Abstract
Bladder cancer remains a huge concern for the medical community because of its incidence and prevalence rates, as well as high percentage of recurrence and progression. Omega-3 polyunsaturated fatty acids and atorvastatin proved anti-inflammatory effects through peroxisome proliferator-activated receptor gamma mechanism. However, their chemopreventive effect still remained to be examined and clarified. In this study, bladder cancer was induced in rats by the chemical carcinogen N-butyl-N-(4-hydroxybutyl)nitrosamine. Omega-3 polyunsaturated fatty acids (docosahexaenoic acid and eicosapentaenoic acid: 2:3 w/w; 1200 mg/kg) and/or atorvastatin (6 mg/kg) were given orally daily to rats for eight consecutive weeks concomitantly with N-butyl-N-(4-hydroxybutyl)nitrosamine and continued for further 4 weeks after cessation of N-butyl-N-(4-hydroxybutyl)nitrosamine administration. The histopathological examination of rat bladder revealed the presence of tumors and the absence of apoptotic bodies in sections from N-butyl-N-(4-hydroxybutyl)nitrosamine group, while tumors were absent and apoptotic bodies were clearly observed in sections from rat groups treated with omega-3 polyunsaturated fatty acids, atorvastatin, or both drugs. The study of the molecular mechanisms illustrated downregulation of COX-2 and P53 (mutant) genes and suppression of transforming growth factor beta-1 and the lipid peroxidation product malondialdehyde in serum of rats of the three treated groups. This chemopreventive effect was confirmed by and associated with lower level of bladder tumor antigen in urine. However, the combined treatment with both drugs exhibited the major protective effect and nearly corrected the dyslipidemia that has been induced by N-butyl-N-(4-hydroxybutyl)nitrosamine. Collectively, omega-3 polyunsaturated fatty acids and atorvastatin, besides having anti-inflammatory properties, proved a chemopreventive effect against bladder cancer, which nominates them to be used as adjuvant therapy with other chemotherapeutics.
Keywords
Introduction
Bladder cancer is one of the most common urinary system malignancies worldwide occurring in both sexes. It is more frequent in men than women, and the incidence and mortality rate of bladder cancer greatly increased in the recent years. 1 Cigarette smoking is the main risk factor for the development of urothelial tumor. Other risk factors may include exposure to arsenic in drinking water, occupational exposure to aromatic amines, and genetic alterations such as mutation in P53 gene.2,3
Omega-3 polyunsaturated fatty acids (n-3 PUFAs), especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are essential very long-chain fatty acids that contribute to either achieving optimal health or protection against many diseases, including cancer. 4 Several studies showed chemopreventive effect of EPA and DHA against several types of cancer such as colorectal cancer, pancreatic cancer, and breast cancer.5–7 Their anti-cancer activities could be attributed to their effect on multiple targets implicated in different stages of cancer development, including cell proliferation, angiogenesis, inflammation, and metastasis. 8
Statins are potent inhibitors of cholesterol biosynthesis and they have showed a great efficacy in decreasing morbidity and mortality from coronary artery disease. 9 They exert their action through inhibition of 3-hydroxy-3-methylglutaryl coenzyme-A (HMG-CoA) reductase, the major rate-limiting enzyme in the mevalonate pathway of cholesterol synthesis. 10 The inhibition of mevalonate pathway by statins and its downstream products which are important in cell signaling, protein synthesis, and cell cycle progression make statins exhibit many other biological activities, known as pleiotropic effects. Statins have shown anti-inflammatory, anti-proliferative, antioxidant, and proapoptotic properties, which could have an important role in cancer prevention. 11
Our target in this work was to investigate the possible chemopreventive role of n-3 PUFAs and atorvastatin (ATOR) in bladder cancer and to elucidate the impact of combined treatment with both drugs on interruption of some molecular mechanisms involved in cancer progression, with emphasis on oxidative stress, proliferation, and apoptosis.
Materials and methods
Experimental design
The study was performed in accordance with the guidelines for the care and use of laboratory animals approved by Research Ethics Committee (Faculty of Pharmacy, Tanta University, Egypt). Male albino rats were utilized in the study, 120–150 g each. Rats were purchased from National Research Center, Dokki, Giza, Egypt. Rats were weighed and housed in aluminum cages for 2 weeks under identical environmental conditions and allowed free access to standard pellet diet and water ad libitum.
After acclimatization period, rats were weighed and randomly divided into five groups: Group 1 (normal control group; n = 6)—rats in this group were given the vehicle for 12 weeks. Group 2 (N-butyl-N-(4-hydroxybutyl)nitrosamine (BBN) group; n = 10)—rats in this group received 150 mg/rat orally of BBN (Sigma-Aldrich, Inc, Tokyo, Japan) dissolved in ethanol:water (1:3 v/v) twice weekly for 8 weeks according to Prasain et al. 12 and vehicle for 4 weeks after the last dose. Group 3 (n-3 PUFAs group; n = 8)—rats in this group received oral dose of 1200 mg/kg/day of n-3 PUFAs (eBioChem, Shanghai, China) for 8 weeks concomitant with BBN and continued for 4 weeks after cessation of BBN. n-3 PUFAs contained DHA and EPA in a ratio of 2:3 w/w dissolved in orange juice. Group 4 (ATOR group; n = 8)—rats in this group received oral dose of 6 mg/kg/day of ATOR (Pfizer Inc, New York, NY, USA) for 8 weeks concomitant with BBN and continued for 4 weeks after the cessation of BBN. Group 5 (co-treatment group; n = 8)—rats in this group received both n-3 PUFAs and ATOR as in groups 3 and 4.
At the end of the experiment (12 weeks), rats were weighed, left in metabolic cages for 12 h for urine collection, and then anesthetized by halothane (Delta Pharma, Heliopolis, Cairo, Egypt). Blood samples were immediately collected by vein puncture from the inferior vena cava and serum was separated. Then, the rats were sacrificed by cervical dislocation and bladders were dissected. Each bladder was washed twice with saline and carefully opened. The lumen was blindly inspected by a pathologist for grossly visible lesions, and the number of tumors per rat was calculated. The length and the width of each tumor were measured by using a caliber, and then the tumor volume was calculated using the following equation: tumor volume = (width2 × length)/2. 13 The bladder was then divided into two portions: one portion was preserved in 10% formalin for histopathological examination and the other portion was immediately frozen and stored in liquid nitrogen.
Histopathological examination
Bladder sections were prepared (3−5 µm thick), stained with hematoxylin and eosin (H&E), and examined blindly by a pathologist. The slides were viewed and images were recorded using Olympus microscope equipped with spot digital camera and computer program MATLAB software in Pathology Department, Faculty of Veterinary Medicine, Mansoura University, Mansoura, Egypt.
Biochemical analysis of serum
Transforming growth factor beta-1 (TGF-β1) was measured in rat serum by rat TGF-β1 enzyme-linked immunosorbent assay (ELISA) kit (Hangzhou Sunlong Biotech Company, Ltd., Mainland Shanghai, China). The concentration of TGF-β1 was determined according to manufacturer procedure and expressed as picogram/milliliter. The lipid peroxidation marker malondialdehyde (MDA) was measured according to Yoshioka et al. 14 using colorimetric kit (Biodiagnostic, Cairo, Egypt), which depends on the reaction of thiobarbituric acid with MDA in acidic medium at the temperature of 95°C for 30 min to form thiobarbituric acid reactive product. Serum cholesterol and triglycerides were measured according to Savoldi et al. 15 and Nagele et al., 16 respectively, using commercial colorimetric kits (Spinreact, S.A., Girona, Spain). High-density lipoprotein (HDL)-cholesterol (HDL-C) was measured according to Austin et al. 17 after precipitation of very-low-density lipoprotein (VLDL)-cholesterol and low-density lipoprotein (LDL)-cholesterol (LDL-C) by phosphotungstate in the presence of magnesium ions. LDL-C was calculated from Friedewald equation as follows: LDL-C = total cholesterol − HDL-C − triglycerides/5. 18
Determination of urinary bladder tumor antigen
Bladder tumor antigen (BTA) was measured in rat urine by rat BTA ELISA kit (Mlbio Biotechnology Company). The concentration of BTA was determined according to manufacturer procedure and expressed as U/mL urine.
Quantitative reverse transcription polymerase chain reaction analysis for COX-2 and P53
Total RNA was extracted from bladder tissue lysed by RNeasy Plus Mini Kit (Qiagen, Maryland, North America), and 0.5 µg of total RNA was converted to complementary DNA (cDNA) by using Thermo Scientific RevertAid First Strand cDNA Synthesis Kit as the initial step of two-step reverse transcription polymerase chain reaction (RT-PCR) protocol. A volume of 1 µL of the cDNA was then used for quantitative PCR using Thermo Scientific Maxima SYBER Green QPCR Master Mix (2×). The target gene Ct values were normalized to the Ct value of the housekeeping gene, glyceraldehyde-3-phosphate dehydrogenase (Rat GAPDH), and expressed as relative copy number (RCN). Primers used in RT-PCR are presented in Table 1. The primers of P53 (mutant form) and GAPDH were prepared according to Ibrahim et al., 19 and those for COX-2 were prepared according to Zuo et al. 20 The relative content of the gene amplification product was calculated using the 2−ΔΔCt method. The real-time PCR instrument (Thermo Fisher Scientific, Waltham, MA, USA) was adjusted according to the program: 90°C for 1 min (pre-denaturation), and then, 35 cycles (95°C for 30 s, 60°C for 30 s, and 60°C for 30 s).
Primers for the studied genes.

Statistical analysis
Analysis of data was performed with Statistical Package for Social Science (SPSS) software version 20.0. All data are presented as mean ± standard error of the mean (SEM). Statistical comparison among groups was performed by one-way analysis of variance (ANOVA). Statistical significance was obtained at p < 0.05.
Results
Survival rate
The survival rate was 60% in BBN group and 75% in treated groups compared to 100% in the normal control group (Figure 1). The increased mortality rate in treated animals may be due to the elevated temperature and long experimental period (12 weeks).

Survival rate in the studied groups.
Macroscopic and microscopic examination
The results of macroscopic and microscopic examination are presented in Table 2 and Figures 2–8. In the BBN group, all survived rats (n = 6) had preneoplastic lesions, including hyperplasia (100%), dysplasia (100%), and metaplasia (66.7%) (Table 2), and 83% of the rats had grossly visible bladder tumors (Figure 2(b)). The bladder walls were thicker than those of the control rats, with new or enlarged small vessels. In total, 11 tumors were detected in five rats (with a mean of tumors 2.2 ± 0.37 per rat). In these five rats, the mean tumor volume was 4.29 ± 0.48 mm3 and the mean tumor volume per tumor was 3.6 ± 1.23 mm3 (Table 2). Histopathological evaluation of BBN group revealed that there were malignant lesions, including carcinoma in situ (CIS), urothelial carcinoma, adenocarcinoma, and squamous cell carcinoma in addition to the preneoplastic lesions (Table 2, Figure 5). All bladders from the control animals were normal (Figure 2(a)), with translucent and tiny bladders and without any abnormal mass or vascularization. Histopathologically, rats of the control group showed normal urothelium with no preneoplastic lesions (Table 2, Figure 4). The n-3 PUFAs group, ATOR group, and co-treatment group showed no abnormal masses or tumors, but some of them showed thicker wall than normal and preneoplastic lesions mainly hyperplasia with a ratio of 50%, 33.3%, and 16.7%, respectively (Table 2, Figure 3). The histopathological examination of rats treated with n-3 PUFAs and/or ATOR concomitant with BBN showed no neoplastic lesions in bladders but few preneoplastic lesions were found in some rats. The treated rats showed the presence of apoptotic bodies which were not observed in the BBN group (Table 2, Figures 6–8).
Macroscopy and microscopy examination results.
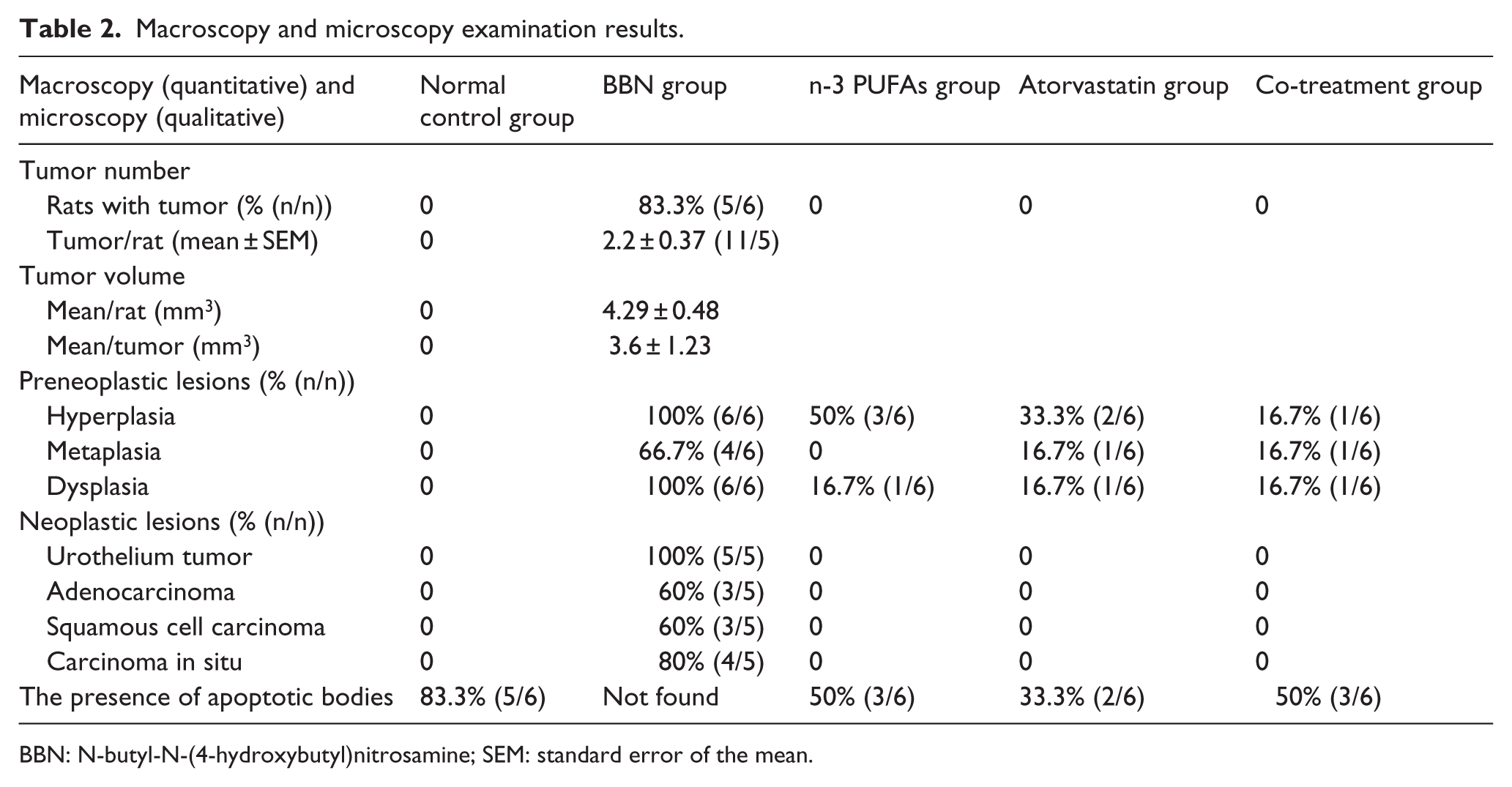
BBN: N-butyl-N-(4-hydroxybutyl)nitrosamine; SEM: standard error of the mean.

Gross urinary bladder. (a) Normal bladder showing normal color (arrow) and normal smooth surface of bladder mucosa with normal consistency. (b) Bladder of BBN group showing intense hemorrhagic and ulcerated mucosa (arrow) with severe thickening in consistency.

Preneoplastic lesions in various treated groups. (a) Hyperplasia (arrow): whitish color of bladder mucosa with the presence of mild nodularity on the surface which is hard in consistency. (b) Marked hyperplasia (arrow): bladder mucosa is slightly red in color with nodularity on the surface. (c) Dysplasia (arrow): bladder mucosa showing hardness in consistency, abnormal red colored foci, and nodularity on the surface.

Sections of urinary bladder from normal control group showing normal urothelium lining bladder with normal submucosa (H&E, 100×).

Sections of urinary bladder from BBN group. (a) Carcinoma in situ where neoplastic cells are showing marked dysplastic and neoplastic alterations with tendency to forming cell nests (arrow) without invasion underlying lamina propria and marked esinophilic infiltrate in the lamina propria (H&E, 100×). (b) Squamous cell carcinoma where neoplastic cells have large nucleus, vacuolated with prominent esinophilic nucleoli forming characteristic cell nests (arrow), and has keratin pearl in the center (H&E, 400×). (c) Adenocarcinoma where small densely basophilic neoplastic cells forming acini (arrow) and papillary projections into the lumen of the acini (H&E, 400×). (d) Invasive-type squamous cell carcinoma where nests of neoplastic cells with criteria of malignancy and the presence of the characteristic keratin pearl (arrow) invade the submucosa and muscle layer (H&E, 100×).

Sections of urinary bladder from n-3 PUFAs group. (a) Simple hyperplasia of epithelium (arrow) lining urinary bladder (H&E, 400×). (B) Apoptotic body (arrow) present in the epithelium lining urinary bladder (H&E, 400×).

Sections of urinary bladder from atorvastatin group. (a) Dysplastic alteration (arrow) of transitional epithelium lining bladder (H&E, 100×). (b) Simple hyperplasia (arrow) of lining epithelium of bladder (H&E, 100×).

Sections of urinary bladder from co-treatment group. (a) Squamous metaplasia (arrow) of transitional epithelium lining urinary bladder (H&E, 400×). (b) Apoptosis of hyperplastic cells (arrow) of transitional epithelium lining urinary bladder (H&E, 400×).
Serum transforming growth factor-β1 (TGF-β1)
In the BBN group, there was a significant increase (701 ± 25.4 pg/mL) in serum TGF-β1 (p < 0.05), compared with normal control group (445 ± 13.5 pg/mL) (Figure 9). Treated groups showed a significant reduction in TGF-β1 (p < 0.001) when compared with the BBN group. In n-3 PUFAs, the serum level of TGF-β1 was reduced to 548 ± 14.9 pg/mL; in ATOR group, the serum level was 565 ± 6.3 pg/mL; and in co-treatment group, the serum level was 518 ± 25.7 pg/mL (Figure 9).

Serum levels of transforming growth factor β1 in the studied groups. Data are expressed as mean ± SEM (n = 6 for each group).
Lipid peroxidation
Serum MDA concentration showed a significant high values (p < 0.05) in the BBN group (10.4 ± 1.62 nmol/mL) when compared with control rats (3.7 ± 0.5 nmol/mL). Treated groups showed a significant decrease (p < 0.001) in serum MDA compared with the BBN group. The serum level of MDA in n-3 PUFAs group was reduced to 2.8 ± 0.46 nmol/mL, in ATOR group was 3.6 ± 0.51 nmol/mL, and in co-treatment group was 3.6 ± 0.22 nmol/mL (Figure 10).

Serum levels of malondialdehyde (MDA) in the studied groups. Data are expressed as mean ± SEM (n = 6 for each group).
Lipid profile
The lipid profile showed increased level of total cholesterol and LDL-C in BBN group compared with normal control group. ATOR treated group and co-treatment group showed a significant increase in HDL-C and decrease in LDL-C compared with each of BBN, n-3 PUFAs, and normal control groups. n-3 PUFAs treated group and co-treatment group showed a significant decrease in triglycerides’ level versus the BBN group (Table 3).
Lipid profile in the studied groups.

HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TGs: triglycerides; BBN: N-butyl-N-(4-hydroxybutyl)nitrosamine; SEM: standard error of the mean; ATOS: atorvastatin.
Data are expressed as mean ± SEM (n = 6 for each group).
Significant versus normal control.
Significant versus BBN group.
Significant versus n-3 PUFAs group.
BTA
In the BBN group, there was a significant increase (84.5 ± 5.38 U/mL) in urinary BTA (p < 0.05) compared with normal control group (40 ± 3.72 U/mL). n-3 PUFAs group showed a significant reduction in urinary level of BTA (55.3 ± 5.4 U/mL) than the BBN group (p < 0.001). BTA was also reduced significantly in ATOR group to 64.4 ± 0.84 U/mL (p < 0.01) and in co-treatment group to 60.5 ± 2.8 U/mL (p < 0.001) versus BBN group (Figure 11). Although decreased in treated groups, urinary BTA remained higher than its value in the normal control group.

Urinary levels of bladder tumor antigen (U/mL) in the studied groups. Data are expressed as mean ± SEM (n = 6 for each group).
Real-time PCR for COX-2 and P53 (mutant) in bladder tissue
COX-2 gene expression as a marker of inflammation in bladder cancer tissues showed marked increase in BBN group up to 7.71 fold (p < 0.05) compared with the normal control group. In the same way, P53 (mutant form) gene expression was upregulated in BBN group to 11.63 fold (p < 0.05) (Table 4). The gene expression of COX-2 and P53 in rats treated with n-3 PUFAs, ATOR, or both drugs was significantly decreased (p < 0.001) when compared with BBN group, with the greatest suppression of P53 gene expression observed in the co-treatment group rather than mono-treatment groups (Table 4).
Cyclooxygenase-2 and P53 gene expression in the studied groups.

RCN: relative copy number; BBN: N-butyl-N-(4-hydroxybutyl)nitrosamine; SEM: standard error of the mean; ATOS: atorvastatin.
Data are expressed as mean ± SEM (n = 6 for each group). (−) Indicates percent decrease.
Significant versus negative control.
Significant versus BBN group.
Significant versus n-3 PUFAs group.
Significant versus ATOR group.
Discussion
Bladder cancer is the fifth most frequent tumor in men and ninth in women in the United States. 21 Due to a high likelihood of recurrence, effective chemoprevention is a significant unmet need. 22 This work aims to examine the chemopreventive effect of n-3 PUFAs and ATOR in male rats exposed to induction of bladder cancer and to evaluate the anti-proliferative, anti-inflammatory, proapoptotic, and antioxidant properties of both treatments. Bladder cancer was induced in rats in this study by BBN, which is metabolized by liver cytochrome P450 into the bladder carcinogen N-butyl-N-(3-carboxy propyl)nitrosamine (BCPN). 23 BCPN reaches the urinary bladder through urine and comes into contact with the urothelium, binding covalently to cellular macromolecules, specifically DNA, and initiating the carcinogenic process. 24
Our results indicated that BBN administration twice weekly for 8 weeks induced neoplastic changes in bladder of rats which was associated with elevated level of the urinary marker BTA. The bladder tissues obtained from all rats of BBN group showed marked dysplasia, hyperplasia, CIS, urothelial carcinoma, and squamous cell carcinoma. These histopathological changes were greatly corrected by treatment of rats with n-3 PUFAs (1200 mg/kg/day) and/or ATOR (6 mg/kg/day) for 12 weeks. Regarding the treated groups, the bladder sections showed no transitional or squamous cell carcinoma but the presence of few preneoplastic lesions. The improvement in the histopathological changes and the cancerous manifestations in the treated groups, herein, was associated with and confirmed by the significant decrease in the urinary BTA, the effect which was highly apparent in the co-treatment group rather than mono-treatment groups.
Our gene expression study supported the histopathological findings and indicated that the tumorigenic effect of BBN was associated with a significant increase in gene expression of COX-2 and the mutant form of P53. Vasconcelos-Nobrega et al. 25 demonstrated that BBN-induced rodent tumors, particularly murine tumors, have P53 mutations or mutations in genes related to the P53 pathway. Mutation in the P53 gene results in the production of an abnormal and usually dysfunctional mutated P53 with prolonged half-life compared to the wild type, and accumulates in the cell nucleus were proposed to confer a growth advantage to tumor cells. 26
Prominent COX-2 expression has been described in bladder cancer, including urothelial and squamous cell carcinomas, and this expression correlates with tumor grade and invasion. 27 COX-2 overexpression by BBN has been implicated in enhanced angiogenesis, which plays a role in carcinogenesis, and is correlated with increased production of vascular growth factors and formation of capillary networks. 28 COX-2 overexpression may also lead to DNA damage that may eventually lead to carcinogenesis. 29
Our data showed that the n-3 PUFAs administration to BBN-exposed rats significantly decreased COX-2 and mutant P53 gene expression, thereby supporting the antitumorigenic effect of n-3 PUFAs. Downregulation of P53 by n-3 PUFAs could be explained by the fact that n-3PUFAs decrease the expression of the mutant form of P53 (mtp53) gene and help the mutant form to restore its normal function and thus leading to enhancement of apoptosis in cancerous cells. 30 This effect was evidenced in this study histopathologically by increased number of apoptotic cells in bladder tissues of n-3 PUFAs treated rats. The proapoptotic properties of n-3 PUFAs were previously examined by Matsuda et al., 31 who found that n-3 PUFAs can modulate phosphatase and tensin homolog (PTEN), the expression of which in cancer cell lines upregulates the apoptotic signaling by decreasing nuclear factor kappa B (NF-κB) expression and hence induces tumor regression. 32
n-3 PUFAs, in this work, downregulated COX-2 gene expression, which could be attributed to the activation of peroxisome proliferator-activated receptor gamma (PPAR-γ) as DHA and EPA act as PPAR-γ ligands. 33 Moreover, DHA and EPA act as competitive inhibitors of arachidonate’s binding to the active site of COX-2, and EPA acts as an alternative substrate for this enzyme. 34 EPA and DHA were also reported to have anti-inflammatory role as they can inhibit the production of a range of other inflammatory proteins, including inducible nitric oxide synthase; TNF-α; IL-1, IL-6, IL-8, and IL-12 in cultured endothelial cells; monocytes; macrophages; and dendritic cells. 35 Thus, inhibition of COX-2 expression by n-3 PUFAs could be one of the mechanisms underlying their antitumorigenic effect.
This work showed that ATOR downregulated P53 (mtp53) gene expression in bladder of rats exposed to BBN and exhibited proapoptotic properties confirmed by histopathological results. Previous studies showed that statins alter Murine double minute 2 (Mdm2) expression and the P53 response to DNA damage, as statins induce mammalian target of rapamycin (mTOR)-dependent Mdm2 phosphorylation at Ser166. 36 Phosphorylation of Mdm2 at Ser166 has been shown to activate Mdm2 and enhance its ubiquitination ligase function and destabilize P53 and therefore may help P53 to restore its normal function and induce apoptosis to cancer cells. 37 Statins could also induce apoptosis through inhibition of RhoA activity, which in turn activates caspase-dependent mitochondrial pathway in tumor cells. 38
It has been demonstrated that suppression of COX-2 is one of the pathways required for inhibition of tumor progression. 39 Treatment of rats exposed to BBN with ATOR, herein, caused downregulation of COX-2 gene expression in bladder tissue. The mechanism underlying this effect is probably related to inhibition of NF-κB activity secondary to a decrease in isoprenylation of proteins involved in intracellular signal transduction. COX-2 gene expression is controlled by NF-κB, and it has been documented that statins can directly decrease this transcription factor. 40 Our results were in agreement with many previous studies which reported that statins downregulate COX-2 expression in human endothelial cells 41 and in different cell types. 42
Elevated expression levels of TGF-β1 have been reported in several types of cancer such as colorectal, prostate, endometrial, pancreatic, and breast cancer.43,44 TGF-β1 has a negative impact on tumor-surrounding cells, including the host immune cells, and allows tumor cells to evade from the immune system. 45 TGF-β1 may promote tumor cell invasion and metastasis by increasing fascin-1 expression. 46 A close relationship has been reported between reactive oxygen species (ROS) and TGF-β1 in many cancers. 47 ROS upregulates TGF-β1 secretion in rat bladder cancer and activates latent TGF-β1 by oxidation and/or cleavage of latency-associated protein in a complex with TGF-β1 when secreted from the cells. 48 Moreover, induction of TGF-β1 responsive genes is mediated by ROS via mitogen-activated protein kinase (MAPK) pathways and Smad pathway. 49
Our laboratory data confirmed the relation between TGF-β1 and ROS and showed elevation of serum TGF-β1 together with the presence of the oxidative stress marker MDA in cancer control group. A previous study by Oliveira et al. 50 showed that BBN increases the susceptibility of mitochondrial permeability transition pore to induction by calcium and this can be a consequence of increased ROS production. Furthermore, when the nitrosamine compounds were metabolized by the liver, they stimulate Kupffer cells leading to the generation of ROS and precipitation of oxidative stress. 51
The current data also showed that n-3 PUFAs significantly reduced serum MDA levels which could be correlated with their reported antioxidant properties. n-3 PUFAs were demonstrated to modulate antioxidant status in diabetic rats 52 and to inhibit lipid peroxidation in liver and kidney of rats intoxicated with lead acetate. 53 The antioxidant properties may be related to the assembly of n-3 PUFAs in membrane lipids and lipoproteins making the double bonds less available for free radical attack, inhibition of the pro-oxidant enzyme phospholipase A2, and stimulation of antioxidant enzymes. 54 Meanwhile, ATOR also reduced the level of MDA, in this work, when compared with the cancer control group, the effect which may be attributed to inhibition of Rac1/NADPH oxidase activity by ATOR that leads to reduced levels of ROS and hence, decreased oxidative stress. 55
The observed antioxidant effect of n-3 PUFAs and ATOR, herein, may explain the suppression of TGF-β1 levels in the rat groups treated with n-3 PUFAs and/or ATOR. Moreover, the inhibitory effect of ATOR on the serum levels of TGF-β1 could be attributed to the inhibitory effect of ATOR on Smad pathway and also through the inhibition of MAPK/extracellular signal-regulated kinase (ERK) signaling cascade, which is the major pathway controlling cellular processes including growth proliferation and survival. 56
Regarding lipid profile, the BBN group showed significantly elevated serum level of total cholesterol, LDL-C, and TGs when compared with normal control group. The BBN-induced dyslipidemia was corrected by the co-treatment with ATOR and n-3 PUFAs. The effects of statins on lipid profile are through the competitive, reversible inhibition of HMG-CoA reductase, the rate-limiting step in cholesterol biosynthesis. 57 The resultant reduction in cholesterol concentration within hepatocytes triggers upregulation of LDL-receptor expression, which promotes the uptake of LDL and LDL-precursors from systemic circulation. 58 Statins also inhibit the synthesis of apolipoprotein B100 and reduce the synthesis and secretion of triglyceride-rich lipoproteins. 59 Our findings were in line and showed that ATOR treatment produced a significant reduction in serum total cholesterol and LDL-C and significantly increased the serum HDL-C when compared with the BBN group.
The current results showed that n-3 PUFAs significantly decreased the serum level of triglycerides. The effect of n-3 PUFAs on triglyceride metabolism primarily includes the suppression of hepatic VLDL synthesis and discharge with enhancement of VLDL metabolism. 60 Since VLDL is metabolized into intermediate-density lipoprotein (IDL) and then LDL, the increased metabolic rate of VLDL by n-3 PUFAs may account for the increased level of LDL-C in rats treated with n-3 PUFAs as observed by our data. The beneficial effect of n-3PUFAs mono-treatment in reducing serum triglyceride levels was associated with elevation of the cardiovascular risk factor LDL-C. However, the combined treatment with both n-3 PUFAs and ATOR produced a protective effect where all total cholesterol, LDL-C, and TGs were significantly lowered, whereas HDL-C was significantly elevated.
Conclusion
n-3 PUFAs and ATOR, at the studied doses, proved anti-tumorigenic effect and prevented bladder cancer progression, which could be through anti-proliferative, anti-inflammatory, antioxidant, and proapoptotic mechanisms. Combined treatment was more effective than either drug alone in interrupting some molecular targets involved in tumor growth. Being of different pharmacological and chemical classes, n-3 PUFAs and ATOR could augment each other, exhibiting high therapeutic value. Further investigations are required to examine the anticancer effect of the combined treatment with n-3 PUFAs and ATOR in other cancer models.
Footnotes
Acknowledgements
The authors gratefully acknowledge Dr Mohamed Fawzey, Professor of Histochemistry and Cell Biology, Pathology Department, Faculty of Veterinary Medicine, Mansoura University, Mansoura, for conducting and interpreting the histopathological examination.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
