Abstract
5-Fluorouracil has been considered as a cornerstone therapy for colorectal cancer; however, it suffers from low therapeutic response rate and severe side effects. Therefore, there is an urgent need to increase the clinical efficacy of 5-fluorouracil. Recently, fish oil rich in n-3 polyunsaturated fatty acids has been reported to chemosensitize tumor cells to anti-cancer drugs. This study is designed to understand the underlying mechanisms of synergistic effect of fish oil and 5-fluorouracil by evaluation of tumor cell–associated markers such as apoptosis and DNA damage. The colon cancer was developed by administration of N,N-dimethylhydrazine dihydrochloride and dextran sulfate sodium salt. Further these animals were treated with 5-fluorouracil, fish oil, or a combination of both. In carcinogen-treated animals, a decrease in DNA damage and apoptotic index was observed. There was also a decrease in the expression of Fas, FasL, caspase 8, and Bax, and an increase in Bcl-2. In contrast, administration of 5-fluorouracil and fish oil as an adjuvant increased both DNA damage and apoptotic index by activation of both extrinsic and intrinsic apoptotic pathways as compared to the other groups. The increased pro-apoptotic effect by synergism of 5-fluorouracil and fish oil may be attributed to the incorporation of n-3 polyunsaturated fatty acids in membrane, which alters membrane fluidity in cancer cells. In conclusion, this study highlights that the induction of apoptotic pathway by fish oil may increase the susceptibility of tumors to chemotherapeutic regimens.
Introduction
Colorectal cancer (CRC) is the most common gastrointestinal malignancy worldwide and accounts for significant morbidity and mortality in patients. 1 Chemotherapy with 5-fluorouracil (5-FU) is considered as first-line therapeutic modality for colon cancer.2,3 5-FU inhibits thymidylate synthase (TS) which affects de novo synthesis of thymine. This further blocks DNA synthesis and cell proliferation and also primes the cells for apoptosis.3,4 However, the major shortcoming of 5-FU treatment is its low effective response rate and severe side effects such as, hematological suppression, and gastrointestinal, hepatic and renal damages.5,6 Another area of concern is the development of drug resistance to conventional therapies by tumors. Therefore, there is a dire need for the development of new therapeutic strategies or modification of the current regimens in order to increase their efficacy. One promising possibility is to use dietary fats particularly n-3 polyunsaturated fatty acids (PUFAs) that chemosensitize tumor cells to the chemotherapeutic drugs. It has been reported that 5-FU and n-3 PUFAs have distinct anti-cancer capacity and combination of the two may be more effective.6–9 However, no distinct mechanism for this synergy has been elucidated. This warrants further investigation, as understanding these mechanisms may provide insight into the beneficial effect of fish oil (FO) as a chemotherapeutic adjuvant.
Apoptosis or programmed cell death plays an important role in the development and regulation of tissue homeostasis. 10 The signaling cascades leading to apoptosis are extrinsic or death receptor pathway and intrinsic or mitochondrial pathway. In the extrinsic pathway, apoptosis is initiated by binding of cell surface death receptor to its ligand, particularly Fas/FasL. 11 The intrinsic pathway, on the contrary, is characterized by the release of mitochondrial apoptogenic factors such as cytochrome c, apoptosis-inducing factor (AIF) into the cytosol. Their release in turn is regulated by the members of Bcl-2 family proteins. Therefore, an accurate evaluation of apoptosis and its key regulators may provide valuable information on the tumor cell dynamics.
It has been reported that long-chain n-3 PUFAs may get incorporated in the membrane and alter its physicochemical properties. Any alteration in the membrane fluidity could further dictate the apoptotic outcome of the cell. Not only n-3 PUFAs but also various anti-cancer drugs alter the cell membrane fluidity, which may play a role in the associated tumor cell sensitivity.
Therefore, this study was designed to understand the effect of combinatorial regimen of 5-FU and FO on the apoptotic markers and membrane properties in experimental colon carcinogenesis.
Materials and methods
Chemicals used
N,N-dimethylhydrazine dihydrochloride (DMH), 5-FU, protease inhibitor cocktail and phenylmethylsulfonyl fluoride (PMSF) were purchased from Sigma (St. Louis, USA). Dextran sulfate sodium salt (DSS) and ethidium bromide (EtBr) were obtained from MP Biomedicals (Santa Ana, USA). M30 cytoDEATH monoclonal antibody was purchased from Boehringer Ingelheim (Mannheim, Germany). Monoclonal antibodies against Bax and caspase 8 procured were from Santa Cruz Biosciences (Santa Clara, USA). Monoclonal antibodies against Bcl-2, Fas, Fas ligand, fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse IgG1, phycoerythrin (PE)-streptavidin labeled-secondary antibody, and FITC Annexin V Apoptosis Detection Kit were purchased from BD Biosciences (San Jose, USA). FO under the brand name Maxepa containing 180 mg/mL eicosapentaenoic acid (EPA) and 120 mg/mL docosahexaenoic acid (DHA) was purchased from Merck Chemicals Limited (Goa, India). All other chemicals used in the study were of analytical grade (AR).
Experimental animals
Male BALB/c mice weighing 28–35 g were obtained from Central Animal House, Panjab University, Chandigarh, India. The experimental protocols were approved by the Institutional Ethics Committee and conducted according to Indian National Science Academy Guidelines for the use and care of experimental animals. The animals were acclimatized and fed with standard pellet diet and water ad libitum.
Experimental design
The acclimatized animals (n = 150) were divided into the following groups:
Group A: The animals in this group were administered with a single dosage of 1 mM ethylenediaminetetraacetic acid (EDTA; vehicle of DMH) intraperitoneally (i.p.) and maintained for 22 weeks.
Group B: The animals received a single i.p. dosage of DMH [20 mg/kg body weight (b.w.), pH 6.5]. After 1 week, animals were given 3% DSS dissolved in distilled water for 7 days, followed by normal drinking water for 14 days. The animals were subjected to three such alternate cycles of DSS. These animals were further maintained on standard pellet diet for a period of 20 weeks for the development of colon cancer. After this period, animals were further subdivided according to the treatment regimen: DMH/DSS group: No further treatment was given in this group. 5-FU + DMH/DSS group: The animals were administered 5-FU (12.5 mg/kg b.w.) orally for 14 days. The criteria for selection of dosage and route of administration have been reported in our earlier study.
6
FO + DMH/DSS group: The animals were treated with 0.2 mL of FO orally for 14 days. This dosage of FO was selected based on a previous study.
9
5-FU + FO + DMH/DSS group: The animals were given combined oral dosage of 5-FU (12.5 mg/kg b.w.) and 0.2 mL FO for 14 days.
After the treatment, the animals were sacrificed and the samples were collected for the analysis.
Isolation of colonocytes
The colonocytes were isolated by the method of Sanders et al. 12
Estimation of DNA damage
DNA damage in isolated colonocytes was evaluated using comet assay according to the method of Singh et al. 13 The comets were scored per field, and the olive tail moment was measured as the product of the tail length and the fraction of total DNA in the tail using Autocomet software.
Measurement of apoptotic index
The apoptosis in isolated colonocytes was estimated by the following methods.
M30 CytoDEATH method
M30 CytoDEATH antibody is a unique tool for the easy and reliable determination of very early apoptotic events. The isolated colonocytes were fixed with 70% ice cold ethanol and then washed with phosphate-buffered saline (PBS). The cell suspension was incubated with M30 CytoDEATH monoclonal antibody followed by FITC-conjugated anti-mouse IgG1 in the dark. The percentage of positive cells, as a measure of apoptosis, was estimated with BD FACS Canto Flow Cytometer (BD Biosciences, San Jose, USA) equipped with an excitation and emission laser at 495 and 520 nm, respectively. The results were reported as percentage of apoptotic cells.
Annexin–PI staining method
The percentage of apoptotic cells was quantitatively determined using FITC Annexin V Apoptosis Detection Kit (BD Biosciences, San Jose, USA) as per the protocol mentioned by the manufacturer. Approximately 1 × 107 isolated colonocytes were washed twice with cold PBS at 630g for 5 min. The cell pellet was resuspended in 1X binding buffer and stained with FITC labeled Annexin V and propidium iodide (PI) for 15 min at room temperature in the dark. After staining, cells were washed and resuspended in 400 µL of 1X binding buffer. The acquisition from each sample was conducted on BD FACS Canto Flow Cytometer (BD Biosciences, San Jose, USA), and the collected data were analyzed using the BD FACSDiva software. The important controls (i.e. unstained cells, cells stained with FITC Annexin V alone and PI alone) were also run simultaneously. The untreated cell population was used to define the basal level of apoptotic and dead cells. The percentage of apoptotic cells was then determined by subtracting the percentage of apoptotic cells in the untreated population from percentage of apoptotic cells in the treated population. Cells that stain positive for FITC Annexin V and negative for PI were considered to be early apoptotic cells. Cells that stain positive for both FITC Annexin V and PI were either in the end stage of apoptosis, or necrosis, or already dead. Cells that stain negative for both FITC Annexin V and PI are considered as viable.
Hoechst staining method
The formation of apoptotic bodies in the nucleus was evaluated on morphological examination with fluorescence microscopy after Hoechst (H33342) staining. The isolated colonocytes were incubated with Hoechst dye and then washed with PBS. After staining, the formation of apoptotic bodies in cell nuclei was visualized and scored in three fields of 100 cells for each sample using fluorescence microscope (Nikon Eclipse 80i, Nikon, Japan) and its corresponding Northern Eclipse imaging Elements-D (NIS-D) software. Cells with dark blue color represent apoptotic cells having condensed/fragmented DNA, whereas faint blue all over were considered to be the normal cells.
Measurement of Fas, FasL, caspase 8, Bax, and Bcl-2
The extrinsic apoptotic pathway was analyzed by estimating Fas, FasL, and caspase 8 expression and the intrinsic pathway by estimating Bax and Bcl-2 levels using flow cytometric and western blot method.
Flow cytometry
The expression of Fas, FasL, caspase 8, Bax, and Bcl-2 was estimated using flow cytometric method as described by Somma et al. 14 Briefly, the colonocytes were fixed in 2% paraformaldehyde (PFA) and washed with PBS. The fixed colonocytes were permeabilized in saponin buffer specifically for the estimation of intracellular proteins such as caspase 8, Bax, and Bcl-2. The different aliquots of colonocytes were subsequently incubated with diluted anti-mouse caspase 8, Fas, FasL, Bax, and Bcl-2 monoclonal antibody. The colonocytes with Fas, caspase 8, Bcl-2, and Bax were counterstained with FITC-conjugated anti-mouse secondary IgG1 antibody and Fas ligand–treated cells with PE-streptavidin secondary antibody. The acquisition of each sample was conducted on BD FACS Canto Flow Cytometer (BD Biosciences, San Jose, USA), and the data were analyzed using BD FACSDiva software. The important controls for the different antibodies were also run simultaneously. The results were depicted as percentage of positive cells (percentage of positive cells treated with antibody − percentage of positive cells treated without antibody).
Western blot
Approximately 60 mg of tissue was homogenized in chilled lysis buffer comprising 50 mM Tris–HCl (pH 7.4), containing 1% Triton-X, 2 mM dithiothreitol (DTT), 1 mM phenylmethylsulfonyl fluoride (PMSF), 0.1% sodium dodecyl sulfate (SDS), 100 mM sodium chloride (NaCl), 5 mM EDTA, and 0.2% of protease inhibitor cocktail, and incubated on ice. After centrifugation at 12,000 r/min for 20 min, the supernatants were collected as protein samples. Protein concentration was measured with standard Bradford 15 assay. 100 µg of lysate protein was separated by SDS–polyacrylamide gel electrophoresis (PAGE) using 12% polyacrylamide gel and then transferred to polyvinylidene difluoride membranes (Bio-Rad, Hercules, USA). The membranes were then blocked using 3% bovine serum albumin (BSA) in Tris-buffered saline (TBS) containing 0.1% Tween-20. The membranes were probed with anti-mouse CD95 primary antibody, Biotin Hamster Anti-Mouse and Rat CD178, mouse anti-human caspase-8 primary antibody, mouse anti-human Bcl-2 primary antibody, and β-actin primary antibody for 3 h. The membranes were then washed with TBS containing 0.1% Tween-20 and incubated with horseradish peroxidase (HRP)-labeled goat anti-mouse IgG, anti-rabbit-IgG, or mouse PE-streptavidin at room temperature for 1 h. β-actin was used as a loading control. The immunoblots were visualized on FluorChemM (ProteinSimple, San Jose, USA) by using the Enhanced Chemiluminescence Detection Kit (Bio-Rad, Hercules, USA).
Measurement of membrane characteristics
The membrane fluidity of isolated colonocytes was estimated using 1,6-diphenyl-1,3,5-hexatriene (DPH) dye according to the method of Grunberger et al. 16 The fluorescence polarization (p) and anisotropy (r) of the sample were measured using Perkin Elmer Luminescence Spectrometer LS-55 (Beaconsfield, UK). Order parameter (S) was calculated using the formula: S2 = (4/3r − 0.1) / ro. Here, ro is a limiting anisotropy for DPH which is equal to 0.362. 17 Membrane fluidity was measured in terms of reciprocal of fluorescence polarization (1/p) values. The membrane lateral diffusion in isolated colonocytes was estimated using pyrene dye by the method of Massey et al. 18 The results were expressed as Excimer fluorescence/Monomer fluorescence (E/M) ratios that are inversely related to microviscosity (η).
Estimation of n-3 PUFAs content
The lipids were isolated from the colon tissue using the method of Folch et al. 19 and estimated according to the method of Fringes and Dunn. 20 In isolated lipids, EPA and DHA content were estimated by the method of Wilson and Sargent 21 using high-performance thin layer chromatography (HPTLC). EPA and DHA standards in different concentrations ranging from 0 to 20 µg were run along with the samples. The results were expressed as micrograms of EPA or DHA per milligram of total lipids.
Statistical analysis
The results are expressed as mean ± standard deviation (SD). The differences between the groups were assessed by one-way analysis of variance (ANOVA) using Statistical Package for Social Sciences (SPSS) 18.0 software package for Windows. Post hoc testing was performed for inter-group comparisons using the least significant difference (LSD). A value of p < 0.05 was considered to indicate a significant difference between groups.
Results
Effect of 5-FU and/or FO on DNA damage
The results of DNA damage are represented in Figure 1. In DMH/DSS-treated group, no significant alteration in olive tail moment of DNA was observed in comparison to control group. As expected, the treatment with 5-FU significantly increased olive tail moment of DNA in the isolated colonocytes as compared to DMH/DSS-treated group. FO by itself did not lead to any DNA damage; however, its combination with 5-FU significantly increased olive tail moment of DNA in comparison to the other treated groups.

Effect of 5-FU and/or FO on DNA damage using comet assay. (a) Representative images showing DNA damage by comet assay in isolated colonocytes of different groups. → represents head and - -> represents tail of DNA comet. (b) Data analysis of olive tail moment in different groups.
Effect of 5-FU and/or FO on apoptotic index
The results of flow cytometric and immunofluorescence studies for the evaluation of apoptosis correlated well and are depicted in Figures 2–4. There was a significant decrease in apoptotic index in carcinogen-treated animals as shown by a decrease in M30 CytoDEATH and Annexin–PI stained cells and a decrease in the presence of apoptotic bodies. However, treatment with 5-FU or FO led to an increase in apoptotic index as compared to DMH/DSS-treated group, with a significant potentiation in apoptotic index in 5-FU + FO group.

Effect of 5-FU and/or FO on M30 CytoDEATH levels. (i) Representative images showing percentage of apoptotic cells (P3) using M30 CytoDEATH antibody on flow cytometer. A: cells without antibody (Ab), B: control group, C: DMH/DSS group, D: 5-FU + DMH/DSS group, E: FO + DMH/DSS group, and F: 5-FU + FO + DMH/DSS group. (ii) Graphical representation of M30 CytoDEATH levels in different groups.
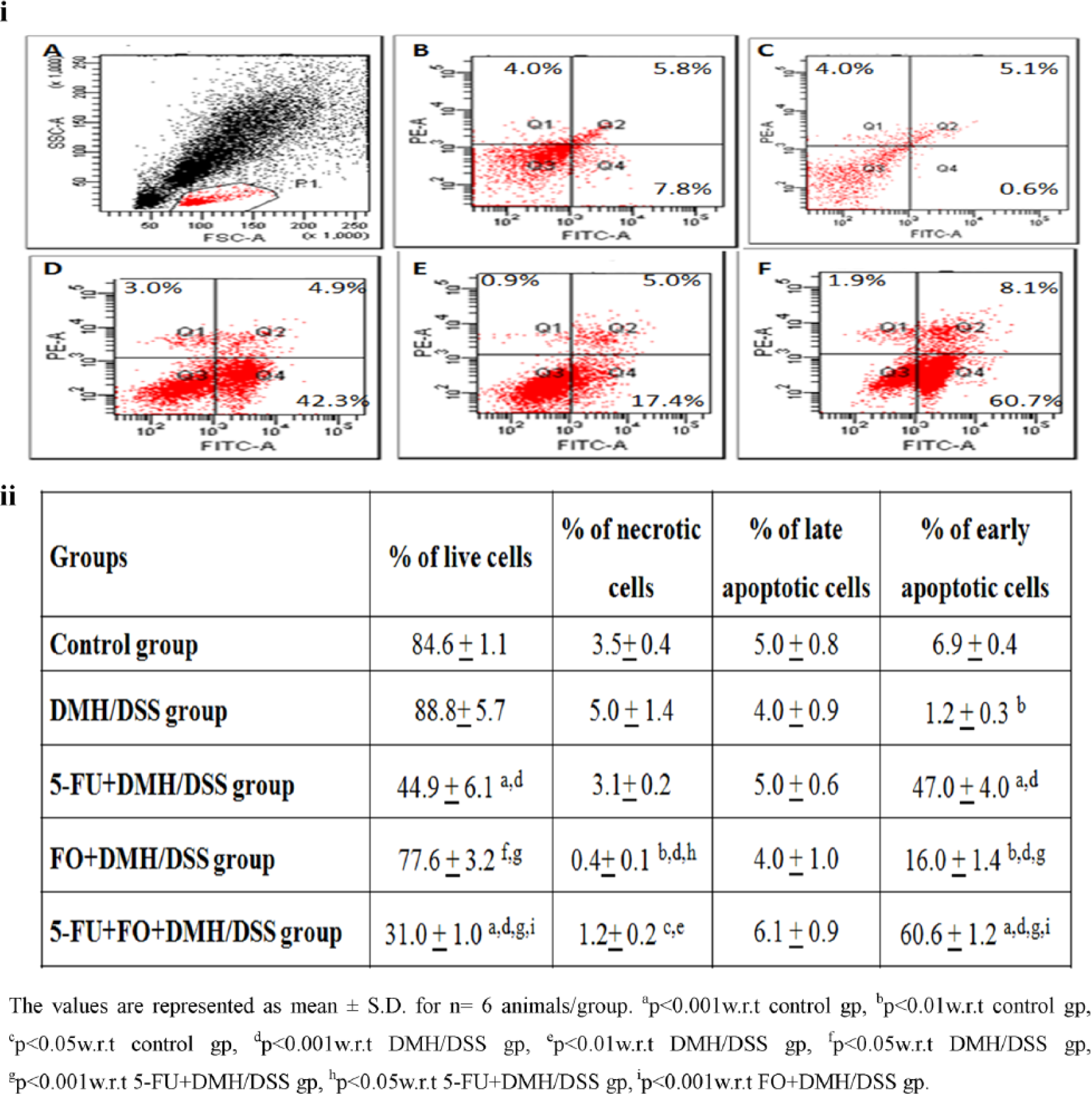
Effect of 5-FU and/or FO on apoptotic index using Annexin–PI staining. (i) Representative photograph showing percentage of apoptotic cells using Annexin–PI staining by flow cytometric method. A: gated cells, B: control group, C: DMH/DSS group, D: 5-FU + DMH/DSS group, E: FO + DMH/DSS group, and F: 5-FU + FO + DMH/DSS group. Q1 quadrant represents the percentage of necrotic cells; Q2 quadrant represents the percentage of late apoptotic cells; Q3 quadrant represents the percentage of live cells; and Q4 quadrant represents the percentage of early apoptotic cells. (ii) Flow cytometric analysis of apoptotic index in different groups.

Effect of 5-FU and/or FO on apoptotic index using Hoechst staining. (i) Representative images showing immunofluorescence staining of Hoechst dye in the isolated colonocytes. A: control group, B: DMH/DSS group, C: 5-FU + DMH/DSS group, D: FO + DMH/DSS group, and E: 5-FU + FO + DMH/DSS group. Here → represents live cells and - -> denotes fragmented and the formation of apoptotic bodies, which are the classic characteristics of apoptosis. (ii) Data analysis of apoptotic cells in different groups.
Effect of 5-FU and/or FO on apoptotic pathways
Extrinsic pathway
The role of extrinsic apoptotic pathway was evaluated by analyzing the expression of Fas, FasL, and caspase 8 by both flow cytometry and western blotting, and the results are represented in Figure 5. There was no significant alteration in the expression of Fas and caspase 8, whereas FasL levels were downregulated in DMH/DSS-treated group. Treatment with 5-FU and/or FO led to an increase in Fas, FasL, and caspase 8 expression as compared to DMH/DSS-treated group. However, the combined treatment of 5-FU and FO resulted in a significant increment in the levels of Fas, FasL, and caspase 8 in comparison to the other treated groups.

Effect of 5-FU and/or FO on Fas, FasL, caspase 8, bax, and bcl-2 levels using different monoclonal antibodies on flow cytometer and western blot method. (i) Graphical representation of Fas, FasL, and caspase 8 levels in isolated colonocytes of different groups. (ii) Data analysis of Bax and Bcl-2 levels in isolated colonocytes of different groups. The values are represented as mean ± SD for n = 6 animals/group. (iii) Representative image showing western blot analysis for different apoptotic proteins—Lane 1: control group; Lane 2: DMH/DSS group; Lane 3: FO + DMH/DSS group; Lanes 4 and 5: FU + DMH/DSS group; and Lane 5: 5-FU + FO + DMH/DSS group. β-actin has been used as an internal standard. The data were observed for n = 3 animals/group.
Intrinsic pathway
To understand the involvement of intrinsic pathway, we decided to analyze the expression of two major regulatory proteins, which determine the cell fate (i.e. Bcl-2 and Bax). Bax levels could not be detected by western blot so only flow cytometric evaluation was done. DMH/DSS treatment led to an increase in the expression of Bcl-2 with no significant alteration in Bax levels in isolated colonocytes as compared to control animals (Figure 5). On the contrary, 5-FU treatment exhibited an increase in the expression of Bax and a decrease in Bcl-2 levels as compared to DMH/DSS-treated group. FO treatment by itself led to a decrease in the expression of Bcl-2 but had no major effect on Bax expression, but its administration with 5-FU resulted in a significant increase in the Bax expression and a decrease in Bcl-2 expression.
Effect of 5-FU and/or FO on the membrane characteristics
The results of membrane characteristics of isolated colonocytes are depicted in Table 1. DMH/DSS treatment led to an increase in membrane fluidity and E/M ratios as well as a decrease in anisotropy in comparison to control group. Treatment with 5-FU increased E/M ratios and decreased anisotropy as compared to DMH/DSS-treated group. On the other hand administration of FO led to an increase in membrane fluidity and E/M ratios along with a decrease in anisotropy and order parameter as compared to DMH/DSS-treated group. However, the combination of 5-FU and FO significantly increased the membrane fluidity and E/M ratios with a concomitant decrease in anisotropy and order parameter as compared to 5-FU + DMH/DSS-treated group.
Effect of 5-FU and/or FO on membrane characteristics and n-3 PUFAs’ content.

5-FU: 5-fluorouracil; FO: fish oil; n-3 PUFAs: n-3 polyunsaturated fatty acids; EPA: eicosapentaenoic acid; DHA: docosahexaenoic acid; DMH: dimethylhydrazine dihydrochloride; w.r.t: with respect to.
The values are represented as mean ± SD for n = 6 animals/group.
p < 0.001, bp < 0.01, cp < 0.05 w.r.t control group.
p < 0.001, ep < 0.01, fp < 0.05 w.r.t DMH group.
p < 0.001, hp < 0.01, ip < 0.05 w.r.t 5-FU + DMH group.
p < 0.001, kp < 0.01 w.r.t FO + DMH group.
Effect of 5-FU and/or FO on n-3 PUFAs content
The standard peaks of EPA and DHA are shown in Supplementary Figure 1. A significant decrease in EPA and DHA content was observed in DMH/DSS-treated group with respect to control group (Supplementary Figure 1, Table 1). Although treatment with 5-FU increased the content of DHA, no significant change was observed in EPA levels as compared to DMH/DSS-treated group. On the contrary, the supplementation of FO alone or as an adjuvant resulted in a significant increase in the content of EPA and DHA in comparison to DMH/DSS-treated group. But the increase was more pronounced in 5-FU + FO + DMH/DSS treated group in comparison to 5-FU + DMH/DSS group (Supplementary Figure 1, Table 1).
Discussion
Substantial evidence supports the contention that dietary intake of FO rich in n-3 PUFAs plays an important role in the prevention and treatment of colon cancer.22–24 In addition, n-3 PUFAs also sensitize tumor cells to anti-cancer drugs and enhance tumor responsiveness to chemotherapy.8,25–28 In an earlier study, we had reported an augmentation in the therapeutic efficiency of 5-FU when given in combination with FO. The combined strategy not only decreased the tumor burden but also significantly reduced the associated side effects.6,29 Various mechanisms have been hypothesized to explain the modulation of the tumor cell response to chemotherapeutic drugs by FO which includes an alteration in the expression of pro-apoptotic proteins and membrane characteristics by incorporation of n-3 PUFAs in the membrane. However, no experimental data to validate this hypothesis are available till date, which necessitates further investigation. This study is designed to delineate the mechanistic action of FO (rich in n-3 PUFAs) as a dietary adjuvant with 5-FU in DMH/DSS-induced colon carcinogenesis.
It is well established that 5-FU treatment elicits cell death by causing DNA damage, specifically double-strand breaks due to misincorporation of its active metabolites. Consequently, 5-FU not only suppresses both DNA synthesis and repair but also augments DNA damage in the cells. An increase in DNA damage further results in the blockage of DNA replication and triggers cell death.3,25 Our results also support this contention as 5-FU treatment led to significant DNA damage, which in turn induced apoptosis. FO by itself, on the contrary, did not cause any significant DNA damage but when given with 5-FU potentiated its effects. It has been suggested that FO supplementation may enrich cell membrane with DHA and/or EPA and alter the accumulation and/or retention of chemotherapeutic drugs, which would impact the drug response in tumor.30–33 An increased drug uptake at the target site may also be related to the incorporation of n-3 PUFAs into tumor cell membranes which can affect the biophysical properties of cell membranes and alters the membrane fluidity which indeed was observed in this study.24,34–37 As the drug concentrates in the tumor tissue, it would lead to an increase in DNA damage and therefore augmented cytotoxic effect.
The increased effectiveness of 5-FU in combination with FO was further substantiated by measuring apoptotic index. In DMH/DSS-treated animals, a decrease in apoptotic index was observed as compared to control animals. Diminution in apoptosis on progression of colon cancer has been reported earlier also.24,38,39 Treatment with 5-FU and/or FO led to a marked increase in apoptosis in carcinogen-treated animals, and the effect was further augmented using the combinatorial regimen. Both 5-FU and FO have been shown to enhance apoptosis by regulating the interplay between pro- and anti-apoptotic members of Bcl-2 family.40,41 Therefore, we decided to investigate the effect of these treatments on the intrinsic and extrinsic apoptotic pathways. The extrinsic apoptotic pathway is stimulated by death receptors such as Fas and TNFR1, which constitutes a prominent mechanism in the defense against tumors. 42 The present results indicated that there was no alteration in the levels of Fas and caspase 8, whereas FasL expression was downregulated in DMH/DSS group as compared to control group. Deregulated Fas/FasL apoptotic pathway has also been implicated in immune evasion and acquired drug resistance in tumor cells. Downregulation of FasL in the carcinogenic group would allow the tumor cells to escape immune surveillance and evade its recognition by Fas-positive lymphocytes. 43 Furthermore, the results demonstrate that on administration of 5-FU and/or FO, there was a significant increase in Fas, FasL, and caspase-8 expression, and the increase was more pronounced with combinatorial dose regimens. As expression of Fas, FasL and caspase-8 is necessary for apoptosis, the results, in the present context, indicate significant activation of extrinsic apoptotic pathway. The results are also corroborated by other studies which have shown activation of Fas pathway and subsequently caspase 8 by 5-FU and n-3 PUFAs.44,45
DMH/DSS treatment led to a major increase in the expression of anti-apoptotic Bcl-2 protein; however, pro-apoptotic Bax expression was not altered. An increase in Bcl-2 would lead to inhibition of apoptosis and this also corroborates our observations of a decreased apoptosis in this group. 46 On treatment with 5-FU and/or FO, a decrease in expression of Bcl-2 and an increase in Bax expression were observed with an enhanced effect in combinatorial regimen. An increase in Bcl-2 levels by 5-FU may be related to DNA damage, which induces the expression of a large number of pro-apoptotic genes, such as Bax, Noxa, and Puma, and represses Bcl-2 expression.40,47,48 FO has been postulated to alter the structure of mitochondrial membrane by modifying its fatty acid composition and thereby the interaction between different Bcl-2 members.49–51 Our results suggest Bcl-2 as a major player in regulation of apoptosis by both 5-FU and/or FO.
As the observations in this study confirmed the beneficial effect of FO with 5-FU treatment, it was imperative to determine the content of EPA and DHA in the colon and its effect on membrane biophysical properties to prove our hypothesis. DMH/DSS-treated animals had low levels of EPA and DHA in colon tissue. Incorporation of long-chain n-3 PUFAs in membranes is known to induce apoptosis and thereby decrease tumor growth. 37 Therefore, a decrease in EPA and DHA levels should hamper apoptotic pathway and thereby facilitate the process of colon carcinogenesis. These results are also supported by a decrease in apoptosis in this group. As expected, this group also exhibited an increase in membrane fluidity (1/p) and a decrease in microviscosity. An increase in membrane fluidity of tumor cells has been associated with enhanced cell proliferation, metastasis, chemoresistance, and poor prognosis.24,37,52,53 In 5-FU + DMH/DSS group, there was an increase in DHA levels only with a minor increase in membrane fluidity (1/p). Membrane microviscosity which is inversely proportional to membrane fluidity was, however, significantly decreased. The chemotherapeutic drugs have been documented to alter membrane fluidity by modulating the degree of unsaturation in the membrane. Nutritional interventions by FO with or without 5-FU increased EPA and DHA content in the colon tissue. It has been reported that phospholipid fraction of tumor cell undergoes rapid turnover that permits its fatty acid composition to be altered through dietary regimens.54,55 The increased unsaturated fatty acid content is associated with an increase in membrane fluidity (1/p) and a decrease in microviscosity as compared to DMH/DSS and 5-FU + DMH/DSS groups. This confirms our hypothesis that dietary n-3 PUFAs supplied by FO get incorporated into the membrane and alter its physicochemical properties. A high degree of unsaturation of these n-3 PUFAs alters not only membrane fluidity but also membrane-mediated functions such as modulation of eicosanoids and lipid second messengers as well as transcription factor activity.56–60 It has also been reported that Non-steroidal anti inflammatory drugs (NSAIDs) exhibit their anti-tumoral effect by decreasing membrane fluidity and subsequently modifying epidermal growth factor receptor (EGFR) and its downstream signaling in HCA-7 colon cancer cells. 53 This further strengthens the hypothesis given in this study. In an initial study conducted in our laboratory, we had reported that administration of FO modified mitochondrial membrane characteristics, which resulted in dissipation of mitochondrial membrane potential. 24 We also observed a decrease in cardiolipin levels and subsequent release of cytochrome c, which promoted apoptosis. 61
In summary, the evidence presented in this preclinical study gives new insight into the combined anti-neoplastic activity of 5-FU and FO in DMH/DSS-induced CRC model. Our findings indicate that the use of FO as an adjuvant with 5-FU chemosensitizes tumor cells by incorporation of long-chain n-3 PUFAs into the tumor cell membrane, which increase the effectiveness of 5-FU as shown by upregulation of apoptotic pathways. However, further studies investigating the mechanism pertaining to the adjuvant effect of FO on chemotherapeutic regimens must be carried out before its implementation in clinical trials.
Footnotes
Acknowledgements
The authors acknowledge the assistance from University Grants Commission and Department of Science &Technology as the department is supported under UGC SAP and DST-PURSE program. The authors would like to acknowledge Mrs Sandhya, Senior Technician, CSIC Department, Postgraduate Institute of Medical Education and Research, Chandigarh, for her assistance in flow cytometric estimations. The authors would also like to acknowledge Department of Science and Technology (DST)-Innovation in Science Pursuit for Inspired Research as Ms Isha Rani was SRF-DST INSPIRE (IF10173).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by a grant from Department of Science and Technology (DST), Government of India (Ref. No. SR/SO/BB-0138/2012), India.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
