Abstract
The differential diagnosis of malignant pleural effusion and benign pleural effusion remains a clinical problem. Reactive oxygen species modulator 1 is a novel protein overexpressed in various human tumors. The objective of this study was to evaluate the diagnostic value of joint detection of reactive oxygen species modulator 1 and carcinoembryonic antigen in the differential diagnosis of malignant pleural effusion and benign pleural effusion. One hundred two consecutive patients with pleural effusion (including 52 malignant pleural effusion and 50 benign pleural effusion) were registered in this study. Levels of reactive oxygen species modulator 1 and carcinoembryonic antigen were measured by enzyme-linked immunosorbent assay and radioimmunoassay, respectively. Results showed that the concentrations of reactive oxygen species modulator 1 both in pleural fluid and serum of patients with malignant pleural effusion were significantly higher than those of benign pleural effusion (both p < 0.05). The diagnostic sensitivity and specificity of pleural fluid reactive oxygen species modulator 1 were 61.54% and 82.00%, respectively, with the optimized cutoff value of 589.70 pg/mL. However, the diagnostic sensitivity and specificity of serum reactive oxygen species modulator 1 were only 41.38% and 86.21%, respectively, with the cutoff value of 27.22 ng/mL, indicating that serum reactive oxygen species modulator 1 may not be a good option in the differential diagnosis of malignant pleural effusion and benign pleural effusion. The sensitivity and specificity of pleural fluid carcinoembryonic antigen were 69.23% and 88.00%, respectively, at the cutoff value of 3.05 ng/mL, while serum carcinoembryonic antigen were 80.77% and 72.00% at the cutoff value of 2.60 ng/mL. The sensitivity could be raised to 88.17% in parallel detection of plural fluid reactive oxygen species modulator 1 and carcinoembryonic antigen concentration, and the specificity could be improved to 97.84% in serial detection.
Introduction
As a common clinical syndrome, pleural effusion remains a public health problem, of which the etiology is very diverse and complicated. A research showed that cancer (27%), heart failure (21%), pneumonia (19%), and tuberculosis (9%) were four main causes of pleural effusion. 1 Malignant pleural effusion (MPE) is the most common pleural effusion which has an estimated annual incidence of 150,000 to 175,000 in the United States. 2 Patients with MPE have poorer life quality and shorter life expectancy. Therefore, the diagnosis of MPE is very significant but still difficult with the currently available parameters derived from thoracocentesis. The golden standard for diagnosing MPE is the observation of pathological changes of malignancies in pleural biopsy, but only about 60% cases can be diagnosed by golden standard. 3 Thus, more invasive procedures such as thoracoscopy or thoracotomy will be necessary to obtain the diagnosis with higher expense, increased trauma, and prolonged hospital stay. 4 In the past few years, numerous researches have investigated the role of biomarkers to improve the diagnostic ability of pleural fluid analysis, but no consensus has been achieved for a highly efficient marker because of varied test accuracies. Therefore, a more efficient marker of pleural fluid is required urgently.
Reactive oxygen species modulator 1 (Romo1), a novel mitochondrial membrane protein with molecular weight of about 8.9 kDa, was first cloned after screening from a human lymph node complementary DNA (cDNA) library in 2006. 5 Then, it was first discovered in the tumor tissue of a patient with head and neck cancer who became resistant to chemotherapy after recurrence. 6 The gene encoding this protein is localized in chromosome 20. Romo1 is responsible for increasing the level of reactive oxygen species (ROS) in cells and also has antimicrobial activity against a variety of bacteria by inducing bacterial membrane breakage. Numerous researches demonstrated that Romo1 is up-regulated in a variety of human tumors, including lung cancer, gastric cancer, and hepatic tumor cells.7–9 Recently, Romo1 expression in tissue and concentration in serum have been the focus of researches as diagnostic and prognostic markers for various human malignancies. However, the expression of Romo1 in pleural effusion was not researched yet and data on its expression in patients with malignancy were also very limited. Therefore, we evaluated the value of Romo1 as a potential diagnostic biomarker in MPE in this study. We hypothesized that Romo1 might be a useful marker for the differential diagnosis of MPE and benign pleural effusion (BPE).
Materials and methods
Patients
A total of 102 consecutive patients with pleural effusion from the First Affiliated Hospital of Anhui Medical University between April 1, 2015 and April 1, 2016 were enrolled in the study.
All pleural effusion was definitely diagnosed by history, clinical manifestation, examination of effusion biochemistry or cytology or bacteriology, pleural biopsy, endoscopic examination, and percutaneous biopsy.
The protocol of this study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University. All patients signed written informed consent before enrollment.
Diagnostic criteria for pleural effusion
The pleural effusions were first classified as exudate or transudate not only using Light’s criteria 10 but also according to clinical data and laboratory or imaging examination.
MPE was diagnosed if pathological changes of malignancies were observed in pleural biopsy or malignant cells were found in biopsy specimen and on cytologic examination. Diagnosis of different malignancies was based on histology or cytology.
Tuberculous pleural effusion was determined if one of the following criteria was met: acid-fast bacilli were identified at least in one of the following specimens such as pleural fluid or sputum or pleural biopsy tissue, pleural effusion was absorbed as a result of antituberculous therapy, and unexplained lymphocytic exudate (lymphocyte >50%) with a high level of pleural fluid adenosine deaminase (>40 U/L), which responded to antituberculous therapy.
Parapneumonic effusion was diagnosed when there was a positive gram stain or culture of the pleural fluid or an acute febrile illness with purulent sputum, pulmonary infiltration, and response to antibiotic treatment.
Pleural effusions were split into two groups depending on diagnosis (pleural effusion of malignant origin vs pleural effusion of benign origin). Pleural fluid and serum samples from patients prior to any therapy were collected in tubes containing ethylenediaminetetraacetic acid (EDTA) for detection of Romo1 and carcinoembryonic antigen (CEA) levels. In addition, pleural fluid samples were collected in sterile containers for microbiology studies. The serum and supernatant of the pleural fluid obtained by centrifugation at 3000g for 10 min were stored at −80°C until assayed.
Measurements of Romo1 and CEA
Levels of Romo1 in pleural fluid and serum were determined by enzyme-linked immunosorbent assay (ELISA) method with the Romo1 ELISA kit (Wuhan EIAab Science). First, 100 µL of standards and pleural fluid or serum samples was added to ELISA plate per well with incubation for 2 h at 37°C, and the liquid of each well was removed but not washed. Second, 100 µL of detection reagent A working solution was added to each well and covered with the plate sealer followed by 1 h incubation at 37°C. Then, each well was aspirated and washed and the process was repeated for three times. Third, 100 µL of detection reagent B working solution was added to each well and incubated for 1 h at 37°C. Then, 90 µL of substrate solution was added to each well and allowed to incubate within 15–30 min at 37°C, causing the conversion of the colorless solutions to blue solutions, the intensity of which was proportional to the levels of Romo1 in the samples. Finally, 50 µL of stop solution was added to each well causing the color turned yellow. The color change was measured spectrophotometrically at a wavelength of 450 ± 2 nm, and the concentration of Romo1 in the samples was then determined by comparing the optical density (OD) of the samples with the standard curve.
The levels of CEA in pleural fluid and serum were measured using radioimmunoassay (RIA) by the Department of Laboratory of the First Affiliated Hospital of Anhui Medical University.
Statistical analysis
All data were analyzed by Statistical Package for the Social Science Version 16.0 (SPSS, Inc., Chicago, IL, USA) statistically. Kolmogorov–Smirnov and Shapiro–Wilk tests were used to determine whether sample data met normal distribution or not. Normally distributed variables were expressed as mean ± standard deviation, while variables with non-Gaussian distribution were expressed as median (interquartile range). For continuous variables, comparison between groups was conducted using t test for normally distributed. The Kruskal–Wallis or Mann–Whitney U test was used for non-Gaussian distribution, while chi-square test or Fisher’s exact test was used for categorical variables. The areas under the curves (AUCs) of the biomarkers were compared, and the Youden indices by conducting receiver operating characteristic (ROC) curves were calculated to evaluate experimental data and select the cutoff values of Romo1 and CEA. The Youden index was defined as follows: sensitivity + specificity − 1. The index was defined for all points of an ROC curve. The optimal cutoff value has the highest Youden index and clinical significance. To analyze the relationship between variables, the Pearson correlation test was used for bivariate normal distribution, while the Spearman correlation test was used for non-normal distribution. Values of p < 0.05 were considered statistically significant.
Results
Patients’ clinical characteristics
One hundred two patients were enrolled in the study including 52 patients with MPE and 50 patients with BPE. Among 102 patients of the study, 75.50% (77/102) were male and 24.51% (25/102) were female. All were Chinese and their average age was 59.83 years. There were no significant differences in the distribution of ages, genders, and main symptoms among groups (all p > 0.05). The causes of the pleural effusions of these 102 patients were presented in Table 1. Demographic and clinical characteristics and biochemical data of the studied groups were summarized in Table 2.
Etiology of pleural effusions.

Demographic, clinical, and biochemical characteristics of the studied groups.

MPE: malignant pleural effusion; BPE: benign pleural effusion; ADA: adenosine deaminase; BNP: brain natriuretic peptide; Hb: hemoglobin; LDH: lactate dehydrogenase; CRP: C-reactive protein; PF: pleural fluid.
p < 0.05 (significant difference vs the BPE group); **p < 0.001(significant difference vs the BPE group).
Romo1 levels
Table 3 and Figure 1 show that the concentration of Romo1 in pleural fluid of patients with MPE (651.48 (353.48–1000.50) pg/mL) was significantly higher than in those with BPE (350.98 (89.41–534.64) pg/mL) (p < 0.001). The ROC curve and diagnostic value for pleural fluid Romo1 were presented in Figure 2 and Table 5, which show an AUC of 0.757 with a value of 589 pg/mL as the best threshold of pleural fluid Romo1 for distinguishing MPE and BPE. At a cutoff value of 589.70 pg/mL, pleural fluid Romo1 had a sensitivity of 61.54% (32/52), a specificity of 82.00% (41/50), an accuracy of 71.57% (73/102), a positive predictive value (PPV) of 78.05% (32/41), a negative predictive value (NPV) of 67.21% (41/61), a positive likelihood ratio (PLR) of 3.14, and a negative likelihood ratio (NLR) of 0.47.
Levels of Romo1 and CEA in both pleural effusion and serum.
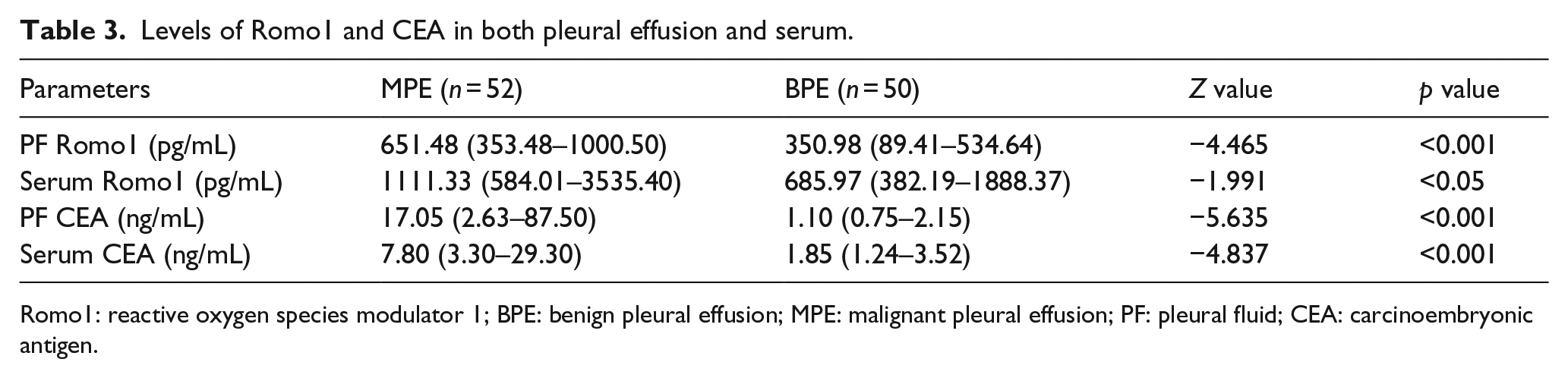
Romo1: reactive oxygen species modulator 1; BPE: benign pleural effusion; MPE: malignant pleural effusion; PF: pleural fluid; CEA: carcinoembryonic antigen.

Box-whisker plot representing the levels of pleural fluid Romo1 in MPE group and benign BPE group. The concentration of pleural fluid Romo1 in malignant group was higher than that in benign group (651.48 (353.48–1000.50) pg/mL vs 350.98 (89.41–534.64) pg/mL) (p < 0.001).

ROC curves of pleural fluid Romo1 (PF Romo1), pleural fluid CEA (PF CEA), and serum CEA for predicting malignant pleural effusion (MPE), with the areas under the curves 0.757, 0.830, and 0.778.
With a cutoff value of 27.22 ng/mL, serum Romo1 had a sensitivity of 41.38% (12/29), a specificity of 86.21% (25/29), an accuracy of 63.79% (37/58), a PPV of 75.00% (12/16), an NPV of 59.52% (25/42), a PLR of 3.00, and an NLR of 0.68. The area under the Romo1 ROC curve was 0.652. These results showed that serum Romo1 had little value in differentiating malignant from benign pleural effusions.
CEA levels
Table 3 also show that patients with MPE (17.50 (2.63–87.50) ng/mL) had significantly higher pleural fluid CEA concentration as compared with BPE (1.10 (0.75–2.15) ng/mL) (p < 0.001) and the concentration of serum CEA of patients with MPE (7.80 (3.30–29.30) ng/mL) was similarly higher than that in those with BPE (1.85 (1.24–3.52) ng/mL) (p < 0.001).
The ROC curves and diagnostic value for CEA were presented in Figure 2 and Table 5, respectively. The diagnostic threshold afforded by the ROC analysis for pleural fluid CEA was 3.05 ng/mL, and the area under the pleural fluid CEA ROC curve was 0.830 which was higher compared with the area of Romo1. With a cutoff point of 3.05 ng/mL, pleural fluid CEA had a sensitivity of 69.23% (36/52), a specificity of 88.00% (44/50), an accuracy of 78.43% (100/102), a PPV of 85.71% (36/42), and an NPV of 73.33% (44/60), a PLR of 5.77, and an NLR of 0.35. Meanwhile, with a cutoff point of 2.60 ng/mL, serum CEA had a sensitivity of 80.77% (42/52), a specificity of 72.00% (36/50), an accuracy of 76.47% (78/102), a PPV of 75.00% (42/56), and an NPV of 78.26% (36/46), a PLR of 2.88, and an NLR of 0.27. The area under the serum CEA ROC curve was 0.778.
Table 4 show the concentration of pleural fluid Romo1 and CEA in different causes of pleural effusion. No significant differences were found in concentration of pleural fluid Romo1 among patients with lung cancer and patients with breast cancer, gastrointestinal cancer, ovarian cancer, lymphoma, and unknown primary site (p > 0.05). However, there were significant differences in levels of pleural fluid CEA among different causes of pleural effusion (p < 0.05).
Levels of Romo1 and CEA in different diagnosis of pleural effusion.
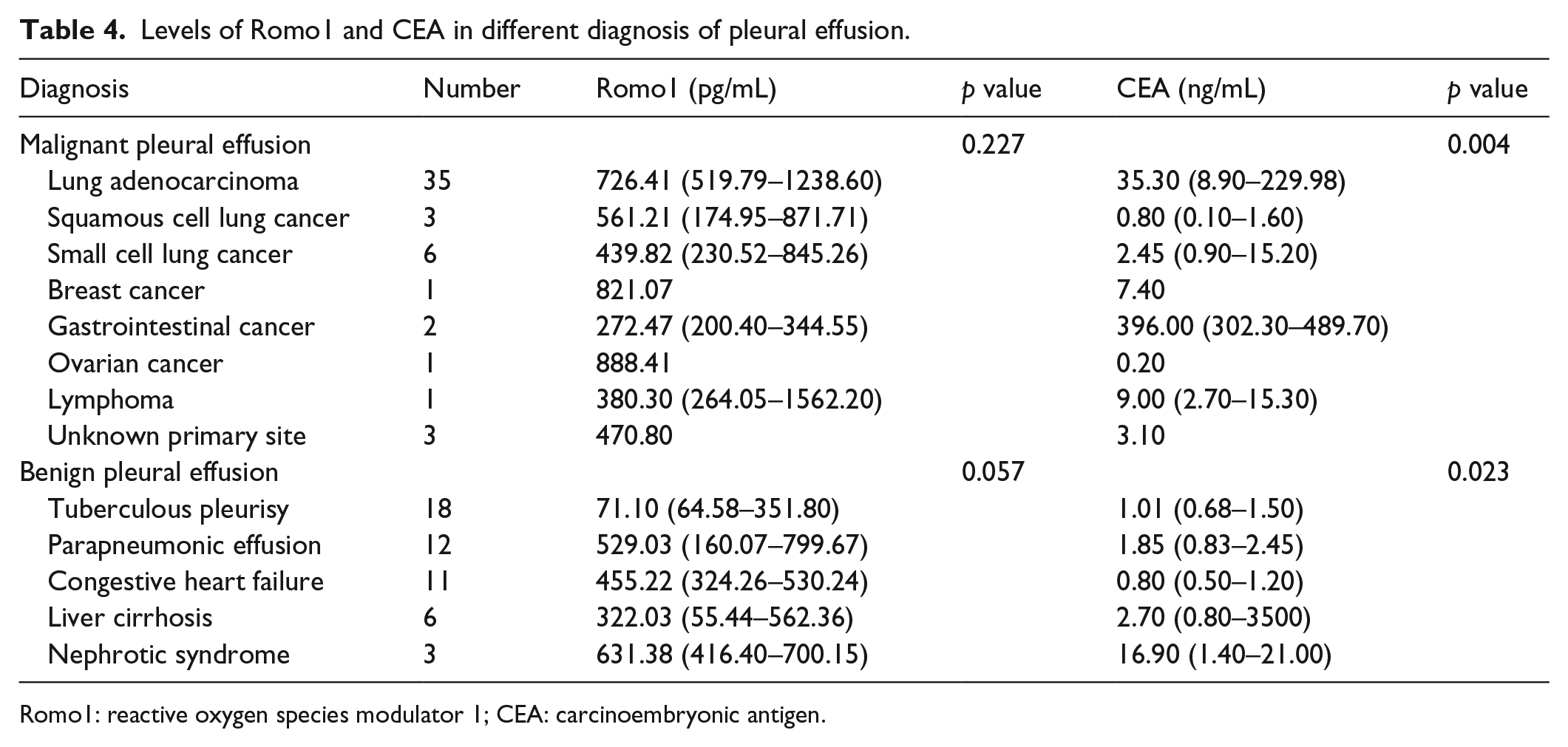
Romo1: reactive oxygen species modulator 1; CEA: carcinoembryonic antigen.
The diagnostic value of combined detection of Romo1 and CEA in MPE and BPE was further analyzed (Table 5). A parallel test of the two indices can improve diagnostic sensitivity, while a serial test may elevate specificity. The joint detection of pleural fluid Romo1 and CEA revealed a sensitivity of 88.17% and a specificity of 97.84%, which were higher than Romo1 and CEA alone. To determine the relationship between concentration of Romo1 in pleural fluid and serum, Spearman’s correlation analysis was performed in paired pleural fluid and serum. Results showed that there was no relationship between Romo1 expression in pleural fluid and serum (p = 0.714).
Diagnosis values of Romo1 and CEA for predicting MPE.
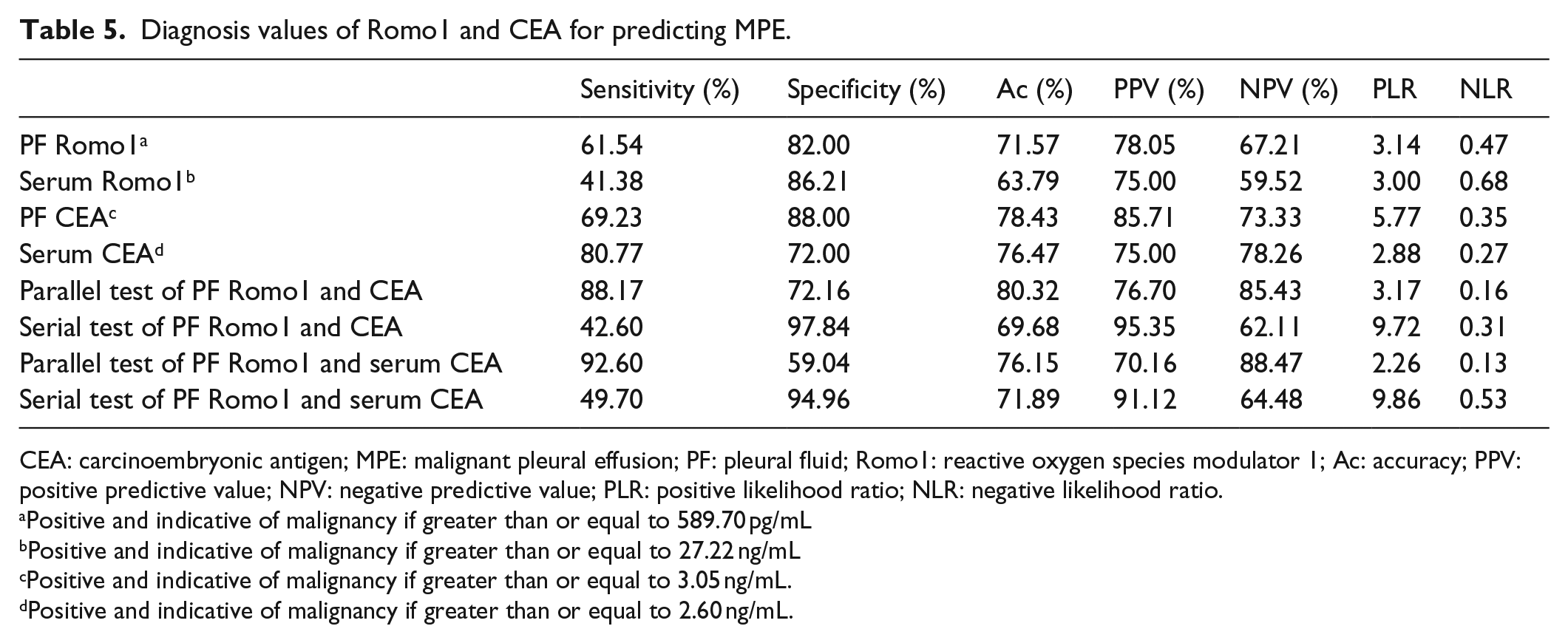
CEA: carcinoembryonic antigen; MPE: malignant pleural effusion; PF: pleural fluid; Romo1: reactive oxygen species modulator 1; Ac: accuracy; PPV: positive predictive value; NPV: negative predictive value; PLR: positive likelihood ratio; NLR: negative likelihood ratio.
Positive and indicative of malignancy if greater than or equal to 589.70 pg/mL
Positive and indicative of malignancy if greater than or equal to 27.22 ng/mL
Positive and indicative of malignancy if greater than or equal to 3.05 ng/mL.
Positive and indicative of malignancy if greater than or equal to 2.60 ng/mL.
Romo1 and smoking
It is well known that cigarette smoking is related to both intracellular oxidative stress and lung cancer. Therefore, we further categorized smoking intensity into four groups (0, 1–20, 21–40, and over 40 pack-months) to analyze the relationship between Romo1 concentration and smoking intensity in MPE patients. Results showed that there were no significant differences in pleural fluid Romo1 concentration and smoking status. Meanwhile, no significant difference was found between or among different groups when malignant effusions were classified according to age or gender (Table 6).
The levels of PF Romo1 in MPE patients with different demographic characteristics.

PF: pleural fluid; Romo1: reactive oxygen species modulator 1; MPE: malignant pleural effusion.
Discussion
MPE, as one of the most common complications of advanced cancer, is also the first symptom of malignant tumors or recurrent symptom after cancer treatment. The main mechanism of MPE is the invasion and metastasis of malignant tumor on pleura. More and more studies confirmed that almost all kinds of malignant tumors can develop MPE, but lung cancer is thought to be the most common cause. 11 Approximately 15% of lung cancer patients have a pleural effusion at the time of initial diagnosis, and 50% develop a pleural effusion in their courses. 12 Development of lung cancer is a complicated process that is still not fully understood, but many researches firmly indicated that oxidative stress is an important mechanism of various human cancers. 13 Moreover, Romo1-induced ROS up-regulation was observed in different kinds of cancer including lung cancer.
Romo1 is a novel protein located in the mitochondrial membrane which can regulate intracellular ROS generation. Researches demonstrated that Romo1 induced ROS generation through the complex III of the mitochondrial electron transport chain. 14 Na et al knocked down Romo1 by small inhibitory RNA transfection, resulting in growth inhibition of both normal and cancer cells. They also found that cell proliferation could be inhibited by using extracellular signal–regulated kinase (ERK) inhibitor. 15 Romo1 was also reported to be a molecular bridge between tumor necrosis factor-α (TNF-α) signaling and the mitochondria for intracellular ROS generation. Meanwhile, a recent study showed that Romo1 played an important role in Myc turnover and the Myc protein was known to play an important role in cell proliferation. Myc protein was up-regulated in response to growth-stimulating signals that triggered the cell cycle progression. 16 Above all, Romo1-induced ROS can regulate normal and cancer cells proliferation mainly through the signal transduction pathway such as ERK signaling pathway. High levels of ROS can induce oxidative stress in cells, causing gene mutation, which ultimately leads to the occurrence and development of tumor.13,17 Overexpression of Romo1 by all kinds of carcinogenic factors increases intracellular ROS levels, resulting in cellular DNA damage and genomic instability, which can contribute to the initiation, promotion, and malignancy of tumors. In addition, Romo1 was reported to be correlated with tumor resistance to chemotherapeutic agents such as 5-fluorouracil (5-FU). Romo1-induced ROS production could cause massive cell death, but tumor cells adapt to oxidative stress by increasing manganese superoxide dismutase (MnSOD) and peroxiredoxin I (Prx I), and B-cell lymphoma 2 (Bcl-2) showed drug resistance to 5-FU. 18 Therefore, the expression of Romo1 may have a new clinical significance for the choice of tumor chemotherapy scheme. Recently, several researches have indicated that Romo1 was overexpressed in many kinds of human tumors, suggesting that this protein might be a new molecular biomarker for tumor diagnosis and prognosis. A recent study by Lee et al showed that Romo1 expression in lung cancer tissues was significantly increased compared with non-tumorous tissues (p < 0.001) and serum Romo1 concentration in patients with non–small cell lung cancer (NSCLC) was higher than that of healthy population or patients with benign lung diseases (both p < 0.001). Serum Romo1 revealed a sensitivity of 81.9% and a specificity of 89.8% for the diagnosis of NSCLC using an optimal cutoff value of 329.7 pg/mL. 7 In this study, we successfully demonstrated that Romo1 expression in MPE was up-regulated compared with BPE. Results showed that pleural fluid Romo1 had a sensitivity of 61.54% and a specificity of 82.00% for diagnosis of MPE at the cutoff value of 589.70 pg/mL. Furthermore, we found no relationship among concentration of Romo1 in MPE and ages, genders, and smoking status. From these results, Romo1 played an important role in differential diagnosis of BPE and MPE. To the best of our knowledge, this was the first study on pleural effusion in which Romo1 levels were investigated for its diagnostic value of MPE.
Our data demonstrated the possibility of Romo1 as a potential diagnostic biomarker for MPE. In recent years, different tumor biomarkers have been studied in numerous researches to improve the value of pleural fluid analysis in the diagnosis of MPE including antibodies, messenger RNA (mRNA) and micro RNA (miRNA), DNA methylation products, and circulating tumor cells. However, because of the scarce quantity of the biomarker, complexity of measurements, high cost of measurements, and lack of a well-verified assay technique, their clinical application was limited. 19 Thus, Romo1 may be a potential and suitable biomarker for the diagnosis of MPE because of its large quantity of expression, considerable discriminating performance, low cost, and facility of measurement using a widely applied assay technique.
The results of our study again confirmed CEA as a useful tumor marker in pleural fluid. In our study, CEA had a sensitivity of 69.23% and a specificity of 88.00% at the cutoff value of 3.05 ng/mL. A meta-analysis showed that the summary sensitivity and specificity for CEA in the diagnosis of MPE were 54.00% and 94.00%, respectively. 20 CEA, one of the most widely used tumor markers, is elevated in many malignancies such as lung cancer, gastrointestinal cancer, and breast cancer. 21 CEA is also considered to be an independent prognostic factor for NSCLC. 22 However, single tumor marker is hard to achieve the desired level of sensitivity and specificity in detecting lung cancer and MPE. Combination of CEA and other potential biomarkers can get a more precise diagnosis of malignancies. In this study, we found that 16 of the 52 patients with MPE did not show elevated levels of CEA in pleural fluid. However, in these 16 cases, the levels of Romo1 in pleural fluid were significantly increased (p < 0.001), indicating that Romo1 might be a sufficiently good marker for diagnosis of MPE when CEA was not elevated.
Diagnostic tests used in clinical work have different specificity and sensitivity. We can combine multiple diagnostic tests to achieve better diagnosis. There are two methods for the joint of multiple diagnostic tests: parallel test and serial test. A parallel test indicates that any test positive can be used as evidence of the presence of the disease, while a serial test indicates that all tests positive can be considered as evidence of the existing disease. Our study demonstrated that both CEA and Romo1 had a certain value in the differential diagnosis of BPE and MPE. The combined diagnostic value of Romo1 and CEA in pleural effusion was also analyzed. Combination detection of the two indices gained a sensitivity of 88.17% and a specificity of 72.16% in parallel test, and a specificity of 97.84% and a sensitivity of 42.60% in serial test. Parallel test of Romo1 and CEA can significantly improve the diagnostic sensitivity and effectively avoid missed diagnosis for high-risk population screening MPE, while serial test can improve the diagnostic specificity, reduce misdiagnosis rate, and provide a reliable basis for clinical diagnosis. The golden standard for diagnosing MPE is histopathologic diagnosis of primary neoplasm combined with observation of pathological changes of malignancies in pleural biopsy. However, only about 60% cases can be diagnosed by the golden standard. In the remaining 40% of undiagnosed cases, a significant elevated level of Romo1 or CEA in pleural fluid may suggest the possibility of MPE. Moreover, the levels of both Romo1 and CEA were significantly increased, which more likely to indicate MPE. Therefore, the combination of Romo1 and CEA in pleural fluid is useful in assessing the risk of MPE and in determining whether further invasive measures are needed to prevent missed diagnosis.
In conclusion, the results of our study indicated the possibility of Romo1 as a potential biomarker in the differential diagnosis of MPE and BPE. Moreover, the combination of pleural fluid Romo1 and CEA improved the sensitivity and specificity in the diagnosis of MPE. At present, there is no other report on the detection of Romo1 in pleural effusion. However, our study only used pre-chemotherapeutic specimens and did not analyze follow-up change of the biomarker after chemotherapy, and we demonstrated the diagnostic value of Romo1 but did not evaluate the prognostic utility of Romo1 simultaneously. Therefore, further studies are required to assess the real clinical utility of Romo1 in the diagnosis of pleural effusion.
Footnotes
Acknowledgements
X.C. and N.Z. are equal contributors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Key Clinical Specialist Construction Programs of China (No. 3101005005025).
