Abstract
5-fluorouracil in combination with the folate leucovorin is the cornerstone in treatment of colorectal cancer. Transport of leucovorin into cells, and subsequent metabolic action, require expression of several genes. The aim was to analyze if tumoral expression of genes putatively involved in leucovorin transport, polyglutamation, or metabolism was associated with outcome of patients with stage III colorectal cancer treated with adjuvant chemotherapy. A total of 363 stage III colorectal cancer patients who received adjuvant bolus 5-fluorouracil + leucovorin alone, or in combination with oxaliplatin according to Nordic bolus regimes were included. Expression of 11 folate pathway genes was determined in tumors using quantitative real-time polymerase chain reaction and related to disease-free survival. The median follow-up time was 5 years. During follow-up, 114 (31%) patients suffered from recurrent disease. A high tumoral expression of the genes SLC46A1/PCFT, SLC19A1/RFC-1, ABCC3/MRP3, GGH, and MTHFD1L, which are involved in folate transport, polyglutamation, or metabolism, was associated with longer disease-free survival of the patients. Each of these genes either encodes mitochondrial enzymes or is being regulated by mitochondrial transcription factors. Expression of the SLC46A1/PCFT gene was most strongly associated with disease-free survival, regardless of treatment regimen. In conclusion, the results show that expression of folate pathway genes are associated with outcome of colorectal cancer patients treated with adjuvant 5-fluorouracil in combination with leucovorin. A prospective study needs to be conducted to determine if expression of these genes can be used to predict response to leucovorin and other folates that are now being tested in clinical studies. Moreover, there seems to be a link between folate metabolism and mitochondrial biogenesis and respiration that deserves further exploration.
Introduction
Colorectal cancer is ranked as the third most common cancer worldwide, and each year approximately 1.4 million people are diagnosed with the disease. 1 Although novel combination treatments have been developed during the last years, 5-fluorouracil (5-FU) in combination with the folate leucovorin ([6R,S]-5-formyltetrahydrofolate) is still a cornerstone in treatment of colorectal cancer. 2
Within the cells, leucovorin is converted to the cofactor 5,10-methylenetetrahydrofolate (methyleneTHF). This folate forms a complex together with the target enzyme, thymidylate synthase (TS; EC 2.1.1.45), and deoxyuridine monophosphate (dUMP) or the fluorinated form of dUMP (FdUMP), which is the active metabolite of 5-FU (Figure 1). In the enzymatic reaction, TS catalyzes the methylation of dUMP to deoxythymidine monophosphate with the transferred methyl group provided by methyleneTHF. 2 It has been shown that in order to achieve maximal inhibition of TS, high levels of methyleneTHF are needed. Transport of leucovorin into the cells, and subsequent metabolic action, require expression of several genes. 3 We previously analyzed the expression of 22 folate-associated genes with putative impact on leucovorin metabolism in a retrospective study on 193 patients with stage III colorectal cancer. 4 Patients were treated with 5-FU + leucovorin (FLV) alone, or in combination with oxaliplatin (FLOX) according to Nordic bolus regimes. Cox regression analysis showed that high tumoral expression of the genes SLC46A1/PCFT (proton-coupled folate transporter) and SLC19A1/RFC-1 (reduced folate carrier 1), which are involved in transport of leucovorin and other folates, 5 was associated with decreased risk of recurrent disease measured as disease-free survival (DFS). High intracellular levels of folates, including the active cofactor methyleneTHF, may be achieved if the expression level of SLC46A1/PCFT and/or SLC19A1/RFC-1 is high. The association between high gene expression and longer DFS may be linked to stabilization of the ternary complex comprising methyleneTHF, TS, and fluorinated dUMP, which is needed for a good response to FLV therapy. Both SLC19A1/RFC-1 and SLC46A1/PCFT act as folate carriers, but function optimally at different pH. 6 In tumor tissue, the pH is considered to be more acidic than in normal tissue and reduced folates are mainly transported by SLC46A1/PCFT, whereas SLC19A1/RFC-1 has its optimum at normal physiological pH. 7

Simplified overview of the folate metabolism in eukaryotic cells. Note that some of the steps can also be catalyzed by enzymes not depicted in the figure. In the cytosol, FPGS converts folate monoglutamates to polyglutamates, whereas GGH converts folate polyglutamates to monoglutamates. The folate metabolism is highly compartmentalized and differs in cytosolic and mitochondrial pools with regard to distribution of folate cofactors and extent of folate polyglutamation. 5-formylTHF (leucovorin) is converted to methyleneTHF in two steps. MethyleneTHF is then used as a methyl donor in the methyl pathway and as a cofactor in the synthesis of dTMP from dUMP. The latter reaction is catalyzed by the enzyme TS. Treatment with 5-FU inhibits dTMP synthesis through formation of FdUMP, which binds TS. Dashed arrows indicate multiple enzyme steps.
The aim of the present study was to analyze the association between tumoral expression of genes putatively involved in leucovorin transport, polyglutamation, or metabolism and DFS in a group of patients with stage III colorectal cancer. The group was larger than in the previous study (363 patients vs 193) and had a longer follow-up time, to enable subgrouping of patients by adjuvant treatment regimens (FLV or FLOX).
Patients and methods
During the period 2001–2013, 669 colorectal cancer patients with stage III disease were subjected to surgery at the Sahlgrenska University Hospital/Östra and received adjuvant FLV (n = 418) or FLOX (n = 251) therapy. Out of these patients, 555 underwent elective surgery and were selected for inclusion. Biopsy samples were obtained from 425 of the 555 patients, and 363 of these were available for the present study. The group of 363 patients consisted of patients from the previous study (n = 193) pooled with a new group of patients (n = 170). The tumor−node−metastasis staging system was used to classify tumors. 8 Patient and tumor characteristics are shown in Table 1. The regional ethical review board in Gothenburg approved the study and informed consent was obtained from all patients.
Patient and tumor characteristics.

FLOX: FLV + oxaliplatin; FLV: 5-fluorouracil + leucovorin.
Median age in FLV group: 68 (range: 36–82) years, FLOX group: 59 (range: 19–72) years, p < 0.0001.
In 92.3% of the patients, ≥12 lymph nodes were analyzed.
Treatment
After surgery, patients received adjuvant FLV (n = 222) or FLOX (n = 141) therapy for 6 months. FLV treatment comprised intravenous bolus injections of 5-FU (500 mg/m2), followed by leucovorin (60 mg/m2) 30−40 min later, on days 1 and 2 every other week, according to the Nordic FLV regimen. 9 FLOX treatment comprised 5-FU (500 mg/m2) and leucovorin (60 mg/m2) bolus on days 1 and 2 every other week, plus an oxaliplatin infusion (85 mg/m2) over 120 min on Day 1 every other week. 10 The patients were followed for a maximum of 5 years or until time of recurrent disease or death.
Preparation of total RNA and cDNA synthesis
Tumor biopsies were snap-frozen in liquid nitrogen after removal and stored at −80°C until used. Total RNA was isolated from 10−30 mg tissue using Qiagen AllPrep DNA/RNA/protein mini kit (no. 80004, Qiagen) according to the manufacturer’s instructions. The samples were kept at −20°C until analysis. cDNA was synthesized from total RNA using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems) and run on Gene Amp PCR System 9600 (Perkin Elmer). The RNA concentration was not assessed; however, to optimize each run, the expression level of β-actin (ACTB) was determined in each sample in a prerun. A second RNA extraction and cDNA synthesis was performed if the Ct value of ACTB was higher than 19.5. The criteria for a sample to be included in the study were Ct values below 19.5 for each of the two housekeeping genes and a variation between duplicates, calculated as ((standard deviation/mean) × 100), no higher than 0.5%.
Real-time quantitative polymerase chain reaction
The relative gene expression of 11 genes (Table 2) putatively involved in leucovorin transport, polyglutamation, and metabolism was quantified in tumors using real-time quantitative polymerase chain reaction (qPCR). The genes selected were the six most strongly associated with DFS in the previous study (ATP-binding cassette, subfamily C (CFTR), member 3 (ABCC3), folylpolyglutamate synthase (FPGS), gamma-glutamyl hydrolase (GGH), MTHFD1L, SLC46A1/PCFT, and SLC19A1/RFC-1), three genes (ATP-binding cassette, subfamily C (CFTR), member 1 (ABCC1), serine hydroxymethyltransferase 1 (SHMT1), and 5,10-methenyltetrahydrofolate synthetase (MTHFS)) associated with leucovorin metabolism according to the scientific literature, and two genes (aminomethyltransferase (AMT) and serine hydroxymethyltransferase 2 (mitochondrial) (SHMT2)) functional in the mitochondrial folate metabolism. TaqMan Gene Expression Assays (Life Technologies, Stockholm, Sweden) were ordered for each gene from Applied Biosystems at http://www.appliedbiosystems.com. The qPCR was set up in triplicates in 384-well plates using a Nanodrop II (GC Biotech) and was carried out in 5 μL reactions with 1× TaqMan® Gene Expression Mastermix (Applied Biosystems), 1× gene-specific TaqMan assay (Applied Biosystems), and 1 µL cDNA. The qPCR was run on a QuantStudioTM12K Flex Real-Time PCR System (Life Technologies) according to a standard protocol. Ct values were obtained when thresholds and baselines had been set in SDS manually. To compensate for between-run variations, values were adjusted to a control sample. Ct values were related to the endogenous housekeeping genes ACTB and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Table 2).
List of analyzed genes with putative impact on leucovorin metabolism (plus two housekeeping genes).

ABCC1/MRP1: ATP-binding cassette, subfamily C (CFTR/MRP), member 1; ABCC3/MRP3: ATP-binding cassette, subfamily C (CFTR/MRP), member 3; ACTB: β-actin; ATP: adenosine triphosphate; CFTR: cystic fibrosis transmembrane conductance regulator; FPGS: folylpolyglutamate synthase; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; GGH: gamma-glutamyl hydrolase; MRP : multidrug resistance protein; MTHFS: 5,10-methenyltetrahydrofolate synthetase; NADP: nicotinamide adenine dinucleotide phosphate; PCFT: proton-coupled folate transporter ; RFC-1: reduced folate carrier 1; SHMT1: serine hydroxymethyltransferase 1 (soluble); SHMT2: serine hydroxymethyltransferase 2 (mitochondrial).
Statistics
Statistical analysis was performed using the “survival” package in the R statistical software.11,12 Cox proportional-hazards regression models were applied to the normalized data deriving from the whole patient group, or from treatment subgroups, to examine the relationship between expression levels of chosen genes and DFS. The regression analysis was performed individually for each gene. Stepwise model selection by Akaike information criterion (AIC) was performed on Cox models, excluding the expression values, to choose between the numerous clinical covariates (gender (male or female), age, tumor differentiation (high/medium or low), tumor location (right-sided colon, left-sided colon, or rectum), lymph node ratio, adjuvant therapy (FLV or FLOX), preoperative radiotherapy (yes or no), elective surgery (yes or no), type of surgery (open or laparoscopic), and number of days between surgery and treatment start). AIC is a measure of goodness of fit and, as long as it improved the AIC value, the covariate that gave the best AIC if removed was deleted. The selected covariates were then included in the final models.
Values of p <0.05 were considered significant. The correlation between the two housekeeping genes ACTB and GAPDH was linear. The expression of the target genes was related to a mean value representing both of these genes in order to keep variance to the minimum. DFS was calculated from start of chemotherapy treatment to the last follow-up, or to the date of recurrence/new colorectal cancer or death by colorectal cancer. Patients without recurrent disease, that is who were disease-free at 5 years, or who died by any other cause than colorectal cancer during follow-up were censored. All-cause mortality and recurrence data were collected from patient journals and the Swedish Population Register. The difference in age between groups was calculated using the nonparametric Wilcoxon/Kruskal–Wallis tests (rank sums).
Results
Recurrence rate
During follow-up, 114 (31%) of the 363 patients suffered from recurrent disease; thus, the recurrence rate was the same as in the previous study. 4 The remaining 249 patients were censored, including three patients who moved away and 17 who died due to other causes than colorectal cancer. The median follow-up time was 5 years (range: 1.6–5 years). No significant differences were found when demographic and clinicopathological data of the 363 patients were compared with those of all stage III colorectal cancer patients who underwent elective surgery and adjuvant treatment during 2001–2013 (n = 555). The recurrence rates were also similar (31% vs 34%). Thus, the 363 patients constituted a representative group of stage III colorectal cancer patients.
Cox regression analysis
Nine patients were considered to be outliers due to very low expression of all analyzed genes, including the housekeeping genes. The nine patients were excluded from the statistical analyses. Cox univariate analysis of clinical and pathological covariates identified lymph node ratio as an important parameter having a hazard ratio of 4.8 (range: 2.0–10.9) per change in regressor over the entire range (p = 0.0005). No other clinical or pathological covariates were found to be significant in the univariate analysis. During stepwise variable selection, the variables that emerged and were included in the model were lymph node ratio, adjuvant therapy, and type of surgery (open or laparoscopic) for the whole group (n = 354); lymph node ratio and type of surgery for the FLV subgroup (n = 220); and lymph node ratio and tumor location for the FLOX subgroup (n = 134).
Disease-free survival
Gene expression results presented as median Ct values are shown in Table 3. An association between longer DFS and high tumoral expression of the genes SLC46A1/PCFT, SLC19A1/RFC-1, ABCC3/MRP3, GGH, MTHFD1L, and FPGS was seen and showed nominally significant p-values (Table 4). As shown in the table, each of these genes, except FPGS, had false discovery rate q-values <0.05. Estimated DFS by level of SLC46A1/PCFT and FPGS expression, that is, those two genes that had the highest and lowest significant nominal p-values, respectively, is shown in Figure 2. IQ ranges for normalized Ct values were determined (Table 4), to assure that acquired hazard ratios (HR) were large enough to be relevant. With HR of 1.3 and an IQ range of 1.8, SLC46A1/PCFT showed a 60% increased risk between the upper and the lower quartiles. For SLC19A1/RFC-1, ABCC3/MRP3, GGH, MTHFD1L, and FPGS, the increase was 58%, 36%, 38%, 35%, and 27%, respectively.
Expression levels of analyzed genes in tumor tissue of colorectal cancer patients.
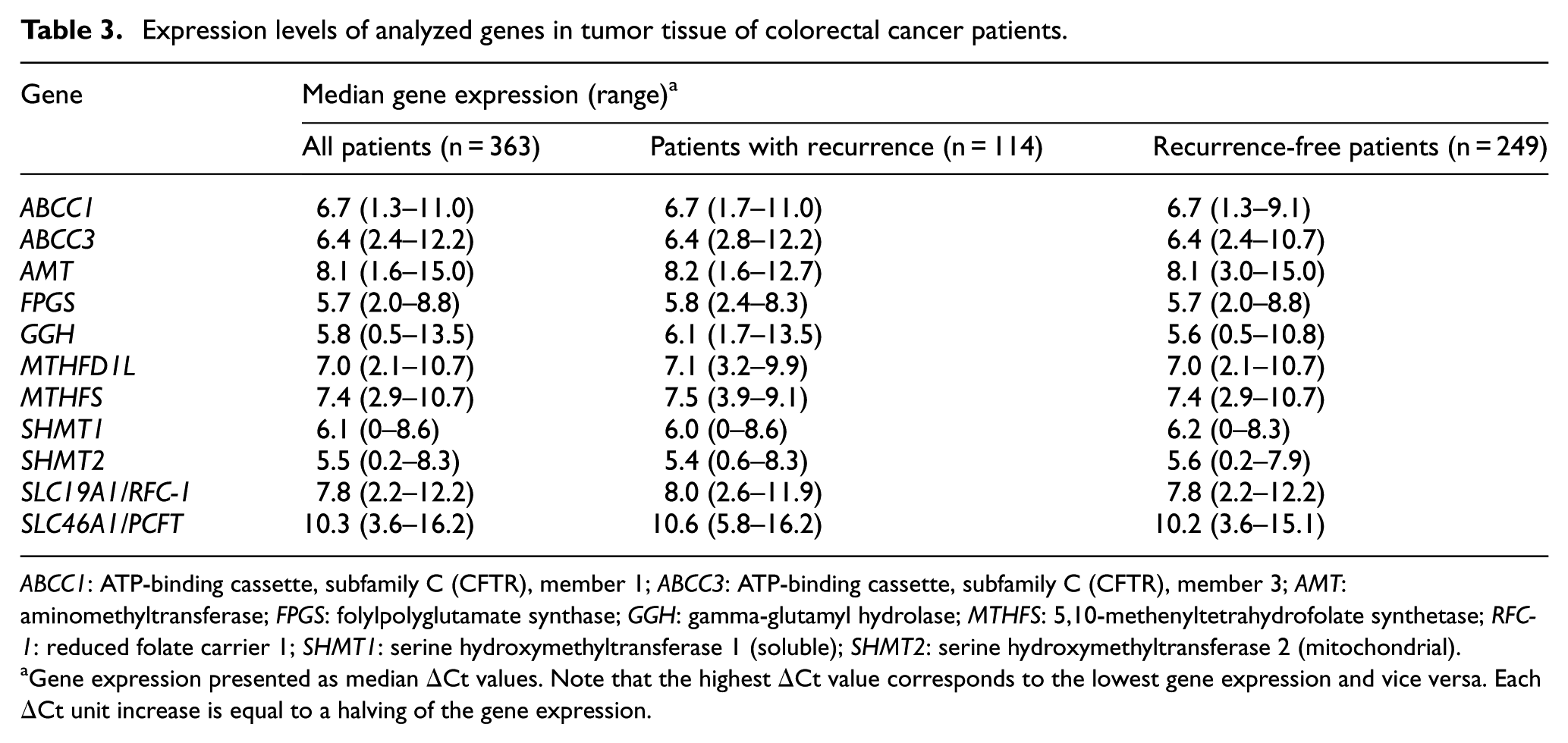
ABCC1: ATP-binding cassette, subfamily C (CFTR), member 1; ABCC3: ATP-binding cassette, subfamily C (CFTR), member 3; AMT: aminomethyltransferase; FPGS: folylpolyglutamate synthase; GGH: gamma-glutamyl hydrolase; MTHFS: 5,10-methenyltetrahydrofolate synthetase; RFC-1: reduced folate carrier 1; SHMT1: serine hydroxymethyltransferase 1 (soluble); SHMT2: serine hydroxymethyltransferase 2 (mitochondrial).
Gene expression presented as median ΔCt values. Note that the highest ΔCt value corresponds to the lowest gene expression and vice versa. Each ΔCt unit increase is equal to a halving of the gene expression.
Results of Cox regression analysis testing the association between gene expression in tumor tissue and disease-free survival.

ABCC1/MRP1: ATP-binding cassette, subfamily C (CFTR/MRP), member 1; ABCC3/MRP3: ATP-binding cassette, subfamily C (CFTR/MRP), member 3; AMT: aminomethyltransferase; FPGS: folylpolyglutamate synthase; GGH: gamma-glutamyl hydrolase; MTHFS: 5,10-methenyltetrahydrofolate synthetase; RFC-1: reduced folate carrier 1; SHMT1: serine hydroxymethyltransferase 1 (soluble); SHMT2: serine hydroxymethyltransferase 2 (mitochondrial); CI: confidence interval.
Hazard ratios were calculated from ΔCt values and correspond to a halving of the expression levels.
Interquartile range, calculated from normalized Ct values.
Nominal p-value.
False discovery rate q-value.
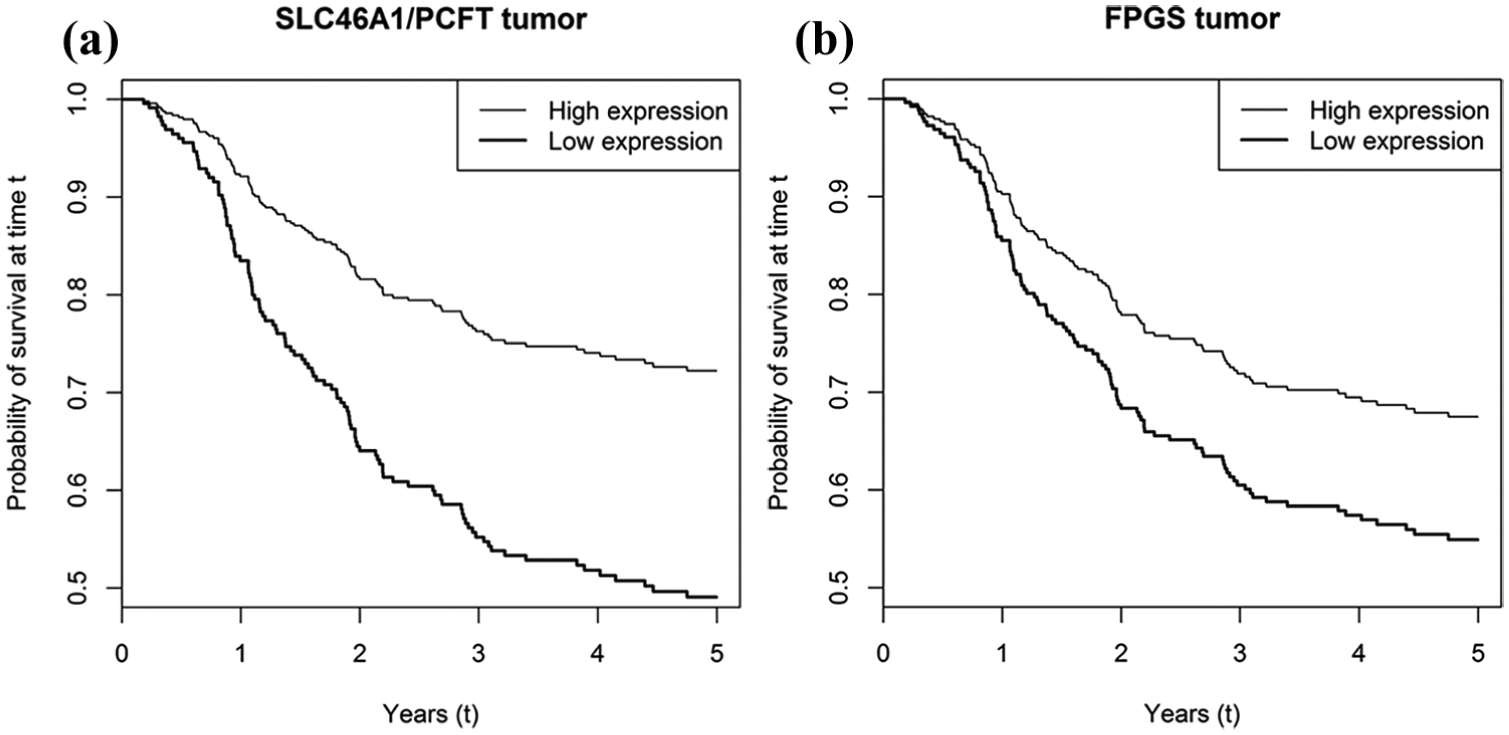
Estimated disease-free (DFS) survival functions by level of (a) SLC46A1/PCFT and (b) FPGS gene expression in colorectal cancer tissue. The two curves in each figure show predicted survival functions for the fitted Cox proportional hazards models. The expression levels for the two curves (high/low) have been set 1 standard deviation above and 1 standard deviation below the mean, where 1 standard deviation corresponds to a fold change in SLC46A1/PCFT and FPGS log expression levels of 1.51 and 1.02, respectively. Lymph node ratio has been set to its mean, surgical procedure has been set to open surgery, and adjuvant therapy has been set to 5-fluorouracil plus leucovorin, as this was the treatment given to most of the patients. Standard deviations and mean for expression levels have been calculated from ΔCt values.
Subgrouping by treatment
Subgrouping by treatment (Table 5) showed nominally significant p-values for an association between longer DFS and high expression of SLC46A1/PCFT (p = 0.002), SLC19A1/RFC-1 (p = 0.005), ABCC3/MRP3 (p = 0.009), GGH (p = 0.03), MTHFD1L (p = 0.01), and FPGS (p = 0.02) in patients treated with FLV, whereas SLC46A1/PCFT and SLC19A1/RFC-1 were nominally significant in patients treated with FLOX (p = 0.007 and p = 0.03, respectively). The q-values were <0.05 for SLC46A1/PCFT, SLC19A1/RFC-1, ABCC3/MRP3, and MTHFD1L in the FLV group, and for SLC46A1/PCFT in the FLOX group.
Results of Cox regression analysis testing the association between gene expression in tumor tissue and disease-free survival of patients grouped by treatment.
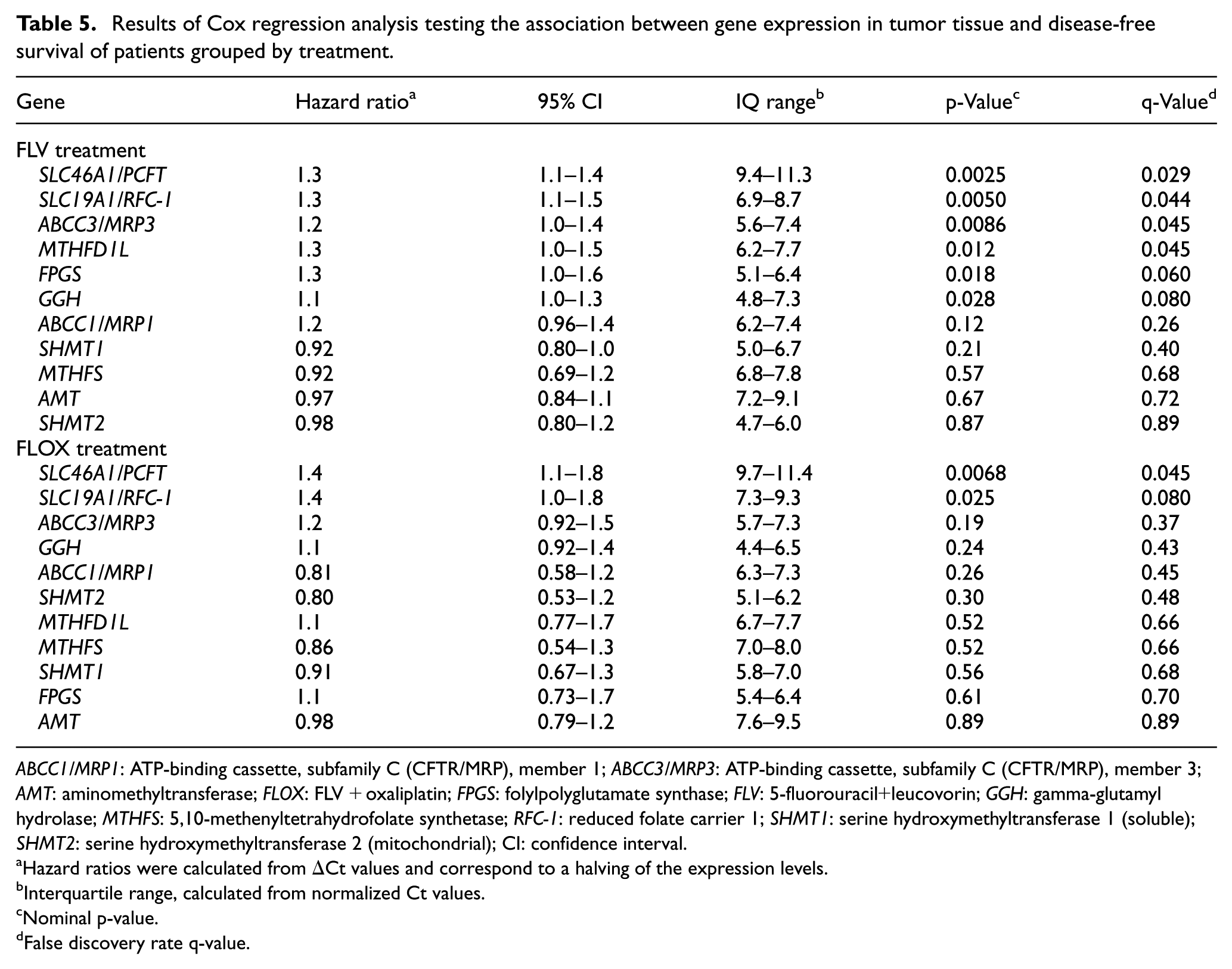
ABCC1/MRP1: ATP-binding cassette, subfamily C (CFTR/MRP), member 1; ABCC3/MRP3: ATP-binding cassette, subfamily C (CFTR/MRP), member 3; AMT: aminomethyltransferase; FLOX: FLV + oxaliplatin; FPGS: folylpolyglutamate synthase; FLV: 5-fluorouracil+leucovorin; GGH: gamma-glutamyl hydrolase; MTHFS: 5,10-methenyltetrahydrofolate synthetase; RFC-1: reduced folate carrier 1; SHMT1: serine hydroxymethyltransferase 1 (soluble); SHMT2: serine hydroxymethyltransferase 2 (mitochondrial); CI: confidence interval.
Hazard ratios were calculated from ΔCt values and correspond to a halving of the expression levels.
Interquartile range, calculated from normalized Ct values.
Nominal p-value.
False discovery rate q-value.
Discussion
The present study analyzed the relative expression of 11 folate-associated genes that may impact on leucovorin metabolism in patients with stage III colorectal cancer subjected to adjuvant bolus 5-FU-based chemotherapy and leucovorin. We chose to analyze snap-frozen tumor tissue, which in our experience yields high concentrations of high-quality mRNA. Analysis of microdissected formalin-fixed paraffin-embedded tissue may be useful when frozen tissue is not available and has the advantage of permitting identification of tissue morphology and cell type. The disadvantage, however, is that the small number of cells isolated may not be as representative of the tumor as a larger biopsy. Furthermore, degradation of RNA leading to loss of measurable transcripts is a common problem when working with formalin-fixed tissue. This degradation is dependent both on time passed from excision to fixation of specimen as well as the formalin fixation itself.
The selected genes encode folate transporters (ABCC1/MRP1, ABCC3/MRP3, SLC19A1/RFC-1, and SLC46A1/PCFT), and enzymes that take part in polyglutamation of folates (FPGS and GGH) or the folate metabolism (AMT, MTHFD1L, MTHFS, SHMT1, and SHMT2). The results showed that the two genes that were most strongly associated with DFS were SLC46A1/PCFT and SLC19A1/RFC-1. Thus, we could strengthen the results presented in a previous study, 4 showing that a high expression level of these two genes, which presumably corresponds to a functional inward transport of folates, is linked to a better outcome.
The ABCC1/MRP1 and ABCC3/MRP3 proteins belong to the superfamily of adenosine triphosphate-binding cassette (ABC) transporters. 13 Both proteins mediate cellular export of monoglutamated forms of folates. The ABCC1/MRP1 protein seems to be a promising predictive factor for reduced folate levels after leucovorin administration based on recent publications. 3 However, as was found in our previous study, expression of the ABCC1/MRP1 gene did not correlate with DFS. 4 In contrast, there was an association between high tumoral expression of the ABCC3/MRP3 gene and a lower risk of recurrence. An increase in ABCC3/MRP3 gene expression is expected to decrease intracellular levels of leucovorin, which, at first glance, seems to counteract the increased levels expected to result from high SLC19A1/RFC-1 and SLC46A1/PCFT expression. However, to convert leucovorin to methyleneTHF in the cells, several enzyme steps are needed. The first step, which is catalyzed by MTHFS, is considered to be “rate-limiting” with regard to the generation of methyleneTHF. The activity of MTHFS is inhibited by 10-formylTHF and studies have shown that the metabolism of leucovorin is dependent on both the activity of MTHFS and the level of 10-formylTHF. 14 Considering that the ABCC3/MRP3 protein has a preference for 10-formylTHF, 13 an increased expression of the ABCC3/MRP3 gene might lead to a lower cellular level of 10-formylTHF and decreased inhibition of MTHFS. This would result in an increased conversion rate of leucovorin to 5,10-methenylTHF and, subsequently, to methyleneTHF. Another possibility would be that ABCC3/MRP3 exports folates to the surrounding cells, which could be advantageous during treatment with leucovorin.
The expression of FPGS and GGH, which are involved in addition and removal of folate glutamates, respectively, was previously found to have impact on antitumor activity of 5-FU by regulating the efficacy of leucovorin. 15 In another study, high expression of FPGS and low expression of GGH were shown to predict high levels of reduced folates after administration of leucovorin in colorectal cancer. 3 Thus, our finding that high tumoral expression of FPGS was associated with a longer DFS was expected. However, the observation that high, rather than low, GGH expression was associated with longer DFS was somewhat unexpected. Possibly, a high expression of both of these genes in tumors indicates a high folate turnover rate, which may be advantageous during FLV-based treatment. The enzyme FPGS converts monoglutamated forms of folates to polyglutamates and is crucial for folate homeostasis and survival of proliferating cells. 16 The best folate substrate for FPGS is dihydrofolate, followed by THF, 10-formylTHF, methylTHF, and leucovorin. 17 Thus, leucovorin is not an ideal substrate for FPGS, and cytosolic monoglutamate forms of this reduced folate will leave the cell rapidly. However, the folate metabolism in eukaryotic cells is highly compartmentalized and there are differences between the cytosolic and mitochondrial pools with regard to distribution of specific folate metabolites, and degree of folate polyglutamation. 18 The mitochondria seem to receive folates from the cytosol in a reduced monoglutamate form (preferably THF or 5-formylTHF), which subsequently is polyglutamated by mitochondrial FPGS. Although only monoglutamated folates can enter the mitochondria, folate polyglutamates can exit the mitochondria without prior cleavage of the polyglutamate chain. The enzyme GGH is primarily located to the lysosomes in the cytoplasm, where it cleaves gamma-polyglutamate chains attached to folates. 19 The fate of the monoglutamated folates generated in the lysosome is not known, but it has been suggested that they may be released into the cytosol via the SLC46A1/PCFT transporter. 20 If this is the case, a high GGH expression may increase the availability of monoglutamated folates for passage into the mitochondria.
Interestingly, folate transport, polyglutamation, and metabolism in tumor tissue might be linked to mitochondrial biogenesis and respiration. The gene SLC46A1/PCFT has been shown to be highly regulated by nuclear respiratory factor 1 (NRF-1), the dominant transcription factor that regulates mitochondrial biogenesis and respiration. 21 NRF-1 silencing leads to downregulation of SLC46A1/PCFT as well as other folate-associated genes, including SLC19A1/RFC-1, FPGS, GGH, SHMT1, and SHMT2. In addition, NRF-2, which is another key mitochondrial transcription factor, regulates ABCC3/MRP3. 22 Furthermore, the present results showed that high tumoral expression of MTHFD1L was associated with a decreased risk of recurrent disease. This gene encodes an enzyme that localizes to the mitochondria, where it catalyzes the reversible conversion of 10-formylTHF to THF. 19 As mentioned, 10-formylTHF is converted to methyleneTHF; however, high levels of 10-formylTHF may inhibit MTHFS, which is active in the first conversion step of leucovorin. Thus, the reaction from 10-formylTHF to THF, and subsequent conversion of THF to methyleneTHF (catalyzed by the enzyme SHMT1) might be favorable during FLV treatment because the inhibition of MTHFS would then decrease. 23
After subgrouping of patients by treatment, the SLC46A1/PCFT, SLC19A1/RFC-1, ABCC3/MRP3, GGH, and MTHFD1L gene expression levels were found to be significantly associated with DFS in the FLV group. Thus, the results of the whole group regarding these folate-associated genes were also reflected in the FLV group. In the FLOX group, however, only SLC46A1/PCFT gene expression was significantly associated with DFS. The difference between the two groups may be due to the fact that the FLOX group consisted of fewer patients, and hence, fewer recurrence events occurred during follow-up compared to the FLV group. The age difference between the groups might also have affected the results. It is known that folate deficiency increases with age 24 and the significantly older patients of the FLV group might have lower tumoral folate levels than patients of the FLOX group. Thus, the expression levels of folate-associated genes might have a higher impact in the FLV group.
Although the analyzed genes were chosen due to a known impact on the folate pathway, additional studies are needed to determine whether the expression of these genes is useful when predicting response to leucovorin and other folates that may be used in the clinical setting, for example methyleneTHF.25,26 The results of the present study will be compared with results of untreated patients with stage III colorectal cancer in order to exclude a possible prognostic effect. The correlation between gene expression and different folate metabolites also needs to be assessed and compared in relation to various colorectal cancer treatment protocols, for example FLV-based treatment given as a bolus versus continuous infusion. It has been suggested that 5-FU given as bolus injection mainly acts by incorporation of 5-FU into RNA, whereas continuous infusion acts through inhibition of TS. However, studies show that although the incorporation into RNA is higher in response to bolus 5-FU compared to infusion, the inhibition of TS seems to be equal between the two administrations.27–29 Thus, studies of folate-associated genes are relevant regardless of the method used for 5-FU administration.
Overall survival was not analyzed in the present study because most patients suffering from recurrent disease during follow-up underwent different palliative treatments, including chemotherapy and, in some cases, surgical removal of metastases. Thus, it is hard to evaluate results regarding overall survival.
Conclusion
The present study showed that high tumoral expression of the genes SLC46A1/PCFT, SLC19A1/RFC-1, ABCC3/MRP3, GGH, and MTHFD1L, which take part in folate transport, polyglutamation, and metabolism, was associated with longer DFS of patients with colorectal cancer. All of these genes either encode mitochondrial enzymes or are being regulated by mitochondrial transcription factors, indicating a possible link between folate pathway genes and mitochondrial biogenesis and respiration. In patients with low tumoral expression of the identified genes, leucovorin may not be the most optimal folate to modulate the efficacy of 5-FU. Expression of the SLC46A1/PCFT gene was most strongly associated with DFS, regardless of treatment regimen. A prospective study needs to be conducted in order to determine if the expression level of SLC46A1/PCFT or other folate-associated genes can be used to predict response to leucovorin and other folates that are now being tested in clinical studies.
Footnotes
Acknowledgements
We thank H. Björkqvist and A-L. Helminen for collection of surgical samples, L. Munro and B. Sjöberg for work with the clinical database, and J. Flach and M. Åkerström for technical assistance. We also thank the personnel at Genomics and Bioinformatics Core Facilities for performing quantitative gene expression analyses and for statistical analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Swedish Cancer Society (CAN 2015/499), the King Gustav V Jubilee Clinic Foundation for Cancer Research (2016:70), the Swedish state under the LUA/ALF agreement (ALFGBG-426941), the IngaBritt and Arne Lundberg Foundation (335/07), and partially funded by Isofol Medical AB. A patent application related to the results of the study is pending. B.G. is a shareholder in Isofol Medical AB.
