Abstract
The protocadherin 7 is a member of the protocadherin family that expressed aberrantly in many types of human cancers. However, its expression, function, and underlying mechanisms are little known in gastric cancer. In this study, we detected protocadherin 7 expression in gastric cancer tissues and non-tumorous gastric mucosa tissues by real-time quantitative polymerase chain reaction and immunohistochemistry. The association of protocadherin 7 expression with the clinicopathological characteristics and the prognosis was subsequently analyzed. MTS ((3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium)) and transwell assays were performed to assess the effect of protocadherin 7 on proliferation, migration, and invasion in gastric cancer cell lines. Moreover, real-time quantitative polymerase chain reaction and western blot were used to detect the expression of epithelial–mesenchymal transition markers. Protocadherin 7 expression was decreased gradiently from normal tissue to gastric cancer, especially in gastric cancer tissue with lymph node metastasis. Low expression of protocadherin 7 was significantly associated with Lauren’s classification (p = 0.0005), lymph node metastases (p = 0.0002), and tumor node metastasis stage (p = 0.0221), as well as poor prognosis (p < 0.05). Furthermore, down-regulation of protocadherin 7 in gastric cancer cell lines significantly increased their migration and invasion abilities (both p < 0.05), while it had no influence on the gastric cancer cell proliferation (p > 0.05). Additionally, our results demonstrated that E-cadherin expression was down-regulated in gastric cancer cells with protocadherin 7 depletion. Our data indicated that protocadherin 7 may play important roles in the invasion and metastasis of gastric cancer, and protocadherin 7 could suppress cell migration and invasion through E-cadherin inhibition. Protocadherin 7 can serve as a novel biomarker for diagnostic and prognosis in patients with gastric cancer.
Introduction
Gastric cancer (GC) is the fourth common gastrointestinal cancer, with a high rate of cancer-related mortality in all kinds of cancers in the world. 1 China is one of the high incidence areas of GC in worldwide. 2 The main factors leading to poor prognosis of GC are invasion and metastasis. 3 Although there are considerable studies on GC, the mechanism of its metastasis or invasion is poorly understood. 4 In this study, we sought to identify novel factors involved in the development and progression of GC.
The protocadherins (PCDHs) are a group of calcium-dependent adhesion proteins that constitute the largest subfamily of the cadherin superfamily. 5 Cadherins are calcium-dependent transmembrane glycoproteins, which form a large family of cell–cell adhesion molecules. 6 To date, more than 70 different PCDH genes have been identified. They are divided into two groups: clustered PCDHs and non-clustered PCDHs. 7 Many studies indicated that non-clustered PCDHs can enhance cell–cell adhesion and other molecules’ function. 8 Moreover, some non-clustered PCDHs have been suggested as tumor-suppressor candidates in human tumors, including PCDH8, PCDH9, and PCDH17.9–11
PCDH7 belongs to the PCDHs and locates at human chromosome 4p15. In human, PCDH7 expresses predominantly in the brain and heart and few are found in the stomach, thyroid, spinal cord, or placenta. 12 PCDH7 encodes an integral membrane protein which plays roles in cell–cell recognition and adhesion. 13 It is well known that loss of adhesion is the first step for tumor invasion and metastasis. Recent studies suggested that PCDH7 can participate in the development of several tumor types, such as breast cancer, colon carcinoma, and prostate cancer.14–16 However, no evidence has been found in its potential role in GC.
Materials and methods
Clinical specimens
Tissue samples for immunohistochemistry (IHC), including 119 cases of GC tissues and 75 cases of non-cancerous gastric mucosa tissues (at least 3 cm away from the surgical border), were collected from the Department of Pathology, Qilu Hospital of Shandong University. None of these patients received any radiotherapy, chemotherapy, or radiofrequency ablation before surgery. The clinicopathological characteristics were also recorded. Staging and differentiation of the tumor were based on the tumor node metastasis (TNM) standard (Union for International Cancer Control, 2002) and Lauren’s method, respectively. GC tissues and non-cancerous tissues were immediately fixed in 10% Formalin Neutral Fixative Manual. The fresh specimens for real-time quantitative polymerase chain reaction (RT-qPCR), which include 47 GC tissues and 25 non-cancerous gastric mucosal tissues, were obtained from Shandong Province Hospital and stored in liquid nitrogen. The study was approved by the Ethical Committee of Shandong University (Jinan, China). Informed consents were obtained from all the patients.
IHC
Immunodetection was performed using the SP Kit (Zhongshan Goldenbridge Biotechnology, Beijing, China). Sections were dewaxed, rehydrated, and then undergone antigen retrieval in ethylenediaminetetraacetic acid (EDTA) buffer (pH 8.0). Next, sections were treated with 3% H2O2 and incubated with rabbit polyclonal antibody PCDH7 (dilution 1:50; Abcam, Cambridge, UK) in a humidified chamber at 4°C overnight. Then, sections were rewarmed at 37°C, incubated with biotinylated IgG secondary antibody and horseradish peroxidase conjugated streptavidin, followed by chromagen detection with DAB (3,3′-diaminobenzidine). After stained with hematoxylin, tissues were dehydrated and mounted. For negative controls (NCs), the primary antibody was replaced with phosphate-buffered saline (PBS).
The IHC results for the PCDH7 protein were evaluated according to semi-quantitative immunoreactivity scores.17,18 The intensity score was defined as follows: 0, absence of signal; 1, low-intensity signal (light brown); 2, moderate-intensity signal (brown); and 3, high-intensity signal (dark brown). The relative number of positive cells was counted in five randomly chosen 400× microscopic fields (a total of 500 cells were counted in five fields). The percentage score was defined as follows: <5% = 0; 6%–25% = 1; 26%–50% = 2; 51%–75% = 3; >75% = 4. For each specimen, the final score was obtained by the multiplication of the intensity score and the percentage score. The final score ≥2 was defined as PCDH7 positive.
Cell culture
The human GC cell lines BGC-823 and MKN-45 were purchased from the Shanghai Cancer Institute. All cells were cultured in RPMI 1640 (Gibco), added with 10% fetal bovine serum (FBS). Cell lines were maintained at 37°C in a humidified atmosphere of 5% CO2 and were passaged by trypsinization after reaching 80%–90% confluence.
Small interfering RNA transfection
The siRNA against PCDH7 (siPCDH7) and the NC RNA were designed and purchased from RiboBiO (Guangzhou, China). According to the manufacturer’s instructions, GC cells were transfected with siPCDH7 or NC by X-treme GENE transfection reagent (Roche Applied Science, USA). Briefly, 1 × 105 GC cells were seeded in 12-well plates and incubated 12 h before transfection. Then, 4 µL X-treme GENE and 100 nM siRNA were added to each well. Cells were harvested after 48 h. The knockdown efficiency of siRNA was detected by RT-qPCR.
RNA extraction and RT-qPCR
Total RNA from tissues or cell lines were extracted by Trizol (Invitrogen, Carlsbad, CA, USA). Before RNA extraction, fresh gastric tissues were microdissected by freezing microtome. Then, according to the manufacturer’s protocol, RNA was converted to cDNA using the PrimeScript reverse transcription (RT)-PCR kit (Takara, Japan). To analyze mRNA expression level, RT-qPCR was performed using FastStart Universal SYBR Green Master (ROX, Roche, Germany) and 7900HT Fast Real-Time PCR systems (Applied Biosystems). Primers for RT-qPCR are summarized in Supplementary Table S1.
Cell proliferation assay
CellTiter 96® AQueous One Solution Cell Proliferation Assay ((3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) (MTS), Promega) was used to detect the cell proliferation rate. 5 × 103 cells were seeded into 96-well culture plates and incubated for 24, 48, and 72 h, respectively. After washed with PBS, 80-µL serum-free medium and 20-µL MTS solution were added to each well and cell plates were incubated at 37°C for 1 h in the dark. A microplate reader (Bio-Rad, Foster City, CA, USA) was used to measure the absorbance at 490 nm.
Invasion and migration assay
Cancer cell invasion/migration assays were performed using a 24-well transwell insert with 8.0-µm pores (Corning, USA) with/without Matrigel matrix (BD Science, USA), respectively. 1 × 104 cells were resuspended in 200 µL serum-free medium and seeded to the upper chambers. The lower chambers were filled with 600 µL RPMI 1640 medium with 10% FBS. After incubation at 37°C for 24 h, non-invading or non-migrating cells were removed by scrubbing gently using a cotton-tipped swab. The cells that invaded through the membrane to the bottom chamber were fixed in 4% paraformaldehyde for 10 min and then stained with 0.5% crystal violet for 15 min at room temperature. The number of invading or migrating cells was determined by counting five random fields for each insert and calculated as the mean number of cells per field.
Western blot
Total protein was extracted from cells which were transfected with siRNA or NC for 48 h. BCA Protein Assay Kit (Blue Skies, Shanghai, China) was used to determine the protein concentration. The proteins were separated and transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore, Billerica, MA, USA) for immunoblots as previously described. 19 The membranes were incubated with primary antibodies against PCDH7 (1:200, Abcam), E-cadherin (1:1000, Cell Signaling), β-catenin (1:1000, Sangon Biotech), Vimentin (1:1000, Cell Signaling), Snail (1:500, Proteintech), and β-actin (1:1000, OriGene), and then with secondary antibodies and developed with the chemiluminescence method.
Statistical analyses
SPSS 21.0 (SPSS, Chicago, IL, USA) and GraphPad Prism 5 (GraphPad Software, Inc., San Diego, CA, USA) were used to perform statistical analysis. The differences between the two groups were analyzed by Student’s t-test. The relationship between PCDH7 expression level and clinicopathological variables was detected by chi-square test. The Kaplan–Meier method and log-rank test were performed to analyze the survival rates. The Cox proportional hazard model was constructed for the univariate and multivariate analysis of the prognostic value of PCDH7 expression. The diagnostic value of PCDH7 expression was determined by receiver operating characteristics (ROC) curve analysis. p-values <0.05 were considered to be statistically significant.
Results
Expression of PCDH7 was down-regulated in human GC tissues
To assess the potential role of PCDH7 in GC, we examined the mRNA level of PCDH7 in an independent cohort of frozen specimens including 47 cases of GC tissues and 25 cases of non-cancerous gastric mucosal tissues by RT-qPCR. Compared with noncancerous gastric mucosal tissues, PCDH7 mRNA were down-regulated in GC tissues (Figure 1(a), p < 0.0001). Meanwhile, the expression of PCDH7 protein was analyzed by IHC. A total number of 194 paraffin-embedded gastric samples, which included 119 cases of GC tissues and 75 cases of non-cancerous gastric mucosa tissues, were used for IHC. In the 75 cases of non-cancerous gastric mucosa tissues, there were 55 cases of intraepithelial neoplasia gastric tissues and 20 cases of normal gastric tissues. Typical immunostaining profiles of PCDH7 in gastric samples are shown in Figure 1(c)–(f). In GC tissues, PCDH7 presented negative immunoreactivity in most cases and the positive rate of PCDH7 was only 32.8%. In contrast, the positive rate of PCDH7 was high in non-cancerous gastric mucosa. PCDH7 positive was detected in all of the normal gastric tissue (20/20, 100%) and 35 cases of intraepithelial neoplasia gastric tissue (35/55, 63.6%). These data suggested that PCDH7 expression was stepwisely down-regulated from normal tissue to intraepithelial neoplasia tissue and then to GC tissues (Table 1, chi-square, p < 0.05). In addition, our result showed that PCDH7 expression in GC tissues with lymph node metastasis (LNM) was notably lower than that without LNM (Figure 1(b), t-test, p < 0.05), indicating that PCDH7 may be involved in the metastasis of GC.

PCDH7 expression was lower in gastric cancer tissues than non-cancerous mucosal tissues. (a) Quantitative determination of PCDH7 mRNA by real-time PCR in gastric cancer tissues and non-cancerous mucosal tissues. The level of PCDH7 mRNA in gastric tumor tissues was significantly lower than that in non-cancerous gastric mucosal tissues (p < 0.0001). (b) Immunohistochemical analysis showed that PCDH7 protein was progressively down-regulated from non-cancerous tissues, via cancer tissues without lymph node metastasis, to cancer tissues with lymph node metastasis (all p < 0.05). (c–f) Representative images of PCDH7 protein expression in gastric cancer and non-cancerous tissues. PCDH7 positive staining was predominantly localized in the cytoplasm and cytomembrane of gastric cells. PCDH7 showed strong positive expression in noncancerous gastric mucosa tissues (c), moderate expression in intestinal-type gastric adenocarcinomas (d), but negative expression in diffuse-type gastric adenocarcinomas (f). PCDH7 was positive in the mucosa epithelial cells, while negative in cancer glandular epithelium (e).
Expression of PCDH7 protein in gastric cancer and mucosal tissues.

PCDH7: Protocadherins 7.
Difference between gastric tumor tissue and gastric intraepithelial neoplasia tissue.
Difference between gastric intraepithelial neoplasia tissue and gastric normal tissue.
Difference between gastric normal tissue and gastric tumor tissue.
Next, we constructed receiver operator characteristic (ROC) curves to evaluate the diagnostic value of PCDH7 in GC. Between GC and non-cancerous groups, the area under the curve (AUC) was 0.938, suggesting that PCDH7 expression could be used to discriminate these two groups (Figure 2(a), p = 0.000). Moreover, the ROC curves indicated that the PCDH7 expression could discriminate the case with LNM from those without LNM with an AUC of 0.695 (Figure 2(b), p = 0.001).

PCDH7 could be used as diagnostic and prognostic markers for gastric cancer. (a) Area under the ROC curve for PCDH7 in differential diagnosis of gastric mucosa and gastric cancer (AUC = 0.938, p = 0.000). The ROC curves showed that PCDH7 expression could be used to discriminate gastric mucosa and gastric cancer tissues. (b) Area under the ROC curve for PCDH7 in differential diagnosis of gastric cancer with and without lymph node metastasis (AUC = 0.695, p = 0.001). The ROC curves demonstrated that PCDH7 could be used as a diagnostic marker for lymph node metastasis and (c, d) Relationship between PCDH7 expression and patients’ survival in gastric cancer. Kaplan–Meier curves showed that the patients with positive expression of PCDH7 had a better overall survival (c, p = 0.049) and disease-free survival (d, p = 0.0388) than those with negative PCDH7 expression.
Relationship between PCDH7 expression and clinicopathological parameters in GC
To further evaluate the role of PCDH7 in GC, we analyzed the relationship between PCDH7 expression and clinicopathological factors. As shown in Table 2, the PCDH7 expression was significant negatively associated with Lauren’s classification (p = 0.0005), lymph node metastases (p = 0.0002), and TNM stage (p = 0.0221). However, there was no statistical difference between PCDH7 expression and age (p = 0.3009), gender (p = 0.2835), tumor size (p = 0.4465), or distal metastasis (p = 0.2219) (Table 2). All these data suggested that PCDH7 could participate in the progression of GC.
Relationship between PCDH7 expression and clinicopathological characteristics in patients with gastric cancer.

PCDH7: Protocadherins 7; TNM: tumor node metastasis.
Statistically significant (p < 0.05).
Low expression of PCDH7 correlates with poor prognosis
Survival analysis demonstrated that patients with low PCDH7 expression had a poorer overall survival (OS) and disease-free survival (DFS) than those with high PCDH7 expression (Figure 2(c) and (d), p = 0.0490 and p = 0.0380, respectively). To explore the influence of PCDH7 expression and pathological parameters on the patient outcome, we carried out univariate and multivariate analyses through Cox proportional hazard model. The univariate analysis uncovered that PCDH7 expression, depth of invasion, LNM, distal metastasis, and tumor stage were prognostic factors for OS and DFS of GC patients (Table 3, p < 0.05). Multivariate analysis suggested that distal metastasis and tumor stage were independent prognostic predictors for OS and DFS (Table 3, p < 0.05). However, our study revealed that PCDH7 expression was not an independent prognostic factor for OS or DFS of patients with GC. Despite that, it still favors PCDH7 as a protective factor (OS, hazard ratio (HR) = 0.842; DFS, HR = 0.668) for patient survival (Table 3). To sum up, our results demonstrated that low expression of PCDH7 was significantly associated with poor outcome.
Univariate and multivariate analyses for the prognostic value of PCDH7 expression on overall survival and disease-free survival after surgery.

PCDH7: Protocadherins 7; HR: hazard ratio; CI: confidence interval.
PCDH7 had no effect on cell proliferation in GC cells
To study the roles of PCDH7 in GC cell proliferation, we inhibit the expression of PCDH7 using small interfering RNA (siRNA) in MKN-45 and BGC-823 GC cells. Transfection efficiency was measured using RT-qPCR. As shown in Figure 3, the levels of PCDH7 expression were effectively reduced in the siRNA group (Figure 3(a) and (b)). Cell proliferate assay demonstrated that knockdown of PCDH7 could not influence cell proliferation obviously compared with NCs in two GC cell lines (Figure 3(c) and (d), both p > 0.05).
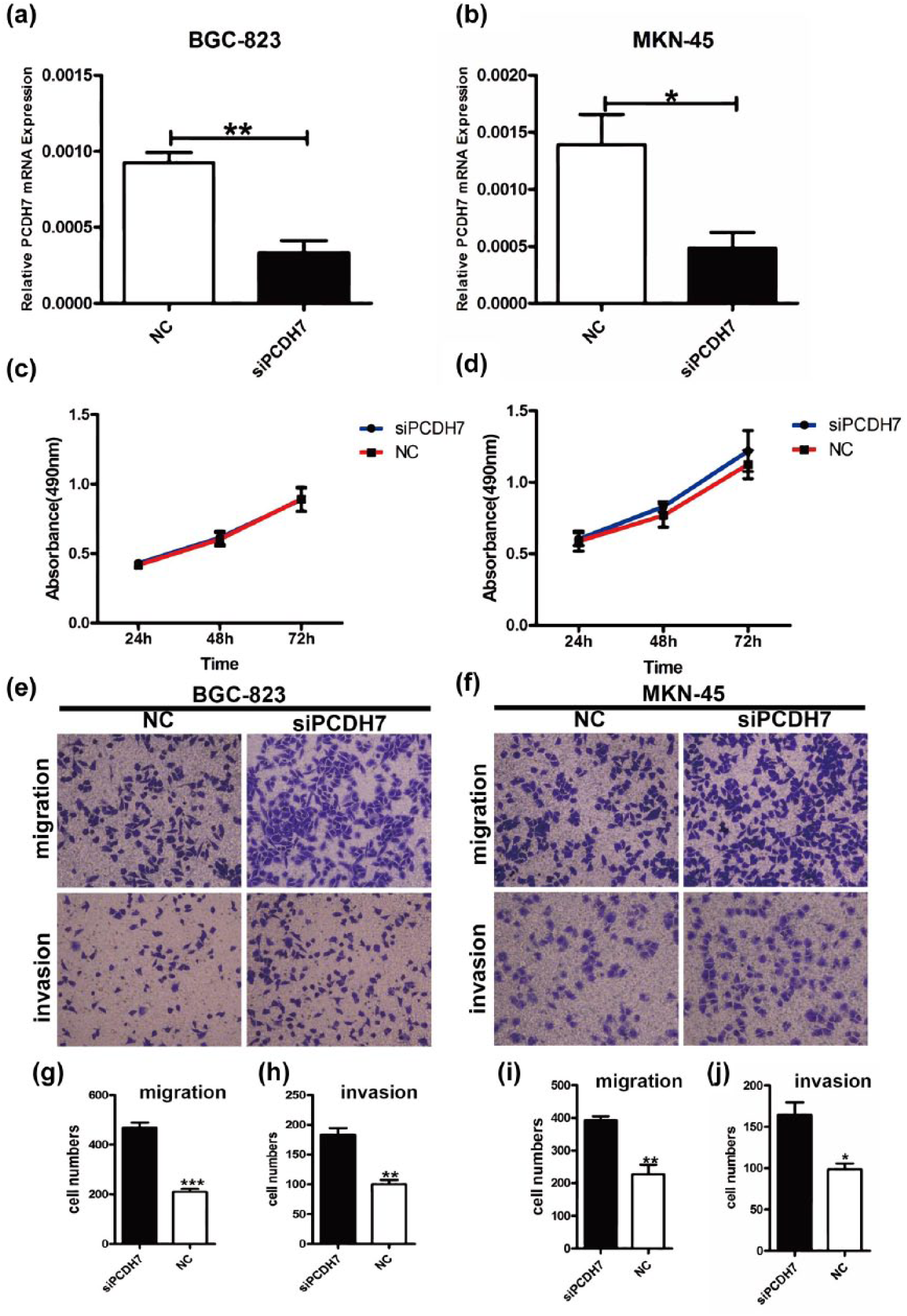
Inhibition of PCDH7 expression enhanced migration and invasion of gastric cancer cells in vitro. (a, b) Gastric cancer cells were transfected with siPCDH7, and real-time PCR was performed to monitor transfection efficiency. (c, d) Cell proliferation rates were measured using MTS assay in BGC-823 and MKN-45 cells following PCDH7 silencing and the results showed that PCDH7 knockdown had no apparent inhibition in the proliferation of gastric cancer cells. (e–i) Transwell assays showed that PCDH7 knockdown increased the migration and invasion ability of BGC-823 (e, g, h) and MKN-45 (f, i, j) cells.
PCDH7 inhibition increases GC migration and invasion
Given that PCDH7 expression was negatively correlated with metastasis to lymph node, we investigated the roles of PCDH7 on cell migration and invasion in GC. Our results showed that after knocking down PCDH7, BGC-823 and MKN-45 cells displayed significantly increased migration and invasion capability, compared with the cells transfected with NC (Figure 3(e)–(j), p < 0.05). Thus, these results suggested that PCDH7 could inhibit cell migration and invasion.
PCDH7 knockdown influenced E-cadherin expression
To explore the possible mechanism of PCDH7 suppression on migration and invasion, the expression of key biomakers in epithelial–mesenchymal transition (EMT) was first detected by RT-qPCR in GC cells. The results suggested that the mRNA expression level of E-cadherin was decreased in BGC-823 and MKN-45 cells after PCDH7 knockdown, whereas no effect was found on the expression of N-cadherin, β-catenin, Vimentin, Twist, Snail, Zeb1, or Zeb2 (Figure 4(a) and (b)). To further confirm this result, we performed western blot. The results showed that PCDH7 knockdown could inhibit the protein expression of E-cadherin and had no effect on β-catenin, Vimentin, or Snail in BGC-823 cells (Figure 4(c) and (e)). Similar results were obtained in MKN-45 cells (Figure 4(d) and (f)). These results indicated that PCDH7 inhibition may promote migration and invasion by reducing the expression of E-cadherin.

Knockdown of PCDH7 inhibited the expression of E-cadherin. (a, b) Real-time PCR analyses showed that PCDH7 knockdown decreased E-cadherin expression in BGC-823 (a) and MKN-45 (b) cells (*p < 0.05). The expression of N-cadherin, β-catenin, Vimentin, Twist, Snail, Zeb1, or Zeb2 did not show significant changes. (c–f) Western blot results showed that with the decrease in the PCDH7, the expression of E-cadherin protein in BGC-823 (c, e) and MKN-45 (d, f) cells were down-regulated (*p < 0.05, **p < 0.01), while the expression of β-catenin, Vimentin, and Snail showed no obvious changes.
Discussion
Recently, many studies have focused on the role of PCDHs during tumor development and progression. PCDH9 could inhibit EMT and cell migration in hepatocellular carcinoma. 20 Zhang et al. 21 had reported that PCDH8 was a gastric tumor suppressor. As a member of the same subfamily, PCDH7 was first identified by Kenichi Yoshida and denoted BH-Pcdh in 1998. 12 Previous studies had showed that PCDH7 was aberrantly expressed in some tumors, such as hepatocellular carcinoma, colon carcinoma, and prostate cancer.15,16,22 For example, low expression of protocadherin 7 (PCDH7) was associated with poor prognosis and could be used as a potential prognostic biomarker in primary non-muscle invasive bladder cancer. 18 However, the expression and biological function of PCDH7 in GC have not yet been explored.
In this study, we first investigated the mRNA expression level of PCDH7 in GC tissues and non-cancerous mucosal tissues. The results demonstrated that PCDH7 mRNA was significantly down-regulated in GC tissues compared to non-cancerous mucosal tissues. Subsequently, the protein expression of PCDH7 exhibited a gradual reduction from normal tissue to intraepithelial neoplasia and to GC. PCDH7 expression was much more lower in GC with LNM. Moreover, we identified that PCDH7 expression was useful to identify different GC tissues through ROC curves.
In addition, our study showed that PCDH7 expression was significantly inverse or negative correlated with Lauren’s classification, lymph node metastases, and TNM stage. More importantly, survival analysis demonstrated that patients with low PCDH7 expression always had poor outcome, which was consistent with the studies of PCDH7 in bladder cancer and prostate cancer.16,18 Unfortunately, univariate and multivariate Cox regressions displayed that PCDH7 was not an independent prognostic factor, but it still indicated that PCDH7 could serve as a protect factor (HR < 1) for GC patients. All these findings demonstrated that PCDH7 could suppress the progression of GC.
The retrieval of published articles revealed that the function of PCDH7 in human cancer was largely unknown.14,23,24 Base on that, we detected the function of PCDH7 in GC. Our results proved that PCDH7 inhibition significantly increased cell migration and invasion abilities in GC in vitro, whereas PCDH7 down-regulation had no effect on GC proliferation. Therefore, we concluded that PCDH7 could serve as tumor suppressor gene in GC. In contrast, Li et al. 14 found that PCDH7 could promote migration and invasion and induced bone metastasis in breast cancer. We considered that differences between breast cancer and GC tissue led to discrepant results. This discrepant phenomenon was also observed in other PCDHs, such as PCDH10. Echizen et al. 25 had reported that PCDH10 could promote proliferation and metastasis in glioblastoma. Conversely, PCDH10 was confirmed to suppressed cell proliferation and migration in GC 26 and colorectal cancer. 27
EMT is a process in which epithelial cells may undergo phenotypic switch to acquire mesenchymal phenotype. 28 The occurrence of EMT plays a vital role in tumor progression. 29 In this process, epithelial markers are repressed, such as E-cadherin. Meanwhile, mesenchymal markers (e.g. vimentin and α-SMA) are up-regulated and cells are loosely arranged with strong mobility. 30 In this study, we analyzed the associations between PCDH7 expression and EMT. RT-qPCR and western blot showed that E-cadherin were down-regulated after PCDH7 knockdown. Many studies demonstrated that the loss of E-cadherin was an essential step during the process of EMT. 31 Thus, we believed that PCDH7 inhibition could promote GC metastasis/invasion by down-regulating E-cadherin. However, the precise regulatory mechanisms between PCDH7 and E-cadherin were unknown. As is well known, E-cadherin could be directly suppressed by Snail. 32 But in our study, PCDH7 knockdown had no effect on the expression of Snail. This indicated that PCDH7 may regulate the expression of E-cadherin through other means and further studies are needed to solve this issue.
In summary, our data confirmed that the low expression of PCDH7 is a pivotal event in the progression of GC, and PCDH7 knockdown could promote cell migration and invasion through inhibiting E-cadherin expression. Therefore, PCDH7 could be taken as a potential diagnostic and prognostic biomarker for GC.
Footnotes
Author contribution
Hong-Fang Chen and Ran-Ran Ma contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from National Natural Science Foundation of China (grant nos 81172351 and 81372856) and Taishan Scholars Program of Shandong Province (ts201511096).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
