Abstract
Pancreatic ductal adenocarcinoma is one of the most lethal cancers. The Hippo pathway is involved in tumorigenesis and remodeling of tumor microenvironments. Hypoxia exists in the microenvironment of solid tumors, including pancreatic ductal adenocarcinoma and plays a vital role in tumor progression and metastasis. However, it remains unclear how hypoxia interacts with the Hippo pathway to regulate these events. In this study, expressions of yes-associated protein 1 and hypoxia-inducible factor-1α were found to be elevated in pancreatic ductal adenocarcinoma samples compared with those in matched adjacent non-tumor samples. Moreover, hypoxia-inducible factor-1α expression was positively correlated with yes-associated protein 1 level in pancreatic ductal adenocarcinoma tissues. The higher expression of nuclear yes-associated protein 1 was associated with poor histological grade and prognosis for pancreatic ductal adenocarcinoma patients. In vitro, yes-associated protein 1 was highly expressed in pancreatic ductal adenocarcinoma cells. Depletion of yes-associated protein 1 inhibited the invasion of pancreatic ductal adenocarcinoma cells via downregulation of Vimentin, matrix metalloproteinase-2, and matrix metalloproteinase-13, and upregulation of E-cadherin. In addition, hypoxia promoted the invasion of pancreatic ductal adenocarcinoma cells via regulating the targeted genes. Hypoxia also deactivated the Hippo pathway and induced yes-associated protein 1 nuclear translocation. Furthermore, depletion of yes-associated protein 1 or hypoxia-inducible factor-1α suppressed the invasion of pancreatic ductal adenocarcinoma cells under hypoxia. Mechanism studies showed that nuclear yes-associated protein 1 interacted with hypoxia-inducible factor-1α and activated Snail transcription to participate in epithelial–mesenchymal transition–mediated and matrix metalloproteinase–mediated remodeling of tumor microenvironments. Collectively, yes-associated protein 1 is an independent prognostic predictor that interacts with hypoxia-inducible factor-1α to enhance the invasion of pancreatic cancer cells and remodeling of tumor microenvironments. Therefore, yes-associated protein 1 may serve as a novel promising target to enhance therapeutic effects for treating pancreatic cancer.
Introduction
Pancreatic cancer, also known as pancreatic ductal adenocarcinoma (PDAC), is one of the most aggressive tumors with a 5-year survival rate lower than 5%. 1 Approximately, 53,070 new PDAC cases are diagnosed and 39,590 patients die of PDAC in the United States, yearly. 2 The incidence and death rates of PDAC increase yearly due to limited treatments. 3 Surgical resection for PDAC is the only therapy with curative potential, but only 15%–20% of patients claim the opportunity for surgery. 1 The mortality of PDAC is mainly caused by delayed diagnosis, early metastasis, and resistance to systemic chemotherapy and radiotherapy.1,4 Therefore, elucidating the underlying mechanisms of PDAC invasion and metastasis and developing novel therapeutic strategies are critical.
The Hippo pathway regulates organ development via inhibiting cell proliferation and promoting apoptosis.5–7 Recent studies have shown that the Hippo pathway is strongly involved in the process of tumor initiation, progression, and metastasis.8–10 The yes-associated protein 1 (YAP1), a transcriptional co-activator, is the most critical component of the Hippo pathway. The Hippo pathway involves a cascade of kinases which mediates YAP1 phosphorylation and leads to YAP1 degradation in cytoplasm. The unphosphorylated YAP1 translocates to the nucleus and transactivates the downstream genes, such as TEADs, SMAD, RUNX, and CTGF. 11 Activated YAP1 is involved in malignant behaviors of tumors, such as cancer stem cell properties, 7 epithelial–mesenchymal transition (EMT), 12 increased potential for migration and metastasis, 13 and resistance to chemotherapy. 14 YAP1 is highly expressed in various malignant tumors, including pancreatic cancer.15–18 The high expression of YAP1 is shown to participate in the development, progression, and recurrence of PDAC.19–21 YAP1 promoted PDAC cells motility and invasion by lysophosphatidic acid receptor 3 (LPAR3)-mediated mitotic phosphorylation. 22 However, few studies have investigated the role of the Hippo pathway in the later stages of tumor metastasis.
Hypoxia commonly exists in the microenvironment of solid tumors and is associated with tumor invasion and distant metastasis,23,24 extracellular matrix (ECM) degradation, 25 and EMT. 26 Hypoxia-inducible factor-1α (HIF-1α) is the most important transcription factor caused by intratumoral hypoxia. 27 Hypoxia microenvironment and HIF-1α are critical drivers of PDAC metastasis and progression. 28 In pancreatic cancer, the expression of HIF-1α is upregulated and associated with tumor progression, metastasis, EMT, and ECM degradation.29,30 YAP1 activity was also linked to oxygen availability. 31 However, the mechanism by which YAP1 regulates tumor invasion and metastasis under hypoxia needs to be further illustrated.
In this study, we first demonstrated that the expression of YAP1 was significantly higher in tumor tissues than that in matched adjacent non-tumor tissues and positively correlated with HIF-1α level. The high expression of the nucleus YAP1 was associated with poor tumor differentiation and prognosis for PDAC patients. Moreover, we focused on the role of YAP1 in PDAC cells migration and invasion. In addition, we investigated the mechanism by which YAP1 interacted with HIF-1α to regulate PDAC cells invasion under hypoxia. We identified the hyperactivated YAP1 as a key regulator to promote PDAC cells invasion via EMT- and matrix metalloproteinase (MMP)-mediated remodeling of tumor microenvironment under hypoxia. Therefore, YAP1 may represent a novel therapeutic target in pancreatic cancer treatment.
Materials and methods
Clinical pancreatic cancer specimens and tissue microarrays
Nineteen PDAC tumor specimens and matched adjacent non-tumor specimens were obtained from the Qianfoshan Hospital of Shandong Province between October 2014 and December 2015 with patients’ consent and approval from the Ethics Committee of Qianfoshan Hospital of Shandong Province. All participants should meet the following criteria: (1) PDAC samples were histologically confirmed as adenocarcinoma; (2) tumor and matched adjacent non-tumor tissues were all obtained; and (3) patients were initially diagnosed as PDAC without other co-existing tumors and not undergoing neoadjuvant chemotherapy. The adjacent non-tumor tissues were obtained from 2 cm around the tumor tissues. All related clinical data were collected simultaneously, including the following: sex and age, tumor size, location and differentiation, vascular, perineural and extrapancreatic invasion, lymph node metastasis, and tumor–node–metastasis (TNM) stages.
Human pancreatic cancer tissue microarrays (TMAs) were purchased from Shanghai Biochip Company Ltd. (Shanghai, China). The TMAs contained 63 tumor tissues, and 57 matched adjacent non-tumor tissues. The TMAs contained the detailed clinicopathological features and follow-up data. All patients underwent a complete surgical resection of the PDAC as an initial treatment between January 2009 and August 2013. The last follow-up time was November 2014.
All experiments were performed in accordance with the approved guidelines of the Qianfoshan Hospital of Shandong Province, affiliated to Shandong University.
Immunohistochemistry
The YAP1 expression in the tissues was evaluated with a YAP1 antibody (1:200, Santa Cruz Biotechnology, USA) by immunohistochemistry (IHC). Immunohistochemical staining was performed as previously described. 32 The scoring was done by two independent observers who were unaware of the clinical data. We separately assembled the nucleus and cytoplasm YAP1 expression according to the established rules.32,33 The cytoplasmic expression of YAP1 was assessed by assigning a proportion score and an intensity score as previously described. The nuclear expression of YAP1 was scored by estimating the proportion of tumor cells with positive nuclear staining (0: none, 1: ≤10%, 2: 10% to ≤50%, and 3: >50%).
Cell lines and culture conditions
The human PDAC cell lines BxPC-3, MIAPaCa-2, and Panc 02.03 were purchased from American Type Culture Collection (ATCC, USA). The human pancreatic ductal cells (HPDE6) were obtained from Fuheng Biology (Shanghai, China). The PANC-1 and SW1990 cells were obtained from Chinese Academy of Sciences Typical Culture Collection (CTCC) (Shanghai, China). All cells were cultured in an RPMI-1640 or Dulbecco’s Modified Eagle’s Medium (HyClone, USA) containing 10% fetal bovine serum (Gibco, USA) in a humidified incubator containing 5% CO2 at 37°C. The cells for hypoxic treatment were first cultured in normoxic conditions to obtain the desired subconfluence level (65%–70%). Then, the cells were incubated in an incubator in an atmosphere consisting of 94% N2, 5.0% CO2, and 1% O2.
Quantitative real-time polymerase chain reaction
Total tissues or cellular RNAs were extracted using TRIzol reagent (Invitrogen, USA) according to the manufacturer’s protocol. Total RNAs were reverse transcribed using a reverse transcription kit (FastQuant RT Kit, Tiangen, China) according to the manufacturer’s guidelines. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed using the SYBR Green Master Mix (Tiangen, China). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the internal control. The primer sequences used for the quantitative PCR (qPCR) were presented in Table 1.
Genes examined and the primers used for RT-PCR.

RT-PCR: real-time polymerase chain reaction; YAP1: yes-associated protein 1; HIF-1α: hypoxia-inducible factor-1α; MMP: matrix metalloproteinase; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Western blot analysis
Total protein was extracted as previous described. 34 Nuclear protein extraction was performed according to the manufacturer’s guidelines (Thermo Scientific NE-PER; Thermo Fisher Scientific, USA). Accordingly, 20 to 50 µg of the total cell lysates were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, USA). The membranes were blocked with 5% non-fat milk for 2 h at room temperature and probed with primary antibodies at 4°C overnight according to the manufacturer’s protocol. The membranes were then incubated with horseradish peroxidase (HRP)-conjugated anti-mouse or -rabbit immunoglobulin G (IgG) secondary antibodies (Beyotime, Beijing, China) for 2 h at room temperature, followed by detection with the enhanced chemiluminescence immunoblotting detection reagents (Millipore). The protein band intensities were quantified by ImageJ software (National Institutes of Health, USA).
The primary antibodies and diluted concentration were as follows: YAP1 (1:2000; Santa Cruz Biotechnology), phosphorylated YAP1 (pYAP1; 1:5000; Abcam, USA), E-cadherin (1:1000; Abcam), Vimentin (1:1000; Abcam), MMP2 (1:1000; Abcam), MMP13 (1:500; Abcam), HIF-1α (1:1000; Abcam), TEAD (1:1000; Abcam), Snail (1:1000; Proteintech, China), Histone H3 (1:1000; Proteintech), and GAPDH (1:3000; Proteintech).
Confocal fluorescence microscopy
Cells were briefly seeded on chamber slides overnight. The plates were fixed in 4% paraformaldehyde and permeabilized in 0.1% Triton X-100. The plates were incubated with primary antibodies overnight at 4°C and then incubated with fluorescein isothiocyanate–labeled or Alexa Fluor® 488–conjugated IgG secondary antibodies (Thermo Fisher Scientific). The cells were mounted on microscope slides with a 4′,6-diamidino-2-phenylindole (DAPI) mounting solution (Abcam). The fluorescent images of the cells were photographed and analyzed using a confocal laser scanning (Carl Zeiss LSM 780, Germany).
Gene expression vectors and transfection
The YAP1 short hairpin RNA (shRNA; 5′-CCCAGTTAAATGTTCACCAAT-3′) and scrambled control shRNA (GenePharma, China) were transfected into PANC-1 and BxPC-3 cells. The HIF-1α expression was suppressed by shRNA interference (Santa Cruz Biotechnology), and plasmid-A shRNA (Santa Cruz Biotechnology) was used as a negative control. The cells were selected by 1 mg/mL of G418 or 1 µg/mL of puromycin for 2 weeks to form stable clones after transfection in vitro with PolyJet™ DNA transfection reagent (SignaGen Laboratories, USA) according to the manufacturer’s protocol.
Wound healing assay
The cells were seeded at a density of 2 × 106/well onto six-well plates and cultured to 90% confluence. The cell layers were scratched using a sterile 10 µL pipette tip to form wounded gaps. The wounded monolayers were gently washed with phosphate-buffered saline (PBS) and cultured under normoxia or hypoxia for 48 h. The wounded areas were photographed at the indicated time points and the results were quantified by ImageJ. The experiments were repeated at least in triplicate.
Transwell invasion assay
The invasion assays used to evaluate the pancreatic cancer cells invasion were performed with polycarbonate filters with 8.0 µm pore inserts (Corning, USA) in a 24-well transwell. The upper surface of the transwell insert membrane was coated with 50 µL of ¼ diluted Matrigel (BD Biosciences, USA). Accordingly, 3 × 105/mL cells were resuspended in a serum-free medium. Subsequently, 200 µL of cell suspensions were seeded in the upper chamber, whereas 500 µL of the complete medium with 10% fetal bovine serum was added to the lower well. After incubating under normoxia or hypoxia for 48 h, the cells were fixed with methanol and stained with 0.1% crystal violet. The numbers of migrated cells in five randomly selected fields (magnification: ×100) were counted under microscopy. The experiments were repeated at least in triplicate.
Statistical analysis
The data were expressed as mean ± standard deviation (SD) with SPSS software (SPSS version 17.0, Shanghai, China). The correlation between HIF-1α and YAP1 messenger RNA (mRNA) expressions was analyzed by Pearson test. The chi-square or Fisher’s exact test was used to analyze the correlation between the YAP1 expression and the clinicopathological features. The Kaplan–Meier method and log-rank test were used to test the survival curves. The Student’s t test was used to determine the significance of the differences in the cell migration and invasion assays in vitro. A p value less than 0.05 was considered statistically significant.
Results
Expressions of YAP1 and HIF-1α are upregulated in PDAC
The expressions of YAP1 and HIF-1α mRNAs were examined in 19 PDAC tumor samples and matched adjacent non-tumor samples by qRT-PCR. YAP1 and HIF-1α mRNA levels were upregulated in tumor tissues (Figure 1(a) and (b)). In addition, HIF-1α mRNA expression was positively correlated with that of YAP1 in tumor tissues, analyzed using a Pearson test (Figure 1(c)). The results were further confirmed by Western blot analysis (Figure 1(d)). In accordance, the results of immunofluorescence also demonstrated that YAP1 and HIF-1α expressions were upregulated in tumor tissues (Figure 1(e)).
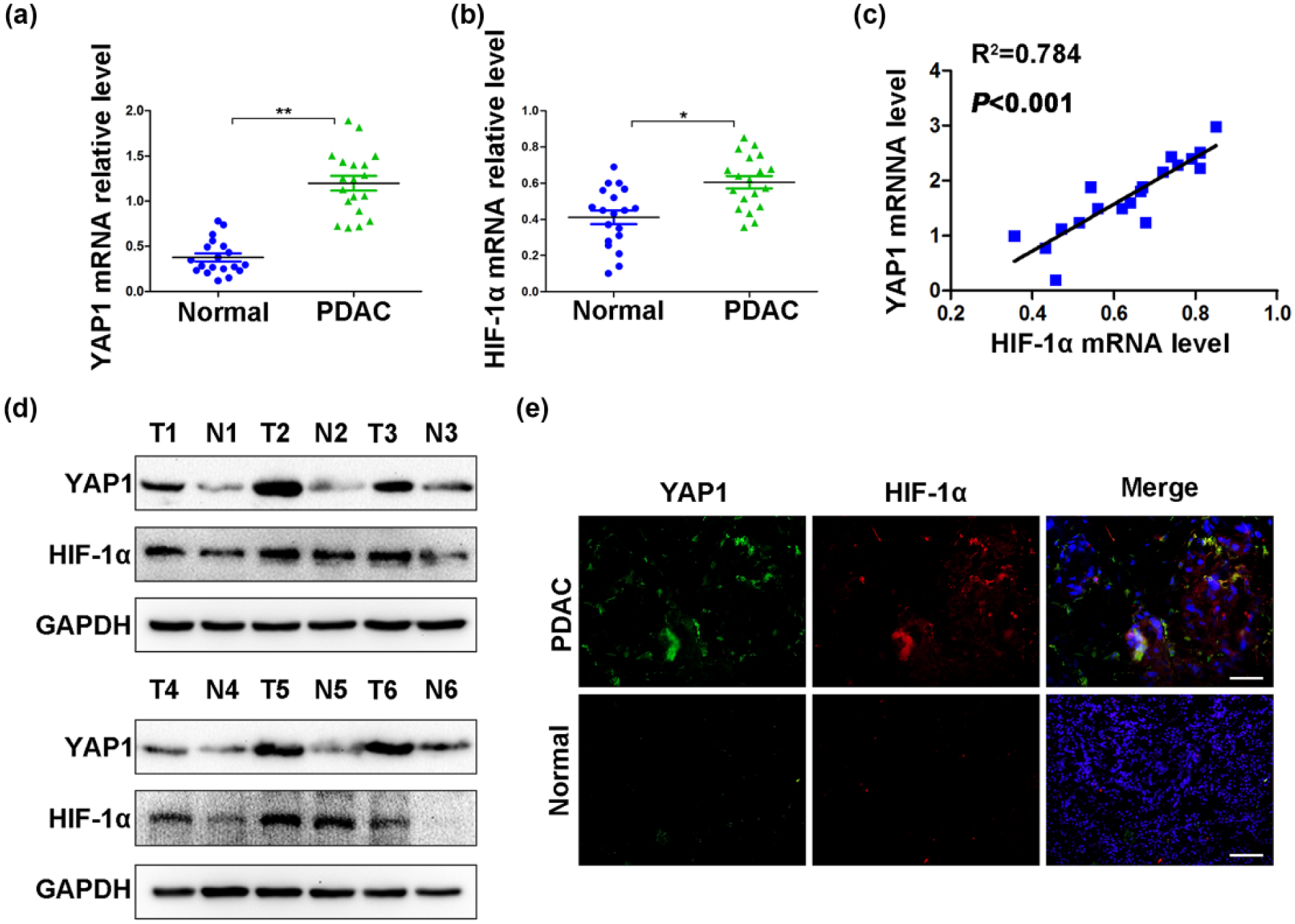
Expressions of YAP1 and HIF-1α are upregulated in PDAC. The expressions of (a) YAP1 mRNA and (b) HIF-1α mRNA in 19 tumor (T) and matched adjacent non-tumor tissues (N) were detected by qRT-PCR. (c) The correlation between HIF-1α and YAP1 mRNA expressions in PDAC tissues was analyzed using a Pearson test. (d) The expressions of YAP1 and HIF-1α protein were detected in six pairs of PDAC patients by Western blot analysis. (e) The expressions of YAP1 and HIF-1α in tumor and matched adjacent non-tumor tissues were examined by immunofluorescence (magnification: ×200, bar = 100 µM).
Nucleus YAP1 upregulation correlates with poor tumor differentiation and prognosis for PDAC patients
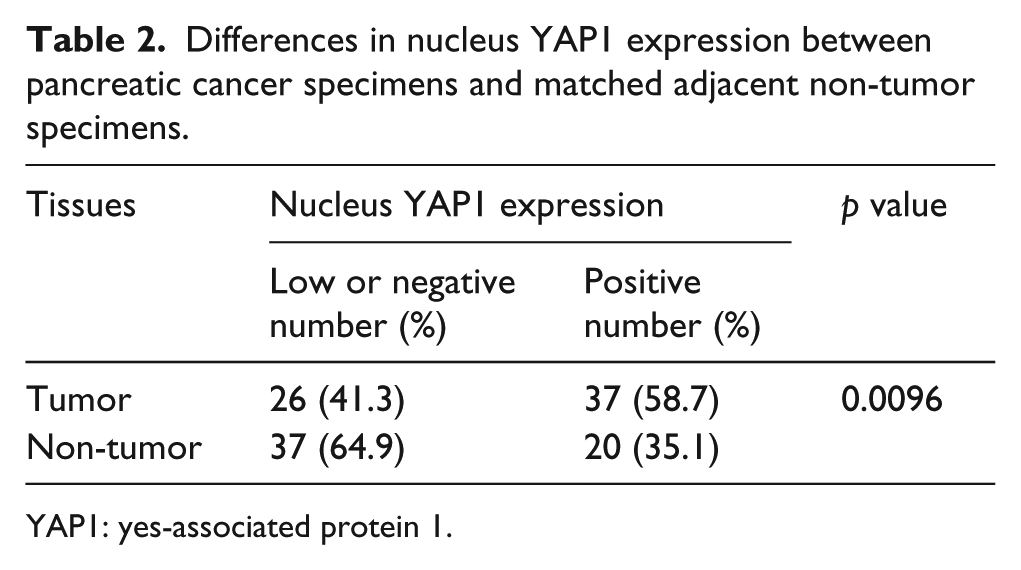
We further investigated the YAP1 expression and subcellular location by examining the pancreatic cancer TMAs containing 63 tumor samples and 57 matched adjacent non-tumor samples. YAP1 was detected in both cytoplasm and nucleus by IHC. Expression of nucleus YAP1 in advanced stage tumors was higher than that in early stage tumors (Figure 2(a) and (b)). The adjacent non-tumor tissues had low YAP1 expression (Figure 2(c)). YAP1 activation is characterized by accumulation of the nucleus YAP1. 16 The PDAC patients were divided into two groups according to the status of the nucleus YAP1 expression. The nucleus YAP1 was positively expressed in 58.7% of the tumor tissues and 35.1% of the adjacent non-tumor tissues. A significant difference in the positive rate of the nucleus YAP1 was found between the tumor tissues and non-tumor tissues (Table 2, p < 0.05). We further investigated the associations between nucleus YAP1 expression and clinicopathological characteristics of pancreatic cancer patients. Table 3 indicated that the nucleus YAP1 expression was correlated with tumor differentiation. However, staining for nucleus YAP1 showed no significant associations with gender, age, tumor location, tumor size, TNM stages, or clinical stage (p > 0.05). According to Kaplan–Meier method, the overall survival time of patients with high nucleus YAP1 expression was significantly shorter than those with negative and low nucleus YAP1 expression (Figure 2(d)). These results indicate that the nucleus YAP1 overexpression is correlated with poor differentiation and prognosis for PDAC patients.
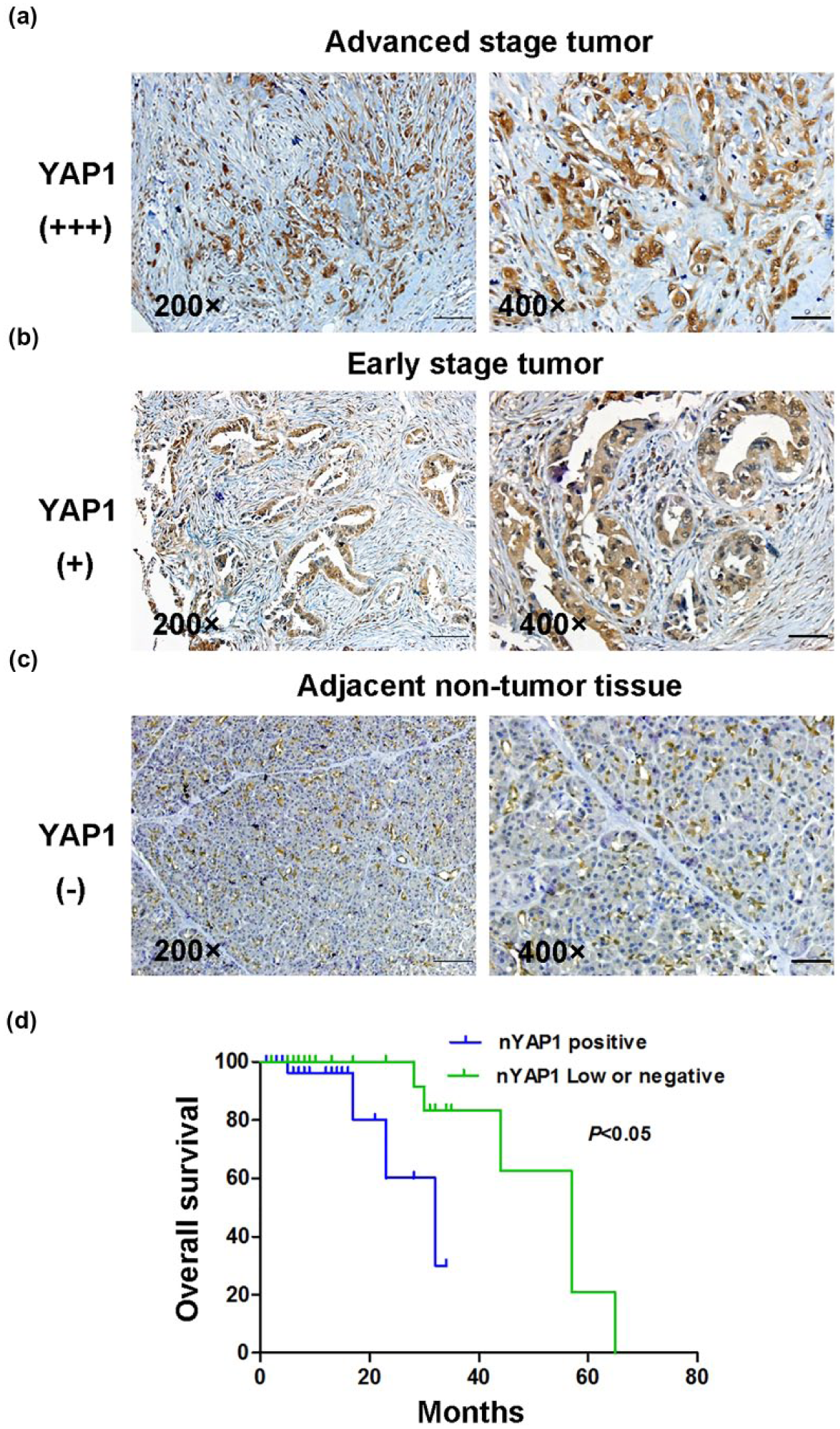
YAP1 is upregulated in PDAC and nucleus YAP1 upregulation correlates with a poor prognosis. The expression of YAP1 was detected in (a) advanced, (b) early stage tumor tissues, and (c) adjacent non-tumor tissues by immunohistochemistry (magnification: ×400, bar = 25 µM). (d) Kaplan–Meier survival curves showed that high expression of nucleus YAP1 was significantly correlated with poor survival.
Differences in nucleus YAP1 expression between pancreatic cancer specimens and matched adjacent non-tumor specimens.
YAP1: yes-associated protein 1.
Correlation between nucleus YAP1 expression and the clinicopathological features of pancreatic cancer.

YAP1: yes-associated protein 1; TNM: tumor–node–metastasis.
The differential TNM stages of 16 patients were not recorded.
The differential lymphnodes of three patients were not recorded.
The clinical stages of nine patients were not recorded.
Expression of YAP1 is upregulated in PDAC cells
We first examined YAP1 expression in five PDAC cell lines and HPDE6 in vitro by qRT-PCR and Western blot analysis. YAP1 mRNA and protein levels were higher in PDAC cells than those in HPDE6 (Figure 3(a) and (b)). Two PDAC cell lines (PANC-1 and BxPC-3) with YAP1 overexpression were selected for further studies in vitro. As expected, nucleus YAP1 protein levels in PANC-1 and BxPC-3 cells were significantly increased (Figure 3(c)). We also detected the expression and location of YAP1 in PANC-1 and BxPC-3 cells by confocal microscopy. The YAP1 expression was significantly increased in both cytoplasm and nucleus (Figure 3(d)). These results show that YAP1 is upregulated in PDAC cells and enhances PDAC progression.

Expression of YAP1 is upregulated in PDAC cells. (a and b) The expressions of (a) YAP1 mRNA (b) and protein in PDAC cells and HPDE6 were determined by qRT-PCR and Western blot analysis. (c) The expression of nucleus YAP1 was detected in PANC-1, BxPC-3, and HPDE6 by Western blot analysis and quantified. (d) The expression and location of YAP1 was detected in PANC-1, BxPC-3, and HPDE6 by confocal microscopy (bar = 10 µΜ). Data were presented as mean ± SD.
Knockdown of YAP1 inhibits PDAC cells migration and invasion in vitro
YAP1 is associated with various malignant behaviors of cancers 8 and plays an important role in metastasis of pancreatic cancer in the clinical stages. 22 Therefore, we investigated the role of YAP1 in pancreatic cancer metastasis. The results of qRT-PCR and Western blot analysis revealed that the shYAP1 significantly inhibited YAP1 expression (Figure 4(a) and (b)). The wound healing assay and transwell assay demonstrated that depletion of YAP1 decreased the cells migration and invasion (Figure 4(c) and (d)). The EMT- and MMP-mediated ECM degradation plays an important role in the tumor invasion and metastasis. 35 MMP2 and MMP13 are the key (MMPs) that enhance PDAC cells invasion.36,37 We examined the expressions of the EMT-related markers, MMP2, and MMP13 to investigate the mechanism by which YAP1 regulated PDAC cells invasion. Western blot analysis showed that depletion of YAP1 downregulated Vimentin, Snail, MMP2, and MMP13 and upregulated E-cadherin (Figure 4(e)). These results indicate that YAP1 promotes PDAC cells invasion via EMT- and MMP-mediated ECM degradation.
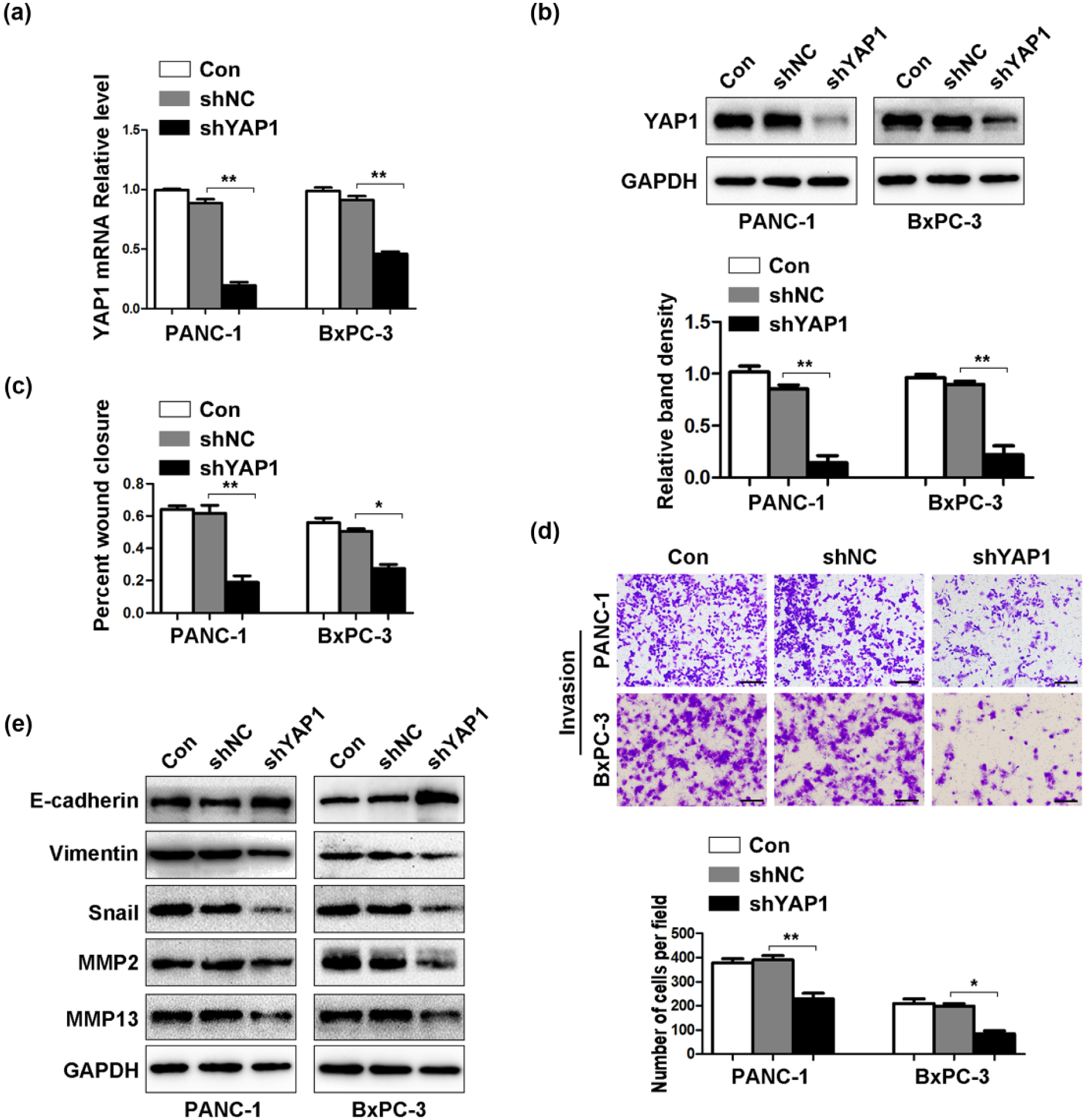
Knockdown of YAP1 inhibits PDAC cells migration and invasion in vitro. (a and b) Compared with untreated and control groups, the expressions of (a) YAP1 mRNA and (b) protein were inhibited in knockdown group by qRT-PCR and Western blot analysis. (c and d) The ability of PDAC cells migration and invasion was examined by wound healing assay and transwell assay (magnification: ×100, bar = 200 µM). The results were quantified. (e) Depletion of YAP1 suppressed the expressions of Vimentin, Snail, MMP2, and MMP13 and increased the expression of E-cadherin by Western blot. Data were presented as mean ± SD.
Hypoxia promotes PDAC cells invasion via EMT
Hypoxia promotes various tumor cells migration and invasion. 23 The wound healing assay and transwell assay showed that hypoxia significantly increased PDAC cells migration and invasion (Figure 5(a) and (b)). We examined the mRNA and protein levels of EMT-related markers, MMP2, and MMP13 by qRT-PCR and Western blot analysis. As expected, the expressions of Vimentin, Snail, MMP2, and MMP13 were increased and the expression of E-cadherin was decreased under hypoxia (Figure 5(c) and (d)).

Hypoxia promotes PDAC cells invasion via EMT. The cells were cultured under normoxia or hypoxia for 48 h. (a and b) Hypoxia promoted PDAC cells migration and invasion by wound healing assay and transwell assay (magnification: ×100, bar = 200 µM). The results were quantified. (c and d) The expressions of E-cadherin, Vimentin, Snail, MMP2, and MMP13 mRNA and protein were detected by qRT-PCR and Western blot. Data were presented as mean ± SD.
Hypoxia deactivates Hippo pathway and induces YAP1 nucleus translocation
HIF-1α is a critical transcriptional factor in pancreatic cancer metastasis under hypoxia. 28 We proved that YAP1 played an important role in remodeling of tumor microenvironment. Therefore, we investigated whether hypoxia affects the Hippo pathway. Western blot analysis suggested that hypoxia induced HIF-1α upregulation and von Hippel-Lindau (VHL) downregulation. The expression of pYAP1 was decreased and the expression of TEAD was increased under hypoxia (Figure 6(a)). Nucleus YAP1 expression was elevated under hypoxia, while cytosolic YAP1 expression was decreased (Figure 6(b)). The ratio of nucleus YAP1 to cytosolic YAP1 was also increased under hypoxia (Figure 6(c)). In addition, the confocal microscopy demonstrated that both HIF-1α and YAP1 expressions were increased and specially located in nucleus under hypoxia (Figure 6(d)). Therefore, we transfected the shRNA targeted HIF-1α to silence HIF-1α expression (Figure 6(e) and (f)). These results suggest that hypoxia deactivates the Hippo pathway and contributes to YAP1 nucleus translocation.
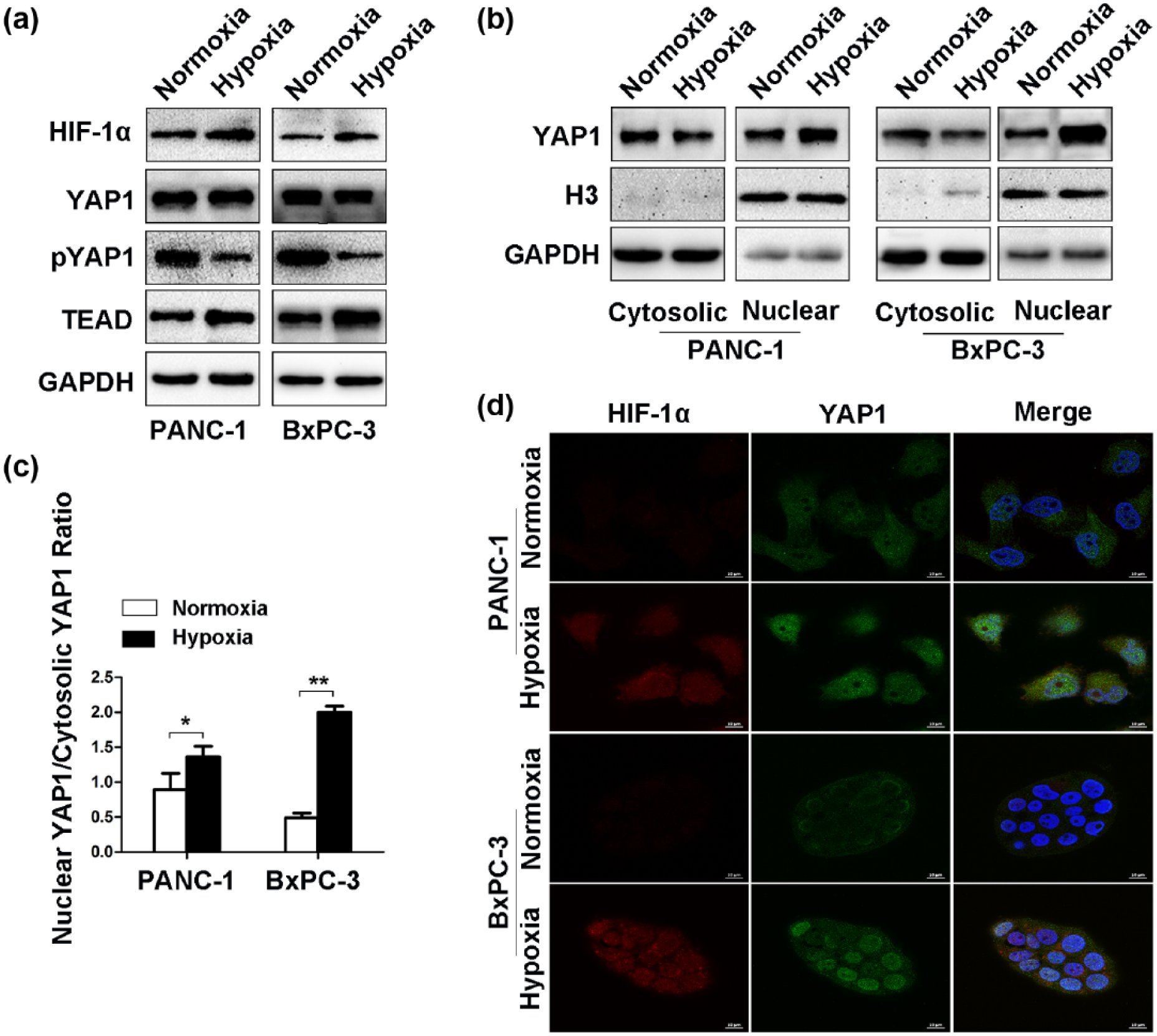
Hypoxia deactivates Hippo pathway and induces YAP1 nucleus translocation. The cells were cultured under normoxia or hypoxia for 48 h. (a) Effects of hypoxia on HIF-1α, VHL, YAP1, pYAP1, and TEAD expressions were measured by Western blot analysis. (b) The expressions of nuclear YAP1 and cytosolic YAP1 were detected by Western blot. (c) Nuclear YAP1 was quantified and normalized to cytosolic YAP1. (d) The expressions and locations of YAP1 and HIF-1α were detected by confocal microscopy (bar = 10 µM). (e and f) The expressions of HIF-1α mRNA and protein were inhibited in knockdown group by qRT-PCR and Western blot analysis. Data were presented as mean ± SD.
YAP1 interacts with HIF-1α and activates Snail transcription to promote PDAC cells invasion under hypoxia
The transwell assay showed that depletion of HIF-1α or YAP1 inhibited PANC-1 and BxPC-3 cells invasion under hypoxia (Figure 7(a)). Mechanism studies indicated that knockdown of HIF-1α or YAP1 decreased Vimentin, Snail, MMP2, and MMP13 mRNA and protein expressions and increased E-cadherin mRNA and protein expressions as shown by qRT-PCR and Western blot analysis (Figure 7(b) and (c)). The invasive ability of cancer cells was dependent on HIF-1α and YAP1 expressions under hypoxia. In addition, we further explored the mechanism by which HIF-1α and YAP1 interacted to promote PDAC cells invasion under hypoxia. We examined the Hippo pathway and HIF-1α protein levels under the condition of YAP1 or HIF-1α depletion. Western blot analysis demonstrated that the expressions of total YAP1, pYAP1, and TEAD showed no significant difference between shNC1 and shHIF-1α group. However, depletion of YAP1 significantly inhibited the expressions of total YAP1, pYAP1, and TEAD compared with the control under hypoxia. The expression of HIF-1α was also notably reduced in shYAP1 group (Figure 7(d)). These results demonstrate that YAP1 plays a critical role in HIF-1α transcriptional activation. Nucleus YAP1 interacts with HIF-1α and activates Snail transcription to participate in EMT- and MMP-mediated remodeling of tumor microenvironment (Figure 8).

YAP1 interacts with HIF-1α and activates the Snail transcription to promote PDAC cells invasion under hypoxia. The cells were cultured under hypoxia for 48 h. (a) The numbers of invaded cells in control groups were measured compared with those in HIF-1α or YAP1 silencing groups by transwell assay (magnification: ×100, bar = 200 µM). The results were quantified. (b and c) The expressions of E-cadherin, Vimentin, Snail, MMP2, and MMP13 mRNA and protein were determined by qRT-PCR and Western blot analysis. (d) The effects of silencing HIF-1α and YAP1 on HIF-1α and Hippo pathway under hypoxia were measured by Western blot analysis. Data were presented as mean ± SD.

Schematic presentation of crosstalk between Hippo pathway and hypoxia to promote PDAC metastasis. The high expression of nucleus YAP1 promotes PDAC cells migration and invasion via regulating EMT-related markers, MMP2, and MMP13. Hypoxia deactivates Hippo pathway and induces YAP1 nucleus translocation. In addition, nucleus YAP1 interacts with HIF-1α and activates the Snail transcription to participate in EMT- and MMP-mediated remodeling of tumor microenvironment.
Discussion
Hippo-YAP1 signaling pathway is initially identified as a mechanism involved in the regulation of organ development and tissue growth 7 and is more recently implicated in cell proliferation, migration, and stem cell self-renewal. 10 The hyperactivated YAP1 expression is upregulated in various malignancies, including gastrointestinal cancers15–17 and pancreatic cancer.18,19 Expression of YAP1 is elevated in pancreatic cancer 21 and implicated in tumorigenesis and recurrence in a genetic mouse model of pancreatic cancer.19,20 In this study, we confirmed that the YAP1 mRNA and protein levels in PDAC specimens were upregulated compared with those in matched adjacent non-tumor specimens. Although non-tumor tissues were also stained positive for YAP1, the overall rate of YAP1 expression was significantly higher in the tumor tissues, in consistent with previous studies. 21 The expression of nucleus YAP1 was positively associated with tumor differentiation. These data indicate that nucleus YAP1 is essential for pancreatic cancer progression. YAP1 is an independent predictor of short overall survival for hepatocellular carcinoma 15 and cholangiocarcinoma. 16 Our study also revealed, for the first time, that high expression of nucleus YAP1 was negatively associated with the overall survival for PDAC. These results suggest that YAP1 is a novel prognostic marker for PDAC patients.
YAP1 is required for tumor cell proliferation and apoptosis, which has been widely studied in various types of cancers.8,10 In addition, the role of hyperactivated YAP1 in tumor migration and invasion is an important process for tumor metastasis. The overexpression of YAP1 promotes proliferation, invasion, metastasis, and inhibits apoptosis in multiple cancers.38,39 Meanwhile, Yang et al. 22 revealed that YAP1 promoted pancreatic cancer cells invasion and motility through LPAR3-mediated mitotic phosphorylation–dependent manner. However, few studies have investigated the role of the Hippo pathway in the later steps of cancer metastasis. Furthermore, YAP1 is implicated in EMT. The overexpression of YAP1 overcomes cell contact inhibition and contributes to metastatic properties via EMT in cholangiocarcinoma, 16 colon cancer, 38 and breast cancer. 40 In this study, depletion of YAP1 resulted in increased E-cadherin expression and decreased Vimentin and Snail expressions. The MMPs are also essential factors related to cell migration and invasion and are considered as markers for malignant tumors invasion and metastasis.41,42 The MMPs participate in the initial steps of degrading the tumor microenvironment and contribute to tumor invasion and metastasis. Previous studies showed that YAP1 knockdown downregulated the MMP2 and MMP9 expressions and inhibited invasion of osteosarcoma 43 and gastric adenocarcinoma. 44 MMP13 is a matrix metalloproteinase produced by various malignant tumors, including pancreatic cancer 37 and liver cancer. 45 We found that knockdown of YAP1 inhibited PDAC cells migration and invasion and significantly decreased the MMP2 and MMP13 expressions. Based on these data, YAP1 facilitates PDAC cells invasion via EMT- and MMP-mediated remodeling of tumor microenvironment.
Hypoxia commonly exists in the tumor microenvironment of solid tumors and is associated with aggressive features, such as local invasion and distant metastasis. 25 This study confirmed that the expression of YAP1 was positively correlated with HIF-1α level in PDAC tissues. Hypoxia induced HIF-1α expression enhanced the EMT to promote tumor migration and invasion. 46 HIF-1α also activates nuclear factor–kappa Β, Notch signaling, and Hedgehog signaling to induce EMT in several human epithelial cancer cells.47–49 In this study, hypoxia enhanced PDAC cells migration and invasion. Hypoxia also suppressed E-cadherin expression and increased Vimentin, Snail, MMP2, and MMP13 expressions. Yan et al. 50 found that hypoxia induced downregulation of pYAP in prostate, colon, and breast cancer cells and promoted proliferation. However, the way by which hypoxia interacts with Hippo pathway to promote tumor metastasis needs to be investigated. We demonstrated that hypoxia induced HIF-1α expression and VHL proteasomal degradation lead to HIF-1α accumulation. In addition, hypoxia suppressed pYAP1 expression and induced YAP1 nucleus translocation. The ratio of nucleus YAP1 to cytosolic YAP1 was also increased. These results demonstrate that hypoxia deactivates the Hippo pathway to promote PDAC cells invasion.
Under hypoxia, HIF-1α stimulated the expressions of two E3 ubiquitin ligases (seven in absentia homolog (SIAH)1/2). SIAH1/2 promoted ubiquitination and degradation of large tumor suppressor kinase 2 (LATS2), leading to decrease in YAP1 phosphorylation and nucleus translocation. The activated YAP1 interacted with and stabilized HIF-1α to enhance the transcription of HIF-1α targeted genes to induce breast cancer tumorigenesis. 31 The transcription factor Snail also initiated EMT during tumor progression. 51 In addition, YAP1 induced EMT to participate in tumor growth, invasion, and metastasis via regulating Snail in colorectal cancer. 38 Hypoxia also induced EMT through the activation of Snail expression in PDAC. 52 In this study, depletion of HIF-1α or YAP1 suppressed PDAC cells invasion via downregulation of E-cadherin and upregulation of Vimentin, Snail, MMP2, and MMP13. HIF-1α silencing did not change the expressions of YAP1, pYAP1, and TEAD, while YAP1 silencing dramatically reduced the accumulation of HIF-1α under hypoxia. The results suggest that YAP1 regulates HIF-1α expression and activates Snail expression to participate in EMT- and MMP-mediated remodeling of tumor microenvironment. However, the regulatory role of YAP1 and HIF-1α on these molecules was not validated in PDAC cells by chromatin immunoprecipitation.
In summary, our data confirmed that YAP1 and HIF-1α expressions were elevated in PDAC. The high expression of nucleus YAP1 was associated with poor histological grade and prognosis for PDAC patients. The hyperactivated YAP1 contributed to the PDAC cells invasion via regulation of EMT-related markers, MMP2, and MMP13 expressions. In addition, hypoxia promoted PDAC cells invasion via regulating the targeted genes. Hypoxia deactivated the Hippo pathway and induced the YAP1 nucleus translocation. Nucleus YAP1 interacted with HIF-1α and activated Snail transcription to participate in EMT- and MMP-mediated remodeling of tumor microenvironment. Furthermore, depletion of YAP1 or HIF-1α suppressed PDAC cells invasion under hypoxia. Taken together, we linked the two pathways with very similar oncogenic effects and investigated the pleiotropic roles of YAP1 in tumor progression and metastasis. Overall, we identify YAP1 is a new promising target for better treatment of pancreatic cancer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent
The study was performed according to the ethical standards of the institutional research committee and the standards set in the Declaration of Helsinki 1975. All samples were collected with informed consent and the experiments were approved by the institutional ethics committee of the Qianfoshan Hospital of Shandong Province, affiliated to Shandong University. This article does not contain any studies with animals performed by any of the authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Major Research and Development Program of China (2016YFC0106004), Shandong Provincial Science and Technology Development Planning, China (2015GSF121040), and Shandong Provincial Natural Science Foundation, China (ZR2012HL05 and ZR2015HL080).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
