Abstract
Beta-1,4-galactosyltransferase II is found to be associated with the alterations of tumor-related glycosylation. However, the clinical significance of beta-1,4-galactosyltransferase II in non-metastatic clear-cell renal cell carcinoma has not been reported up to now. Herein, our researches suggested that the expression level of beta-1,4-galactosyltransferase II was first found to be positively associated with tumor size, Fuhrman grade, lymphovascular invasion, rhabdoid differentiation, tumor necrosis and poor overall survival and recurrence-free survival of patients with non-metastatic clear-cell renal cell carcinoma, both in training set and validation set. Moreover, beta-1,4-galactosyltransferase II expression was identified as an independent adverse prognosticator for overall survival and recurrence-free survival of patients with non-metastatic clear-cell renal cell carcinoma. Ultimately, prognostic accuracy of the nomogram integrating beta-1,4-galactosyltransferase II with other independent prognostic parameters was dramatically improved for overall survival and recurrence-free survival of patients with non-metastatic clear-cell renal cell carcinoma. Taken together, beta-1,4-galactosyltransferase II is a potential independent adverse prognostic factor for postoperative recurrence and survival, which could be developed as a useful biomarker for non-metastatic clear-cell renal cell carcinoma by a series of further independent and retrospective studies, so as to help the postsurgical management of clear-cell renal cell carcinoma patients.
Keywords
Introduction
Renal cell carcinoma (RCC) is the most common solid lesion within kidney, accounts for 2%–3% of all adult malignancies. 1 The incidence of RCC is increasing in most areas of the world for which statistics are available for recent decades. 2 Clear-cell renal cell carcinoma (ccRCC) is the most common subtype of RCC, accounting for 60%–80% of all RCCs. 3 Although most patients with localized tumors can be cured with nephrectomy, 20%–30% of the patients without any evidence of metastasis will die of relapse and/or metastatic RCC in the future. 4 Therefore, it is extremely urgent to discover potential prognostic markers to monitor recurrence or progression for localized RCC patients and also required to establish a new prognostic model of clinical outcomes so as to make optimized treatment plans and improve the overall prognosis through in-depth analysis.
Glycosylation, as a quite common posttranslational modification, is found to play a critical role in immune recognition, cell signaling, and cell–cell interaction because glycan branches confer structure variability and binding specificity to lectin ligands. 5 Growing evidence demonstrates that aberrant glycosylation can modulate tumor growth and malignant transformation in different cancer types, and glycosylation itself is tremendously dynamic during the development of cancer. 6 As a matter of fact, aberrant glycosylation is associated with differential expressions of glycosyltransferase and glycosidases, which subsequently result in cancer cells to produce glycoproteins with specific cancer-associated aberrations in glycan structures. 7 Therefore, aberrant glycosylation and/or glycosyltransferase represents a hallmark of cancer. In recent decades, it is already well confirmed that tumor-associated carbohydrate antigens have drawn increasing attention from all over the world in the development of tumor vaccines and diagnostic reagents.8,9 The ability to distinguish differences in glycosylation of proteins or activity of glycosyltransferases between cancer and control patients contributes to develop a promising field for potential biomarker. 10
Beta-1,4-galactosyltransferase II (beta4GalT-II), a member of beta-1,4-galactosyltransferase family, is responsible for the biosynthesis of N-acetyllactosamine on N-glycans by transferring uridine diphosphate (UDP)-galactose to the terminal N-acetylglucosamine (N-GlcNAc) residues with the formation of a beta-1,4-linkage. 11 Beta4GalT-II has been reported to play an intensely important role in cellular functions such as adhesion, endocytosis, and receptor signaling. 12 Increasing studies have revealed that the abnormal activity of beta4GalT-II contributes to the tumor progression.13,14 Besides, the results of The Cancer Genome Atlas (TCGA) database showed that patients with high beta4GalT-II messenger RNA (mRNA) expression tend to have adverse outcomes for overall survival (OS) comparing with those with a low beta4GalT-II mRNA expression (p = 0.025). However, clinicopathological features of beta4GalT-II in tissues of patients with ccRCC and its prognostic value have not been reported up to now.
In this study, we first confirmed the up-regulation of beta4GalT-II in non-metastatic ccRCC tissue microarray (TMA) and subsequently dichotomize the patients into low and high groups according to the cutoff value. Furthermore, the correlation of beta4GalT-II expression with clinicopathological features and the prognostic value of beta4GalT-II in non-metastatic ccRCC patients were evaluated. Finally, a nomogram integrating beta4GalT-II with other independent prognostic parameters was generated to help predict prognosis and guide management for non-metastatic ccRCC patients after surgery.
Materials and methods
Patients and specimens
We retrospectively recruited 585 non-metastatic ccRCC patients who underwent radical nephrectomy or nephron-sparing surgery at Zhongshan Hospital in 2008 and 2009. Inclusion criteria included the following: no history of previous anticancer therapy, no history of other malignancies, patients after radical or partial nephrectomy, and histopathologically proven ccRCC. Exclusion criteria were as follows: mixed type of primary renal cancer as confirmed histopathologically, tumors with necrosis >80%, patients died within the first month after surgery due to surgical complications, and patients with N1 or M1 tumors were considered to have metastatic disease. Patients were staged according to radiographic reports and postoperative pathological data and were reassigned according to 2010 American Joint Committee on Cancer (AJCC) tumor/node/metastasis (TNM) classification. 15 OS and recurrence-free survival (RFS) were calculated from the date of surgery to the date of death (or the last follow-up) or to the date of recurrence (or the last follow-up). This study was approved by the Clinical Research Ethics Committee of Zhongshan Hospital, Fudan University (Shanghai, China), and written informed consent was obtained from each patient.
The overall cohort of 585 patients was subdivided into a training set (n = 293) and a validation set (n = 292) according to the computer-generated random numbers. The median follow-up was 68 months (range: 39–74 months) and 67 months (range: 40–74 months) for the training set and validation set, respectively. Among the overall cohort of 585 patients, 113 (19.32%) patients had recurrence, while 102 (17.44%) patients died of all causes during the follow-up period. The two sets were well matched for pathological characteristics.
TMA, immunohistochemistry, and evaluation of immunohistochemical intensity
Formalin-fixed, paraffin-embedded surgical specimens were used for TMA construction and for subsequent immunohistochemistry (IHC) study. Two cores were taken from each surgical sample by using punch cores that measured 1.5 mm in the greatest dimension from the center of the tumor foci. Tissue blocks were mounted on glass slides by sequencing. The IHC protocols were as described previously. 16 The primary antibody against beta4GalT-II (1:50 dilution; Abcam, Cambridge, MA, USA) was used.
The intensity of immunohistochemical staining of beta4GalT-II was scored by two independent pathologists using the semiquantitative immunoreactivity scoring (IRS) system as described previously. 16 A semiquantitative score which ranged from 0 to 300 was derived by multiplying the staining intensity (0: negative, 1: weak, 2: moderate, 3: strong) by the percentage of immunoreactive cells (percentage of positive staining cancer cells, 0%–100%) at each intensity level for each sample. According to the “minimum p value” approach conducted by X-tile, 105 was determined as the cutoff of IRS value, and 54.95% (132/293) and 55.48% (162/292) specimens were scored as high beta4GalT-II expression in the training set and validation set, respectively.
Western blot
Western blot was performed as described previously. 17 Anti-beta4GalT-II antibody (diluted 1:500; Abcam) was used for western blot. Beta4GalT-II expression was detected in three pairs of tumor and corresponding peri-tumor tissues from ccRCC patients by western blot.
Statistical analysis
The optimum cutoff of the IRS-score was selected by X-tile software v3.6.1 (Yale University, New Haven, CT, USA). Student’s t-test or χ2-test, Kaplan–Meier survival curves, univariate and multivariate Cox analysis were performed by MedCalc Software 11.4.2.0 (MedCalc Software, Mariakerke, Belgium). Nomogram and calibration plot were analyzed by R software 3.0.2 with the “rms” package (R Foundation for Statistical Computing, Vienna, Austria). All significance tests were two-sided and p < 0.05 was considered statistically significant.
Results
Immunohistochemical beta4GalT-II intensity and its association with clinicopathological characteristics in patients with non-metastatic ccRCC
To investigate whether the expression level of beta4GalT-II was altered during the development and progression of non-metastatic ccRCC, we evaluated beta4GalT-II expression by IHC staining analysis and western blot. As shown in Figure 1(a), the specificity of anti-beta4GalT-II antibody was first confirmed by IHC staining with IgG on ccRCC tissues as negative control. Beta4GalT-II positive staining was mainly located in the cytoplasm of tumor cells in non-metastatic ccRCC specimens, and its intensity in tumor tissues was dramatically higher than that in corresponding peri-tumor tissues, which was consistent with the results of western blot (Supplementary Figure 1).
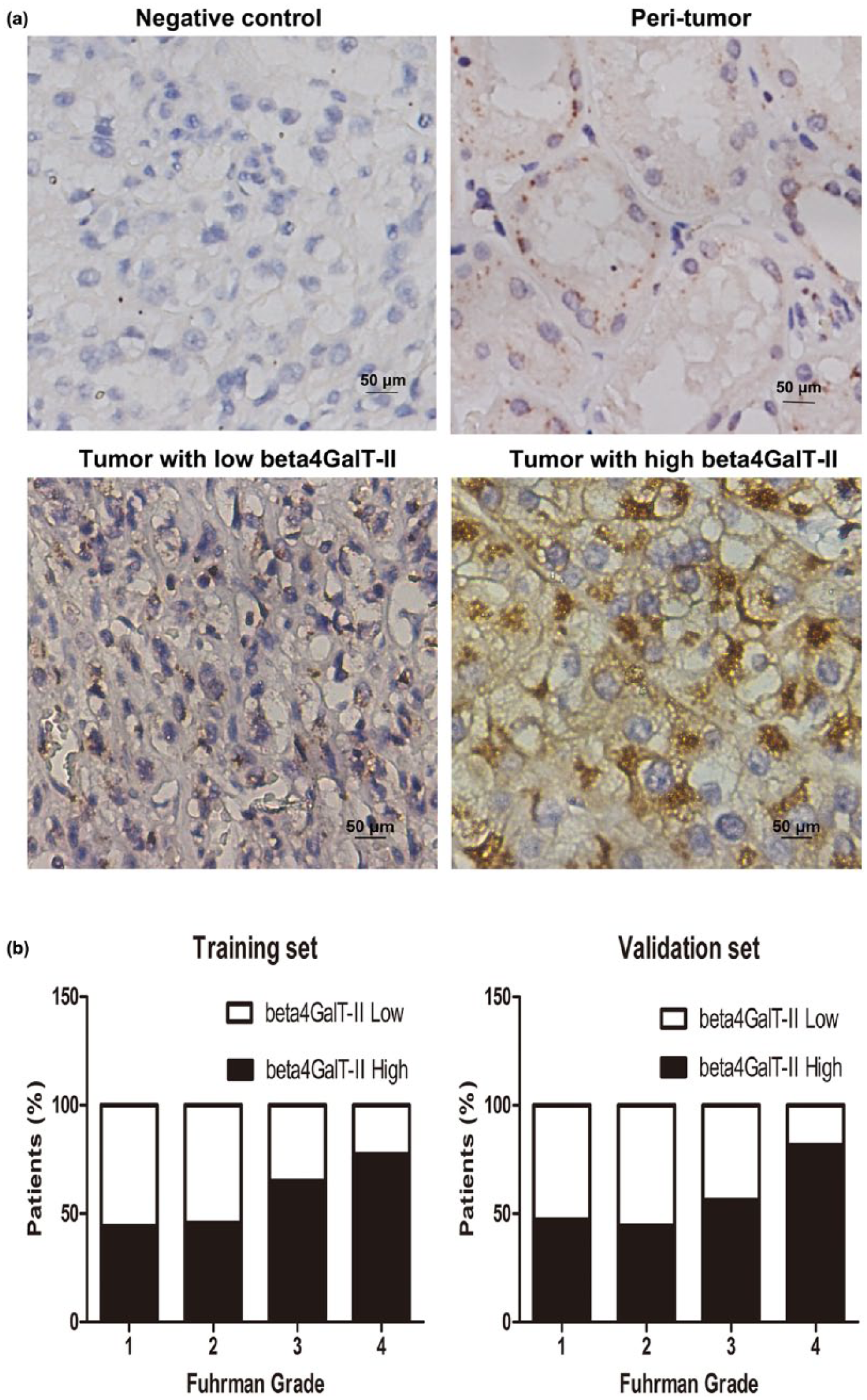
The analysis of beta4GalT-II expression by immunostaining in ccRCC tissues. (a) Representative photographs of beta4GalT-II immunostaining: IHC staining with IgG on ccRCC tissues as negative control at the upper left corner, IHC staining with anti-beta4GalT-II antibody on peri-tumor tissues at the upper right corner, low beta4GalT-II expression in tumor tissue at the lower left corner, and high beta4GalT-II expression in tumor tissue at the lower right corner (original magnification: 400×). (b) The percentage of patients with beta4GalT-II high or low expression according to the Fuhrman grade in the training (left panel) and validation (right panel) sets.
The IRS value of beta4GalT-II in training set (n = 293) and validation set (n = 292) is shown in Supplementary Figure 2. The relationship of beta4GalT-II expression level and Fuhrman grade is shown in Figure 1(b); higher expression level of beta4GalT-II was more easily found in advanced Fuhrman grade.
The correlation between beta4GalT-II expression and clinicopathological features is shown in Supplementary Table 1. Expression level of beta4GalT-II was positively associated with tumor size (p < 0.001 and p = 0.001 in the training set and validation set, respectively), Fuhrman grade (p < 0.001 and p < 0.001, respectively), lymphovascular invasion (LVI; p = 0.002 and p = 0.046, respectively), rhabdoid differentiation (p = 0.002 and p = 0.027, respectively), tumor necrosis (p = 0.004 and p = 0.017, respectively), and recurrence (p < 0.001 and p = 0.003, respectively). In contrary, no significant correlation was found between beta4GalT-II expression and other clinical variables of the patients, such as age of patients, gender, pT-stage, sarcomatoid features, and Eastern Cooperative Oncology Group performance status (ECOG-PS).
Beta4GalT-II expression correlated with OS and RFS of the patients with non-metastatic ccRCC
The relationship between beta4GalT-II expression and clinical outcome of patients with ccRCC was analyzed by Kaplan–Meier method and log-rank test. As shown in Figure 2(a) and (b), patients with high beta4GalT-II expression level tend to have adverse outcomes for OS comparing with those with a low beta4GalT-II expression in the training set and validation set (p < 0.001 and p < 0.001, respectively). Similarly, patients with high beta4GalT-II expression level presented a poorer RFS comparing with those with a low beta4GalT-II expression (p < 0.001 and p = 0.003, respectively; Figure 2(c) and (d)). Therefore, higher beta4GalT-II might mean earlier recurrence and shorter OS of patients with non-metastatic ccRCC.

Kaplan–Meier analysis for overall survival (OS) and recurrence-free survival (RFS) according to beta4GalT-II expression. Kaplan–Meier analysis for OS in (a) the training set and (b) the validation set and Kaplan–Meier analysis for RFS in (c) the training set and (d) the validation set. The p value was calculated by log-rank test.
Furthermore, to determine whether patients with different Fuhrman grades of non-metastatic ccRCC could be stratified by beta4GalT-II expression, we evaluated the prognostic value of beta4GalT-II expression according to Fuhrman grade in the overall cohort of patients so as to distribute enough cases in each Fuhrman grade. As shown in Figure 3, high expression of beta4GalT-II was positively associated with OS of patients with Fuhrman grade 3 (hazard ratio (HR), 2.668; 95% confidence interval (CI), 1.086–6.552; p = 0.033; Figure 3(c)) and Fuhrman grade 4 (HR, 3.730; 95% CI, 1.344–10.350; p = 0.012; Figure 3(d)), according to further Univariate cox analysis. Analogously, an obvious positive correlation was found between higher beta4GalT-II expression and RFS of patients with Fuhrman grade 3 (HR, 2.154; 95% CI, 1.009–4.599; p = 0.049; Figure 3(g)) and Fuhrman grade 4 (HR, 2.728; 95% CI, 1.074–6.929; p = 0.036; Figure 3(h)). No significant correlation was found between beta4GalT-II expression and OS or RFS of patients with Fuhrman grade 1 (Figure 3(a) or (e)) and grade 2 (Figure 3(b) or (f)). Taken together, beta4GalT-II expression might be contributed to stratification of non-metastatic ccRCC patients with Fuhrman grades 3 and 4 for OS and RFS analysis.

Subgroup analysis to assess prognostic value of beta4GalT-II by Fuhrman grade in non-metastatic ccRCC patients. Kaplan–Meier analysis of overall survival (OS) in patients with (a) Fuhrman grade 1, (b) Fuhrman grade 2, (c) Fuhrman grade 3, and (d) Fuhrman grade 4. Kaplan–Meier analysis of recurrence-free survival (RFS) in patients with (e) Fuhrman grade 1, (f) Fuhrman grade 2, (g) Fuhrman grade 3, and (h) Fuhrman grade 4. The p value was calculated by log-rank test.
Beta4GalT-II expression was identified as an independent adverse prognosticator in patients with non-metastatic ccRCC
To further investigate whether beta4GalT-II expression was an independent prognostic predictor of OS and RFS, univariate and multivariate Cox analyses were performed. According to the results of univariate Cox analysis in Supplementary Table 2, beta4GalT-II was significantly associated with OS (HR, 3.646; 95% CI, 2.261–5.879; p < 0.001) and RFS (HR, 3.073; 95% CI, 1.998–4.727; p < 0.001). Besides, age of patients, tumor size, pT-stage, Fuhrman grade, LVI, rhabdoid differentiation, sarcomatoid features, tumor necrosis and ECOG-PS were all significantly associated with OS. All these factors (except age of patients) also have a significant impact on RFS.
Subsequently, all the significant factors above were brought into the multivariate Cox analysis of OS and RFS, respectively. As shown in Figure 4, beta4GalT-II expression was identified as an independent adverse prognostic factor for OS (HR, 2.595; 95% CI, 1.557–4.324; p < 0.001) and RFS (HR, 2.383; 95% CI, 1.511–3.760, p < 0.001). Besides, tumor size, pT-stage, Fuhrman grade, LVI, sarcomatoid features, tumor necrosis, and ECOG-PS were also independent prognostic factors for OS and RFS.

Multivariate Cox analysis for overall survival (OS) and recurrence-free survival (RFS) in non-metastatic ccRCC patients. Independent prognostic factors for OS and RFS were identified and presented according to multivariate analysis. The p value was calculated by log-rank test.
Prognostic nomogram of non-metastatic ccRCC
To predict the OS and RFS in ccRCC, a prognostic nomogram via integrating all these independent prognostic indicators including beta4GalT-II was established. As shown in Figure 5(a), total points were calculated to evaluate the clinical outcomes, with higher point indicating more adverse outcome probability for OS. The calibration plot for the probability of OS at 3 or 6 years after surgery presented an optimal agreement between actual observation and the prediction by nomogram (Figure 5(b) and (c)); predictive accuracy of 3-year RFS probability and 6-year RFS probability was 95.99% and 89.63%, respectively. Similarly, the prognostic nomogram for RFS is shown in Figure 5(b); predictive accuracy of 3-year RFS probability and 6-year RFS probability was 96.01% and 87.68%, respectively (Figure 5(e) and (f)).

Nomogram for predicting 3- and 6-year overall survival (OS) and recurrence-free survival (RFS) in patients with non-metastatic ccRCC. (a) Nomogram for predicting OS rate integrated with tumor size (continuous variable), pT-stage (pT1 + pT2 + pT3 + pT4), Fuhrman grade (1 + 2 + 3 + 4), LVI (absent/present), sarcomatoid (absent/present), necrosis (absent/present), ECOG-PS (0/⩾1), and beta4GalT-II expression (low/high). (b) Calibration plot for nomogram predicted and observed 3-year OS rate. (c) Calibration plot for nomogram predicted and observed 6-year OS rate. (d) Nomogram for predicting RFS rate integrated with tumor size (continuous variable), pT-stage (pT1 + pT2 + pT3 + pT4), Fuhrman grade (1 + 2 + 3 + 4), LVI (absent/present), sarcomatoid (absent/present), necrosis (absent/present), ECOG-PS (0/⩾1), and beta4GalT-II expression (low/high). (e) Calibration plot for nomogram predicted and observed 3-year RFS rate. (f) Calibration plot for nomogram predicted and observed 6-year RFS rate. Gray line indicates ideal model and vertical bars indicate 95% confident interval. Higher total point indicates a more adverse outcome probability.
Superior performance of prognostic nomogram model including beta4GalT-II for non-metastatic ccRCC
To evaluate whether prognostic model including beta4GalT-II improve predictive accuracy, Harrell’s concordance index (C-index) and Akaike information criterion (AIC) of University of California Los Angeles Integrated Staging System (UISS); the Mayo Clinic stage, size, grade, and necrosis (SSIGN); Leibovich; and a prognostic nomogram via integrating all these independent prognostic indicators including beta4GalT-II for OS and RFS were performed. As given in Table 1, for OS, the C-index of UISS, SSIGN, and Leibovich was 0.752, 0.789, and 0.808. The C-index of newly established prognostic model was improved to 0.896. The AIC of UISS, SSIGN, and Leibovich was 1164.15, 1115.38, and 1105.80. The AIC of newly established prognostic model was reduced to 986.94. For RFS, the C-index of newly established prognostic model was improved to 0.877, and the AIC of was reduced to 1125.09. All these results were proved to generate a better predictive model for OS and RFS of non-metastatic ccRCC patients.
Comparison of the prognostic accuracy of the prognostic models.

C-index: Harrell’s concordance index; AIC: Akaike information criterion; UISS: University of California Los Angeles Integrated Staging System; SSIGN: the Mayo Clinic stage, size, grade, and necrosis.
Discussion
In this study, beta4GalT-II was first found to be upregulated in patients with non-metastatic ccRCC and positively related to tumor size, Fuhrman grade, LVI, rhabdoid differentiation, tumor necrosis, and recurrence. Moreover, beta4GalT-II was positively correlated with OS and RFS and then identified as a potential independent adverse prognosticator of patients with non-metastatic ccRCC. Besides, beta4GalT-II expression might be contributed to stratification of non-metastatic ccRCC patients with Fuhrman grades 3 and 4 for OS and RFS analysis. Ultimately, prognostic accuracy of the nomogram integrating beta4GalT-II with other independent prognostic parameters was dramatically improved for OS and RFS of patients with non-metastatic ccRCC, compared with UISS, SSIGN, and Leibovich models.
Beta4GalT-II is one of the enzymes transferring galactose to the terminal N-acetylglucosamine of complex-type N-glycans, which exerts an extensive influence on pathology and physiology function. 18 Beta4GalT-II is a key regulator of glycosylation of the proteins involved in neuronal development. Beta4GalT-II is confirmed to be mainly localized in Schwann cells and be gradually increased at proximal and distal stumps of rat-injured sciatic nerves, suggesting that beta4GalT-II affects the galactosylation of glycoproteins in injured nerve regeneration. 19 To our surprise, the enhancement of the beta4GalT-II expression in tumors is one of the promising approaches to suppress tumor growth and angiogenesis and promote apoptosis in mouse melanoma and human hepatocellular carcinoma. 14 Beta4GalT-II might serve as a target gene of p53 transcription factor during adriamycin-induced HeLa cell apoptosis, which elucidated a new mechanism of p53-mediated cell apoptosis in cervix cancer. 20 Interestingly, specific enzyme complex of beta4GalT-II and glucuronyltransferase-P facilitates biosynthesis of N-linked human natural killer-1 carbohydrate. 21 In ccRCC, beta4GalT-II was found to be correlated with Fuhrman grade, tumor size, LVI, rhabdoid differentiation, tumor necrosis, and recurrence (Supplementary Table 1) and promotes the growth of the tumor, suggesting this pathway as a potential therapeutic target. However, underlining mechanisms need to be addressed in further research.
For an understanding of tumor-related alterations of the complex carbohydrate pattern of carcinomas, it is indispensable to monitor the expression profile of the various glycosyltransferases. 22 Expression of beta4GalT-II has been reported to be significantly altered in a variety of tumors. Upregulation of beta4GalT-II is correlated with the malignant degree of astrocytoma and results in the increase of galactosylation in astrocytoma. 11 High expression level of beta4GalT-II seems to be of relevance for the aberrant biosynthesis of membrane-bound carbohydrates during colonic carcinogenesis and metastasis. 13 On the contrary, the expression of beta4GalT-II is markedly downregulated during the malignant transformation of mouse melanoma and human hepatocellular carcinoma. 14 Herein, beta4GalT-II was demonstrated to be upregulated in patients with non-metastatic ccRCC (Figure 1). It is worth mentioning that our results are consistent with the results of beta4GalT-II mRNA in TCGA database, although the proportion of high expression is inconsistent (55% vs 4%).
It is reported that increased beta-1,4-galactosyltransferase IV (beta4GalT-IV) expression is strongly associated with tumor metastases and poor prognosis in colorectal cancer. 23 Beta-1,4-galactosyltransferase III (Beta4GalT-III) predicts an unfavorable prognosis for neuroblastoma, 24 negatively associated with colorectal cancer metastasis. 25 These studies suggest that the family of beta4GalT may be closely related to the prognosis of tumors. However, there is no report about association between beta4GalT-II and prognosis of patients with tumors up to now. In our research, beta4GalT-II was found to be correlated with OS and RFS and then identified as a potential independent adverse prognosticator of the patients with non-metastatic ccRCC for the first time (Figures 2 and 4; Supplementary Table 2). As a matter of fact, the expression level of beta4GalT-II mRNA is positively associated with OS according to TCGA database, which is consistent with our results. The higher the Fuhrman grade, the poorer the prognosis of ccRCC. 26 Our results suggested that beta4GalT-II expression might be contributed to stratification of non-metastatic ccRCC patients with Fuhrman grades 3 and 4 for OS and RFS analysis (Figure 3), which shows an important significance for postoperative management and prognosis of non-metastatic ccRCC patients.
Genetic changes underlying ccRCC include alterations in genes controlling cellular oxygen sensing (e.g. VHL) and the maintenance of chromatin states (e.g. PBRM1). 27 In current clinical practice, TNM stage, Fuhrman grade, rhabdoid differentiation, sarcomatoid features, tumor necrosis, LVI, and ECOG-PS have been widely used as predictors of clinical outcomes for patients with ccRCC.28,29 Several outcomes of prognostic prediction algorithms like UISS, which incorporates TNM stage, Fuhrman grade, and ECOG-PS, have been proposed previously to stratify the clinical outcomes after nephrectomy.17,30 However, the biological nature of ccRCC is kind of complex with an unpredictable course; even tumors with the comparable stage or the same pathological type could show a wide variation in biological behavior and clinical outcomes.29,31 Therefore, the improvement in the predictors of clinical outcomes for RCC is needed. In this research, the nomogram integrating beta4GalT-II with other independent prognostic parameters was established, and its predictive accuracy was dramatically improved for OS and RFS of non-metastatic ccRCC (Figure 5, Table 1), compared with UISS, SSIGN, and Leibovich models, which indicated a profound clinical significance.
Although the clinical significance of beta4GalT-II in ccRCC has been illuminated, there are still several limitations of this study to be discussed. First, functional studies should be carried out to reveal the potential molecular mechanisms of beta4GalT-II in the development and progression of ccRCC in the future, because this study was mainly performed on the immunohistochemical staining of beta4GalT-II. Second, we will further explore whether the alteration of beta4GalT-II, as a glycosyltransferase, is given rise to the change in quantity and structure of carbohydrate chain, whether the latter can also be used as diagnostic and prognostic markers. Third, the independent adverse prognostic value of beta4GalT-II and the predictive accuracy of the nomogram integrating beta4GalT-II with other independent prognostic parameters are necessary to be confirmed in multicenter, large sample research.
In conclusion, the data demonstrated that increased beta4GalT-II was a potential independent adverse prognostic factor for OS and RFS of patients with non-metastatic ccRCC. The nomogram integrating beta4GalT-II with other independent prognostic variables is found to improve the predictive accuracy for the prognosis of patients with non-metastatic ccRCC. Beta4GalT-II could be developed as a useful biomarker for non-metastatic ccRCC by a series of further independent and retrospective studies, so as to help the postsurgical management of ccRCC patients.
Footnotes
Acknowledgements
The authors would like to thank Dr Yuan Ji, Dr Jun Hou, and Ms Haiying Zeng (Department of Pathology, Zhongshan Hospital of Fudan University) for diagnosis confirmation and technical assistance. H.Z., Y.L., and H.X. contributed equally to this work.
Author contributions
H.Z. helped in acquisition of data, analysis and interpretation of data, statistical analysis, and drafting of the manuscript; Y.L., H.X., Q.F., Z.L., Y.Z., L.X., and W.Z. provided technical and material support; and Y.Y. and J.X. helped in study concept and design, analysis and interpretation of data, drafting of the manuscript, and obtaining funding and study supervision. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by grants from National Basic Research Program of China (2012CB822104), National Key Projects for Infectious Diseases of China (2012ZX10002012-007 and 2016ZX10002018-008), National Natural Science Foundation of China (31100629, 31270863, 81372755, 31470794, 81401988, 81402082, 81402085, 81471621, 81472227, 81472376, 31570803, 81501999, and 81572352), and Program for New Century Excellent Talents in University (NCET-13-0146). All these study sponsors have no roles in the study design and in the collection, analysis, and interpretation of data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
