, 40, or 80 µmol/L) of ABT-737 for 24 h. Cell viability was detected by MTT assay. Data are presented as mean ± SD, n = 6. *p < 0.05, **p < 0.01, ***p < 0.001 vs control group, and (d) the percentage of apoptotic cells of HepG2/ADM cells induced by different concentrations (0, 5, 10, 20, or 40 µmol/L) of ABT-737 for 24 h was detected by flow cytometry analysis.
As known, HCC cells have high levels of Bcl-2, and we examined whether ABT-737 has an effect on the apoptosis of HepG2/ADM cells. For this, we treated cells with different concentrations (0, 5, 10, 20, and 40 µmol/L) of ABT-737 for 24 h and evaluated apoptosis using PI-Annexin V double dye assays. These results show that ABT-737-induced apoptosis rate increased in a dose-dependent manner, suggesting that ABT-737 induced apoptosis in HepG2/ADM cells (Figure 1(d)).
To confirm the apoptotic mechanism induced by ABT-737, we evaluated the expression of the apoptotic-related proteins caspase 3, cleaved caspase 3, Bax, and Bcl-2, in response to various concentrations of ABT-737 (0, 2.5, 5.0, and 10.0 mol/L) for 24 h and the expression of apoptotic-related proteins caspase 3 and cleaved caspase 3 in response to different times (10 µmol/L; 0, 8, 12, and 24 h). ABT-737 induced the expression of Bax/Bcl-2 ratio and cleaved caspase 3 in a dose-dependent manner (Figure 2(a)–(c)) and the expression of cleaved caspase 3 in a time-dependent manner (Figure 2(d) and (e)). Together, these results suggest that ABT-737 inhibited HepG2/ADM cell growth and induced apoptosis in a time- and dose-dependent manner. It is a remarkable fact that the small dose of ABT-737 (10 µmol/L) induced mild apoptosis, and we speculate there is some mechanism of resistance to ABT-737 in HepG2/ADM cells.
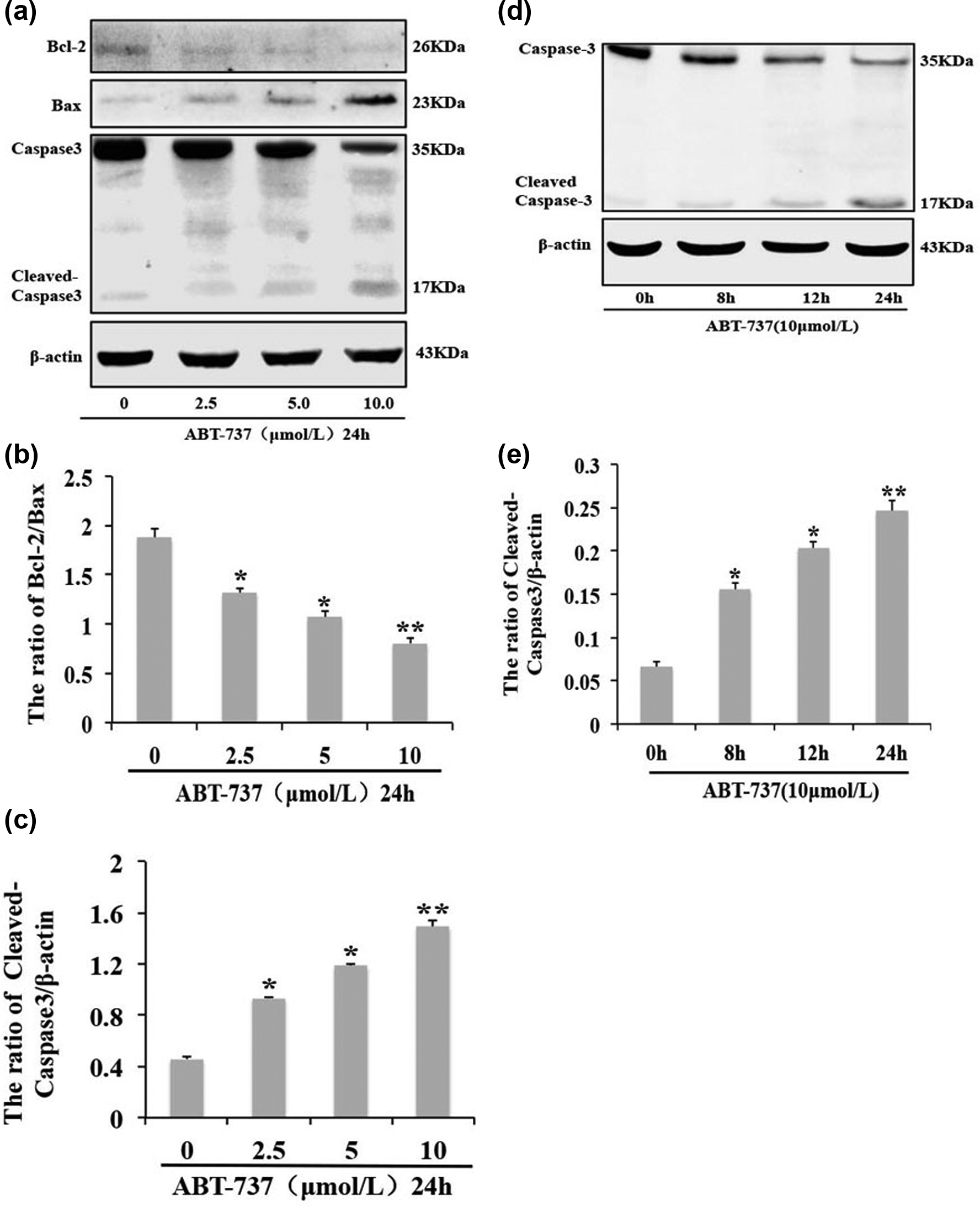
ABT-737 induces the apoptosis of HepG2/ADM cells. (a) Western blot analysis of Bcl-2, Bax, caspase 3, and cleaved caspase 3 expression in HepG2/ADM cells treated with different concentrations (2.5, 5, and 10 µmol/L) of ABT-737 for 24 h, (b) and (c) quantitative analysis of Bax, Bcl-2, and cleaved caspase 3 proteins levels from (a). Data are presented as mean ± SD, n = 3; *p < 0.05, **p < 0.01 vs control group; (d) western blot analysis for the expression of caspase 3 and cleaved caspase 3 in HepG2/ADM cells treated with 10 µmol/L ABT-737 for 8, 12, and 24 h, and (e) quantitative analysis of cleaved caspase 3 proteins levels from (d). Data are presented as mean ± SD, n = 3; *p < 0.05, **p < 0.01 vs control group.
ABT-737 induces Beclin1-dependent autophagy in Adriamycin-resistant human HCC HepG2/ADM cells
Several studies have shown that besides regulating apoptosis, Bcl-2 protein also regulates autophagy. 15 Therefore, we further examined whether ABT-737 could induce autophagy in Adriamycin-resistant human HCC HepG2/ADM cells. Beclin1 has an important function in the initiation of autophagy, 25 and LC3 (Atg8) is a component of the ubiquitin-conjugating system and plays an indispensable role in autophagosome formation.10,26 Our results show that Beclin1 expression increased in a dose- and time-dependent manner in HepG2/ADM cells treated with ABT-737 (Figure 3(a)–(d)). When autophagy occurs, LC3 protein has dot aggregation, and the soluble form of LC3 (LC3-I) is converted into a lapidated and autophagosome-associated form (LC3-II). 15 HepG2/ADM cells were treated with different concentrations of ABT-737 (0, 2.5, 5.0, and 10.0 µmol/L) for 24 h or with 10.0 µmol/L ABT-737 for different times (0, 8, 12, and 24 h). We observed that the expression of LC3-II was increased in a dose- and time-dependent manner (Figure 3(a)–(d)). We also found that LC3 dot aggregation increased in the cytoplasm in a time-dependent manner in HepG2/ADM cells treated with 10 µmol/L ABT-737 for different times (0, 8, 12, and 24 h) (Figure 4(a)). Furthermore, gene silencing revealed that autophagy induced by ABT-737 is Beclin1-dependent in HepG2/ADM cells (Figure 4(b) and (c)). These results indicate that ABT-737 induced Beclin1-dependent autophagy, and this autophagy is dose- and time-dependent in HepG2/ADM cells treated with ABT-737.

ABT-737 induces Beclin1 and LC3-I/II in HepG2/ADM cells. (a) Western blot analysis of Beclin1 and LC3-I/II expression in HepG2/ADM cells treated with different concentrations of ABT-737 (0, 2.5, 5, and 10 µmol/L) for 24 h; (b) western blot analysis of Beclin1 and LC3-I/II expression in HepG2/ADM cells treated with 10 µmol/L ABT-737 for 8, 12, and 24 h; and (c) and (d) quantitative analysis of Beclin1 and LC3-II proteins levels from (a) and (b). Data are presented as mean ± SD, n = 3; *p < 0.05 vs control group.
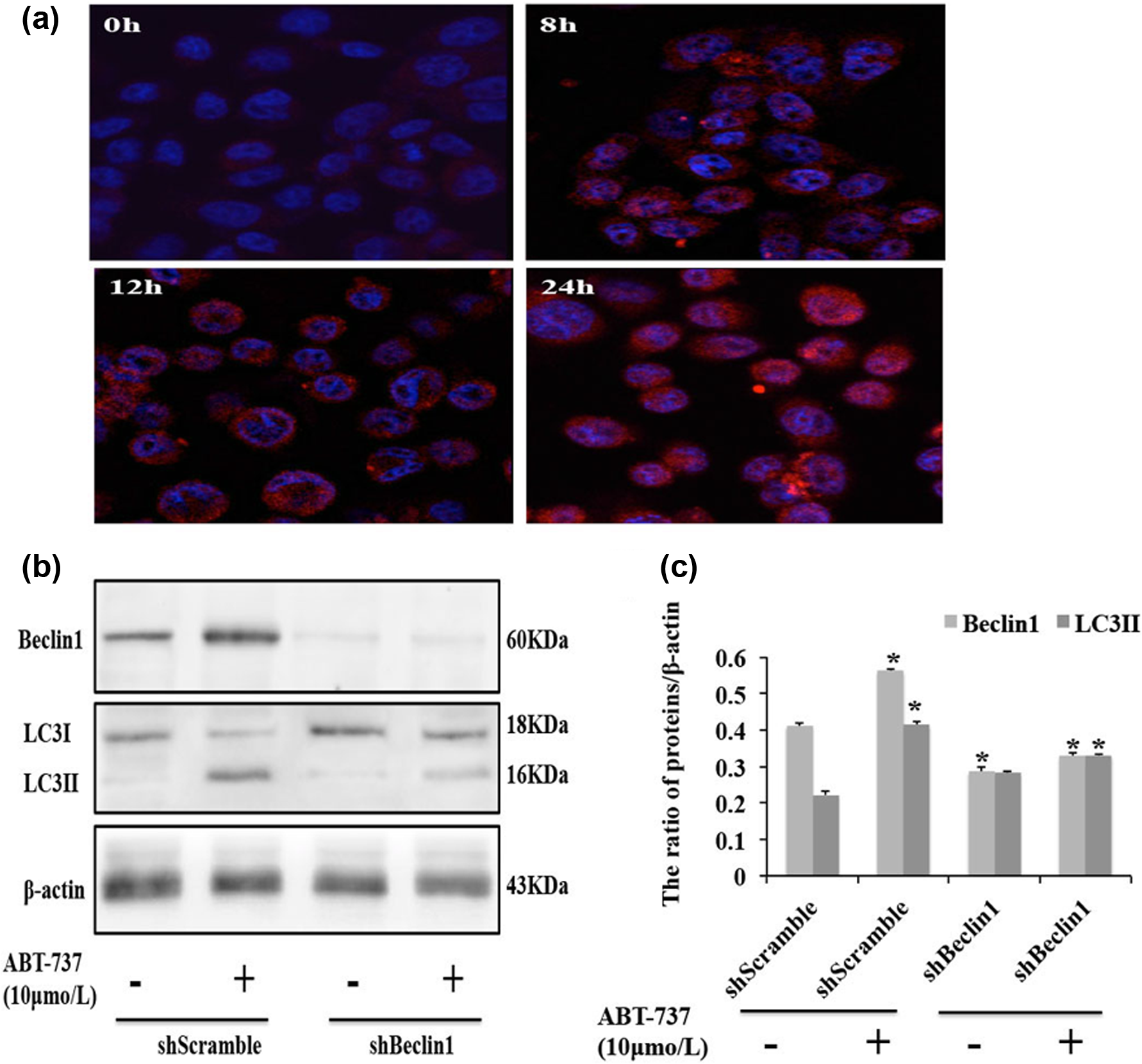
ABT-737 induces Beclin1-dependent autophagy in HepG2/ADM cells. (a) Distribution of LC3 dot aggregation in the cytoplasm of HepG2/ADM cells treated with 10 µmol/L of ABT-737 for 8, 12, and 24 h was observed by confocal microscopy; (b) western blot analysis of Beclin1 and LC3-I/II expression in Beclin1 knockdown HepG2/ADM cells. HepG2/ADM cells were transfected with shScramble or shBeclin1 for 24 h, and the cells were treated with 10 µmol/L ABT-737 for an additional 24 h, and (c) quantitative analysis of Beclin1 and LC3-II proteins levels from (b). Data are presented as mean ± SD, n = 3, *p < 0.05 vs control group.
ABT-737 induces the production of ROS and ROS-mediated autophagy in Adriamycin-resistant human HCC HepG2 cells
Recent studies have reported that BH3 mimetics can activate many pro-autophagy pathways beside Beclin1-dependent autophagy; 27 for example, ApoG2 can induce ROS production and ROS-mediated autophagy in HCC cells. 23 Therefore, we next examined the intracellular level of ROS in order to confirm whether ABT-737 can induce ROS production. Our results show that ABT-737 induced the increase of the intracellular level of ROS in a time-dependent manner in HepG2/ADM cells (Figure 5(a)). Moreover, we determined whether ROS induced by ABT-737 can induce autophagy in HepG2/ADM cells. Therefore, we examined the visible LC3 dot aggregation in HepG2/ADM cells treated with ABT-737 alone and with ABT-737 combined with NAC, which is a ROS inhibitor. We observed that LC3 dot aggregation decreased in HepG2/ADM cells treated with ABT-737 combined with NAC, compared with HepG2/ADM cells treated with ABT-737 alone (Figure 5(b)). In addition, we examined the expression of LC3-II protein in HepG2/ADM cells treated with ABT-737 alone and with ABT-737 combined with NAC. Compared with HepG2/ADM cells treated with ABT-737 alone, the expression of LC3-II protein decreased in HepG2/ADM cells treated with ABT-737 combined with NAC (Figure 5(c) and (d)). These results indicate that ABT-737 induced ROS production and ROS-mediated autophagy in HepG2/ADM cells.
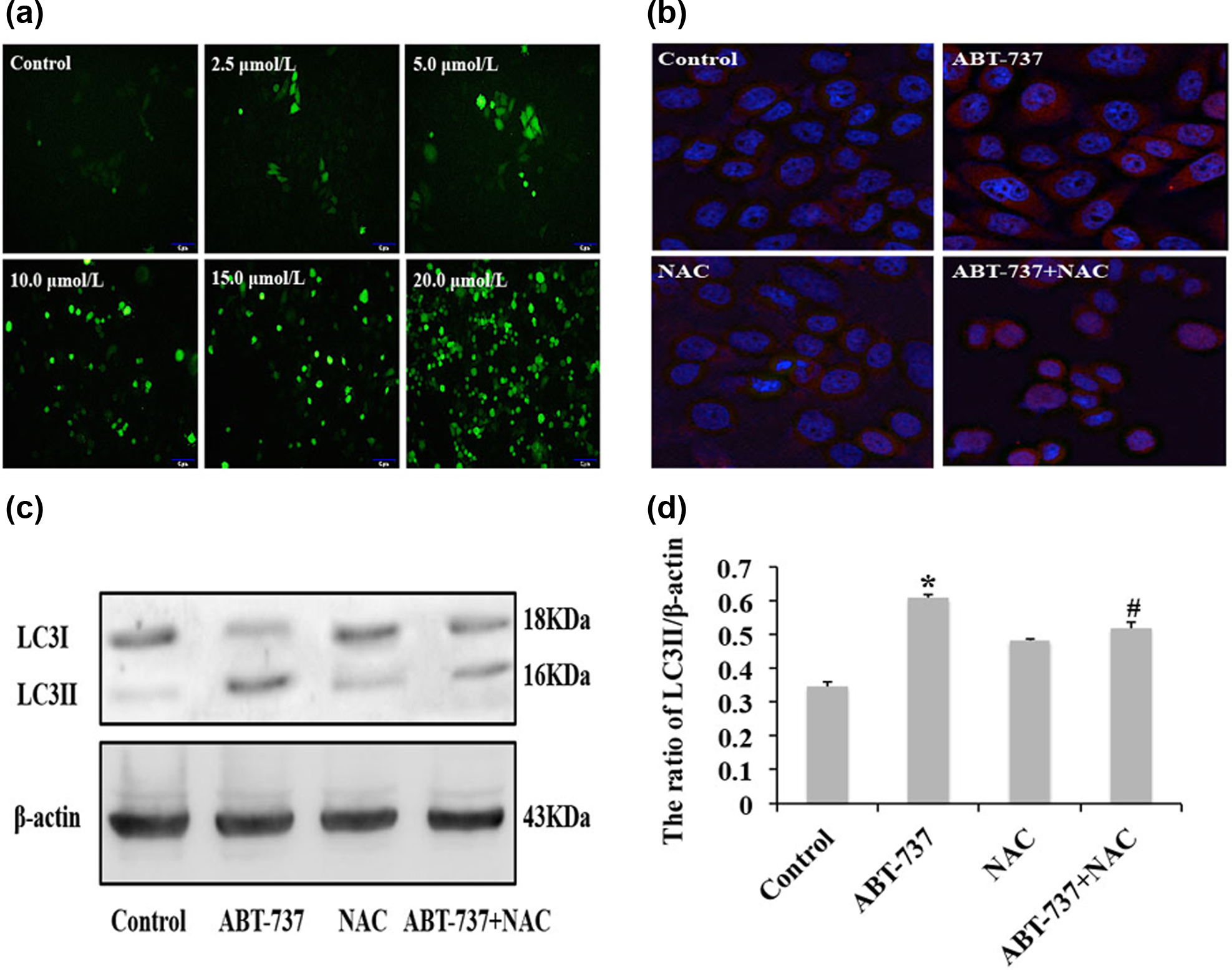
ABT-737 induces ROS production and ROS-mediated autophagy in HepG2/ADM cells. (a) DCFH-DA fluorescent probe detected total intracellular ROS in HepG2/ADM cells treated with different concentrations of ABT-737 (0, 2.5, 5, 10, 15, and 20 µmol/L) for 24 h; (b) distribution of LC3 dot aggregation was observed by confocal microscopy in the cytoplasm of HepG2/ADM cells treated with 10 mmol/L NAC for 30 min, and then incubated in 10 µmol/L ABT-737 for 24 h; (c) western blot analysis of LC3-I/II expression in HepG2/ADM cells treated with 10 mmol/L NAC for 30 min, and then incubated in 10 µmol/L ABT-737 for 24 h; and (d) quantitative analysis of LC3-II protein level from (c). Data are presented as mean ± SD, n = 3; *p < 0.05 vs control group, #p < 0.05 vs ABT-737 group.
Inhibition of ROS enhances Adriamycin-resistant human HCC HepG2/ADM cell apoptosis induced by ABT-737
To examine whether autophagy mediated by ROS can enhance HepG2/ADM cell death induced by ABT-737, HepG2/ADM cells were treated with ABT-737, NAC, or ABT-737 combined with NAC. Our result show that compared with ABT-737 alone, ABT-737 combined with NAC obviously decreased cell viability (Figure 6(a)) and increased cell apoptosis in HepG2/ADM cells (Figure 6(b)–(d)). In addition, we observed that the expression of apoptotic-related cleaved caspase 3 protein increased in HepG2/ADM cells treated with ABT-737 combined with NAC compared with HepG2/ADM cells treated with ABT-737 alone (Figure 6(e) and (f)). These results indicate that autophagy mediated by ROS can enhance HepG2/ADM cells apoptosis induced by ABT-737.

ROS inhibitor NAC enhances HepG2/ADM cell apoptosis induced by ABT-737. HepG2/ADM cells were treated with 10 mmol/L of NAC for 30 min and then incubated in 10 µmol/L ABT-737 for 24 h. (a) Cell viability was detected with MTT assay. Data are presented as mean ± SD, n = 6. *p < 0.05 vs control group, #p < 0.05 vs ABT-737 group; (b) cell apoptosis was detected by flow cytometry; (c) TUNEL assay for detection of apoptosis in HepG2/ADM cells, (d) quantitative analysis of the number of TUNEL-positive cells in (c). Data are presented as mean ± SD, n = 3. *p < 0.05 vs control group, #p < 0.05 vs ABT-737 group; (e) western blot analysis of caspase 3 and cleaved caspase 3 expression and (f) quantitative analysis of cleaved caspase 3 protein level from (e). Data are presented as mean ± SD, n = 3. *p < 0.05 vs control group, #p < 0.05 vs ABT-737 group.
Inhibition of autophagy increases Adriamycin-resistant human HCC HepG2/ADM cells apoptosis induced by ABT-737
To examine the effect of autophagy on cell apoptosis induced by ABT-737, we used the autophagy inhibitor 3-MA, which inhibits the initiation of autophagy. MTT results show that 3-MA increased the inhibition of cell viability induced by ABT-737 in HepG2/ADM cells (Figure 7(a)). To further confirm the effect of 3-MA on HepG2/ADM cell apoptosis induced by two different concentrations (10 and 25 µmol/L) of ABT-737. The PI-Annexin V double dye assay showed that 3-MA can increase cell apoptosis rate in HepG2/ADM cells treated with ABT-737 (Figure 7(b) and (c)). In addition, we observed that 3-MA decreased the expression of autophagy-related proteins LC3-II and Beclin1, and at the same time increases the expression of apoptotic-related cleaved caspase 3 protein (Figure 7(d) and (e)). The above results show that inhibition of autophagy can increase cell apoptosis induced by ABT-737 in HepG2/ADM cells.
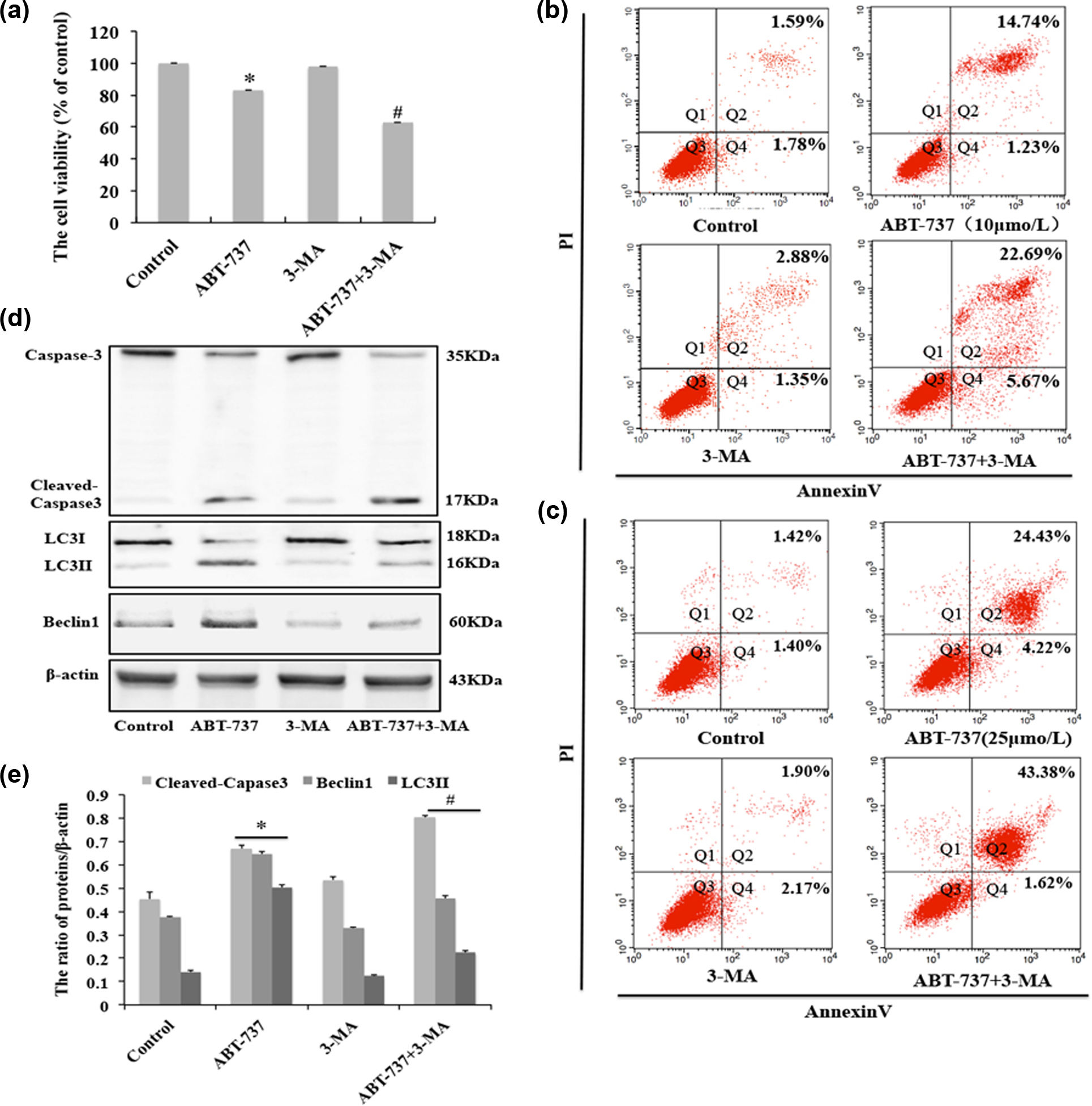
Inhibition of autophagy enhances HepG2/ADM cells apoptosis induced by ABT-737. HepG2/ADM cells were treated with 5 mmol/L 3-MA for 30 min and then incubated in 10 µmol/L ABT-737 for 24 h. (a) Cell viability was detected with MTT assay. Data are presented as mean ± SD, n = 6. *p < 0.05 vs control group, #p < 0.05 vs ABT-737 group; (b) and (c) cell apoptosis was detected by flow cytometry; (d) western blot analysis for the expression of caspase 3, cleaved caspase 3, LC3-I/LC3-II, and Beclin1; and (e) quantitative analysis of cleaved caspase 3, LC3-I/LC3-II, and Beclin1 protein levels from (d). Data are presented as mean ± SD, n = 3; *p < 0.05 vs control group, #p < 0.05 vs ABT-737 group.
Discussion
In this study, we demonstrated for the first time that the BH3 mimetic ABT-737 induced autophagy via Beclin1- and ROS-dependent pathways in HepG2/ADM cells. In addition, our results show that both autophagy inhibitor 3-MA and ROS inhibitor NAC enhanced cell death induced by ABT-737 in HepG2/ADM cells. Therefore, we further investigated the crucial role of autophagy in therapeutic strategies for HCC resistance.
Several studies showed that Bcl-2 protein has dual functions in cell apoptosis and cell autophagy.28–30 ABT-737, a BH3 mimetic of BH3-only protein Bad, not only induces cell apoptosis by suppressing the interaction of anti-apoptotic protein Bcl-2 and apoptotic protein Bax but also induces autophagy by promoting the release of autophagy initiation protein Beclin1 from Bcl-2. 28 Some studies have reported that ABT-737 induces apoptosis in different cancers, including small-cell lung carcinoma (SCLC), breast cancer, and several lymphoid malignancies.16,31 Here, we have also found that ABT-737 induced mitochondria-mediated apoptosis by inhibiting the interaction of Bcl-2 and Bax in HepG2/ADM cells in a time- and dose-dependent manner. In addition, we wanted to confirm whether ABT-737 induced autophagy and whether this autophagy has an effect on cell apoptosis induced by ABT-737 in HepG2/ADM cells.
Here, we also studied the effect of ABT-737 on autophagy in HepG2/ADM cells. We observed that ABT-737 induced Beclin1-dependent autophagy in a time- and dose-dependent manner in HepG2/ADM cells. Recent studies have reported that BH3 mimetics can induce diverse pro-autophagic pathways, except Beclin1-mediated autophagy, 27 and both ApoG2 and ABT-737 can trigger ROS-mediated autophagy in HCC cells.23,24 Therefore, we were interested in finding whether ABT-737 can induce ROS-mediated autophagy in HepG2/ADM cells. As expected, ABT-737 induced the production of ROS in a dose-dependent manner in HepG2/ADM cells. Furthermore, we demonstrated that the ROS inhibitor NAC decreased autophagy induced by ABT-737 in HepG2/ADM cells. These results suggest that ABT-737 induced Beclin1- and ROS-mediated autophagy in HepG2/ADM cells.
As we mentioned before, autophagy plays a very important role in the entire process of cancer development. Furthermore, Yanxin Chang et al. 32 have reported that autophagy is activated under starvation condition and chemotherapy, with the overexpression of autophagy-related proteins Beclin1 and ATG5. In addition, we show that autophagy suppressed by an autophagy inhibitor or knockdown of autophagy-related gene obviously increased HCC cell apoptosis. These results indicate that autophagy has a protective function under some conditions in HCC. Therefore, in our study, we further detected the effect of autophagy induced by ABT-737 on cell apoptosis in HepG2/ADM cells. As shown, ABT-737 induced ROS production, and ROS triggered autophagy in HepG2/ADM cells. Several papers reported that ROS plays dual functions in apoptosis and autophagy. 33 Furthermore, it has been demonstrated that low levels of ROS induce autophagy and high levels of ROS result in apoptosis. 33 In our study, we found that the inhibition of ROS increased cell death induced by ABT-737 in HepG2/ADM cells. This result demonstrates that ABT-737 induced low levels of ROS and protective autophagy in HepG2/ADM cells, which is consistent with a previous study in HCC cells with high levels of Bcl-2. 24 This may be attributed to the fact that the expression levels of Bcl-2 in HepG2/ADM cells were higher than those in HepG2 cells, as shown in our previous study. In addition, we found that the inhibition of autophagy increased ABT-737-induced apoptosis in HepG2/ADM cells. Both these results show that Beclin1- and ROS-mediated autophagy has a protective role against ABT-737-induced apoptosis in HepG2/ADM cells.
Collectively, our results confirm that the Bcl-2 inhibitor ABT-737 induced protective autophagy mediated by Beclin1- and ROS- dependent pathways in HepG2/ADM cells, which show higher expression levels of Bcl-2 than those in HepG2 cells. Furthermore, autophagy inhibitors increased the sensitivity of Bcl-2 inhibitors on HCC cells with high Bcl-2 expression levels. Our study further demonstrates that autophagy will be an effective target for chemotherapy in tumors with high Bcl-2 expression.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
