Abstract
Hepatocellular carcinoma is one of the most common cancers, and approximately 80% develop from cirrhotic livers. We have previously shown that the aspartate salt of adenosine prevents and reverses carbon tetrachloride–induced liver fibrosis in rats. Considering the hepatoprotective role of this adenosine derivative in fibrogenesis, we were interested in evaluating its effect in a hepatocarcinogenesis model induced by diethylnitrosamine in rats, where multinodular cancer is preceded by cirrhosis. Rats were injected with diethylnitrosamine for 12 weeks to induce cirrhosis and for 16 weeks to induce hepatocarcinogenesis. Groups of rats were treated with aspartate salt of adenosine from the beginning of carcinogen administration for 12 or 18 weeks total, and another group received the compound from weeks 12 to 18. Fibrogenesis was estimated and the proportion of preneoplastic nodules and tumors was measured. The apoptotic and proliferation rates in liver tissues were evaluated, as well as the expression of cell signaling and cell cycle proteins participating in hepatocarcinogenesis. The adenosine derivative treatment reduced diethylnitrosamine-induced collagen expression and decreased the proportion of nodules positive for the tumor marker γ-glutamyl transferase. This compound down-regulated the expression of thymidylate synthase and hepatocyte growth factor, and augmented the protein level of the cell cycle inhibitor p27; these effects could be part of its chemopreventive mechanism. These findings suggest a hepatoprotective role of aspartate salt of adenosine that could be used as a therapeutic compound in the prevention of liver tumorigenesis as described earlier for hepatic fibrosis.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer in the world. The HCC is a complex pathology associated with chronic liver disease and cirrhosis caused by diverse etiologies. 1 The mortality rate is very high mainly due to the difficulty in achieving early detection and efficient therapies for its treatment. There is an urgent need for safe chemoprevention against HCC. Animal models of hepatocarcinogenesis are crucial for understanding the cellular and molecular aspects of tumor development and for pharmacological testing. Although genetically engineered models of HCC are useful for demonstrating the oncogenic and tumor-suppressive roles of cellular and viral genes, 2 chemically induced hepatocarcinogenesis models can cause both cirrhosis and HCC. Specifically, the diethylnitrosamine (DEN)-induced hepatocarcinogenesis model produces HCC in a background of fibrosis resembling the liver microenvironment of almost all HCC patients. 3
Adenosine is a purine nucleoside that exerts its function by activation of adenosine receptors (A1AR, A2aAR, A2bAR, and A3AR) or by its uptake into cells via nucleoside transporters. Inside the cell, adenosine is phosphorylated, deaminated, degraded to uric acid, or transformed into S-adenosylhomocysteine, and its metabolism is very active, resulting in a short half-life. 4 IFC-305 is the aspartate salt of adenosine that has a longer half-life than adenosine in the liver, which increases its effects. 5 This adenosine derivative compound prevents and reverts carbon tetrachloride (CCl4)-induced cirrhosis from structural and physiological points of view and also a recovery of lipid droplets in stellate liver cells as observed by transmission electron microscopy. 6 Transcriptome studies have shown that more than half of the dysregulated genes in cirrhosis are normalized by IFC-305 treatment, favoring the recovery of hepatic functions, including the liver regenerative capacity lost in cirrhosis, and promoting the recovery of damaged DNA through proliferative cell nuclear antigen (PCNA) and p53. 7 IFC-305 showed a cancer chemopreventive effect in the Solt–Farber protocol, which induces HCC without liver cirrhosis. 8 Considering the hepatoprotective effects of IFC-305 in fibrogenesis and hepatocarcinogenesis, we were interested in evaluating the effect of IFC-305 in a model that combines cirrhosis and multifocal HCC, through weekly treatment with DEN.9–11 We measured the fibrosis level and the proportion of preneoplastic nodules through histological determination of two known tumor markers of hepatocarcinogenesis in the rat: γ-glutamyl transferase (GGT) and glutathione S-transferase pi (GSTP). 12 The proliferation and apoptotic rates in liver tissues were evaluated, as well as the expression of cell signaling and cell cycle proteins participating in HCC.
Materials and methods
Chemicals
IFC-305 is the aspartate salt of adenosine prepared with adenosine free base (MP Biomedicals, LLC, Illkirch, France) and
Animal treatment and experimental groups
Male Wistar rats (200 g) were obtained and housed at the Animal Facility of the National Autonomous University of Mexico (UNAM). All procedures were conducted according to our institutional guidelines for laboratory animals. Rats (n = 6) were intraperitoneally injected with DEN at 50 mg/kg body weight once a week for 12 weeks to induce cirrhosis (12-DEN), and another group received DEN for 16 weeks followed by a 2-week wash-out period for HCC development (18-DEN). 9 One group was intraperitoneally treated with IFC-305 at 50 mg/kg body weight three times weekly from the beginning of DEN administration for 12 (12-D + IFC) or 18 (18-D + IFC) weeks. Another group received IFC-305 from weeks 12 to 18 (12-18-D + IFC), as described in Figure 1. Age-matched normal rats were used as controls. Rats were weighed every week. At the end of the treatments, animals were deeply anesthetized for euthanization with pentobarbital sodium (PISA Agropecuaria, Hidalgo, México), blood was collected, livers were removed and weighed, one liver fraction was frozen for further experiments, and another liver fraction was fixed in paraformaldehyde.
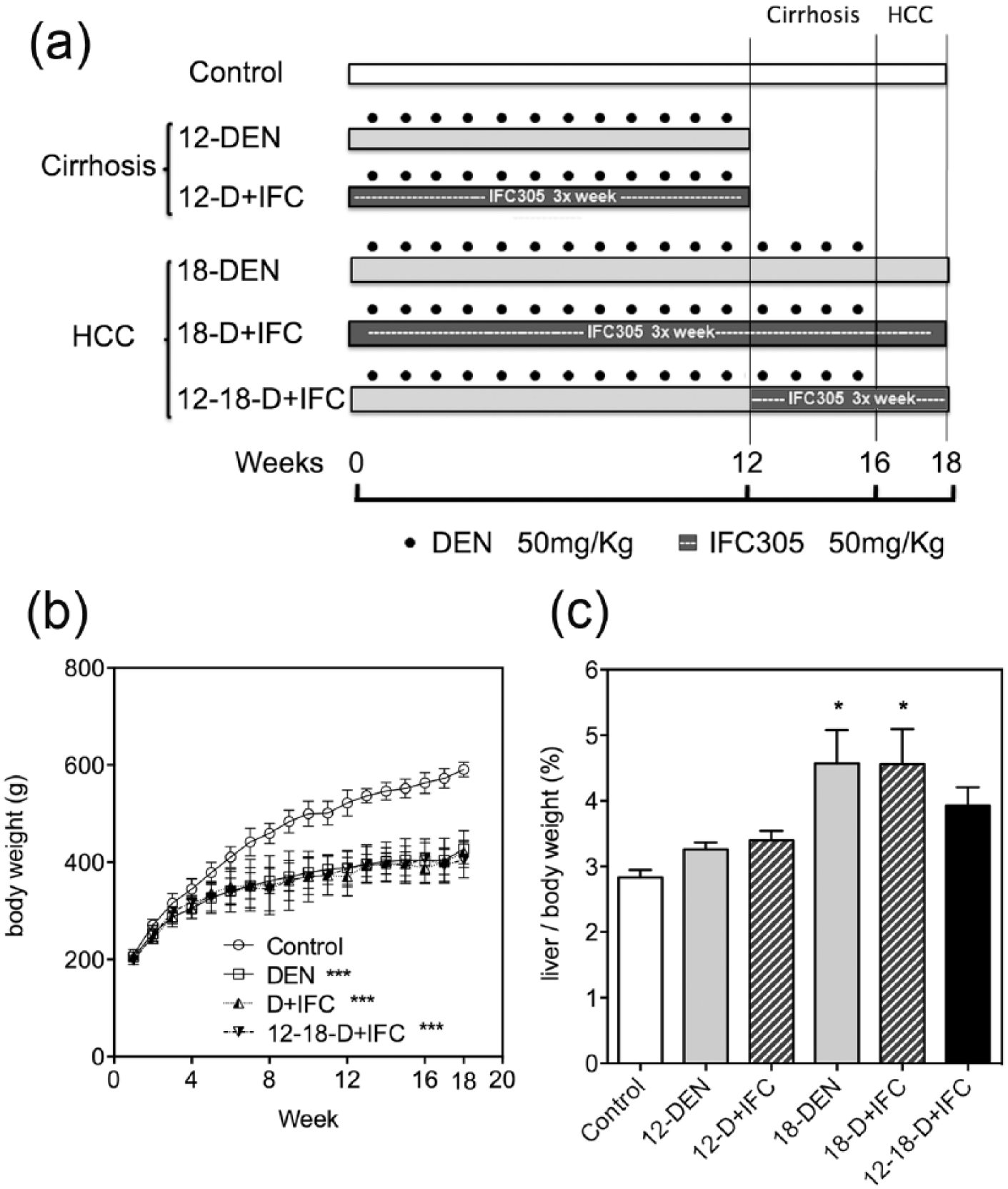
Experimental groups to evaluate the effect of IFC-305 on a sequential rat model of cirrhosis-HCC induced by DEN. (a) Rats were injected with DEN for 12 weeks to induce cirrhosis (12-DEN) and for 16 weeks to induce HCC development (18-DEN). IFC-305 was administered from the beginning of DEN injection as a prevention scheme (12-D + IFC; and 18-D + IFC). IFC-305 was administered from weeks 12 to 18 of DEN administration as a cirrhosis reversion and cancer prevention scheme (12-18-D + IFC). (b) Effects of DEN and IFC-305 on body weight measured each week. (c) Effects of DEN and IFC-305 on liver/body-weight ratios at the end of the treatments. The data represent mean ± standard error of the mean (SEM; n = 6 rats/group).
GGT activity in liver tissue
Frozen liver sections were processed following Rutenberg et al.’s 14 protocol. 15 Sections were fixed for 10 min in 96% ethanol and incubated with substrate solution (125 µg/mL γ-glutamyl-4-methoxy-2-naphtyl-amine (GMNA), 500 µg/mL glycyl-glycine, and 500 µg/mL Fast Blue BB salt in 10 mM Tris, pH 7.4). Then, the slides were washed with deionized water and incubated with 0.1 M CuSO4 for 5 min. Once the slides were dried, they were scanned and the number of lesions positive for GGT activity and their cumulative areas were quantified using ImageJ software.
Quantitative reverse transcription–polymerase chain reaction analysis
Total RNA was obtained from frozen liver samples using an RNeasy Mini Kit (Qiagen, Inc., Valencia, CA). RNA quality was verified by agarose gel electrophoresis, and rRNA 28S/18S > 1.7 ratios were utilized. cDNA was synthesized from 2 µg of total RNA using a High-Capacity RNA to cDNA Kit (Applied Biosystems, Inc., Foster City, CA). mRNA determinations were carried out using TaqMan® gene expression assay with the following FAM dye–labeled probes for rat (Applied Biosystems, Inc.): GSTP (Rn00561378_gH), thymidylate synthase (Tyms; Rn00569849_m1), and hepatocyte growth factor (HGF; Rn00566673_m1). The housekeeping genes used for normalization were VIC dye–labeled probes for 18S rRNA (Rn03928990) and actin-β1 (Rn00667869). Data analysis was performed using the 2−ΔΔCt method.
Apoptosis assay
Apoptosis was analyzed by terminal deoxynucleotidyl transferase–mediated dUTP nick end labeling (TUNEL) assay following the manufacturer’s protocol (DNA Apoptosis TUNEL Kit; Roche Diagnostics Corp., Indianapolis, IN). The apoptosis index was calculated as the percentage of TUNEL-positive nuclei after counting at least five random fields at 40× magnification.
Western blot
Liver samples were homogenized with radioimmunoprecipitation assay (RIPA) buffer containing protease and phosphatase inhibitors (Roche Diagnostics Corp.). Western blotting was performed as described previously. 7 The following antibodies were used: PCNA (Upstate Biotechnology, Inc., Lake Placid, NY); cyclin D1, cyclin E, cyclin A, Cdk2, Cdk4, Cdk6, p16, p27, p21, pRb, gankyrin, and β-actin (Santa Cruz Biotechnology, Santa Cruz, CA); Met, Mdm2 (murine double minute 2), and p53 (Chemicon International, Temecula, CA); E2F1 and poly(ADP-ribose) polymerase (PARP; BD PharMingen Corp., San Jose, CA); and cleaved caspase-3 (Asp175; Cell Signaling Technology, Inc., Danvers, MA). Densitometry of bands was performed with Quantity One software (Bio-Rad Laboratories, Hercules, CA).
Immunohistochemical analysis
Immunohistochemical staining of PCNA, GSTP, A2aAR, and A3AR protein expression
Paraffin-embedded liver sections (4-µm thick) were analyzed using the Dako EnVision® + System-HRP (DAB; Dako, Carpinteria, CA) as previously described. 7 Primary antibodies directed against the following proteins were diluted 1/100: PCNA clone PC10 (Dako), GSTP (MBL International Corporation, Woburn, MA), and A2aAR and A3AR (Alpha Diagnostic International, San Antonio, TX). Images of five random fields (n = 4 rats) at 40× magnification were captured using an Evolution™/QImaging Digital Camera (Media Cybernetics, Inc., Bethesda, MD), and the percentage of PCNA-positive hepatocytes was counted using Image-Pro Plus 7 (Media Cybernetics, Inc.).
Statistical analysis
The data are expressed as the mean ± standard error of the mean (SEM) of at least four animals, as indicated in the figure legends. Statistically significant differences among the experimental groups were determined by one-way analysis of variance (ANOVA) with multiple comparisons. Significance was set at p < 0.05. Graphs were created with the GraphPad Prism software version 6.0.
Results
Effect of IFC-305 on liver fibrogenesis and body weight in the hepatocarcinogenesis model
In the model of cirrhosis-HCC induced by DEN cirrhosis is predominantly observed in the liver at 12 weeks, and at multifocal HCC appears at 18 weeks. All groups of rats treated with DEN exhibited significantly (p < 0.01) lower body-weight gain than did rats in the control group (Figure 1(b)). When liver/body-weight ratios were compared, only the 18-DEN and 18-D + IFC groups demonstrated a significantly increased proportion of 42% compared to controls (Figure 1(c)). Fibrosis status in experimental groups was visualized by quantitative analysis of histological sections with Masson’s trichrome staining and by measuring gene expression of collagen type 1 alpha 1 (
IFC-305 treatment inhibits preneoplastic lesion development in DEN-induced hepatocarcinogenesis
The effect of IFC-305 treatment on preneoplastic lesions development in DEN-treated rats was determined by examining the expression of two enzymes of glutathione metabolism and oxidative stress: GGT and GSTP commonly used as tumor markers in liver chemical carcinogenesis.16,17 In the 12-DEN group, the GGT-positive nodular area at the cirrhosis stage demonstrated an accumulated tissue area of 6.5%, whereas in the 12-D + IFC group, this area was reduced to 3.3% (Figure 2(a) and (b)). During the multinodular HCC period, the GGT-positive tissue area significantly increased, reaching 37.5% in the 18-DEN group. Both IFC-305-treated groups (18-D + IFC and 12-18-D + IFC) showed a statistically significant reduction in GGT-positive areas compared to the 18-DEN group (Figure 2(b)). The highest level of
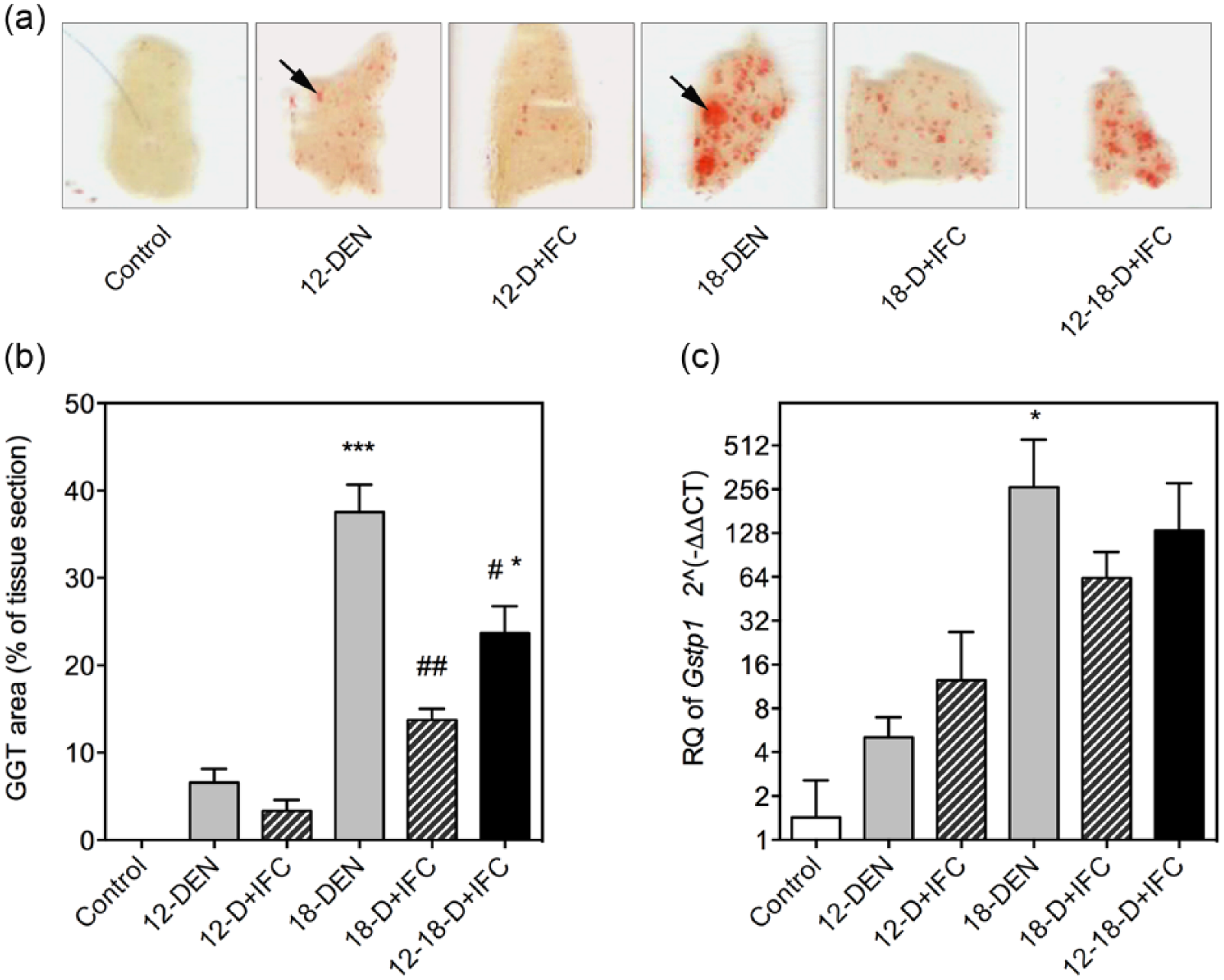
IFC-305 treatment inhibits preneoplastic lesion development in DEN-induced hepatocarcinogenesis. (a) Histological sections of livers with GGT+ lesions were the activity of the enzyme is stained in red (black arrow). Scale bar corresponds to 2 mm. (b) Quantitative analysis of GGT+ areas. (c) Expression of the
Effect of IFC-305 on apoptosis in HCC groups
To determine whether tumor reduction by IFC-305 was due to apoptosis induction, the number of apoptotic hepatocytes was evaluated by TUNEL assay in serial sections of GSTP-positive and GSTP-negative immunostained areas. All DEN-treated groups presented greater hepatocyte apoptosis compared to the control group, although the differences were not statistically significant among them, regardless of the IFC-305 treatment or GSTP-positive and GSTP-negative areas (Figure 3(a) and (b)). To further evaluate cell death, we studied the expression of apoptosis-related proteins by western blot analysis of total liver homogenates. The 18-DEN group showed the highest level of cell death compared to the control group, as indicated by increases in the cleavage of caspase-3 and PARP. Both IFC-305-treated groups exhibited fewer parameters of apoptosis than did the 18-DEN group (Figure 3(c) and (d)); thus, diminution of nodules by IFC-305 treatment does not appear to be related to increased apoptosis.

Effect of IFC-305 on cell apoptosis in DEN-induced hepatocarcinogenesis. (a) Immunohistochemical analysis in serial sections of GSTP and hepatocyte apoptosis evaluated by TUNEL assay in HCC groups. A strong GSTP signal is seen in large areas of DEN-treated groups (GSTP+). GSTP+ areas are delineated with a dotted line in the middle panels of the TUNEL assay. Scale bar corresponds to 200 µm under 10× magnification and 50 µm under 40× magnification. (b) Quantification of the percentage of TUNEL-positive hepatocytes (as exemplified with a black arrows in 40× panels) was performed in GSTP+ and GSTP− areas as described in the “Materials and methods” section. (c) Expression levels of active caspase-3 and cleaved PARP were evaluated by western blot analysis of total liver homogenates; a representative experiment is shown. (d) Densitometry quantification of bands expressed as the mean ± standard error of the mean (SEM; n = 6 rats/group); values were normalized to β-actin.
Effect of IFC-305 on cell proliferation in DEN-induced hepatocarcinogenesis
To evaluate whether diminution of nodules by IFC-305 could be related to a decrease in cell proliferation, we studied proliferative activity by analyzing PCNA immunostaining of hepatocytes (Figure 4(a)–(e)). PCNA staining was analyzed in serial sections considering both GSTP-positive and GSTP-negative immunostained areas. In GSTP-positive areas of the 18-DEN group, a marked increase in the number of hepatocytes with PCNA-positive nuclei (proliferating cells) was evident and tended to decrease with IFC-305 treatment, with a statistically significant reduced number in the 12-18-D + IFC group (Figure 4(b)). PCNA expression in total liver homogenates, where all liver cells are evaluated, was reduced in the IFC-305-treated groups (Figure 4(c) and (d)). This was further tested measuring Tyms expression; an enzyme required for DNA synthesis, the diminution of its expression in the presence of IFC-305 confirmed this possibility (Figure 4(e)). Thus, a more plausible explanation for reduced tumor presence would be an anti-proliferative effect of IFC-305 on tumor cells.

Effect of IFC-305 on cell proliferation in DEN-induced hepatocarcinogenesis. (a) Immunohistochemical analysis of GSTP and PCNA, as hepatocyte proliferation marker, in serial sections of HCC groups. GSTP+ areas are delineated with a dotted line in the middle panels of the PCNA assay. Scale bar corresponds to 200 µ m under 10× magnification and 50 µm under 40× magnification. (b) Quantification of the number of PCNA-positive hepatocytes (as exemplified with black arrows in 40× panels) was performed in GSTP− and GSTP+ areas as described in the “Materials and methods” section. (c) Representative western blot analysis of protein expression of PCNA and β-actin from total liver homogenates of HCC groups. (d) Densitometry quantification of PCNA expressed as the mean ± standard error of the mean (SEM; n = 6 rats/group); values were normalized to β-actin. (e) Expression of Tyms in experimental rat livers as measured by quantitative reverse transcription–polymerase chain reaction (qRT-PCR). The mRNA level was measured and compared to that in control liver. Arbitrary expression values were normalized to β-actin. The data represent the mean ± SEM (n = 6 rats/group).
Effect of IFC-305 on the HGF/c-Met pathway
To understand the possible pathway involved in the reduced cell proliferation of tumor cells by IFC-305 in HCC groups, we studied the expression of the c-Met receptor and its liver ligand, HGF, which plays an important role in liver carcinogenesis and which promote cell survival and proliferation by the Ras/MAPK and PI3K pathways. The 18-DEN group had the highest mRNA
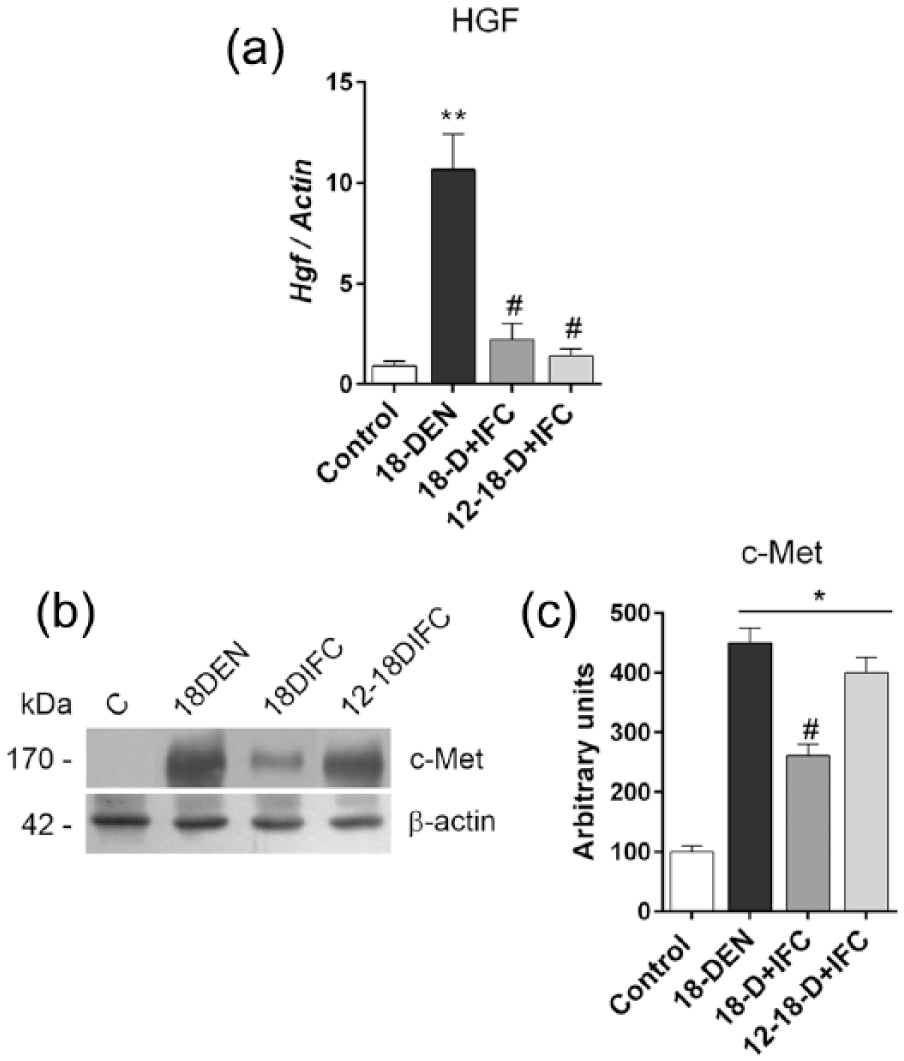
Effect of IFC-305 on the HGF/c-Met pathway. (a)
Effect of IFC-305 on cell cycle–related proteins
To further characterize the effect of IFC-305 on liver cell proliferation, the expression of cell cycle proteins was studied. Cyclin D1 showed a higher expression level in all DEN-treated groups than in the control group, independent of IFC-305 treatment. Cyclin E was reduced in the 18-D + IFC group compared to the 18-DEN group, whereas cyclin A exhibited a significant reduction in the 18 DEN-treated groups. In both IFC-305-treated groups, its expression was restored to control level, while Cdk2 expression was only significantly increased in the 18-DEN group (Figure 6(a) and (b)). In contrast, DEN treatment increased E2F1 protein expression that was significantly reduced in the 18-D + IFC group (Figure 6(a) and (b)). The most notable expression change induced by IFC-305 in DEN-treated groups was the significantly increased level of the cell cycle inhibitor p27, whereas the p21 protein exhibited a statistically higher expression level in all DEN-treated groups compared to the control group (Figure 6(a) and (c)). No changes were observed for Cdk4, Cdk6, and p16 protein expression (data not shown). To further understand the effects of IFC-305 on cell cycle regulation, we evaluated the expression of other cell cycle–related proteins. An increase in the oncoprotein gankyrin was observed in the 18 DEN-treated groups, whereas in both IFC-305-treated groups, its expression was similar to that of the control group (Figure 6(a) and (c)). The expression of the Mdm2 protein was increased in the three DEN-treated groups. The increases in gankyrin and Mdm2 expression in the 18-DEN group were accompanied by a significant reduction in p53 expression. No such effect was observed in both IFC-305-treated groups, in which p53 was expressed as in the control group. In contrast, the DEN-induced increase in pRB expression did not change upon IFC-305 administration (Figure 6(a) and (c)). Overall, the most important effects of IFC-305 treatment on DEN-induced liver tumors were the increase in p27 expression and the recovery of p53 levels lowered by DEN. This could be associated to inhibition of the cell cycle through suppression of the HGF/c-Met pathway and reduction in Tyms expression by IFC-305 in tumor cells.
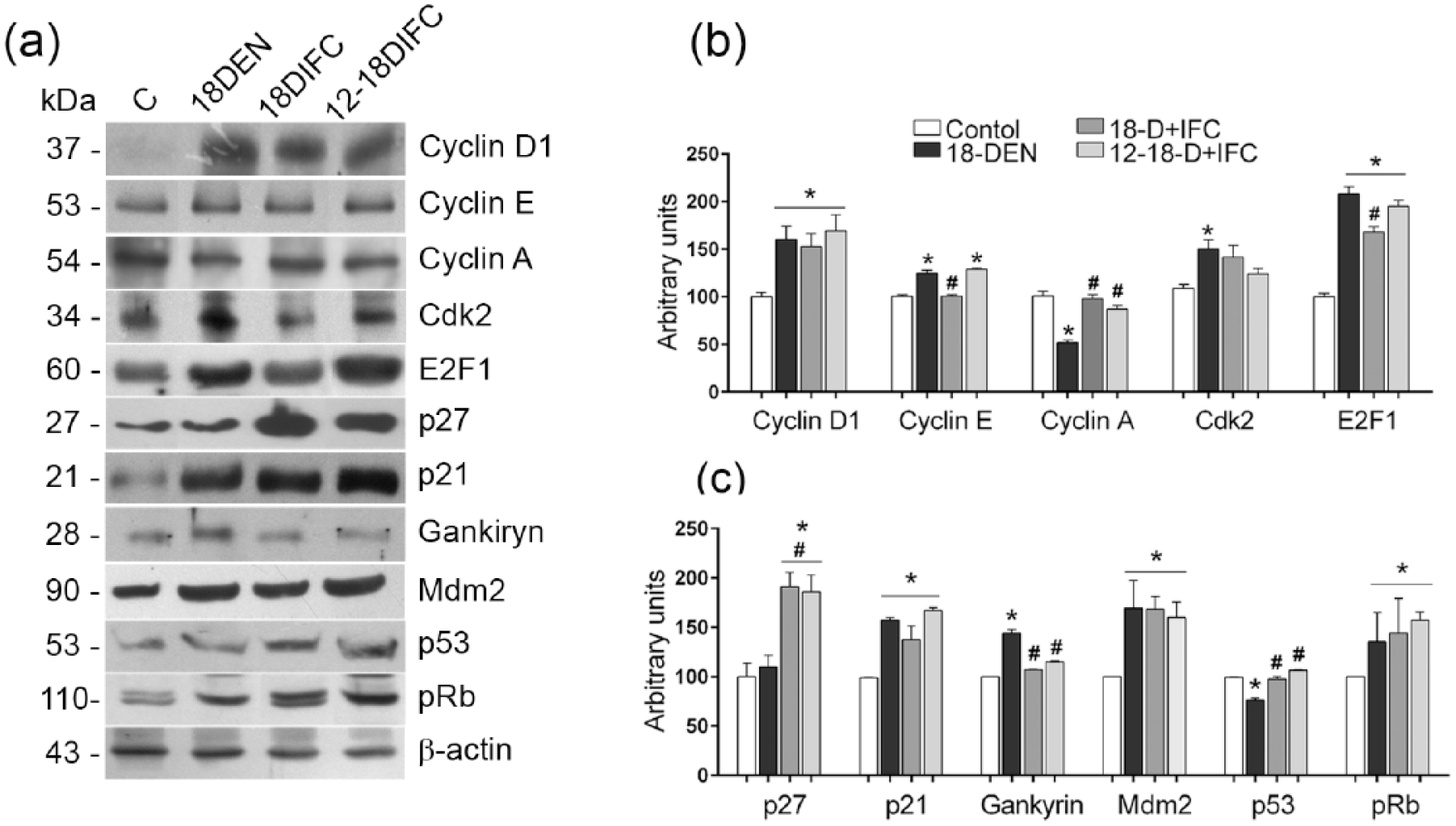
Expression of cell cycle–related proteins in liver samples of HCC groups. (a) Representative western blots of cell cycle protein expression in total liver homogenates of HCC groups. (b, c) Densitometry quantification of bands expressed as the mean ± standard error of the mean (SEM; n = 6 rats/group); values were normalized to β-actin and expressed as a percentage of the control.
Effect of IFC-305 on the expression of adenosine receptors
The adenosine receptors A2aAR and A3AR have been highly implicated in cirrhosis and liver cancer.18,19 To further understand the mechanism underlying IFC-305-induced growth suppression, we evaluated the expression of both receptors in liver through immunohistochemistry. In the control livers, staining for A2aAR was not detected. In 12-DEN livers, a strong staining signal for A2aAR was observed in the fibrotic septa surrounding hepatic nodules. In the 12-D + IFC group, reduced presence of fibrotic septa was accompanied with A2aAR expression reduction, although the level of expression was maintained in the hepatocytes (Figure 7, top left, and Table 1). In the 18-DEN group, a more strong staining for A2aAR was observed in hepatocytes from tumor foci (T) than in hepatocytes from non-tumor adjacent regions (NT) (Figure 7, middle, and Table 1). A2aAR expression was reduced in both IFC-305-treated groups compared to 18-DEN livers (Figure 7, lower part). Regarding A3AR, a low staining signal could be detected in the endothelial cells of control livers. A signal was observed in the 12-DEN groups in the fibrotic septa surrounding the hepatic nodules; these septa decreased in the IFC-305-treated livers rats. In tumor regions, A3AR expression increased in hepatocytes of the 18 DEN-treated groups and a diminution was observed in the groups treated with IFC-305.

Inmmunostaining of A2aAR and A3AR in rat livers. Immunohistochemical analysis in serial sections for adenosine receptor expression in livers of all experimental rats; a representative image of each group is presented. Tumor foci (T, outlined by arrowheads) and non-tumor adjacent regions (NT). Photographs were taken at 20× magnification. Scale bar corresponds to 100 µm.
Analysis of adenosine receptors immunostaining in the liver.

“−” No staining; “+” weakly staining; “++” moderate staining; “+++” strong staining.
Discussion
Previous studies have demonstrated that the adenosine aspartate compound IFC-305 prevents and reverses cirrhosis induced by CCl4 in rats.
6
To explore the possible hepatoprotective role of this compound in HCC, we characterized the effect of IFC-305 in a sequential model of DEN-induced cirrhosis followed by HCC.9–11 IFC-305 inhibited preneoplastic lesion development and that this protective effect could be associated with decreased cell proliferation. The capacity of IFC-305 to reverse fibrosis has been well characterized in CCl4-treated rats.
6
Although the fibrotic septa area induced by DEN was not statistically reduced upon IFC-305 administration (Supplementary Figure 1), a significant decrease in
Regarding liver carcinogenesis, the IFC-305 caused a significant reduction in the number of HCC nodules. To explore the possible mechanism of tumor reduction by IFC-305 treatment, we analyzed the overall effect of IFC-305 on proliferation and apoptosis at the HCC stage, namely, at 18 weeks of DEN-induced liver damage. Our studies indicate that apoptosis significantly increased in all DEN-treated groups compared to the control group. Concordantly, an increase in apoptosis has been related with liver damage induced by DEN. 21 The cancer chemopreventive effect of IFC-305 treatment was not explained by increased apoptosis when compared to the DEN-treated group. In contrast, both IFC-305 treatments reduced the cell proliferation increased upon DEN administration in GSTP-positive areas, especially in the 12-18-D + IFC group. We found that the IFC-305 reduced cell proliferation could be associated principally to changes in the expression of Tyms, HGF, c-Met, and the cell cycle inhibitor p27.
Tyms is a crucial enzyme in the DNA biosynthesis that has an important role in liver cancer proliferation and is an important target for certain chemotherapeutic drugs such as 5-Fluorouracil. This enzyme is a downstream target of the transcription factor E2F1. 22 DEN administration induced Tyms mRNA and E2F1 protein levels. Notably, in the 18-D + IFC group, both were down-regulated, and in the 12-18-D + IFC, decreased Tyms expression was observed. These could be associated to the IFC-305 tumor growth inhibition effect. Up-regulation of the HGF/c-Met pathway is associated with HCC development and promotes epithelial–mesenchymal transition, an important feature of tumor metastasis.23,24 In this study, IFC-305 inhibits cell proliferation possibly by inhibiting the HGF/c-Met pathway, which results in tumor reduction and which possibly prevents a metastatic effect. Loss of cell cycle control is an important event in hepatocarcinogenesis. 25 Nearly all analyzed cell cycle proteins had modest expression in control non-treated livers, which increased at 18 weeks of DEN-induced carcinogenesis. The most evident effect of IFC-305 treatment was an increase in the p27 expression level compared to that of the 18 DEN-treated groups. p27 inhibits the cell cycle by binding to the Cyclin E-Cdk2 complex during the G1-S transition. 26 An association of reduced p27 expression with several human cancers has been recently reported, and p27 has been explored as a potential target for cancer therapy. 26 Therefore, increased p27 expression induced by IFC-305 could be associated with a reduction in HCC tumors in DEN-induced liver carcinogenesis.
The p53 protein acts as a central response to cellular stress or DNA damage by inducing cell cycle arrest, apoptosis, senescence, and other tumor-suppressive actions.25,27 The pRB protein is implicated in the progression through G1 into S phase. 25 Gankyrin is an important oncoprotein that is overexpressed in human HCC and that binds Mdm2, promoting proteasomal degradation of p53 and pRb and thus increasing cancer cell proliferation.25,27 In this study, we observed that gankyrin and Mdm2 were overexpressed in the livers of 18-DEN groups, resulting in reduced p53 protein levels. Importantly, under IFC-305 treatment, gankyrin expression was reduced, possibly contributing to restoring p53 protein expression to control levels.
The role of adenosine receptors in cancer is sometimes controversial because pro- and anti-tumor effects of these receptors are well documented. Important roles for A2aAR and A3AR in liver fibrosis and cancer have been described.4,18,19 A2aAR could promote tumor growth by stimulation of angiogenesis and evasion of the immune response that destroys the tumor. 18 A3AR has been shown to be overexpressed in many tumors, including HCC, and has been proposed as a tumor marker. 28 In this study, we observed A2aAR overexpression in the livers of cirrhotic rats (12-DEN group), especially in fibrotic septa, where activated hepatic stellate cells (HSCs) are found. The expression of this receptor was decreased in the IFC-305-treated rats (12-D + IFC group). A2aAR staining also increased in hepatocytes of the tumor area of the 18-DEN group, and IFC-305 treatment lowered this expression. Thus, in general, we observed down-regulation of A2aAR expression afforded by IFC-305 treatment; the same action of this compound was observed in a model of CCl4-induced cirrhosis. 7 In HCC induced by DEN, hepatocyte necrosis triggers the release of inflammatory cytokines, such as Interleukin-1 (IL-1), which are critical mediators of HCC development.21,29 Inflammatory cytokines are important modulators of A2aR expression and function.30,31 The effect of IFC-305 on these cytokines is under investigation in our lab. Regarding A3AR, we found that it is overexpressed in fibrotic septa and in tumor areas, as previously reported.18,28 With the IFC-305 treatment, a less A3AR staining was observed as a result of tumor diminution, according to its tumor marker role. The effect of adenosine or adenosine derivatives could be mediated by activating their receptors or transporters or through their metabolism. Possibly, reduction in A2aAR expression by IFC-305 could contribute to its beneficial effects by avoiding the harmful action mediated by this receptor in fibrosis induction and tumor growth. However, we do not discard the role of IFC-305 through a metabolic action, as described in a previous work, where this compound exerts an anti-proliferative effect on HSC not mediated by any of the adenosine receptors but rather by its uptake into cells by adenosine transporters followed by intracellular conversion to adenosine monophosphate (AMP) by adenosine kinase. 32 Reduced A2aAR expression induced by IFC-305 may increase its transport and its metabolic effects inside the cell. The role of IFC-305 in favoring S-adenosyl methionine (SAM) biosynthesis, a well-known hepatoprotector, through mechanisms that include DNA methylation 33 is under investigation in our lab. However, the molecular mechanism by which IFC-305 causes down-regulation of HGF and Tyms and also increases p27 expression requires further clarification.
In summary, the chemopreventive effect of IFC-305 in a sequential model of cirrhosis-HCC induced by DEN studied here could be associated with decreased hepatocellular proliferation. This occurs through a decreased expression of the HGF/c-Met, Tyms, and A2aAR, also the overexpression of cell cycle inhibitor p27. This study suggests that this compound may have therapeutic potential as a chemopreventive agent against HCC by some different mechanisms to those previously described for its protective effect on liver fibrosis.
Footnotes
Acknowledgements
The authors greatly appreciate the technical assistance of Mariana Domínguez López, Rebeca Pérez Cabeza de Vaca, the comments and revision of Jesús Rafael Rodríguez Aguilera, the pathological advice of Alberto Aranda-Fraustro, and the assistance of our institutional histology unit.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Funding
This work was supported by grants from Consejo Nacional de Ciencia y Tecnología of Mexico (nos 82298 and 240351).
Informed consent
Informed consent was obtained from all individual participants included in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
