Abstract
Paclitaxel (Tx) is one of the first-line chemotherapeutic drugs used against lung cancer, but acquired resistance to this drug is a major challenge against successful chemotherapy. In this work, we have focused on the chronological changes of various cellular parameters and associated effect on Tx (10 nM) resistance development in A549 cell line. It was observed, at initial stage, the cell death percentage due to drug treatment had increased up to 20 days, and thereafter, it started declining and became completely resistant by 40 days. Expressions of βIII tubulin and drug efflux pumps also increased over the period of resistance development. Changes in cellular autophagy and reactive oxygen species generation showed a biphasic pattern and increased gradually over the course of upto 20 days, thereafter declined gradually; however, their levels remained higher than untreated cells when resistance was acquired. Increase in extracellular acidification rates and oxygen consumption rates was found to be directly correlated with acquisition of resistance. The depolarisation of mitochondrial membrane potential was also biphasic; first, it increased with increase of cell death up to 20 days, thereafter, it gradually decreased to normal level along with resistance development. Increase in activity of catalase, glutathione peroxidase and glutathione content over these periods may attribute in bringing down the reactive oxygen species levels and normalisation of mitochondrial membrane potential in spite of comparatively higher reactive oxygen species production by the Tx-resistant cells.
Keywords
Introduction
The major challenge in front of cancer therapy is the development of chemoresistance. The various factors responsible for development of chemoresistance include increased expression of drug efflux pumps [multiple drug resistance (MDR1), multidrug resistance protein (MRP1), etc.], 1 tubulin mutation and increased expression of βIII tubulin isomer 2 (for tubulin targeting drugs), increased ability of the cells to survive under stressed conditions by becoming insensitive to death signals and enhanced autophagy and so on.
Lung cancer is the most common cause of cancer-related death in men and women worldwide (causing 19.4% of all cancer-related death). Chemotherapy is the most widely used method of treating lung cancer followed by radiotherapy and surgery (usually for early stages). Among different chemotherapeutic drugs, paclitaxel (Tx) is most commonly used drug for the treatment of lung cancer. Binding of Tx at high concentration to tubulin polymer leads to stabilisation of spindle microtubule in vivo and prevents its disassembly, thus blocking the cell in G2/M phase leading to apoptosis. 3 Whereas Tx at low concentration stabilises the interphase microtubule of cell causing cell-cycle arrest at G0/G1 phase which may not cause cell death due to activation of repair mechanism. 4 Due to insufficient vascularisation, supply of food, oxygen as well as drug dosages in the median zone of cancer tissue is very limited; this inadequate supply of drugs leads to development of resistance in cancer. 5 Thus, acquisition of chemoresistance is one of the major obstacles for successful long-term chemotherapy. Most of the previous studies reported earlier were based after the development of Tx resistance;6–8 however, in this study, we have investigated the initiation of drug resistance development in chronological manner.
Among the different factors responsible for chemoresistance development, autophagy plays a very interesting dual role, depending upon cancer progression stage. 9 In early phase, autophagy acts as tumour suppressor, 10 while in later stage, autophagy helps cancer cells to survive under various stressed conditions including chemotherapy. 11 Thus, understanding the behavioural role of autophagy in process of chemoresistance development against Tx is essential. Other important factors that aid in Tx resistance development are over-expression of drug efflux pumps (like MDR1, MRP1),12,13 which actively pumps out the drug molecules from the cells there by lowering the affectivity of the drug attributing in Tx resistance. In addition, over-expression of βIII tubulin 14 also contributes in Tx resistance development in lung cancer. 15 Cancer cells are well known for their ‘metabolic shift’ as they show increase in glycolytic rate in hypoxic condition to match the ATP demand, 16 and metabolic shift is the key factor in the development of resistance phenomenon; this phenomenon is reported to play very important role in the development of chemoresistance, 17 although nowadays this theory has been reported to be doubtful as there is another school of thought that suggests that not all types of tumour depend completely on glycolysis for energy production and some may rely on oxidative phosphorylation equally or predominantly depending on condition.18,19 Intercellular reactive oxygen species (ROS) level is a key determining factor for free radical activity of the cells as well as resistance development. Free radical scavenging activity is determined by level of cellular antioxidant activities. 20 However, there are no such reports showing chronological changes in cellular antioxidant activities during the process of Tx resistance development. The number of studies has been reported on end point characterisation of Tx resistance in several cultured cancer cell lines.21,22 But there are no reports of time course study for change in various cellular parameters like ROS formation, autophagy, cell-cycle changes, mitochondrial membrane potential (MMP) changes and energy production rates of the cells during development of resistance. Understanding this phenomenon will possibly lead us to a new window of successful Tx treatment. In this project, we have studied time responsive changes in various cellular parameters like autophagy, cellular morphology, cell-cycle phase distribution, ROS generation, MMP and various antioxidant enzyme’s activity in the course of Tx (10 nM) resistance development in A549 cells. Changes in bio-energetic parameters like oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) during this period were also studied. Relatively low dose of Tx (10 nM) was used for the understanding of the study because it has been reported that low dose of Tx can show tumour control with lesser toxicity to normal cells.23,24 Moreover, due to poor vascularisation, various portions of cancer tissue are known to develop resistance phenomenon for low exposure to drugs; therefore, low drug dose is physiologically more significant than higher doses. At early stage, cancer cells showed sensitivity to exposure with low dose Tx (10 nM) and thereafter those gradually became resistant to it over a period of 40 days. The alterations of different cellular parameters of A549 as mentioned earlier were analysed for 40 days of Tx treatment.
Materials and methods
Materials
Dulbecco’s Modified Eagle Medium (DMEM) nutrient media (supplemented with 1 mM l-glutamine), fetal bovine serum, penicillin–streptomycin mixture, amphotericin B and hydrocortisone were purchased from Hyclone (GE healthcare, India). Trypsin–ethylenediamine-tetraacetic acid (EDTA) was purchased from Hi Media, India. Tx, monodancylcadverine (MDC), Trypan blue, propidium iodide (PI), dimethyl sulfoxide (DMSO), 2′,7′–dichlorofluorescin diacetate (DCF-DA), and RNaseA were purchased from SIGMA (USA). JC-1 (5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazol-carbocyanineiodide) and MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) were obtained from Life Technologies (Thermo Fischer). Mouse monoclonal anti–β actin antibody (catalogue No.- sc-47778; clone-C4), mouse monoclonal Beclin 1 (catalogue No.-sc-48341; clone-E8), rabbit polyclonal anti-Bcl-2 antibody (catalogue No.-sc-492; clone-N-19), mouse monoclonal anti-MRP1 antibody (catalogue No.-sc-7773; clone-E20) and rabbit monoclonal anti-MDR-1 antibody (catalogue No.-sc-390883; clone-10) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Rabbit monoclonal cleaved caspase-3 antibody (catalogue No.-9661; clone-Asp-175), rabbit monoclonal anti-LC-3 antibody (catalogue No.-3868; clone-D11) and rabbit monoclonal anti-βIII antibody (catalogue No.-4466; clone-TU-20) were purchased from Cell Signalling. Mouse monoclonal anti-BAX antibody (catalogue No.-B3428) was obtained from Sigma Aldrich. Caspase 3 activity assay kit was obtained from Cell Signalling. The Bradford protein estimation kit, goat anti-mouse IgG-horseradish peroxidase (HRP), rabbit anti-goat IgG-HRP and goat anti-rabbit IgG-HRP were purchased from Genei (India). All the other reagents and chemicals were analytical grade and were purchased from Sisco Research Laboratories (India). Human non-small lung epithelium cell line (A549) was obtained from cell repository of National Centre for Cell Science (NCCS, Pune, India).
Cell culture and maintenance
Human lung adenocarcinoma cells (A549) were cultured in DMEM supplemented with 1 mM
Cell treatment
The A549 cells were seeded and cultured till they are about 40%–50% confluent. The cells were treated with 10 nM Tx for 48 h, then the media were changed and the treatment was repeated. At 80% confluency, the cells were trypsinised and kept in same culture condition as before. Different cellular parameters of the living cells were monitored at every 48-h time point till complete resistance development.
Cell viability assay
Cultured cells were collected and re-suspended in 1 mL phosphate-buffered saline (PBS). Cells were stained with 0.05% (w/v) Trypan blue for 5 min and visualised under a haemocytometer. The total number of cells versus number of blue stained cells was counted and percentage of viable cells was calculated by using following formula 25
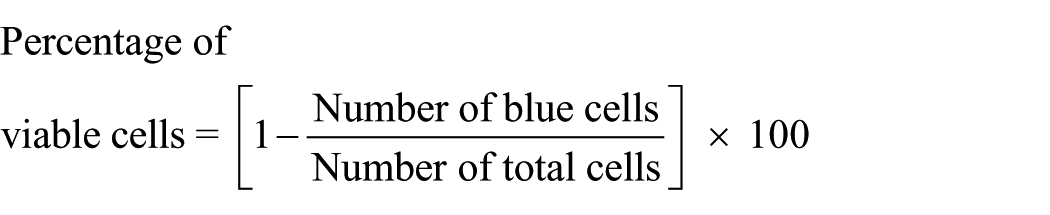
Cytotoxicity assay
Cytotoxicity induced in A549 cells due to Tx treatment was determined by MTT assay. Cells were seeded in 96-well flat bottom microtiter plates at a density of 1 × 104 cells/mL incubated for 24 h before 48-h drug treatment. After treatment, 10% (v/v) filter sterilised MTT solution (2 mg/mL) was added to each well and incubated in a humidified 5% CO2 incubator at 37°C for 4 h. Then 100 µL DMSO/well was added to dissolve the violet precipitate. The absorbances of the solutions were measured at 570 nm with a microtiter plate reader (Bio-Tek ELX800) and percentage of cell viability was calculated according to the following formula

where At and As indicated the absorbance of the test and solvent control, respectively. 26
Cell-cycle assay
After treatment as mentioned before, cells were fixed in chilled methanol (100%). Thereafter, cells were incubated with RNase solution (500 U/mL) at 37°C for 3 h. Nuclear DNA was stained using PI before flowcytometric cell-cycle analysis by BD FACScalibur. The data were analysed with Cell Questpro software (Becton Dickinson). 27
Detection of ROS generation
For detection of ROS generation, treated and control cells were taken at various time points and stained with DCFDA (10 µM) at 37°C for 15 min followed by flowcytometric analysis as before. 28 For microscopic study, cells were grown on cover slips as before and stained with DCFDA for 15 min, then the pictures were taken by Olympus fluorescence microscope.
Measurement of catalase activity
The experimental cells after collection were re-suspended in 200 µL of assay buffers. Cells were sonicated in ice to extract cellular proteins. For blank, 120 µL of sample buffer [50 mM potassium phosphate buffer (pH 7)] was mixed with 180 µL of 30 µM H2O2. For other samples, 20 µL cell extract was added to 100 µL of sample buffer and mixed with 180 µL of 30 µM H2O2. The decomposition of H2O2 was monitored at 240 nm for 4 min at room temperature in spectrophotometer (JASCO V-630). Three mutually independent experiments were done for each sample. Catalase activity of samples was expressed as number of µmol of H2O2 consumed per minute per milligram of protein. 29
Measurement of glutathione peroxidase activity
For each sample, 10 µL of cell extract (isolated as before) was added to 170 µL of potassium phosphate buffer with 1 mM EDTA and 2 mM sodium azide (pH 7). In blank, 10 µL of sample buffer was added instead of cell extract. To each sample, 90 µL master mix (composed of 30 µL (10 mM) glutathione (GSH), 30 µL (2.4 U/mL) glutathione reductase enzyme and 30 µL nicotinamide adenine dinucleotide phosphate (NADPH; 1.5 mM) dissolved in 0.1% sodium bicarbonate) was added, mixed well and incubated at 37°C for 10 min. To start the reaction, 30 µL (2 mM) H2O2 was added to each set before taking absorption. The consumption of NADPH was monitored at 340 nm at 37°C for 10 min in spectrophotometer (JASCO V-630). Three mutually independent experiments were done for each sample. NADPH (in nM) consumed per minute per milligram of protein was calculated for each sample. 30
Measurement of GSH content
The cells were processed and supernatant was stored as mentioned above. A reaction mixture was prepared with 0.84 mM DTNB (5,5′-dithiobis-(2-nitrobenzoic acid)) and 0.28 mM NADPH dissolved in 100 mM sodium phosphate buffer (pH 7.5) with 5 mM EDTA. For each sample, 200 µL of this above-mentioned reaction mixture was added with 20 µL of sample in a quartz cuvette (for blank, 20 µL of sample buffer was added instead of sample) and incubated at room temperature for 5 min, the reaction was started by adding 40 µL of glutathione reductase and the formation of TNB was monitored over 10 min using absorption spectrophotometer (JASCO V-630) at 412 nm. Standard curve for GSH was prepared using known concentration of GSH (0–50 µM) by the same technique; 31 with the aid of this, the GSH content of the experimental samples was determined.
Detection of autophagy using MDC by flowcytometry and fluorescence microscopy
Treated and control cells were stained with 50 µM of MDC (a marker for autophagic vacuoles) at 37°C for 15 min. 32 After washing three times with PBS, data for autophagic vacuole formation were collected using BD FACScalibur. Data were processed and analysed using Cell Questpro software (Becton Dickinson). For imaging, the cells were seeded on coverslips and treated as before. Cells were processed similarly with MDC and washed three times before collecting data using Olympus fluorescence microscope via 380 nM excitation filter. 33
Measurement of MMP
Change in MMP was determined by JC-1, a cationic lipophilic dye, which accumulates and aggregates forming orange cluster in active mitochondria. Monomeric JC-1 in cytosol shows green colour. Therefore, a shift is observed in red versus green fluorescence if any change in MMP takes place. 34 Experimental cells were stained with JC-1 (1 µg/mL) for 15 min at 37°C and subjected to flowcytometric analysis in BD FACScalibur instrument. 35
Caspase 3 activity assay
Equal amount of protein containing cell lysates was dissolved in 1× activity assay buffer. Then 25 µL of sample (lysate) was mixed with 200 µL of substrate (solution B). The samples were incubated at 37°C for 1 h in dark. Thereafter, their fluorescence excitation maximum at 460 nm was measured using excitation wavelength at 380 nm. For each sample, three independent readings were taken, then the average fluorescence of the samples at 460 nm was plotted. 36
Western blot analysis
Total protein extraction of experimental cells was done using radioimmunoprecipitation assay (RIPA) buffer. Western blot for different cellular pathways like apoptosis or autophagy regulatory proteins (p62, Bcl-2, BAX, LC3II, β-actin, cleaved caspase 3, and Beclin I), drug efflux pump proteins (MDR1, MRP1) and βIII tubulin protein of untreated and treated cells was performed. Primary antibody dilutions were done following the manufacturer’s instructions. For loading control, β-actin was used. 37 The protein concentrations were measured by Bradford’s method. 38
ECAR and OCR measurements
The OCR (indicator of mitochondrial respiration) and ECAR (indicator of proton loss due to glycolysis) of A549 cells at three different time points of Tx treatment (0, 20 and 40 days) were measured by Seahorse XFe24 Extracellular Flux Analyser. The cells were evenly seeded (2.5 × 104 cells per well) in XFe24 cell culture plate and allowed to attach for 24 h. Before beginning the experiment, the cell culture media were replaced with XF base media (seahorse, bioscience) and were kept in a non-CO2 incubator at 37°C. Three basal measurements of ECAR and OCR were taken before injection of pharmacological manipulators of mitochondrial respiratory chain proteins (used for measurement of several bioenergetic parameters). After recording basal OCR and ECAR readings, 1 µM of oligomycin A (oligoA; Sigma) was injected to each sample to inhibit the proton flow through ATP synthase and blocking of all ATP-linked oxygen consumption. Thereafter, 750 nM of carbonyl cyanide-
Statistical analysis
All the data are presented as the mean of at least three independent experiments along with standard error of the mean (SEM). Statistical analysis of data was done by using Student’s t test in MS Excel and the measurements with the p value ⩽0.05 were considered to be statistically significant.
Results
Chronological changes in cell viability and expressions of resistance markers in course of Tx resistance development
Human lung non-small adenocarcinoma (A549) cells were treated with 10 nM Tx as described before, and the changes in cell viability with time were measured using Trypan blue assay (after every 48 h of treatment until development of resistance). Initially, the cell death percentage was observed to increase after every 48 h; this pattern remained the same until about 20 days of treatment (cell death percentage was 20 ± 0.55%). Thereafter, percentage of cell death was observed to decline even after increase in duration of treatment; at 30 days of treatment, the death percentage declined (10 ± 0.41%) and which even further decreased and reached to a level of control by 40 days of Tx (10 nM) treatment (Figure 1(a)). It remained like that even on more prolonged treatment, 100 days (data not shown). Therefore, acquisition of complete resistance was confirmed on 40 days of treatment. Further reconfirmation was done using MTT assay on these cells. The overall drug response pattern found to be changed in the 10 nM Tx-resistant cells. The IC50 value for 48-h Tx treatment increased to 71 nM in case of 10 nM resistance cells (TxR10) in comparison to that of control A549 cells (whose IC50 was 48 nM; Figure 1(b)). Moreover, gradual increase of expression of βIII tubulin, a marker for Tx resistance in lung cancer cells,40,41 as well as MDR1 and MRP1 expressions were observed in course of resistance development (Figure 1(c)).

Time-dependent changes in cell death of A549 cells treated with paclitaxel (10 nM) for different time periods measured by trypan blue exclusion assay: (a) 48-h MTT assay of A549 cells (grey) and cells treated with 10 nM taxol for 40 days (TxR10; dark grey), (b) data represented as the mean ± SD (*p < 0. 05. #p < 0.05 vs control, n = 3), (c) immunoblot assay with βIII tubulin (at three different time point of Tx treatment – 0, 20, 40 days), MRP1, and MDR1 antibodies (at five different points of treatment – 0, 10, 20, 30, 40 days, respectively), β-actin was used as loading control and (d) cell-cycle distribution pattern at five different points (0, 10, 20, 30, 40 days) of treatment in the course of resistance development.
Time responsive biphasic changes in cell-cycle phase distributions in course of Tx resistance development
Cell-cycle phase distribution was monitored after every 48 h during the course of Tx resistance development (till 40 days). Gradually, a steady increase in G0/G1 cell population was observed in response to Tx treatment. And between 20 and 30 days, it reached to maximum of about 87.95 ± 1.23% which is close to 35% higher than that of control A549 cells. But later on, this anomaly also declined and became almost similar as control upon development of complete resistance (Figure 1(d) and Figure S2, Table 1).
Chronological variations in ROS generation, autophagy, MMP loss and cell-cycle phase distribution in A549 cells in the course of 10 nM paclitaxel resistance development.

ROS: reactive oxygen species; DCFDA: 2′,7′–dichlorofluorescin diacetate; MDC: monodancylcadverine; MMP: mitochondrial membrane potential.
Biphasic changes in intracellular ROS levels
ROS is key regulatory factor for number of cellular activities including autophagy, cell-cycle phase distribution, and cellular metabolic activity. Cellular ROS levels were monitored by flowcytometry using DCFDA. For each sample, the mean fluorescence intensity (percentage of MFI) was measured. At initial phase, cellular ROS gradually increased and reached to a maximum of about 10 times more (between 20 and 24 days) than control (untreated) cells. Thereafter, the MFI declined a bit and became stable, once complete resistance developed (at 40 days), although at this point, the DCFDA MFI was found to be significantly lower than cells treated with Tx for 20 days (p < 0.01), but the ROS levels remained higher than the control cells. Similar trends were also observed in fluorescence microscopic study using DCFDA (Table 1 and Figure 2(a) and (b)). For positive control, 0.01% (v/v) H2O2 treatment was done for 6 h before cell processing.

Fluorescence microscope images of DCFDA fluorescence in A549 cells at various time points of Tx treatment (0, 10, 20, 30 and 40 days) in course of resistance development, cells treated with 1 µM hydrogen peroxide (H2O2) were used as positive control for ROS generation: (a) time-dependent changes in mean DCFDA fluorescence in A549 cells treated with paclitaxel in the course of resistance development measured by flowcytometry (taken after every 48 h) and (b) all data are represented as mean ± SD (*p < 0.05, **p < 0.01 vs control n = 3, ##p < 0.01 vs Tx treated cells for 40 days).
Changes in cellular catalase, glutathione peroxidase activities and GSH content of the cells in course of Tx resistance development
The relative specific activities of catalase, glutathione peroxidase (GPx) as well as GSH content in respect to control cells were monitored in course of Tx resistance development; it was observed that the activities of all three antioxidants increased gradually with development of Tx resistance. Initially after 4 days of treatment, the catalase, GPx activity and GSH content of the cells did not show much change in respect to untreated cells; however, after 8 days of treatment, catalase activity increased to 1.6 times in respect to untreated cells although GSH content and GPx activity were found to increase marginally only about 1.14 and 1.23 times of control, respectively; at 12 days after treatment, catalase activity increased to 1.75 times while GSH content and GPx activity were 1.18 and 1.38 times of control, respectively; however, after 24 days of treatment, all three antioxidant levels showed a much significant increase; the catalase and GPx activity as well as GSH content of the cells were found to be about 2.08, 1.7 and 2.02 times of control, respectively, which further increased to 2.83 times (catalase), 2.62 times (GSH) and 1.97 times (GPx) of control cells, after 32 days of treatment and at 40 days when the cells were found to be completely resistant to 10 nM Tx, the GSH content as well as catalase and GPx activity increased further to about 3.03, 3.67 and 2.2 times of control cells, respectively (Figure 3(a)–(c)).

Changes in relative specific activity of (a) catalase, (b) GPx and (c) GSH content in A549 cells, at different time points of Tx (10 nM) treatment in the course of development of resistance in these cells. Data represented as mean ± SD (*p < 0.05, **p < 0.01 vs control, n = 3).
Biphasic changes in cellular autophagy in response to Tx treatment during resistance development
As a measure of autophagy, fluorescence of MDC positive vacuoles of the cells was monitored by flowcytometry and fluorescence microscopy. Serum starved cells (SF-serum free) were used as positive control for autophagic vacuole formation. With progression of study, the number of autophagy positive cells increased in correlation with increase of cell stress percentage. At 2 days of treatment, autophagy vacuole positive cell percentage was about 4.39 ± 0.52%. The cell population, positive to autophagic vacuole, increased to about 10.2 ± 0.35% at 4 days, and this increment of autophagy continued up to 20–22 days and reached up to a maximum of about 31.07 ± 1.63%. Thereafter, the autophagy positive cell population declined slowly, and on 30 days, the percentage cell population positive for MDC staining was found to be about 28.83 ± 1.23%. This trend continued till acquisition of complete resistance (40 days) of Tx (10 nM) treatment with a MDC positive population of 24.19 ± 1.2% (Table 1 and Figure S1), which was found to be significantly lower than the cells treated with Tx for 20 days (p < 0.01). However, no further significant decrease of MDC positive cell populations were found even on 100 days of treatment, and the population remained steady at about 25% (data not shown). Further confirmation of the flowcytometric data was done by fluorescence microscopic study (Figure 4(a)). Western blot of different autophagy regulatory proteins such as LC3, Beclin1 and p62 was also monitored; increase in expressions of Beclin 1 is positively correlated with formation of autophagic vacuoles in cells. The level of Beclin 1 in response to Tx (10 nM) treatment increased, as a result, cellular autophagy level also went up till 20–30 days of treatment. After 30 days, the protein level started declining and as a result autophagy level also declined. But the level remained noticeably higher than the untreated cell, even after acquisition of complete resistance. This observation was further supported by an increase in LC-3II (a marker of autophagosome formation) level. In untreated cells, the level of LC-3II was found to be very low. But gradually, the level went up till 20 days (where maximum autophagy was observed by MDC staining) and thereafter it declined as autophagy level declined. In response to Tx treatment, the level of LC-3I which is the precursor of LC-3II also went up in lung cancer cells. Noticeable decay of p62 level was found at the first phase of treatment which then decreased upon acquisition of resistance (Figure 4(b)). In the positive control sample, upregulation of autophagy was observed in response to serum starvation. Upregulation of autophagy in cancer cell in low Tx treatment is a trademark of stress response. 42 In Tx-resistant cancer cells, autophagy plays critical cytoprotective role in compensating drug-induced stress condition. 43 An enhanced steady autophagic vacuole level had been observed in the resistant cells. This observation goes with previous studies where researchers have found a steady upregulation of autophagy in chemoresistance cells/tissues. However, the phenomenon that was unknown was this pattern of biphasic change in autophagy during Tx resistance development, where first it showed a steady increase and thereafter declined and was stabilised at a higher level than control cells.

Time-dependent changes in autophagy due to Tx (10 nM) treatment visualised via fluorescence microscopy by staining autophagic vacuoles with MDC dye at five different points of treatment in course of 10 nM Tx resistance development: (a) western blot assay showing variation in expression of different proteins associated with autophagy like Beclin 1, LC3, and p62 with time during the course of 10 nM Tx resistance formation in A549 cells, β-actin used as loading control, (b) time-dependent changes in mitochondrial membrane potential in A549 cells at five different points of treatment, in the course of Tx resistance development, measured by flowcytometry using JC-1 dye, (c) immunoblot assay with different pro(BAX, cleaved caspase 3) and anti-apoptotic (Bcl-2) proteins, at five different time points, in course of resistance development, (d) Bar diagram showing caspase3 activity of A549 cells at five different points of Tx treatment in course of resistance development and (e) data represented as mean ± SD (*p < 0.05 vs control, n = 3).
Bi-phasic alteration of MMP during Tx resistance development
Cellular mitochondrial activity is a direct measure of cellular health. JC-1 directly shows activity of cellular mitochondria by showing change in colour. Tx treatment activates mitochondrial apoptosis cascade. At the initial phase, treatment cell death due to Tx treatment happened due to loss of MMP. Close to 20 days treatment, a maximum loss of 7.59 ± 0.12% MMP was observed. Thereafter, the percentage of depolarised mitochondria started to decline at and on 40 days (complete resistance); unlike ROS level, the percentage of depolarised mitochondria were found to be 0.4 ± 0.031% which was almost near to Tx untreated cells (Figure 4(c), Table 1, Figure S3).
Time-dependent changes in expressions of pro and anti-apoptotic proteins in the course of Tx resistance development in A549 cells
Western blot assay with various pro(BAX, cleaved caspase 3) and anti-apoptotic (Bcl-2) proteins was performed with the cell lysates obtained from various points (0–40 days) of Tx treatment in course of resistance development. Initially, the expression of BAX and cleaved caspase 3 was seen to increase with increase in duration of treatment; it was found to be maximum at 20 days, thereafter their expression reduced, and at 40 days, it was near to control cells. In case of Bcl-2, just the opposite pattern was observed; its expression gradually decreased up to 20 days after which it increased and became near to normal at 40 days of treatment (Figure 4(d)).
Biphasic changes of caspase 3 activity in course of Tx resistance development
The changes in caspase 3 activity also followed a biphasic pattern like MMP. It was observed that caspase 3 activity increased initially at early stage of Tx treatment (till 20 days) and thereafter it declined, and at 40 days of treatment, its activity was almost equal to untreated cells, suggesting resistance development (Figure 4(e)).
Alterations of cellular OCR in course of Tx resistance development
OCR of a cell is directly proportional to the mitochondrial activity of the cells. It gives a direct proof of cellular metabolic rate and of cellular health. The OCR in A549 cells was measured at three different time points (0, 20 and 40 days) of Tx (10 nM) treatment using XFe24 Extracellular Flux Analyser (Figure 5(a)). It was observed that Tx treatment causes increase in cellular OCR level. In control cells, basal mitochondrial OCR level was significantly lower when compared to both 20 and 40 days Tx treated cells (TxR10; Figure 5(c)); thus, most of the basal mitochondrial OCR can be linked to ATP production as seen by injecting the mitochondrial electron transport chain (ETC) inhibitor oligomycin A, while the remaining can be attributed to proton leakage across membrane which followed similar pattern as mitochondrial OCR (Figure 5(d)). The injection of FCCP caused an increase in OCR to maximum in all three samples; however, it was found to be much more in resistant cells (40 days treated; Figure 5(e)), which also showed a higher respiratory reserve capacity than untreated cells (sensitive; Figure 5(f)).

Oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) of A549 cells at three different time points of treatment (0 day (•), 20 days (◾), 40 days (▴)) were measured using Seahorse Extracellular Flux Analyser (XFe24), (Seahorse Bioscience) (a) and (b) respectively Mitochondrial stress test was used to find out different bioenergetic parameters, (c) basal mitochondrial OCR, (d) OCR due to proton leakage, (e) maximum mitochondrial OCR, (f) respiratory reserve, (g) basal ECAR, (h) maximal ECAR and (i) glycolytic capacities of the cells. The data are represented as mean ± SD (*p < 0.05 vs control, n = 3).
Changes in ECAR in course of Tx resistance development
The resistant cells also showed a higher basal ECAR (an indirect measurement of glycolysis) than sensitive cancer cells (0 day; Figure 5(b) and (g)). Moreover, the maximal ECAR (simulated by oligomycin A injection, which shuts down ATP-linked OCR) was also seen to be much higher in resistant cells (Figure 5(h)). In addition, the resistant cells also showed a markedly higher glycolytic reserve capacity (obtained by subtracting basal ECAR from maximal ECAR) than untreated cells (0 day; Figure 5(i)), suggesting that the capacity for glycolysis in these resistant cells are much higher than untreated A549 cells. In order to find the bioenergetic profile of these cells, we plotted the basal ECAR against mitochondrial OCR of the cells, where we observed that the resistant cells (40 days treated (TxR10)) fall in the category of highly energetic cells having both high respiratory and glycolytic rates; the cells treated for 20 days showed intermediate phenotype, although their glycolytic and mitochondrial respiratory capacities were found to be much higher than untreated A549 cells, which were observed to be least energetic cells having both lower glycolytic and respiratory rates (Figure S4).
Discussion
Lung cancer is the second most common cancer in men and women and has been accounted as the leading cause of cancer death worldwide in both men and women. Moreover, among different forms of lung cancer, non-small cell lung cancer (NSCLC) is the most common form; it has been known to account for about 85% of all newly diagnosed cases. 44 Among different chemotherapeutic drugs used for the treatment of NSCLC, Tx is one of the most commonly used drug, but one of the major challenges for successful chemotherapy with this drug is that despite early response to this drug, most of the patients eventually develop resistance to the drug in course of treatment. The reasons behind development of Tx resistance are many which include changes in expressions of the target molecule, tubulin (βIII tubulin over expression), 22 increase in expressions of drug efflux pumps like MDR-1, changes in expressions of proteins involved in regulation of apoptotic pathways and mitotic checkpoint proteins, 45 and increased ability of the cancer cells to survive under stressed condition with the help of autophagy. 11 There are various studies of characterisation of Tx-resistant cells in different cancer cell lines;6,46 however, detail time point study of changes in cancer cells in the course of Tx resistance development has not been reported. Since such time point studies of resistance development in patients and animals are out of scope, therefore, understanding in detail the changes that occur in lung cancer cells with time in the way of development of resistance against Tx could help us in better management of the disease and also aid in prevention of resistance development against Tx.
In this study, we aimed to find out the various time-dependent changes that occur in lung cancer cells in the course of Tx (10 nM) resistance development. We have treated the lung cancer cells (A549) with a low Tx dose (10 nM) and have measured changes in different cellular parameters with time in the course of resistance development. The cells response to the drug differed with difference in duration of treatment. Initially, the cells responded well to the treatment and showed a gradual increase in cell death (measured by Trypan blue assay) with increase in duration of treatment; this pattern was observed up to 20 days of treatment when the death percentage was maximum; thereafter, the percentage of cell death declined with increase in duration, indicating decrease in sensitivity of the cells towards the drug; at 40 days, the cells were found to be completely resistant to this dose of Tx, and this was confirmed by MTT assay; moreover, the cells at this time showed a higher IC50 value than untreated A549 cells, indicating decrease in sensitivity towards Tx (Figure 1(a) and (b)). Western blot analysis with some resistance markers like βIII tubulin, MDR1 and MRP1 also showed an increase in its expression at this time (Figure 1(c)) which is usually observed in Tx-resistant cells.12,40 The cell-cycle distribution phases of the A549 cells at different point of 10 nM Tx treatment initially showed a gradual increase in population percentage in G0/G1 phase; this may be explained by some previous reports which explain an increase in G0/G1 phase of cell cycle with increase in autophagy; 47 moreover, it has been also reported that increased stability of cells in G0/G1 phase of cell cycle increases stability of p-glycoprotein (pgp) thus aiding in resistance development; 48 moreover, earlier reports also suggest that low concentration (5–10 nM) of Tx causes cell-cycle arrest at sub G0 or G0/G1 phase unlike higher concentrations that causes G2/M phase arrest. 49 Although this anomaly in cell-cycle distribution pattern was normalised at 40 days of treatment suggesting resistance development (Figure 1(d) and Figure S2, Table 1), it has been reported in many studies that chemotherapy induces ROS formation in cancer cells which triggers autophagy; 50 in this study, treatment of the A549 cells with Tx initially induced ROS generation that triggered autophagy. The ROS levels increased with increase in duration of treatment up to 22 days; this may be due to therapeutic stress caused by Tx treatment. 51 Thereafter, the ROS levels started declining, and at 40 days, the ROS levels were found to be at an intermediate level between untreated cells and maximum level (22 days); the fluorescence microscopic images with DCFDA also showed similar results (Figure 2(a) and Table 1); this gradual decline in ROS levels may be attributed to increase in autophagy (Table 1, Figure 4(a) and (b)) and increase in antioxidant activities (Figure 3(a)–(c)) of the cells, although the ROS levels in the resistant cells were observed to be much higher than control cells; the reasons for this will be discussed later. During this course of resistance development, a gradual increase in autophagy was observed upto 20 days; thereafter, the autophagy percentage dipped marginally and when resistance was achieved at 40 days, the autophagy percentage was found to be much higher than untreated cells (Table 1, Figure 4(a) and (b) and Figure S1). This phenomenon can be explained by the fact that autophagy has long been known to play cytoprotective role in cancer cells when subjected to chemotherapeutic treatment; 52 thus, it can be said that treatment of the A549 cells with Tx triggered increase in autophagy in the cells as a refuge from the chemotherapeutic stress initially, but later in next phase, when the other stress relieving factors like antioxidant capacities and drug efflux pump activities of the cells got elevated, autophagy levels declined (as excessive autophagy may cause damage to the cells itself); however, the levels of autophagy in resistant cells were found to be much more higher than untreated cells as it acted continuously as a stress removal factor in the Tx-resistant lung cancer cells; the detailed study of this is being performed in our future work. The depolarisation of MMP followed a different pattern; initially the percentage of depolarised mitochondria increased with increase in duration of the drug treatment up to 20 days, thereafter the percentage of depolarised mitochondria declined, and after 40 days of treatment, the MMP of the cells was found to be almost near to untreated cells with very little percentage of depolarised mitochondria (Table 1, Figure 4(c) and Figure S3). Immunoblot assay showed a gradual increase in pro apoptotic proteins like BAX and cleaved caspase 3 with decline of anti-apoptotic Bcl-2 along with increase in caspase 3 activity until 20 days of treatment, which could be triggered due to increase in depolarisation of mitochondrial membrane during this period; however, after this with decrease in depolarisation of mitochondrial membrane, the amount of pro apoptotic proteins (BAX, cleaved caspase 3) and caspase 3 activity declined with increase in anti-apoptotic protein Bcl-2, indicating resistance development, and their levels were almost similar to untreated cells after 40 days of Tx treatment indicating complete resistance development (Figure 4(d) and (e)). The stress induced by the drug treatment caused depolarisation of mitochondrial membrane, triggering ROS generation which in turn triggered autophagy, which helped to overcome the chemotherapeutic stress. Although the ROS levels were observed to decline after 20 days of treatment, when the percentage of depolarised mitochondria started declining, its level was still found to be much higher than untreated cells, this can be explained by the fact that apart from depolarisation of MMP, we also observed an increase in basal and maximal ECAR levels (an indicator of glycolysis) of the cells when resistance was achieved (Figure 5(b), (g) and (h)); the reason behind this could be the increase in energy demand of the cells during resistance development. These resistant cells also showed to have a higher glycolytic reserve capacity (Figure 5(i)) indicating much higher glycolytic capacity of resistant cells compared to untreated A549 cells. The higher ECAR levels in resistant cells may be a direct contributor of ROS in the cells; moreover, it has been shown in earlier reports that in many cancer cells, Tx-resistant cells show an increased ROS levels which may aid in resistance development.53,54 The resistant cells were also observed to show higher basal mitochondrial OCR levels (Figure 5(c)); moreover, the resistant cells also had higher maximal and respiratory reserve capacity compared to untreated A549 cells (0 day; Figure 5(e) and (f)). The bio-energetic profile (Figure S4) also showed that the resistant cells were highly energetic cells (having both high glycolytic and respiratory rates) as compared to untreated A549 cells which were found to be much less energetic; although this fact is slightly in contrast to the proposed Warburg effect, but presently many reports have shown that the Warburg effect is applicable mainly in hypoxic condition 55 in cancer cells, and nowadays many reports have suggested that the cancer cells may use both oxidative phosphorylation and glycolysis as predominant energy source, depending on condition. In situation of high energy demand, it may take refuge to high aerobic glycolysis in addition to oxidative phosphorylation,56,57 thus showing high metabolic activity and efficient utilisation of both energy pathways. However, another question that arises is that how these Tx-resistant cells (showing both increased rate of oxidative phosphorylation and glycolysis) could tolerate these higher levels of ROS; for this, relative specific activity of different cellular antioxidants like catalase, GPx and GSH content was measured, and it was observed as discussed earlier that with increase in duration of treatment, the relative specific activity of these antioxidants was observed to rise and was very high in cells treated with Tx for 40 days, that is, the resistant cells (Figure 3(a)–(c)), thus helping the cells to tolerate the moderately higher ROS levels; this correlates with earlier reports that resistance to Tx is positively correlated with cellular total antioxidant capacity, 58 although some more detail studies in this field would help us in better understanding of the various cellular factors that aid in development of resistance against this drug, aiding in prevention of chemoresistance development.
Thus, from this study, we could conclude that the development of Tx resistance in A549 cells was associated with a gradual biphasic change in autophagy, ROS production as well as mitochondrial membrane depolarisation pattern, with gradual upregulation of antioxidant activity levels in the cells, which helped the cells to tolerate the therapeutic stress of the drug. Increased autophagy and energy production with both upregulated OCR and ECAR (glycolysis) capacity also helped the cells to meet the extra energy demand of the resistant cells produced by the chemotherapeutic stress; as a result, the expressions of ATP-dependent drug efflux pumps (MDR1, MRP1) were also upregulated aiding in resistance development. Thus, this study could provide us a scenario of chronological alterations in various cellular parameters that take place in lung cancer cells in the course of Tx resistance development.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The work was supported by grants from Department of Science and Technology, Govt. of India (No. SR/SO/BB-14/2008) and Department of Biotechnology, Government of India (No. BT/PR12889/AGR/36/624/2009) to G. Chakbarati. FACS and fluorescence microscope instruments facility are developed by grants from National Common Minimum Project, Government of India. S Datta, D Das Mukherjee and N Das are supported by Senior Research fellowship from Council of Scientific and Industrial Research (CSIR), Government of India.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
