Abstract
Granulocyte-macrophage colony-stimulating factor has been widely used as an adjuvant therapy for cancer patients exhibiting myelosuppression induced by chemotherapy or radiotherapy. However, the effects of granulocyte-macrophage colony-stimulating factor on tumor growth, as well as its precise mechanism, are still controversial due to inconsistent evidence. This study investigated the effect of exogenous granulocyte-macrophage colony-stimulating factor on the growth of B16 melanoma, S180 sarcoma, and U14 cervical carcinoma in mice. The angiogenesis and recruitment of bone-marrow-derived cells were analyzed in tumor tissues. Interactions among granulocyte-macrophage colony-stimulating factor, bone-marrow-derived cells, and B16 tumor cells were investigated in vitro. Proangiogenic types of bone-marrow-derived cells in blood were assessed both in vivo and in vitro. The results showed that granulocyte-macrophage colony-stimulating factor markedly facilitated the growth of B16 and S180 tumors, but not U14 tumors. Granulocyte-macrophage colony-stimulating factor increased the densities of blood vessels and the number of bone-marrow-derived cells in B16 tumor tissues. The granulocyte-macrophage colony-stimulating factor–induced enhancement of tumor cell proliferation was mediated by bone-marrow-derived cells in vitro. Meanwhile, a distinct synergistic effect on endothelial cell function between granulocyte-macrophage colony-stimulating factor and bone-marrow-derived cells was observed. After separating two types of bone-marrow-derived cells, granulocyte-macrophage colony-stimulating factor–induced enhancement of tumor growth and angiogenesis in vivo was mediated by proangiogenic cells in granulocytes, but not monocytes, with CD11b+, vascular endothelial growth factor receptor 2, and C-X-C chemokine receptor 4 granulocytes possibly involved. These data suggest that granulocyte-macrophage colony-stimulating factor contributes to the growth and angiogenesis of certain types of tumor, and these mechanisms are probably mediated by proangiogenic cells in granulocytes. Applying granulocyte-macrophage colony-stimulating factor may attenuate the antitumor effects of chemotherapy and radiotherapy in certain types of tumor.
Introduction
Granulocyte-macrophage colony-stimulating factor (GM-CSF), which belongs to the family of hematopoietic cytokines, is an endogenous glycoprotein that mobilizes hematopoietic cells from bone marrow (BM) and induces BM hematopoietic progenitors to proliferate and differentiate into specific types of mature blood cells. 1 GM-CSF is usually expressed with several different cell types, including T cells, B cells, macrophages, mast cells, endothelial cells, fibroblasts, and adipocytes as part of the host response to infection or injury. 2 As a bilineage factor, the function of GM-CSF includes the activation and augmentation of the functions of granulocytes (GRANs), monocytes (MNCs), macrophages, and dendritic cells in a dose-dependent manner.3,4 GM-CSF not only promotes the proliferation of progenitors committed to GRANs and MNCs production but also has a limited capacity to stimulate megakaryocytic and erythroid progenitors. GM-CSF is vital to the maturation of macrophages and invariant natural-killer T cells and is involved in the development and function of dendritic cells. 5 These properties have resulted in GM-CSF being used clinically as an immunological adjuvant.
GM-CSF has also been found to stimulate the mobilization and release of various hematopoietic stem cells—such as multipotent progenitors, myeloid-committed progenitors, and stem cells from BM—into the circulating blood through its receptors expressed on hematopoietic cells. 6 One of the clinical applications of GM-CSF is therefore to ameliorate cancer-therapy-induced and potentially life-threatening febrile neutropenia. Since the mobilization of myeloid populations and their precursors from the BM can modulate the pathology of certain diseases, the administration of GM-CSF, particularly systemically, is also believed to ameliorate diseases such as neurological diseases and atherosclerosis. GM-CSF is mostly administered to patients who receive myelosuppressive chemotherapy, to mobilize peripheral blood progenitor cells, and used for myeloid reconstitution after autologous or allogeneic BM transplants.7,8
To be an immunological adjuvant for enhancing immunity, the pharmacological action of GM-CSF implies a potential application in inhibiting the occurrence and development of tumors, especially as an adjuvant tumor therapy in patients after radiotherapy or chemotherapy.9–11 It was reported that GM-CSF inhibited breast cancer growth in mice through re-educating macrophages to reduce angiogenesis and metastases. 12 The intratumoral administration of GM-CSF was found to stimulate macrophages in low-O2 environment to secrete high levels of a soluble form of the vascular endothelial growth factor receptor 1 (VEGFR1), which neutralized VEGF and inhibited its biological activity, thereby resulting in reductions in tumor growth and angiogenesis in vivo. 13
However, the evidence for the benefits of adjuvant therapy GM-CSF for patients is unclear. On one hand, the constitutive expression of GM-CSF—frequently together with its receptor—is found in numerous solid tumors, such as carcinoma, gliomas, and meningiomas. 14 GM-CSF was found to stimulate proliferation, migration, and invasion in several tumor cell lines and to promote tumor progression and metastasis in various tumors via the autocrine stimulation of tumor cells as well as via the paracrine activation of tumor-supportive stroma.15–18 On the other hand, GM-CSF induces the proliferation and migration of endothelial cells, thus contributes to angiogenesis.17,19,20 As a result, GM-CSF has also been described as a tumor-promoting factor.
Another study showed that GM-CSF exerts dose-dependent opposing effects on tumor growth, with high levels of GM-CSF expression promoting tumor growth and low levels of GM-CSF expression inhibiting tumor growth. 3 GM-CSF overexpression and treatment were also variously reported to lead to inhibition of both the proliferation and the growth of tumor cells and to enhancement of the migration, invasion, and angiogenesis of tumor cells. 18 Therefore, as a widely used adjuvant medicine in tumor therapy for attenuating the adverse effects of radiotherapy and chemotherapy, the effects of GM-CSF on tumor growth in vivo are still confounded by the lack of solid empirical evidence.
The growth, metastasis, and recurrence of solid tumors depend on angiogenesis and vasculogenesis. The ability of GM-CSF to stimulate neovascularization, including angiogenesis and arteriogenesis, has been demonstrated in both animal models and clinical patients.19–23 It has been established that GM-CSF is able to induce and support the proliferation of endothelial cells that may lead to the formation of endothelial capillaries.24,25 GM-CSF appears to drive the epithelial secretion of VEGF—which is the main mediator in angiogenesis—via autocrine and/or paracrine circuitry. 26 Meanwhile, other studies have shown that bone-marrow-derived cells (BMDCs) released into the circulating blood from BM and recruited into angiogenic local tissues play a dominate role in angiogenesis.27,28 Although GM-CSF was reported to increase circulating progenitor cells and cause neovascularization in ischemic tissues by directly incorporating progenitor cells into the endothelium of blood vessels, there is emerging evidence that BMDCs are recruited in close proximity to angiogenic vessels as accessory cells to enhance proliferation of endothelial cells via secreting proangiogenic activities distinct from locally induced activities.27,29 To act as a hematopoietic growth factor as originally identified, GM-CSF is administered to mobilize MNCs and GRANs from BM, which leads to both systemic and local increases in the numbers of myeloid cells. 4 However, although the angiogenic potency of GM-CSF has been known for some time, the main action of GM-CSF is to promote angiogenesis directly via endothelial cells or indirectly via BMDCs, which has not yet been evaluated. The precise mechanisms underlying the proangiogenic activity of GM-CSF therefore remain unclear.
The widespread use in rescuing GRANs, the inconsistent findings concerning the effects on tumor growth, and the obscure angiogenesis mechanisms warrant a careful investigation of the role of GM-CSF as an adjuvant medicine in radiotherapy and chemotherapy. This study therefore investigated the effects of GM-CSF on tumor growth both in vivo and in vitro. To clarify the underlying mechanisms, the direct action of GM-CSF on tumor cell proliferation and the indirect action of GM-CSF on angiogenesis were assessed both with or without the presence of BMDCs. Considering that BMDCs in tumor tissues are recruited from circulating blood and that GM-CSF can mobilize BMDCs from BM into the bloodstream, white blood cells (WBCs) and BM cells were isolated and tested as BMDCs in the experiments. The types of BMDCs were then preliminary identified in vivo and in vitro.
Materials and methods
Animals
Male C57BL/6J mice and KM mice (6–8 weeks, purchased from the Experimental Animal Center, Xi’an Jiaotong University) were housed under constant temperature, humidity, and lighting (12-h light per day) and allowed free access to food and water. Animal welfare and experimental procedures were strictly in accordance with the guide for care and use of laboratory animals.
Collection and isolation of BM cells and BMDCs
Mice were anesthetized with urethane (Shanghai Shan Pu Chemical Co., Ltd, Shanghai, China) and then euthanized immediately. BM was obtained from the femurs and tibias of mice. Single BM cell suspension was prepared by passing the BM through a 21-gauge needle. Then the cells were pelleted by centrifugation and suspended in RPMI 1640 medium (Gibco, New York, NY, USA) for BM transplantation or in RPMI 1640 medium containing 2% fetal calf serum (FBS; Zhejiang Tian hang Biological Technology Co., Ltd, Hangzhou, China) for further experiments at a density of 1 × 107 cells/mL.
GRANs and MNCs were isolated from blood with separation medium kits (TBD, Tianjin, China). In brief, peripheral blood was collected into a heparinized tube, and ACK (ammonium-chloride-potassium) lysing buffer (0.15 M NH4Cl, 1.0 mM KHCO3, 0.1 mM Na2EDTA) was added and stayed at room temperature for 5 min to remove red blood cells (RBC). Then the remaining cells (WBCs) were washed twice with phosphate-buffered saline (PBS). After that, the blood cells were diluted with tissue homogenate at the ratio of 1:1 and mixed with E reagent and placed under 20°C–25°C for 30 min. The supernatant was collected and added to A reagent and centrifuged at 400g for 20 min at 20°C. The centrifugal tube was centrifuged into four layers, and the second layer of mononuclear cells and the third layer of GRANs were placed into 4 mL of cell detergent. The mixed solution was centrifuged at 2500 r/min for 20 min at 4°C and the supernatant was discarded, and then RPMI 1640 medium was added to resuspend the cells.
Tumor implantation
Each mouse was injected subcutaneously with 1 × 106 murine tumor cells (0.1 mL) in the right forelimb of the mice. Then the mice were divided into control and GM-CSF groups randomly. Recombinant murine GM-CSF (R&D Systems, USA) was dissolved in 0.9% NaCl and administered subcutaneously with the dose of 17 or 50 µg/kg, respectively. The control group received vehicle only. Tumor size was measured daily using calipers, and tumor volumes were calculated using the formula A × B2 × 0.5, where A and B are the larger and smaller diameter of the tumor, respectively. After a few days of treatment, animals were euthanized by cervical displacement after anesthetization. Tumor tissues were resected, weighted, and then fixed with 4% paraformaldehyde.
Cells isolated from BM or blood, including of BM cells (BMCs, 1 × 107), WBCs (2 × 106), GRANs (1.5 × 106), and MNCs (0.2 × 105), were mixed with 1 × 106 tumor cells, respectively, and then injected subcutaneously into the right flank of mice. The effects of BMDCs on tumor growth were observed in vivo.
Myelosuppressive mouse model
Mice (6–10 weeks old) were irradiated with 6 Gy and then myelosuppressive mouse model was obtained. 30 Tumor implantation was performed one day after irradiation and tumor growth was observed as described above.
BM transplantation
Recipient mice (6–10 weeks old) were lethally irradiated (9 Gy) followed by BM reconstitution by tail vein injection with 1 × 107 BM cells isolated from green fluorescent protein (GFP+)-positive donor femurs. 27 After BM transplantation of 8 weeks, the mice were used for tumor experiments.
Histology and immunohistochemistry
Tissues were fixed with 4% paraformaldehyde and embedded in paraffin. Histological and immunohistochemical staining was performed using standard techniques as described previously. 27 In brief, the embedded tumor tissues were sectioned and the slices were blocked with 10% normal goat serum (Beijing four Zheng Bo Biological Technology Co., Ltd, Beijing, China) and incubated with primary antibody for 3 h at 37°C. Subsequently, the slices were incubated with horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (Beijing four Zheng Bo Biological Technology Co., Ltd) for 1 h under room temperature. After washing with PBS, immunoreactivity was visualized with diaminobenzidine (DAB; Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd, Beijing, China) as chromogen under a light microscope. Rabbit anti-GFP antibody (1:500; AnaSpec, Fremont, CA, USA) and rabbit anti-laminin (1:1000; Sigma-Aldrich, Milwaukee, WI, USA) were used as primary antibodies. Hematoxylin/eosin (H&E) staining was performed after tissues were fixed with 4% paraformaldehyde and embedded in paraffin.
The intratumoral microvessel density (MVD) was determined on H&E- or laminin-stained tumor tissues. Five fields were selected randomly from each tumor tissue section and quantitative analysis of the positively stained density was performed using Optimas image analyzer (Optimas Corporation USA).
Chick chorioallantoic membrane assay
Ex vivo pro-angiogenesis activity of GM-CSF was measured by CAM assay as described elsewhere with minor modification. 31 Fertilized chicken eggs (Xi’an Xinfengyuan livestock breeding specialized cooperatives, Xi’an, China) were incubated at 37.8°C and 55%–65% humidity for 7 days, and the eggs were turned twice a day. On day 8, a 1-cm-diameter window was carefully created on the broader side of the egg, which can candle the egg to assure existence of embryonic blood vessels. Glass coverslips (Jinling Pharmaceutical Co., Ltd, Nanjing, China) (2 mm × 2 mm) saturated with GM-CSF (100 ng/mL), 0.9% NaCl (v/v) or basic fibroblast growth factor (bFGF; 4200 IU/mL; Zhuhaiyisheng Biological Pharmaceutical Co., Ltd, Guangdong, China) were placed in CAM, and a permeable sticky tape was immediately appended to the window. After incubation for another 3 days (until 11 day), the chick chorioallantoic membranes (CAMs) were fixed with 4% formaldehyde and neighboring neovascular zones were photographed with a Canon digital camera. Pro-angiogenesis activity of GM-CSF on CAM was assessed by counting the number of blood vessel branch surrounding the coverslips.
Tube formation assay
The effect of GM-CSF on angiogenesis in vitro was estimated using the HUVECs tube formation assay as described previously. 32 Briefly, 5 × 104 HUVECs were seeded onto a Matrigel-coated (Becton Dickinson) 48-well plate (Costar, USA), HUVECs and different concentration of GM-CSF incubation for 24 h, the tube formation was observed by microscopy and photographed. The length of complete tubes and the number of branch points of individual polygons of the capillary network were evaluated. Five randomly selected fields were determined.
Mouse aortic ring assay
Ex vivo pro-angiogenesis activity of GM-CSF was assayed by culturing rings of mouse aorta in Matrigel. 27 Thoracic aorta from mice were isolated and subsequently cut into 1-mm-thick rings after removal of adventitial fat. Rings were immediately placed into a 48-well plate containing 100-µL Matrigel then incubated at 37°C until the Matrigel polymerized. The 600-µL RPMI 1640 medium containing 20% FBS was added to each well, and the rings were cultured with GM-CSF at a series of concentrations. Vascular sprouting from each ring was observed and photographed on the fifth day using a Leica Inverted Microscope. The angiogenic response was quantified by microvessel counting according to the published criteria.
Flow cytometry
Cells isolated from peripheral blood were incubated with fluorescein isothiocyanate (FITC) anti-mouse CD11b antibody (1:80; R&D Systems, USA), PE anti-mouse Gr-1 (BioLegend; 1:80), PE anti-mouse β3 (BioLegend; 1:150), PE anti-mouse C-X-C chemokine receptor 4 (CXCR4) (BioLegend; 1:80), or PE anti-mouse VEGFR2 (1:80; BioLegend) following a published protocol. 33 The cells were subjected to flow cytometer on a FACScan (BD Bioscience, CA, USA), and data were analyzed with Cell Quest Software (version 3.8, BD).
Statistical analysis
Data are expressed as mean ± standard error of the mean (SEM) values. Statistical analyses among groups were performed using one-way analysis of variance with Fisher’s least-significant-difference test, and statistical analyses between two groups were performed using paired two-tailed Student’s t tests. p value of ≤0.05 was considered to be indicative of statistical significance. Tumor volume and weight were compared using the Kruskal–Wallis test in order to accommodate the smallness of the sample.
Results
GM-CSF promoted tumor growth in normal and myelosuppressive mice
The effects of exogenous GM-CSF on tumor growth in vivo were assessed using mouse S180 sarcoma, B16 melanoma, and U14 cervical carcinoma in the experiments. As shown in Figure 1(a) and (b), 10 days after B16 cell implantation, 50 µg/kg GM-CSF significantly promoted B16 tumor growth by 77.5% compared with the control group (1.58 ± 0.21 vs 0.89 ± 0.07 g). Meanwhile, exogenous 17 and 50 µg/kg GM-CSF markedly enhanced S180 tumor growth by 33.3% and 31.7%, respectively, compared with the control group (0.80 ± 0.06 and 0.79 ± 0.06 vs 0.60 ± 0.07 g, respectively) (Figure 1(c)). However, only slight increases (9.8% and 6.5%) were observed in the U14 tumor model relative to control after treatment with 17 or 50 µg/kg GM-CSF (Figure 1(d)). This difference in the enhancement of tumor growth between the B16 and S180 groups and the U14 group implies that the effects of GM-CSF vary with the tumor cell line involved.
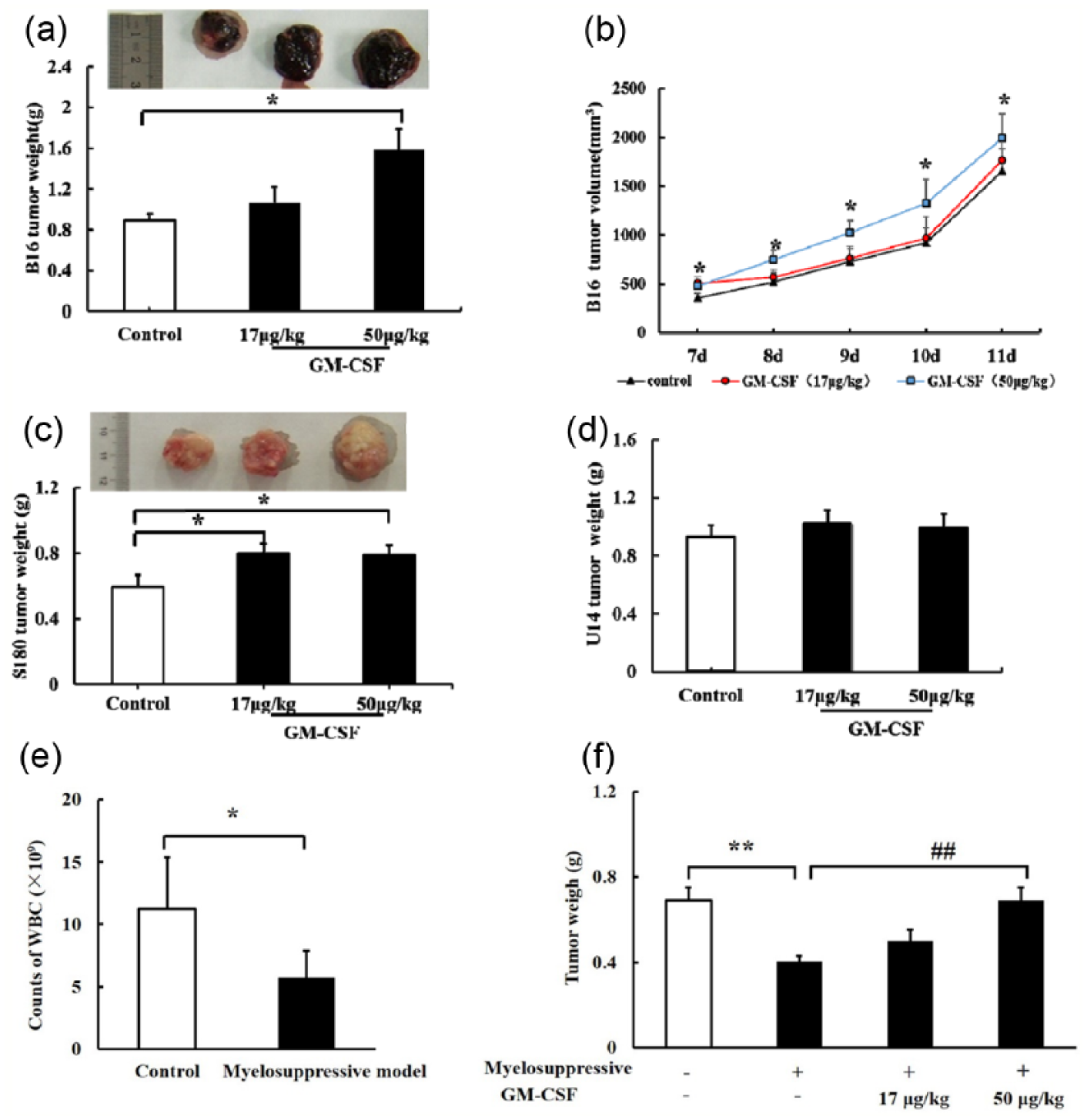
GM-CSF promoted tumor growth in normal and myelosuppressive mice. (a and b) B16 tumor weights at 9 days after implantation, (c) S180 tumor weights at 10 days after implantation, (d) U14 tumor weights at 12 days after implantation, and (e and f) Mice WBCs counts at 3 days after irradiation (6 Gy) and tumor growth in myelosuppressive mice at 9 days after implantation. Tumor weights are indicated as mean and SEM values.
Effects of exogenous GM-CSF on tumor growth were observed in irradiation-induced experimental myelosuppressive mouse models. As shown in Figure 1(e), compared with the control group, irradiation at 6 Gy induced a significant decrease in WBCs by 49.2% at 3 days after the treatment, revealing myelosuppression in irradiated mice. S180 tumor growth was significantly reduced by 41.4% after irradiation (0.40 ± 0.03 vs 0.69 ± 0.06 g). After GM-CSF treatment, tumor growth increased by 25.0% and 72.5% at doses of 17 and 50 µg/kg, respectively, compared with vehicle in myelosuppressive mice (0.50 ± 0.05 and 0.69 ± 0.06 vs 0.40 ± 0.03 g) (Figure 1(f)). The increase in the tumor weight in the 50 µg/kg group was statistically significant. These results suggest that GM-CSF markedly promotes tumor growth in irradiation-induced myelosuppressive mice.
GM-CSF promotion of tumor growth was associated with angiogenesis
As shown in Figure 2(a) and (b), the density of laminin-positive vessels in S180 tumor tissues was markedly enhanced in the 50-µg/kg GM-CSF group by 71.1% compared with the control group (18.33 ± 2.22 vs 31.36 ± 4.35). This implies that GM-CSF contributed to angiogenesis in S180 tumor tissues. In contrast, no obvious changes in blood vessel densities were observed in U14 tumor tissues (data are not shown).

GM-CSF-promoted tumor growth was associated with angiogenesis and BMDCs. (a and b) Densities of laminin- and H&E-positive vessels in S180 tumor sections at magnifications of 100× and 200×, respectively, (c and d) GM-CSF (100 ng/mL) increased numbers of microvessels in mice aortic ring models, and (e and f) GM-CSF promoted angiogenesis in the CAM model.
GM-CSF promoted angiogenesis in vitro
A mouse aortic ring model was employed to examine GM-CSF-induced angiogenesis in vitro. The results showed that GM-CSF significantly stimulated microvessel sprouting from the adventitia of cultured aortic rings by 55.9% and 65.3% at concentrations of 10 and 100 ng/mL relative to each of the controls (12.71 ± 0.56 vs 19.80 ± 1.72, and 12.71 ± 0.56 vs 21.00 ± 1.72, respectively) (Figure 2(c) and (d)).
The CAM model was used to confirm the role of GM-CSF in angiogenesis. As shown in Figure 2(e) and (f), CAM treated with GM-CSF or bFGF exhibited well-developed zones of neovascularization surrounding the sponge and increased densities of new blood vessels and vascular branches, with fewer vessels observed in the negative control group. The number of vascular branches was 1.56-fold higher in the GM-CSF-treated group than in the control group (9.00 ± 1.01 vs 14.00 ± 0.27).
GM-CSF increased BMDCs recruitment in tumor tissues
The density of GFP+ BMDCs in tumor tissues was analyzed in GFP bone-marrow-reconstituted C57BL mice with or without GM-CSF treatment. As shown in Figure 3(a) and (b), the density of GFP+ cells was 39.70 ± 5.07/mm2 in the control group and 46.25 ± 7.40/mm2 in the GM-CSF-treated group (p < 0.05). This 16.5% increase in GFP+ cell numbers induced by GM-CSF in tumor tissues corresponded to a 46.5% enhancement of tumor growth (1.14 ± 0.10 vs 1.67 ± 0.15 g, p < 0.05; Figure 3(c)).

GM-CSF (50 µg/mL) increased the recruitment of GFP+ BMDCs in tumor tissues ((a and b) 200× magnification) and promoted (c) tumor growth in BMT mice. Numbers of GFP+ cells and tumor weights are indicated as mean and SEM values.
BMDCs in tumor tissues promoted tumor growth in both normal and myelosuppressive mice
To confirm the hypothesis that GM-CSF promotes tumor growth via BMDCs in vivo, the effects of BM cells as well as WBCs on the growth of S180 and B16 tumors were further investigated. Compared with the counterpart controls in vivo, 1 × 107 BM cells significantly promoted the growth of both S180 sarcoma and B16 melanoma tumors by 37.3% (0.67 ± 0.08 vs 0.92 ± 0.09 g, p < 0.05) and by 41.2% (1.02 ± 0.09 vs 1.44 ± 0.15 g, p < 0.05) (Figure 4(a) and (b)). WBCs (2 × 106) also markedly enhanced the growth of S180 and B16 tumors (0.68 ± 0.06 vs 0.92 ± 0.09 g, p < 0.05) and (1.13 ± 0.10 vs 1.59 ± 0.15 g, p < 0.05) (Figure 4(c) and (d)). Moreover, in irradiation-induced myelosuppressive mice, 1 × 107 BM cells and 2 × 106 WBCs significantly enhanced the S180 tumor weight compared with the vehicle counterpart (Figure 4(e) and (f)). These results suggest that BMDCs, and especially circulating WBCs, were able to promote tumor growth in vivo.

BMDCs promoted S180 and B16 tumor growth in (a–d) normal and (e and f) myelosuppressive mice. (a and b) BMCs promoted tumor growth in mice, (c and d) WBCs promoted tumor growth in mice, and (e and f) BMCs and WBCs promoted S180 tumor growth in myelosuppressive mice. Tumor cells (1 × 106) were mixed with BMCs (1 × 107) or WBCs (2 × 106) and then implanted subcutaneously. Tumor weights are indicated as mean and SEM values.
GM-CSF promoted B16 tumor cell proliferation mediated by BMDCs in vitro
As shown in Figure 5(a), GM-CSF at concentrations from 0.01 to 781.25 ng/mL slightly increased (but not significantly) the proliferation of B16 tumor cells. Co-culturing 2 × 103 B16 tumor cells with 2 × 103 BM cells resulted in the B16 tumor cell proliferation being slightly increased (with statistical significance) from 7.6% to 19.7% when GM-CSF at concentrations higher than 1.25 ng/mL compared with the B16-tumor-cell group (Figure 5(b)). In contrast, GM-CSF statistically significantly increased B16 tumor cell proliferation (by 10.7%–28.6%) in a concentration-dependent manner at concentrations from 0.25 to 781.25 ng/mL in the presence of 2 × 103 WBCs, though WBCs did not show an obvious effect when they were present alone (Figure 5(c)). These results suggest that the promotion of B16 tumor cell proliferation by GM-CSF might be mediated by BMDCs.
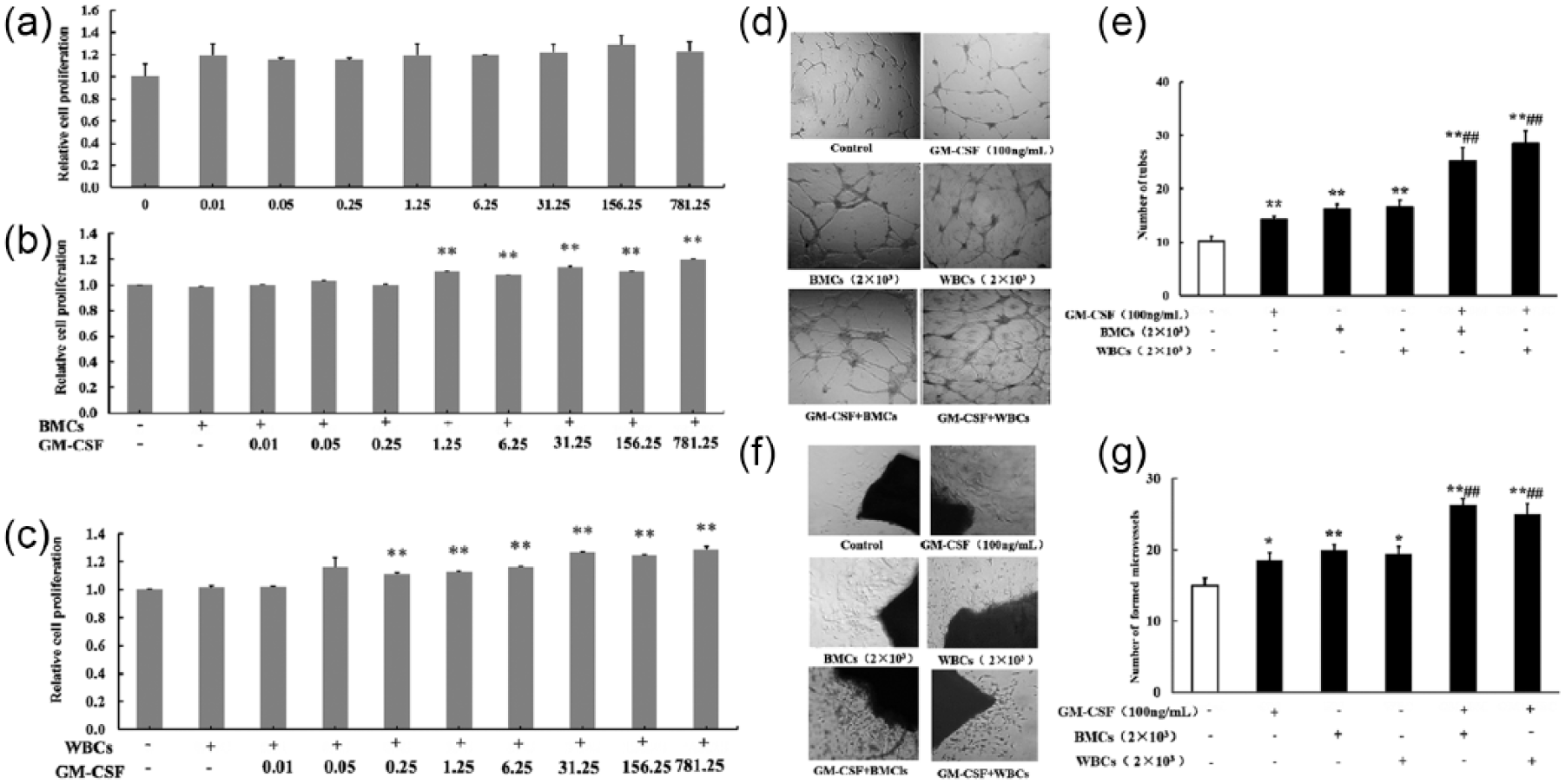
Synergistic action of GM-CSF and WBCs (or BMCs) in promoting B16 tumor cell proliferation and endothelial cell function in vitro. (a–c) Effects of GM-CSF on B16 tumor cell proliferation in the (a) absence of BMDCs and in the presence of (b) 2 × 103 BMCs or (c) 2 × 103 WBCs. (d and e) GM-CSF contributed to endothelial cell tube formation in the presence of 2 × 103 BMCs or WBCs. (f and g) GM-CSF promoted aortic ring sprouting in the presence of 2 × 103 BMCs or WBCs (100× magnification; n = 5 for each group).
GM-CSF increased BMDCs-induced enhancement of endothelial cell function in vitro
Endothelial cells play a key role in angiogenesis. The effects of GM-CSF and BMDCs both alone and in combination on endothelial cell function were evaluated using endothelial cell tube formation and aortic ring models in vitro. The results showed that the number of tubes was significantly increased by 39.2% (10.20 ± 0.86 vs 14.20 ± 0.58) in the presence of 100 ng/mL GM-CSF alone and during co-culturing with BM cells or WBCs compared with controls (10.20 ± 0.86 vs 16.20 ± 0.86 or 10.20 ± 0.86 vs 16.6 ± 1.21) (Figure 5(d) and (e)). The tube numbers in the BMCs and WBCs groups were markedly increased by 147.6% (10.20 ± 0.86 vs 25.25 ± 2.44) or 180.4% (10.20 ± 0.86 vs 28.60 ± 2.26) in the presence of GM-CSF compared with controls, respectively. The tube numbers in GM-CSF group were significantly enhanced, by 77.8% with BMCs (14.20 ± 0.58 vs 25.25 ± 2.44) and by 101.4%with WBCs (14.20 ± 0.58 vs 28.60 ± 2.26) compared with GM-CSF group.
These results were verified by the data obtained in the aortic ring analysis in vitro. When aorta rings were maintained in media containing GM-CSF and/or co-cultured with BMCs or WBCs, microvessel outgrowths were observed on day 3. As shown in Figure 5(f) and (g), GM-CSF, BMCs, and WBCs significantly promoted microvessel sprouting by 23.3% (p < 0.05), 33.3% (p < 0.01), and 29.3% (p < 0.05), respectively, compared with controls. Meanwhile, combined treatment of aortic rings with GM-CSF in the presence of BMCs or WBCs resulted in a significant increase in capillary sprouting by 75.5% or 66.7% (15.00 ± 1.02 vs 26.33 ± 0.79, or 15.00 ± 1.02 vs 25.00 ± 1.50, both p < 0.01), respectively, compared with controls. GM-CSF induced significant enhancements by 31.7% and 28.9% in the BMCs and WBCs groups (20.00 ± 0.71 vs 26.33 ± 0.79, p < 0.01 or 19.40 ± 1.03 vs 25.00 ± 1.50, p < 0.01) compared to BMCs and WBCs, respectively. Meanwhile, the promoting activity of GM-CSF was markedly increased in the presence of BMCs or WBCs, by 42.3% (18.50 ± 1.07 vs 26.33 ± 0.79, p < 0.01) or 35.1% (18.50 ± 1.07 vs 25.00 ± 1.50, p < 0.01) compared with GM-CSF, respectively. These experimental results obtained in vitro suggest the presence of a synergistic action between GM-CSF and BMDCs in promoting endothelial cell function.
GRANs, not MNCs, are the BMDCs that played a dominant role in mediating the enhancement of tumor growth and angiogenesis induced by GM-CSF in vivo
WBCs were separated into GRANs and MNCs using separation medium kits, and then WBCs and each of the subpopulations according to the proportion isolated from WBCs were co-implanted subcutaneously with S180 cells in KM mice or with B16 cells in C57BL/6J mice. The results showed that GRANs and WBCs accelerated the growth of S180 tumors by 40.4% (0.89 ± 0.04 vs 1.25 ± 0.10, p < 0.01) and 33.7% (0.89 ± 0.04 vs 1.19 ± 0.10, p < 0.05), respectively, compared with controls, while an increase of only 15.7% was obtained in the MNCs group (0.89 ± 0.04 vs 1.03 ± 0.11, p > 0.05) (Figure 6(a)). Similar findings were obtained for B16 tumor growth in vivo (Figure 6(b)). The mean tumor weight was 42.5% (1.53 ± 0.07 vs 2.18 ± 0.28, p < 0.01) higher in the GRANs group than in the control group, but there was no obvious difference between the MNC group and the control group (1.53 ± 0.07 vs 1.87 ± 0.29, p > 0.05), implying that GRANs in WBCs played a key role in promoting tumor growth. To verify this result, granulocyte-colony-stimulating factor (G-CSF), a pleiotropic cytokine best known for its specific effects on the proliferation, differentiation, activation, and mobilization of the GRANs lineage, was administered in the S180 sarcoma mice model in parallel. The results showed that G-CSF at 50 µg/mL increased the S180 tumor growth by 20.5% (0.94 ± 0.17 vs 0.78 ± 0.21) compared with the control group, while GM-CSF at 50 µg/mL increased the growth of S180 tumors by 25.6% (0.98 ± 0.20 vs 0.78 ± 0.21) (Figure 6(c)), implying that the effect of G-CSF is similar to that of GM-CSF.
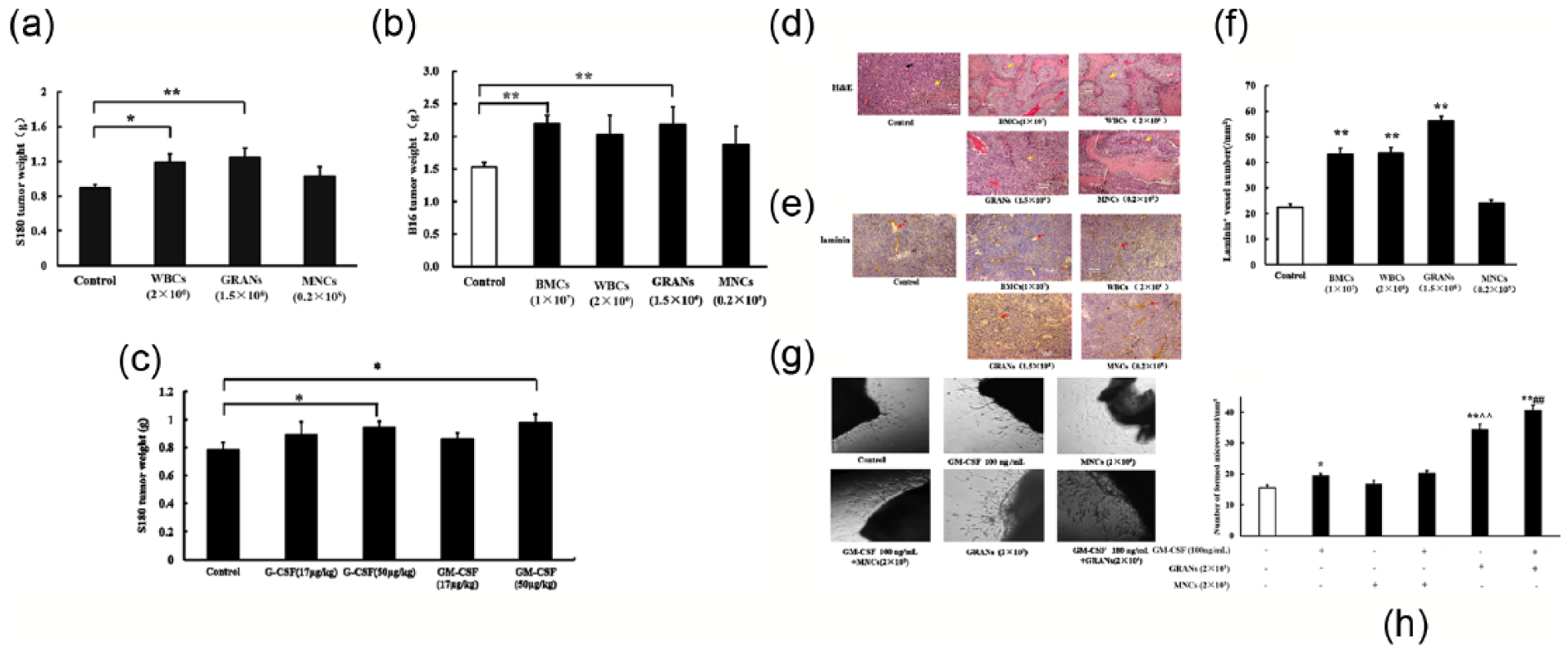
GRANs mediated the enhancement of tumor growth and tumor angiogenesis induced by GM-CSF. (a–c) GRANs and BMCs, WBCs accelerated the growth of (a) S180 and (b) B16 tumors. GM-CSF promoted (c) S180 tumor growth. Tumor weights are indicated as mean and SEM values. (d and f) H&E- and laminin-stained tumor tissue sections, and the promotion of angiogenesis by BMDCs. (g and h) Synergistic action of GM-CSF and GRANs in promoting aortic ring microvessel sprouting (n = 5 for each group, 100× magnification).
The areas of necrosis were observed and blood vessel densities were analyzed in histologically and immunohistochemically stained S180 tumor tissue sections. As shown in Figure 6(d)–(f), the laminin-stained blood vessel densities were significantly increased after GM-CSF administration in all groups except MNCs. The laminin-positive blood vessel densities in the GM-CSF, BMCs, WBCs, and GRANs groups were significantly increased by 97.40% (44.00 ± 1.93 vs 22.29 ± 1.67, p < 0.01), 94.40% (43.33 ± 2.34 vs 22.29 ± 1.67, p < 0.01), 95.92% (43.67 ± 2.34 vs 22.29 ± 1.67, p < 0.01), and 153.16% (56.43 ± 1.68 vs 22.29 ± 1.67, p < 0.01), respectively, compared with the control group; in contrast, the increase was only 8.97% (22.29 ± 1.67 vs 24.29 ± 1.13, p > 0.05) in the MNCs group.
Aortic rings were co-cultured with GRANs or MNCs with or without the presence of 100 ng/mL GM-CSF, and the numbers of sprouting aorta vessels were analyzed in vitro. As shown in Figure 6(g) and (h), there was no obvious difference in the number of sprouting vessels between the MNCs group and the control group or between the GM-CSF-treated MNCs group and the control group (16.75 ± 1.23 vs 15.60 ± 0.81, p > 0.05; 20.40 ± 0.68 vs 15.60 ± 0.81, p > 0.05). In contrast, GRANs significantly increased vessel sprouting by 121.8% relative to controls (34.60 ± 1.57 vs 15.60 ± 0.81), and the number of sprouting vessels was markedly higher in the GRANs group than in the MNCs group. Treatment with GM-CSF led to a significant increase in vessel sprouting by 17.7% (40.75 ± 1.57 vs 34.60 ± 1.57, p < 0.01) in the GRANs group, which produced a distinct increase in the difference in the numbers of sprouting vessels between the GRANs and MNCs groups in the presence of GM-CSF.
Together these results indicate that GRANs play an important role in mediating the activities of GM-CSF in promoting tumor growth and angiogenesis. The results also suggest that the promotion of tumor growth and angiogenesis using GM-CSF is mostly due to the mobilization and recruitment of GRANs.
GM-CSF significantly increased β3+, CXCR4+, VEGFR2+, CD11b+, and CD11b+Gr-1+ GRANs in circulating blood
To identify the dominant subpopulation of GRANs involved in mediating GM-CSF-induced angiogenesis, tumor-bearing mice were injected with 50 µg/kg GM-CSF subcutaneously, and GRANs were subsequently isolated from the circulating blood at days 10 after the injection. The most commonly reported proangiogenic markers in BMDCs, such as β3 integrin, CD11b, CXCR4, and VEGFR2, were utilized to analyze the subpopulations in GRANs with a flow cytometer. The results showed that the counts of β3+ cells significantly increased after GM-CSF treatment, from 58.50 ± 11.10% to 79.21 ± 29.44%, implying increases in the number of adhesive GRANs (Figure 7(a) and (b)). Meanwhile, the counts of CD11b+, CXCR4+, and VEGFR2+ GRANs significantly increased by 1.66-, 3.82-, and 1.72-fold, respectively (from 53.84 ± 1.99% to 89.48 ± 1.85%, from 2.18 ± 0.07% to 8.38 ± 0.63%, and from 2.94 ± 0.96% to 13.65 ± 1.79%, respectively), in the presence of GM-CSF in vivo (Figure 7(c)–(h)). Finally, the levels of CD11b+Gr-1+ cells, the proangiogenic GRANs in circulating blood, greatly increased from 34.25 ± 2.72% to 51.18 ± 3.90% by GM-CSF.

GM-CSF significantly increased proangiogenic BMDCs in tumor-bearing mice circulating blood. (a and b) The number of CD61+ in peripheral blood cells, (c and d) the number of CD11b+in peripheral blood cells, (e and f) the number of CXCR4+ in peripheral blood cells, (g and h) the number of VEGFR2+in peripheral blood cells, and (i and j) the number of CD11b+Gr+in peripheral blood cells. Percentage of are indicated as mean and SEM values.
Discussion
GM-CSF is able to shorten the duration of chemotherapy-induced neutropenia, but a meta-analysis found that GM-CSF does not prolong survival. 34 This has raised questions not only with respect to the clinical benefit of GM-CSF as an adjuvant therapy but also about its role in promoting cancer progression for clinically relevant tumor entities. This situation indicates the need to test the actual contribution of exogenously administered GM-CSF to cancer treatment, as well as its underlying mechanisms.
This study employed normal and experimental myelosuppressive mice models mimicking patients after chemotherapy or radiotherapy to investigate the role of GM-CSF in tumor growth and progression. The results showed that (1) GM-CSF promotes the growth of certain tumors in both normal and myelosuppression mice, by influencing angiogenesis; (2) GM-CSF increases the recruitment of BMDCs in tumor tissues; (3) GM-CSF promotes BMDC-mediated B16 tumor cell proliferation in vitro; (4) GM-CSF increases the BMDC-induced enhancement of endothelial cell function in vitro; (5) GRANs, not MNCs, play a dominant role in mediating the enhancement of tumor growth and angiogenesis induced by GM-CSF in vivo; and (6) GM-CSF significantly increases the levels of a series of proangiogenic BMDCs in circulating blood, such as β3+, CXCR4+, VEGFR2+, and CD11b+Gr-1+ GRANs.
Even though a few studies have found that GM-CSF inhibited the growth of solid tumor by stimulating MNCs and enhancing macrophage antigen presentation and immune responsiveness,10,12,35 many more studies have found that GM-CSF enhanced tumor cell functions in vitro and promoted tumor growth in vivo.3,15 –18 Nevertheless, some studies have also presented evidence for a dose-dependent role of endogenous GM-CSF in tumor growth. 3 The results obtained in this study indicate that GM-CSF promoted the growth of S180 and B16 tumors, but not U14 tumors, in experimental mice models, suggesting a tumor-type-dependent enhancement of tumor growth induced by exogenous GM-CSF in vivo. Considering the extensive usage of GM-CSF as an adjuvant therapy in the clinic and these inconsistent research findings, it is necessary to systematically and comprehensively evaluate the effect of GM-CSF on the growth and recurrence of various type tumors further.
Angiogenesis has long been established as a critical contributor to the growth and progression of solid tumors. Some studies have found that GM-CSF promotes angiogenesis by enhancing endothelial cell function and stimulates the proliferation and migration of tumor cells.16,17 However, angiogenesis is not only attributed to the direct enhancement of endothelial cell function in peripheral resident tissues but also dependent on the recruitment of BMDCs at angiogenic sites from the circulating blood.27,29 There is even evidence of BMDCs playing a dominant role during the process of neovascularization compared with the resident endothelial cells. 27 GM-CSF contributes to the mobilization of a series of BMDCs from the BM into the circulating blood, and thus its proangiogenic effects may lead to both systemic and local increases in the numbers of myeloid cells.36,37 This may result in GM-CSF facilitating angiogenesis and tumor growth by increasing the recruitment of BMDCs in tumor tissues. Therefore, the recruitment of BMDCs in tumor tissues as well as the effects of GM-CSF were analyzed with GFP bone-marrow-reconstituted C57BL mice. The results obtained in this study have confirmed the hypothesis that GM-CSF promotes BMDCs recruitment in tumor tissues, which might increase angiogenesis and tumor growth.
The GM-CSF receptor has been found to be highly expressed on endothelial cells, certain BMDCs, and some tumor cells. 17 However, even though efforts have been made to elucidate the biological actions of GM-CSF in tumor growth, a fundamental gap remains in understanding the effects of exogenous GM-CSF on tumorigenesis and angiogenesis, let alone the role of BMDCs in these processes. Therefore, the effects of GM-CSF on the interactions among cancer cells, endothelial cells, and BMDCs were investigated in this study. The results indicated that the proliferation of B16 cells was only slightly affected by GM-CSF or WBCs directly, but was markedly enhanced by GM-CSF in the presence of WBCs. These data suggest that BMDCs recruited from the circulating blood could mediate the GM-CSF-induced enhancement of tumorigenesis. Meanwhile, a similar distinct synergistic effect between GM-CSF and BMDCs on endothelial cell function was observed in this study. GM-CSF significantly facilitated the WBC- or BMC-induced enhancement of endothelial cell function. Therefore, the promotion of tumorigenesis and angiogenesis by GM-CSF was mediated or enhanced by BMDCs, suggesting that BMDCs recruited in local tissues act as a catalyst. Meanwhile, similar enhancements were obtained with the conditioning media obtained from co-cultures of GM-CSF and BMDCs (data not shown), suggesting that the GM-CSF-induced release of cytokines from BMDCs is involved in this process. The underlying mechanisms need to be researched in detail further. However, the results indicating the promotion of tumor growth and angiogenesis by GM-CSF in vivo were supported by the experimental data obtained in vitro, with the interactions among GM-CSF, BMDCs, and endothelial cells/tumor cells facilitating tumorigenesis and angiogenesis. In brief, GM-CSF enhances the mobilization of BMDCs from BM and the recruitment of BMDCs in tumor tissues, which in turn increase tumor cell proliferation and angiogenesis synergistically with GM-CSF. It can be speculated that the application of GM-CSF attenuates the antitumor effect of chemotherapy and radiotherapy in the clinic.
Several types of BMDCs, such as BM-derived myelomonocytic cells, GRANs, MNCs/macrophages, myofibroblasts, and stem/progenitor cells, reportedly play a pivotal role in tumor angiogenesis.37–42 GM-CSF mobilizes and releases a series of BMDCs, mainly GRANs and MNCs, into the circulating blood. Therefore, effects of GM-CSF, BMCs, WBCs, MNCs, and GRANs on tumor growth were tested in vivo. The results showed that GRANs, but not MNCs, significantly promoted tumor growth, implying that GRANs play a dominant role in this process and that GM-CSF might promote tumor growth and angiogenesis via GRANs. This result was supported by the similar pharmacological action of G-CSF in facilitating tumor growth in vivo. Moreover, the deduction was confirmed in experimental results obtained in vitro, in which GRANs, but not MNCs, increased the stimulation of angiogenesis induced by GM-CSF in mice aortic ring models. It was reported that 70%–80% of the tumor-induced myeloid-derived cell response involved granulocytic-like cells, while 20%–30% of the cells reflected the monocytic lineage.43–45 These findings support the results obtained in this study.
Previous studies found that CXCR4+, VEGFR2+, and CD11b+Gr-1+ BMDCs contributed to tumor growth and angiogenesis in vivo.33,46–48 The subpopulations of BMDCs mobilized by GM-CSF from BM were identified as endothelial progenitor cells, CD11b+Gr-1+, and CXCR4+ cells.33,35,44,48 –54 However, the predominant types of BMDCs involved in promoting tumor growth induced by GM-CSF were still uncertain. On one hand, GM-CSF was found to augment adhesive-molecule β3+ GRANs in this study, which could be at least partially attributable to the increase in BMDC recruitment in tumor tissues after administering GM-CSF. On the other hand, the significantly increased CD11b+, VEGFR2+, and CXCR4+ GRANs, associated with the increases in tumor growth and angiogenesis induced by GRANs, implied that GM-CSF accelerated tumor growth and angiogenesis by mobilizing CD11b+, VEGFR2+, and CXCR4+ GRANs. However, the precise role of these cells and the detailed mechanism underlying the promotion of tumor growth still need to be clarified.
In conclusion, GM-CSF significantly promoted angiogenesis and tumor growth in certain tumor types in this study. GM-CSF markedly enhanced the release of BMDCs from BM and the recruitment of BMDCs in tumor tissues. On one hand, GM-CSF-induced enhancement of tumorigenesis was mediated by BMDCs, while on the other hand, GM-CSF acted synergistically with BMDCs to stimulate the function of endothelial cells. The GM-CSF-induced enhancement of tumor growth and angiogenesis in vivo was mediated by proangiogenic cells in GRANs (but not in MNCs), probably VEGFR2+, CXCR4+, and CD11b+Gr-1+ GRANs.
Footnotes
Author contribution
Q.Z. and X.L. contributed equally to this work. W.F. designed and performed research, collected and analyzed data and wrote the manuscript; Q.Z. and X.L. performed research; X.C. analyzed data and wrote the manuscript; T.C., Y.Z., W.M., and X.Z. performed research; and P.Z. and X.L. collected data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by grants from the Natural Science Foundation of China (81071765 and 81372379).
