Abstract
Sulfiredoxin (Srx), a novel oxidative stress-induced antioxidant protein, has been reported to be expressed in several human tumour tissues. However, the expression and functions of Srx in cervical squamous cell carcinoma remain unknown. Here, we proved that expression of Srx was upregulated in cervical tissues as revealed by immunohistochemistry, and revealed a close correlation between the protein’s expression and the expression level of one core epithelial–mesenchymal transition marker, E-cadherin. We demonstrated that Srx was overexpressed in cervical squamous cell carcinoma and its expression level was closely correlated with lymph node metastasis and invasion of cervical squamous cell carcinoma. Meanwhile, Srx expression was negatively correlated with E-cadherin expression. The remission time (tumour-free status after surgery) of the Srx strong staining group was significantly shorter than that of the Srx weak staining group. We silenced Srx by short hairpin RNA in HeLa and SiHa cells. Diminished Srx expression upregulated E-cadherin expression. The cell invasion and migration activity in the ShSrx group were obviously decreased in HeLa and SiHa cells. Moreover, Srx regulated the expression of the other marker of epithelial–mesenchymal transition, vimentin. In conclusion, the study suggested that Srx was highly expressed in cervical squamous cell carcinoma and may promote invasion and metastasis of cervical squamous cell carcinoma via regulating epithelial–mesenchymal transition.
Cervical cancer is the second most commonly diagnosed cancer and third leading cause of cancer deaths among women in less developed countries. Approximately, 90% of cervical cancer deaths occur in developing parts of the world. 1 Cervical squamous cell carcinoma (CSCC) accounts for nearly 80% of cervical cancer. Metastasis and recurrence seriously affect the quality of life of patients and eventually become the leading causes of death for patients after surgery. 2 Metastasis of the tumour includes various processes, such as the neoplastic cell’s invasion into the basal membrane and the spread into the lymphatic and blood vessels for dissemination into circulation, resulting in metastatic growth in distant tissues. Cancer cell migration and metastasis are typically regulated by integrins, matrix-degrading enzymes, cell-to-cell adhesion molecules and cell-to-cell communications. These adaptation responses include epithelial–mesenchymal transition (EMT). 3 EMT, as the ultimate survival mechanism of cancer cells, has been widely explored in cervical cancer.4–6 Loss of E-cadherin, a core EMT marker, has been known to play an important role in tumour cell invasion and metastasis. 7
Sulfiredoxin (Srx), an endogenous antioxidant protein, was first discovered in yeast as an enzyme that regulates reactive oxygen species signalling. It mainly catalyses reduction of hyperoxidised 2-Cys peroxiredoxins (Prxs) in the presence of ATP, Mg2+ and thiol equivalents such as glutathione and thioredoxins.8–11 Srx has also been reported to catalyse the deglutathionylation of protein phosphatase and PrxI.12,13 In addition, Srx has been identified as an activator protein-1 (AP-1) target gene, which is required for tumour promoter–induced neoplastic transformation, and showed elevated expression in several human cancer tissues, particularly in skin malignancies, rectal carcinoma and lung adenocarcinoma but not in their paired normal tissues. 14 It has been reported that human Prx1 can induce EMT by an unknown mechanism. 15 Numerous evidences have shown that Prxs are selectively reduced by Srx.16–18 However, Srx expression patterns in cervical cancer and the association between the expression of Srx and E-cadherin in cervical cancer patients remain unclear.
In this study, we investigated the expression of Srx and E-cadherin in 123 CSCC tissues, 25 high-grade squamous intraepithelial lesions (HSILs) and 30 normal cervical (NC) clinical samples. The aim was to analyse the association between the expression of Srx and E-cadherin in CSCC patients and to examine the correlation of their expression with clinicopathological characteristics and prognosis of CSCC patients. To further prove the clinical significance of Srx expression in patients with CSCC, a Kaplan–Meier test was adopted. To gain insight into the function of Srx, we knocked down the Srx expression in HeLa and SiHa cells and found that silencing of Srx inhibited invasion and metastasis of both the cell lines. Meanwhile, to explore whether Srx plays an important role in the EMT in CSCC, we investigated the EMT markers in SiHa cells and examined the cell morphology by optical microscopy.
Methods and materials
Patient tissue samples
A total of 178 clinical samples were obtained from patients undergoing surgical resection in the First Affiliated Hospital, Chongqing Medical University, China, during the period March 2009 through May 2015. Samples included 30 NC tissues, 25 high-grade HSILs and 123 CSCC samples. All the patients were diagnosed by two independent pathologists after surgery. The stage and grade of the tumours were evaluated according to the International Federation of Gynecology and Obstetrics staging system. The 123 CSCC patients ranged in age from 30 to 74 years. No patient was given preoperative chemotherapy or radiotherapy before sampling. All participants provided written informed consent. The protocol was approved by the Institutional Ethics Committee of Chongqing Medical University. The procedures were conducted in accordance with the approved guidelines.
Immunohistochemistry
Immunohistochemistry (IHC) staining for Srx protein was performed in 178 surgical specimens consisting of 30 NC tissues, 25 HSILs and 123 CSCC samples. All tissues were fixed in formalin and paraffin-embedded samples. Every tissue was sectioned 5-µm thick and mounted on polylysine-coated slides. Experiments were performed using an immunohistochemical SABC kit (Boster, Wuhan, China), according to the manufacturer’s instructions. Rabbit anti-human Srx antibody (Proteintech, Wuhan, China; Cat. No. 14273-1-AP) and rabbit anti-human E-cadherin antibody (GeneTex, Shanghai, China; Cat. No. GTX100443) were used as the primary antibodies and were diluted 1:200. Negative controls were stained following the same procedure, except that the phosphate-buffered saline (PBS) was used in place of the primary antibody.
To determine the immunoreactivity score (IRS) of Srx or E-cadherin, two independent researchers examined the sections in 10 representative microscopic fields. The IRS was calculated by multiplying the “staining intensity score” by the “fraction of positive cells”. The staining intensity was graded as follows: 0 = no staining, 1 = weak staining (light yellow), 2 = moderate staining (yellow brown) and 3 = strong staining (brown). The fraction of positive cells was scored as 0 = no positive cells, 1 = 10% or less, 2 = 11%–50%, 3 = 51%–80%, or 4 = 80% or more positive cells. The total score ranged from 0 to 12. The IRS was divided into three levels on the basis of the final score: negative immunoreactivity was defined as a total score of 0, low immunoreactivity as a total score of 1–4 and high immunoreactivity as a total score higher than 4. The immunostaining intensity of Srx was assessed as follows: 0 (0) = no staining, 1+ (1–4) = weak staining, 2+ (4–8) = moderate staining and 3+ (8–12) = intense staining. 19
Cell lines and cell culture
Human CSCC cell lines (SiHa) were obtained from Proteintech and were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, USA), supplemented with 10% fetal bovine serum (Life Technologies, USA) at 37°C in a humidified incubator containing 5% CO2. HeLa cells were obtained from the Tumour Laboratory Center of Chongqing Medical University and were cultured in RPMI 1640 (Life Technologies) and supplemented with 10% fetal bovine serum at 37°C with 5% CO2.
Transfection and selection of stable transfectants
A plasmid vector–mediated short hairpin RNA (shRNA) gene knockdown strategy was used to deplete the endogenous expression of Srx in HeLa and SiHa cells. All shRNA constructs included PLK0.1-non-target shRNA, human PLK0.1-ShSrx. Transfection cells with plasmid particles expressing the shRNA targeting in the protein-encoding region of the human Srx gene (ShSrx) effectively depleted the endogenous expression of Srx at both the transcript and the protein levels. In contrast, Srx expression was not affected by transfection of non-target control shRNA (ShNT). Lipofectamine 2000 reagent (Invitrogen, USA) was used to transfect plasmid vectors into HeLa and SiHa cells. Cells were maintained in a puromycin-containing medium to establish stable cells. The protein and messenger RNA (mRNA) levels of Srx, E-cadherin and vimentin in these cells were then evaluated by immunoblot analysis and quantitative real-time polymerase chain reaction (qRT-PCR).
RNA isolation and qRT-PCR
Total RNA from tissues was isolated using an RNA isolation reagent (TaKaRa, Dalian, China), according to the manufacturer’s instruction. The total RNA was then reverse-transcribed using the PrimeScript RT Reagent Kit (TakaRa). RT-PCR was performed with an SYBR® Premix Ex Taq TM II (Tli RNaseH Plus) kit (TakaRa) and CFX96 system (Bio-Rad, USA), based on the manufacturer’s instructions. The primers used were as follows: Srx: forward 5′-AAGGTGCA GAGCCTCGTGG-3′, reverse 5′-GCTACTGCAAGTCTGGTGTGGA-3′; E-cadherin: forward 5′-GGGTTATTCCTCCCATCAGC-3′, reverse 5′-GTCACCTTCAGCCATCCTGT-3′; Vimentin: forward 5′-AGGAAATGGCTCGTCACCTT-3′, reverse 5′-GGGTATCAACCAGAGGGAGTG-3′; and β-actin: forward 5′-CCACGAAACTACCTTCAACTCC-3′, reverse 5′-G TGATCTCCTTCTGCATCCTGT-3′.
The conditions were as follows: pre-denaturation at 95°C for 30 s, 35 cycles of denaturation (95°C for 5 s), annealing (55–60°C for 30 s) and extension (72°C for 1 min); final extension at 72°C for 10 min. The results of qRT-PCR were analysed by the 2−ΔΔCt method. Experiments were repeated three times.
Western blot analysis
Proteins were extracted from different cervical tissues by Protein Extraction Kit (Beyotime, Jiangsu, China). All the proteins were degenerated by boiling, via separation by the sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and transferred onto polyvinylidene fluoride membranes, blocked, and incubated with primary antibodies, including rabbit anti-human Srx antibody (1:500 dilution; Proteintech; Cat. No. 14273-1-AP), rabbit anti-human E-cadherin antibody (1:500 dilution; GeneTex; Cat. No. GTX100443), rabbit anti-human vimentin antibody (1:500 dilution; GeneTex; Cat. No. GTX100619) and mouse anti-human β-actin (1:2000 dilution; Abcam, USA; Cat No. ab205) at 4°C overnight. The membranes were then washed with Tris-buffered saline with Tween and then incubated with a secondary antibody. Blots were visualised using a BeyoECL Plus Western blot detection kit (Beyotime).
Transwell migration assay
Transwell chambers (Corning Costar, USA) coated without Matrigel (BD Biosciences, USA) were used for the migration assay. Stably Srx shRNA or ShNT cells and control cells of HeLa and SiHa cells were re-suspended with serum-free medium in a 24-well transwell invasion chamber, in which the upper surface was coated with Matrigel. Cells (1 × 105) were plated in the upper chamber of the transwells and allowed to invade across the membranes for 24 h. The invading cells were then fixed and stained with hexamethylpararosaniline. Five random fields were photographed, and the invaded cells were then counted by 10 high-power fields on each membrane.
Matrigel invasion assay
Transwell chambers (Corning Costar) coated with Matrigel (BD Biosciences) were used for the invasion assay. Different groups of HeLa and SiHa cells (2 × 105) were plated into the upper chambers. After 24 h of incubation, the cells were removed from the upper chamber by a cotton swab. The cells that migrated to the bottom of the filter were fixed and stained with hexamethylpararosaniline. The mean number of migrated cells was counted in triplicate.
Cell morphology was examined
To observe the cell morphology change, Stably Srx shRNA or ShNT cells and control cells of SiHa cells were re-suspended in six-well plates and cultured for 24 h. The cell morphology was then examined by optical microscopy (×120).
Statistical analyses
All experiments were repeated at least three times. The SPSS version 19.0 software (SPSS, USA) was used for the statistical analysis. Pearson’s chi-square or Fisher’s exact tests was used to compare the different levels of Srx or E-cadherin staining in different cervical tissues. The Spearman rank correlation test was used to analyse the correlation between Srx and E-cadherin expression in CSCC tissues. A one-way analysis of variance was used to analyse the expression of protein and mRNA levels. Data were presented as mean ± standard deviation (SD). Values of p <0.05 were considered statistically significant. Overall remission was analysed using Kaplan–Meier survival curves with log-rank (Mantel–Cox) test, and significance of p < 0.05 was considered significant for all statistical tests.
Results
Srx is overexpressed and E-cadherin is downregulated in human cervical lesion tissue
In our study, 178 cases of cervical tissues were evaluated by IHC, in which Srx-positive staining was rarely found in human NC tissues. However, strong staining of Srx was found in tumour samples from patients with squamous cell carcinoma. Positive staining of Srx was detected mainly in the cytoplasm and some in the cell membrane (Figure 1(a)). The staining was golden or brown in colour. The positive rate of Srx expression in CSCCs (80.5%) and HSILs (76.0%) were significantly higher than in NC (40%). The differences in the positive expression rate of Srx in three groups were statistically significant (p < 0.05; Table 1). E-cadherin strong staining was mainly detected in the cell membrane of the positive tissues and some in the cytoplasm. The results showed that the positive rate of E-cadherin in the NC group (96.7%) was higher than in HSILs (68.0%) and CSCC (29.3%) groups (p < 0.05; Table 1).These findings indicated that Srx expression was markedly upregulated and E-cadherin expression was downregulated in CSCC tissues than in NC tissues (Figure 1).
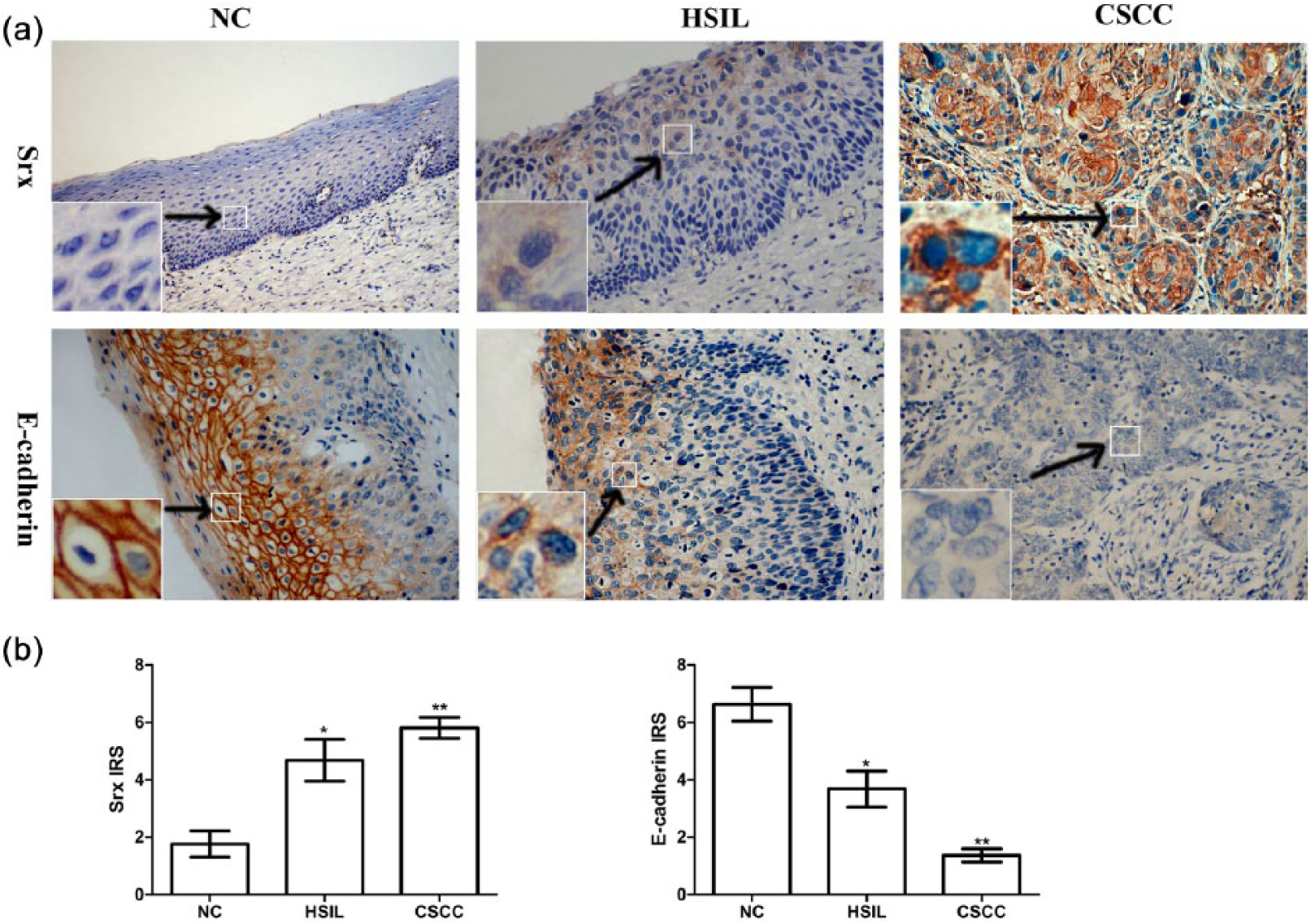
Expression of Srx and E-cadherin in cervical squamous cell carcinoma tissues. (a) IHC for the Srx and E-cadherin protein and negative control (×200) in paraffin-embedded sections of normal cervical (NC) tissues, high-grade squamous intraepithelial lesions (HSILs) and cervical squamous cervical cancer (CSCC) tissues. (b) IRS of Srx and E-cadherin expression in NC, HSIL and CSCC tissues.
Expression of Srx and E-cadherin in different cervical tissues.
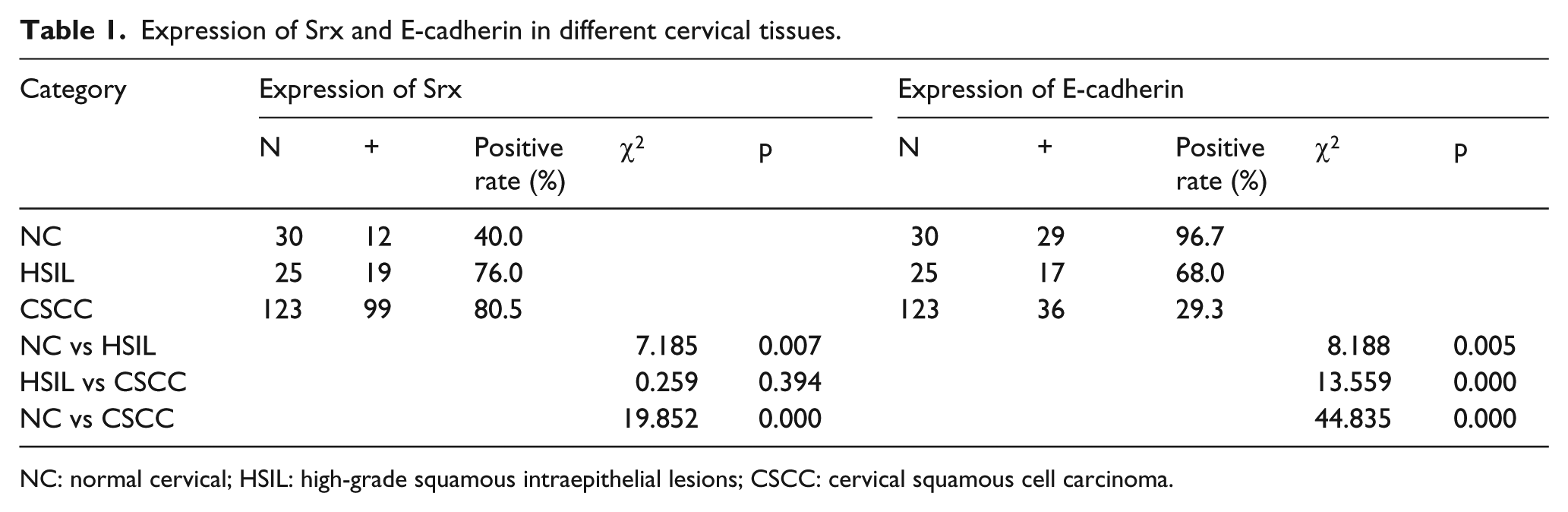
NC: normal cervical; HSIL: high-grade squamous intraepithelial lesions; CSCC: cervical squamous cell carcinoma.
Srx and E-cadherin expression levels are closely correlated with lymph node metastasis and invasion, and their expression is negatively correlated
The Srx protein was positively expressed in 80.5% of CSCC tissues. The positive rate of Srx in the lymph node metastasis group was 100%; however, the ratio was 77.6% in the non-metastasis group. Similarly, the ratio in infiltration of the haemal tube group was 95.7%, non-infiltration of the haemal tube group was 80.5% and the difference between these groups was statistically significant (p < 0.05). There were no significant associations between Srx-positive expression and age, clinical stage, tumour size, degree of histologic differentiation, or depth of cancer invasion (p > 0.05; Table 2). Among the 123 tumour cases, only 36 were detected with positive E-cadherin expression. The study showed the positive rate of E-cadherin in the group with tumour size bigger than 4 cm was 10.3% and, in the smaller size group, 35.1%. The positive ratio in the superior differentiation group (including well-differentiated and moderately differentiated squamous cell carcinoma) was 40.2%, whereas the ratio in the poorly differentiated group was just 2.8%. There was a significant difference between the lymph node metastasis group and the non-metastasis group (p < 0.05; Table 2). The result revealed that Srx and E-cadherin expression were negatively correlated (r = −0.224, p = 0.013) in CSCC.
The relationship between the expression of the two proteins and cancer clinical pathological parameters.

Correlation between the expression of Srx and the overall remission rate in CSCC patients
To further prove the clinical significance of Srx expression in patients with CSCC, a Kaplan–Meier test was adopted. In the Kaplan–Meier survival analysis, Srx expression scores were subgrouped into two categories: weak Srx staining, with scores ranging from 0 to 1+; and strong Srx staining, with scores ranging from 2+ to 3+. The results showed that the remission time (tumour-free status after surgery) of the group with Srx strong staining was significantly shorter than that of the Srx weak staining group. The group with strong Srx staining suffered more relapse and metastasis than the weak group. A log-rank test indicated that the difference between the two groups was statistically significant (χ2 = 4.029, p = 0.0447; Figure 2).

Remission rate of Srx protein expression in cervical squamous cell carcinoma patients.
Srx knockdown efficiency was detected in HeLa and SiHa cells
To observe the effects of Srx in cervical cancer cell lines, we selected two cervical cancer cell lines, HeLa and SiHa transfected with Srx shRNA and obtained stable cell lines. Srx expression was detected in the ShNT, ShSrx stable cells and control cells by qRT-PCR (Figure 3(a)) and western blot (Figure 3(b)) in both the cell lines.

Srx knockdown efficiency was shown both in mRNA and protein level in HeLa and SiHa cells lines. (a) qRT-PCR analyses showing the expression of Srx gene in different group of HeLa cells (left panel) and SiHa cells (right panel). (b) Western blot analysis showing the expression of Srx in HeLa cells (left panel) and SiHa cells (right panel). The results were expressed as mean ± SD.
In HeLa cells, compared to the ShNT and control cells, the Srx mRNA level was significantly reduced by nearly 65% (Figure 3(a), left panel) and the protein level decreased nearly 57% (Figure 3(b), left panel). In SiHa cells, the Srx mRNA level was significantly reduced by nearly 55% (Figure 3(a), right panel) and the protein level decreased nearly 70% (Figure 3(b), right panel). These data suggested that the expression of Srx was significantly downregulated both in mRNA and protein level in the both the cell lines.
Knockdown of Srx decreased the invasion and migration capacities of cervical cancer cells
Cell invasion and migration are the hallmarks of cancer metastasis. To study the function of Srx in the aggressive malignant behaviours of cervical cancer, we performed both Matrigel invasion assay and transwell migration assay to measure the invasion and migration ability of HeLa and SiHa cells following silencing of Srx (Figure 4(a) and (c)). The results showed that downregulation of Srx significantly decreased invasion and migration cell numbers in both the cell lines compared with that of control and ShNT (Figure 4(b) and (d)) cells (p < 0.05). There were no significant differences between the control and ShNT cells. These results indicate that Srx knockdown suppresses the invasive and migratory capacities of HeLa and SiHa cells.

Downregulation of Srx reduces invasion and migration in HeLa and SiHa cells. (a) (upper panels) and (c) (upper panels) Matrigel invasion assay was performed in HeLa and SiHa cells with Srx knockdown. (a) (lower panels) and (c) (lower panels) The effects of Srx downregulation on the migration in HeLa and SiHa cells was assessed by Transwell chamber assay without Matrigel coating. (b) After 24 h of incubation, the number of cell invasion and migration in HeLa cells was counted under the microscope (×120). (d) After 24 h of incubation, the number of invaded cells and migrated cells of SiHa cells was counted under the microscope (×120). The values are expressed as the mean ± SD.
Knockdown of Srx increased the expression of the epithelial marker and decreased the mesenchymal marker
The correlation between Srx and E-cadherin expression in CSCC tissues suggested that Srx might play a role in the process of EMT. To test this hypothesis, we selected SiHa cells, which were established from fragments of a primary tissue sample obtained after surgery from a patient who suffered from CSCC. We performed qRT-PCR and western blot to detect the levels of the epithelial marker E-cadherin and the mesenchymal marker vimentin. Results showed that E-cadherin was increased in ShSrx cells, compared with the control and ShNT cells (p < 0.05), both in mRNA and protein levels (Figure 5). These results in cell lines were similar to the cervical tissues. Conversely, the expression of mesenchymal marker vimentin markedly reduced in Srx shRNA transfected SiHa cells (p < 0.05; Figure 5). These data indicated that Srx was involved in the EMT process in CSCC cells and the knockdown of Srx repressed expression of EMT-related proteins. Meanwhile, Srx knockdown suppressed the invasive and migratory capacities of SiHa cells (Figure 4(c) and (d)). Silencing Srx expression could partly reverse mesenchymal character. As shown in Figure 5(d), cells in the ShSrx group were more cubical in shape than the control group cells. Compared with the control group, Srx downregulation reversed the EMT morphological change.

Srx affected EMT markers and morphology change in SiHa cells. (a) Quantitative RT-PCR showed the expression of EMT-related genes in control, ShNT and ShSrx cells. (b and c) Western blot analysis of the protein levels of Srx, E-cadherin and vimentin in control cells, stable ShNT and ShSrx cells. The results were expressed as mean ± SD (*p < 0.05). (d) The effect of low expression of Srx was examined as cell morphological changes by optical microscopy (×120).
Discussion
Srx is a low-molecular-weight, sulfur-containing protein whose main function is to participate in regulating the reversible sulfinic modification of 2-Cys Prxs enzymes. Srx is encoded by a single gene, and no other isoforms have been found among species varying from bacteria and plants to vertebrates. 20 An evolutionary tree reveals that Srx is present and clusters distinctly in animals, fungi, plants and some protists. 21 These studies have shown that Srx has a critical oncogenic role in cancer development.20,22
In this study, we examined the expression of Srx and E-cadherin in CSCC tissues by IHC. We found that Srx was upregulated in cervical cancer tissues and HSILs compared with NC tissues. Srx-positive expression was mainly detected in the cytoplasm and a few in the cell membrane, which was similar to Merikallio et al.’s 23 findings. By contrast, E-cadherin expression was found to be decreased in cancer tissues compared with NC tissues, and it was expressed in the membrane and cytoplasm of cells of cervical tissues. These observations agree with previous studies showing that E-cadherin was downregulated in cervical cancer.24,25 In addition, Srx and E-cadherin expression were negatively correlated.
We also examined the association of the Srx and E-cadherin expression with the clinicopathological characteristics of CSCC. We have learned that Srx expression was associated with nodal metastasis and infiltration of the haemal tube. However, no significant differences were shown in age, clinical stage, tumour size, histologic differentiation and depth of cancer invasion in cervical cancer. Srx is highly expressed in human lung cancer and is required for cell migration and invasion of lung cancer cells, 22 which provides us evidence that Srx is associated with tumour invasion and metastasis. Correlation between Srx expression and clinical prognosis of patients had been studied in CSCC patients undergoing surgery. Follow-up on these patients indicated that the difference in disease-free survival rate between the stronger and weaker Srx expression groups was statistically significant, and those with a strong expression of Srx had a worse prognosis than those with a weak expression of Srx. The stronger Srx expression groups had higher rates of recurrence and metastasis. Meanwhile, we also found that E-cadherin expression was associated with lymph node metastasis in cervical cancer patients, consistent with previous studies showing that the downregulation of E-cadherin is associated with the invasiveness and metastasis of cervical cancer.4,26,27 We assume that Srx may promote cervical cancer metastasis via the regulation of E-cadherin expression.
To verify this hypothesis, a knockdown strategy was used to deplete the endogenous expression of Srx in SiHa cells. We then examined the protein and mRNA level of Srx and E-cadherin in these cells by immunoblot analysis and qRT-PCR. The results showed that knockdown of Srx by shRNA increased the expression of E-cadherin in both mRNA and protein levels. As we all know, EMT is a key step in cancer invasion and metastasis and E-cadherin is the most important marker of EMT. Increased invasion and migration is a key feature of cells that undergo EMT. 28 In our study, Srx knockdown suppressed the invasive and migratory capacities of HeLa and SiHa cells. We proposed that Srx promotes invasion and migration of CSCC by EMT. To verify this, a mesenchymal marker, vimentin, was tested at both transcript and protein levels by qRT-PCR and western blot analysis. Results showed that vimentin was markedly reduced in Srx shRNA transfected SiHa cells. Furthermore, Matrigel invasion and transwell migration assays were performed to detect the invasion and migration ability of SiHa cells. The cells of the ShSrx group were less invasive than the ShNT and control group in the Matrigel invasion assay and scratch assay. Therefore, knockdown of Srx by shRNA reverses the metastatic behaviour of cervical cancer cells. Several studies have provided evidence of E-cadherin and vimentin regulating EMT during tumourigenesis and cancer progression. 29
Our study provides evidence that Srx may promote cervical cancer tumourigenesis and cancer progression by EMT. Several signalling pathways are important for cancer development by EMT, including the tumour growth factor (TGF)-β, Wnt, Notch, mitogen-activated protein kinase (MAPK) and fibroblast growth factor (FGF) pathways. Qiou Wei et al.
22
revealed that the Srx–Prx IV axis is critical for lung cancer maintenance and metastasis. From their previous study, we came to know that Srx can regulate c-Jun
Footnotes
Acknowledgements
The authors gratefully thank Department of University of Kentucky College of Medicine in the United States and Tumor Central Laboratory of Chongqing Medical University for providing equipment support. H.W. designed the study. X.C., Q.L., K.L., X.Y. and S.W. collected and checked the information of eligible articles included in this meta-analysis. X.C. and H.W. analysed the data. X.C. wrote the main article text. H.W. revised the manuscript. All authors reviewed and approved the manuscript. X.C. and K.L. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
