Abstract
The aspartate aminotransferase-to-platelet ratio index has been reported to predict prognosis of patients with hepatocellular carcinoma. This study examined the prognostic potential of stratified aspartate aminotransferase-to-platelet ratio index for hepatocellular carcinoma patients undergoing curative liver resection. A total of 661 hepatocellular carcinoma patients were retrieved and the associations between aspartate aminotransferase-to-platelet ratio index and clinicopathological variables and survivals (overall survival and disease-free survival) were analyzed. Higher aspartate aminotransferase-to-platelet ratio index quartiles were significantly associated with poorer overall survival (p = 0.002) and disease-free survival (p = 0.001). Multivariate analysis showed aspartate aminotransferase-to-platelet ratio index to be an independent risk factor for overall survival (p = 0.018) and disease-free survival (p = 0.01).
Patients in the highest aspartate aminotransferase-to-platelet ratio index quartile were at 44% greater risk of death than patients in the first quartile (hazard ratio = 1.445, 95% confidence interval = 1.081 – 1.931, p = 0.013), as well as 49% greater risk of recurrence (hazard ratio = 1.49, 95% confidence interval = 1.112–1.998, p = 0.008). Subgroup analysis also showed aspartate aminotransferase-to-platelet ratio index to be an independent predictor of poor overall survival and disease-free survival in patients positive for hepatitis B surface antigen or with cirrhosis (both p < 0.05). Similar results were obtained when aspartate aminotransferase-to-platelet ratio index was analyzed as a dichotomous variable with cutoff values of 0.25 and 0.62. Elevated preoperative aspartate aminotransferase-to-platelet ratio index may be independently associated with poor overall survival and disease-free survival in hepatocellular carcinoma patients following curative resection.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the fifth most commonly occurring malignancy and the third leading cause of cancer-related death worldwide.1–3 Hepatectomy is considered one of the most effective curative treatments for many patients with HCC, yet long-term survival remains less than satisfactory because of the high frequency of postoperative tumor recurrence. 4 Predicting which patients are more likely to experience recurrence after hepatectomy may help guide their postoperative management.
Tumor number and size, serum levels of fetoprotein (AFP), and serum levels of aspartate aminotransferase (AST) have been associated with survival.5–8 Background liver inflammation and fibrosis play critical roles in the process of hepatocarcinogenesis and survival after hepatectomy.9–11 Few studies have aimed to assess the prognostic potential of these and other factors comprehensively and systematically. Research has tended to focus on candidate prognostic markers that can be screened less invasively than by the gold-standard technique of liver biopsy, which is costly and carries a risk of pain, bleeding, and bile duct injury. 12 For example, the degree of liver fibrosis can be assessed using transient elastography and serum biochemical markers.13–15 In particular, the AST-to-platelet ratio index (APRI) has been proposed as a simple, straightforward prognostic test. It has been used to assess fibrosis stage, liver function reserve, and prognosis of HCC patients.16–20 To our knowledge, the prognostic potential of stratified APRI has not been assessed specifically in HCC patients who have undergone hepatectomy.
Therefore, we retrospectively analyzed survival and recurrence in HCC patients who underwent curative hepatic resection at the Affiliated Tumor Hospital of Guangxi Medical University over a 6-year period, and we explored associations between these outcomes and preoperative APRI, stratified by quartiles.
Patients and methods
This research was approved by the Ethics Committee of the Affiliated Tumor Hospital of Guangxi Medical University, and written informed consent was obtained from patients prior to surgery. All treatments were performed in accordance with relevant guidelines and regulations.
Patients
All patients undergoing hepatectomy with curative intent for HCC at the Affiliated Tumor Hospital of Guangxi Medical University between April 2004 and October 2010 were considered for inclusion in our study. To be included, patients were not allowed (a) to have received any treatment for HCC before hepatectomy, (b) to have any other co-malignancies or hematological disorders, or (c) to have any renal, cerebral, or cardiopulmonary dysfunction before hepatectomy.
Diagnosis and definitions
Patients were diagnosed initially with HCC based on preoperative indicators, and this diagnosis was confirmed in all patients based on histological analysis of tissue taken during hepatectomy. Hepatectomy with curative intent was defined as complete resection of all visible tumor tissue, with no residual tumor cells at the surgical margins. 21 Measurements of AST and platelet counts (PLT) were taken within 7 days before surgery and used to calculate preoperative APRI according to the following formula: (AST / ULN) × 100: PLT (109/L). 22
Follow-up visits
All patients were followed up at 1 month after liver resection and subsequently every 3 months during the first postoperative year, and then every 3–6 months in subsequent years. At each follow-up visit, serum biochemistry was analyzed, serum AFP levels were determined, and chest X-ray, ultrasound, and abdominal computed tomography or magnetic resonance imaging were performed.
Statistical analysis
Statistical analysis was conducted using SPSS 19.0 (IBM, USA). Inter-group differences in continuous variables were assessed for significance using one-way analysis of variance (ANOVA) or the non-parametric Kruskal–Wallis test. Differences in categorical data were assessed using the two-sided χ2 test, Fisher’s exact test or Mann–Whitney U test as appropriate.
Based on the observed distribution of preoperative APRI and the size of the study population, we decided to stratify patients into APRI quartiles. Overall survival (OS) and disease-free survival (DFS) rates were compared among patients in different quartiles using Kaplan–Meier survival curves and log-rank tests. Independent predictors of OS and DFS were explored using Cox proportional hazard regression. We also analyzed APRI as a dichotomous variable, in which case we analyzed receiver operating characteristic curves (ROCs) to identify the APRI cutoff that gave greatest sensitivity and specificity. In all statistical analyses, p < 0.05 was defined as the threshold of significance.
Results
Baseline characteristics of patients
During the study period, 858 HCC patients received liver resection with curative intent at our hospital. Of these, 224 (26·1%) were excluded because they had received other treatments before liver resection (183, 21·4%), they had concurrent malignancies (18, 2·1%) or concurrent hematological disorders (13, 1·5%), or they had renal, cerebral, or cardiopulmonary dysfunction before hepatectomy (10, 1·2%). In the end, 661 patients (574 males) were enrolled in the study (Table 1).
Characteristics of HCC patients treated by resection, stratified by APRI quartile.

Data are mean ± standard deviation or median (25th–75th interquartile range) unless otherwise indicated.
HCC: hepatocellular carcinoma; APRI: AST-to-platelet ratio index; HBsAg: hepatitis B surface antigen; AFP: alpha-fetoprotein; BCLC: Barcelona Clinic Liver Cancer; AST: aspartate aminotransferase; ALT: alanine aminotransferase.
Patients were stratified into quartiles of preoperative APRI, since this approach provided several subgroups with a reasonably large sample in each. Each quartile contained the following APRI ranges: Q1, 0.04 ≤ APRI ≤ 0.17; Q2, 0.17 < APRI ≤ 0.25; Q3, 0.25 < APRI ≤ 0.41; and Q4, 0.41 < APRI ≤ 7.52 (Table 1).
Higher APRI quartile was significantly associated with larger tumors, Edmonson grade III–IV and higher levels of AST, alanine aminotransferase (ALT), and total bilirubin (all p < 0.05). At the same time, it was associated with lower Child–Pugh grade, lower AFP, lower PLT and lower levels of albumin (all p < 0.05). Patients in a higher APRI quartile were more likely to be positive for hepatitis B surface antigen (HBsAg) and to have liver cirrhosis (both p < 0.01). The quartiles did not differ significantly in other baseline characteristics.
APRI as predictor of OS and DFS (all patients)
Unadjusted univariate Cox proportional hazard modeling was used to identify factors associated with OS and DFS (Table 2).
Cox proportional hazards regression to identify predictors of overall and disease-free survival in HCC patients treated by resection.

HCC: hepatocellular carcinoma; CI = confidence interval; HR = hazard ratio; APRI: AST-to-platelet ratio index; HBsAg: hepatitis B surface antigen; AFP: alpha-fetoprotein; BCLC: Barcelona Clinic Liver Cancer; AST: aspartate aminotransferase; ALT: alanine aminotransferase.
Patients in the highest APRI quartile were at 44% greater risk of death than those in the first quartile (hazard ratio (HR): 1.445, 95% confidence interval (CI): 1.081–1.931, p = 0.013), as well as 49% greater risk of disease recurrence (HR 1.49, 95%CI 1.112–1.998, p = 0.008). Univariate analysis identified several factors significantly associated with poor OS: APRI quartile, AFP ≥ 400 ng/mL, Barcelona Clinic Liver Cancer (BCLC) stage B or C, multiple tumors, tumor size ≥ 5 cm, incomplete tumor capsule, and AST ≥ 80 U/L (Table 2 and Figure 1). All these factors except AST level were also significantly associated with poor DFS (Table 2 and Figure 1).

Kaplan–Meier survival analysis of (a) disease-free survival and (b) overall survival of patients with hepatocellular carcinoma following curative hepatic resection. Patients were stratified into quartiles based on preoperative aspartate aminotransferase-to-platelet ratio index (APRI). APRI ranges for each quartile are given in Table 1.
Multivariate analysis (Table 2) identified the following independent predictors of poor OS: APRI quartile, BCLC stage B or C, multiple tumors, tumor size ≥ 5 cm, and incomplete tumor capsule. All these factors, together with AFP ≥ 400 ng/mL, were also significantly associated with poor DFS.
To complement this quartile-based analysis, we also considered APRI as a dichotomous variable. ROC analysis identified 0.25 as the APRI cutoff giving the greatest sensitivity and specificity in our cohort (Figure 2). (This value coincided with the border between Q2 and Q3 in our quartile-based analysis.) In addition, previous work in another population of Chinese patients had identified 0.62 as the optimal cutoff value. 17 Therefore, we performed analyses using both cutoff values (Table 3). Multivariate analysis indicated that patients with APRI ≥ 0.25 were at significantly greater risk of poor OS and DFS than those with APRI < 0.25 (both p < 0.01, Table 3). In contrast, multivariate analysis showed similar risk of poor OS for patients with APRI ≥ 0.62 as for patients with APRI < 0.62 (p = 0.262), and univariate analysis showed similar risk of poor DFS (p = 0.441, respectively).

Receiver operating characteristic curve to identify the optimal cutoff value for defining “low” or “high” preoperative aspartate aminotransferase-to-platelet ratio index (APRI). The optimal value of 0.25 was associated with a sensitivity of 59.9% and specificity of 55.6%.
Association between APRI and survival in HCC patients applying different cutoff values.

APRI: AST-to-platelet ratio index; HCC: hepatocellular carcinoma; HR: hazard ratio; CI: confidence interval.
APRI as predictor of OS and DFS (patients with cirrhosis or positive for HBsAg)
To elucidate the subgroups of HCC patients negatively influenced by preoperative APRI, we stratified HCC patients by HBsAg positivity and cirrhosis status. We found APRI quartiles were significantly associated with poor OS and DFS in patients positive for HBsAg or with cirrhosis (both p < 0.05, Figures 3 and 4).
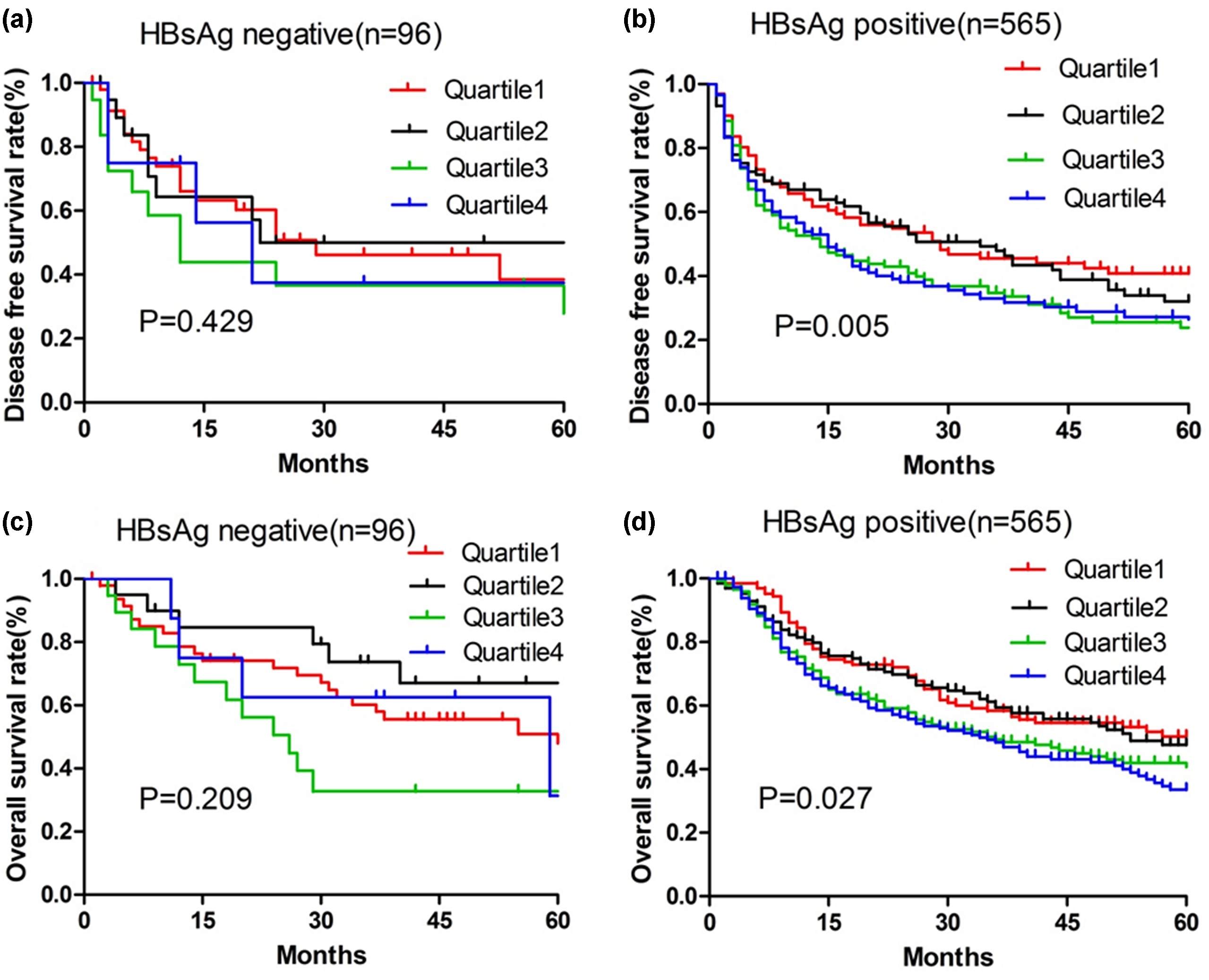
Kaplan–Meier survival analysis of (a and b) disease-free survival and (c and d) overall survival of patients with hepatocellular carcinoma following curative hepatic resection. Patients were stratified into quartiles based on preoperative aspartate aminotransferase-to-platelet ratio index (APRI). In addition, patients were subgrouped according to whether they were (a, c) negative or (b, d) positive for hepatitis B surface antigen (HBsAg). APRI ranges for each quartile are given in Table 1.

Kaplan–Meier survival analysis of (a and b) disease-free survival and (c and d) overall survival of patients with hepatocellular carcinoma following curative hepatic resection. Patients were stratified into quartiles based on preoperative aspartate aminotransferase-to-platelet ratio index (APRI). In addition, patients were subgrouped according to whether they (a, c) did not have cirrhosis or (b, d) did have cirrhosis. APRI ranges for each quartile are given in Table 1.
Discussion
Our study suggests that higher preoperative APRI is significantly and independently associated with poor OS and DFS in HCC patients who have undergone curative resection. These findings are consistent with previous research in other cohorts.16,17,19 We obtained similar results regardless of whether we stratified patients by APRI quartile or whether we treated APRI as a dichotomous variable for which the cutoff value for defining “low” or “high” APRI was 0.25 (our cohort) or 0.62. 17 In addition, we obtained similar results when we looked specifically at the subgroups of patients positive for HBsAg or with cirrhosis. In our cohort, preoperative APRI also showed significant relationships with tumor-related characteristics such as tumor size, as well as with nutritional status, as reflected in albumin levels. The association with nutritional status likely reflects cachexia due to tumor progression. Therefore, it could be concluded that preoperative APRI may be useful for predicting the survival of HCC patients who undergo hepatectomy.
Serum APRI is widely used to assess stage of liver fibrosis and cirrhosis,18,20 liver function reserve, and prognosis of patients with chronic hepatitis. 23 In the correlation analysis, we found preoperative APRI in our cohort to be associated with HBsAg and cirrhosis status (both p < 0.01, Table 1). These results suggest that APRI may be useful for assessing not only hepatitis C virus-related fibrosis and cirrhosis but also hepatitis B virus (HBV)-related cirrhosis. 23 Indeed, our subgroup analysis showed a significant association between APRI quartile and OS or DFS in patients positive for HBsAg or with cirrhosis. Again, the result proved that APRI has a strong connection with HBsAg and cirrhosis.
Why elevated APRI should predict poor survival remains unclear. One possible explanation is that many HCC patients with elevated APRI have low preoperative PLT, which may be caused by progressive destruction of the enlarged spleen24–26 and by progressive liver fibrosis, which decreases thrombopoietin production.27,28 Low preoperative PLT may also be associated with greater risk of major complications, postoperative liver failure, and mortality after hepatectomy. 29 Another possible explanation is that, many HCC patients with elevated APRI may have elevated AST levels, reflecting liver stress or damage arising from, for example, reactivation of HBV replication or progressive liver fibrosis. 30 These events are associated with poor survival in HCC patients. 31
Although our analysis of APRI as a dichotomous variable supports our analysis based on APRI quartiles, we found that APRI ≥ 0.62 was not associated with worse OS than APRI < 0.62 (multivariate model) or with worse DFS (univariate model). This negative result may be an artifact of small sample size, since the number of patients with APRI ≥ 0.62 was >6-fold smaller than the number with APRI < 0.62 (Table 3).
The findings of this study should be interpreted with caution in light of several limitations. First, as the value of APRI in follow-up duration is dynamic, we only use the baseline value to predict the outcomes in the future, so a lot of information during the follow-up may be missing; second, owing to the nature of retrospective study, potential selection bias exists.
In summary, our study suggests that the preoperative APRI is an independent prognostic factor in HCC patients. However, a prospective study with a larger population is needed to justify our researches.
Synopsis
Curative hepatectomy remains one of the most effective treatments for many patients with HCC, yet the rate of postoperative recurrence remains high. Reliable prediction of survival and recurrence after hepatectomy may help clinicians treat and manage these patients more effectively. Several candidate prognostic markers have been identified in the literature, but many require invasive liver biopsy. Candidate serum markers can be tested more easily, but their prognostic potential specifically for HCC patients after curative hepatic resection has yet to be examined systematically and comprehensively. Here, we analyze a relatively large cohort of patients at a major liver treatment center in Southeastern China and provide evidence that the preoperative APRI, which is measured easily in serum, may reliably predict risk of death or recurrence after hepatectomy. We obtained similar results regardless of whether we stratified patients according to APRI quartile or according to a dichotomous classification of “low” or “high” APRI based on cutoff values determined from our own cohort or from previous studies.
Footnotes
Author contribution
H.-J.Y., J.-H.J., and Y.-T.Y. contributed equally to this work; B.-De.X. and L.-Q.L. contributed equally to this work; B.-De.X. and L.-Q.L. designed the research; H.-J.Y., J.-H.J., Y.-T.Y., X.-h.L., F.L. and J.-j.L. performed the research; H.-J.Y, Y.-T. Y., N.-Z.C. and K.-L.Z. evaluated the clinic records and performed the statistical analyses; H.-J.Y. wrote the manuscript; all authors have read and approved the final manuscript.
Data sharing statement
Technical appendix, statistical code, and dataset are available from the corresponding author at
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grants to B.-De.X from the National Natural Science Foundation of China (81260331) and the Key Laboratory for High-Incidence Tumor Prevention and Treatment of the Ministry of Education of China (GKE2015-ZZ05). The funders had no role in study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Informed consent statement
All study participants, or their legal guardian, provided informed written consent prior to study enrollment.
Institutional review board statement
The study was reviewed and approved by the Affiliated Tumor Hospital of Guangxi Medical University Institutional Review Board.
