Abstract
Background
Several therapies are available for psoriasis, including oral, parenteral, and phototherapy. However, these treatments present various drawbacks, like mucosal ulceration, stomatitis, bioavailability issues, patient compliance challenges, and hepatic cirrhosis risk. Therefore, it is crucial to mitigate these impacts associated with psoriasis therapy. The goal is to produce a gel using a new halobetasol–phospholipid complex and test its effectiveness for treating psoriasis using an imiquimod-induced psoriasis model and ameliorate the psoriasis symptoms.
Objectives
The purpose of the research study is to formulate a topical novel halobetasol-phospholipid complex gel for evaluating its ability against psoriatic symptoms through utilization an imiquimod-induced psoriasis model.
Materials and Methods
The halobetasol–phospholipid complex was prepared from the solvent evaporation method. Phospholipid screening of different phospholipids was conducted through solubility studies, and phosphatidylcholine was selected as per its higher solubility for halobetasol. Further, the halobetasol–phosphatidylcholine complex formation and its integration into Carbopol 940 gel formulation were done, and a physical evaluation was performed. The halobetasol–phosphatidylcholine complex was confirmed through various analytical techniques. The estimation of particle size, PDI, and drug content, as well as an assessment of in vitro release and in vivo study, was conducted. The anti-psoriatic efficiency of the halobetasol–phosphatidylcholine complex gel was estimated by the Psoriasis Area Score Index (PASI), and histopathological assessment was analyzed through the imiquimod-induced psoriasis model.
Results
The new halobetasol–phosphatidylcholine complex gel has 200 ± 2 nm particle size, a PDI of 0.224 ± 0.05, and an impressive 84% drug content within the complex. Additionally, the in vitro release of the novel halobetasol–phosphatidylcholine complex demonstrates a sustained release in comparison to halobetasol gel, with 52.34% ± 02% and 90.02% ± 02% values. Finally, the PASI and histopathological evaluation show that psoriasis is getting better, while halobetasol gel shows signs of minor keratosis along with hyperkeratosis.
Conclusion
In conclusion, it is clear that halobetasol–phosphatidylcholine complex gel provides relief from psoriatic symptoms when applied topically.
Introduction
Psoriasis is an autoimmune-mediated inflammatory skin condition affecting around 2%–3% of the global population.1, 2 This condition causes red, swollen plaques and inflammation due to fast keratin growth in the skin stratum corneum, causing pruritus and discomfort on the skin surface. 3 The exact etiology remains unidentified; however, factors like genetic predisposition and environmental conditions may contribute to autoimmune illness. 4 Psoriasis is thought to be caused by immune cells such as T-cells, dendritic cells, and macrophages, which cause keratinocyte proliferation. 5 Approximately 25% of patients have committed suicide due to societal stigma associated with psoriasis symptoms. 6 Psoriasis can be treated using oral, systemic, phototherapy, and topical medicines, with topical corticosteroids being the most common for localized therapy due to their strong inflammation-reducing effects. 7 Gels are often used for topical therapy because they are biocompatible, have a network structure, and provide stability. 8
Nanotechnology offers promising treatments for dermatological conditions like psoriasis, offering advantages like lower dosing, better effectiveness, encapsulation, controlled release, higher bioavailability, and improved stratum corneum permeability.9, 10 Nanocarriers like ethosomes, niosomes, and liposomes are being used in traditional therapies to enhance their effectiveness and minimize side effects. 11 Clobetasol propionate (CP), a potent glucocorticoid, is commonly applied to the skin to alleviate inflammation and itching. 12 Clinical settings have used halobetasol, a highly potent topical corticosteroid, for psoriasis therapy for approximately 30 years (Figure S1 in the supplemental material). 13 The US Food and Drug Administration (USFDA) licensed it for psoriasis treatment in the early 1990s. 14 Halobetasol is known to reduce psoriasis inflammation, but it is poorly absorbed and has side effects. 15 Drug–phospholipid complexes effectively reduce problems with corticosteroids, such as their inability to dissolve, stay stable, be bioavailable, or be absorbed into the skin, leading to effective psoriasis treatment. 16 Phosphatidylcholine (Figure S1 in the supplemental material), a phospholipid, is crucial for the rapid breakdown of halobetasol, its penetration into the skin, and its prolonged duration. Phosphatidylcholine, an essential component in medicine, enhances solubility, penetration, stability, and release by creating a stable bilayer vesicle at the action site. 17 The imiquimod (IMQ)-induced psoriasis BALB/c model shows many skin inflammatory disorders similar to psoriatic inflammation in humans. 18 Applying IMQ, a ligand for toll-like receptors, causes psoriasis-like dermatitis similar to human psoriasis, providing a valuable test for the efficacy of new treatments. 19 Research on the gel formulation of this drug with phospholipid in a mouse model for psoriasis treatment remains undocumented. We are going to make a new gel out of halobetasol and phospholipids and then test its anti-psoriatic effects in a model rat of IMQ-induced psoriasis, both in vitro and in vivo.
Materials and Methods
Materials
Halobetasol and phosphatidylcholine were obtained from North India Pharma, Karnal, and Lipoid GmbH (Ludwigshafen, Germany). Carbopol 940, ethylenediaminetetraacetic acid (EDTA), methylparaben, glycerol, propylene glycol, propylparaben, and triethanolamine were obtained from Loba Chemie Pvt. Ltd., Chemigens, and Central Drug House Pvt. Ltd., in New Delhi. The marketed product, Imiquad cream, was obtained from Glenmark Pharmaceutical Ltd., Mumbai, India.
Methodology
In Silico Studies Docking Methodology
We used a molecular docking simulation 20 for making ligands and proteins and testing how well the drug halobetasol worked with phosphatidylcholine polymer. For the docking study, the relevant ligands and polymeric structures were taken from the PubChem database 21 and changed into *pdb format using the molecular mechanics (MM2) force field of Chem3D software, 22 Discovery Studio software, and CDOCKER, conducting the docking analysis for 30 iterations to determine the binding pose as well as docking procedure.23, 24
Screening of Phospholipids
Phospholipids like glycerophospholipid, phosphatidylcholine, phosphatidylserine, and sphingophospholipids were tested for compatibility with halobetasol. Phospholipids were added to culture flasks, liquefied at 80°C for 2 h, centrifuged for 48 h, and a 100 µL aliquot was extracted, diluted with methanol, and analyzed using a UV method at 235.5 nm wavelength. 25
Preparation of Halobetasol–Phosphatidylcholine Complex
Halobetasol was mixed with phosphatidylcholine in various ratios, refluxed for 4 h, and dried utilizing solvent for evaporation, as seen in Figure S2 in the supplemental material. The residue was treated with n-hexane, filtered, and dried for 12 h. The final complex was wrapped with parafilm and stored in a refrigerator. 26
Solubility of Halobetasol–Phosphatidylcholine Complex
A mixture of halobetasol–phosphatidylcholine was combined in n-octanol and water, subjected to three trials, followed by continuous agitation, centrifugation, and filtration. The supernatant was collected and analyzed using UV spectroscopy with proper dilution. 27
Characterization of Halobetasol–Phosphatidylcholine Complex
Particle Size.
The Malvern Zetasizer Nano ZS 90 was used to measure the particle size of halobetasol–phosphatidylcholine, with photon correlation spectroscopy performed at 25°C and 90° detection angle for standard deviation determination in a triplicate manner. 28
Polydispersity Index
PDI is a polymer science instrument that quantifies the molecular weight distribution (MWD), with values ranging from 0 to 0.5, indicating monodisperse or wide particle sizes. 29
Drug Content
The drug content was determined by dissolving 10 mg of halobetasol–phosphatidylcholine in methanol, diluting it in methanol, filtering, and analyzing it using a UV spectrophotometer at 235.5 nm. 30
Differential Scanning Calorimetry
The study investigated thermal behaviors using a DSC-60 with a refrigerated cooling system, freeze-dried samples in an aluminum pan, and heating at 20°C–250°C, analyzing differential scanning calorimetry (DSC) thermograms with 0.2 mL/min nitrogen purging rates. 31
Fourier Transform Infrared Spectroscopy (FTIR)
The medication compatibility with excipients was assessed using a Fourier transform infrared spectrophotometer, with FTIR spectra analyzed using the NaCl cell method in a range of 500–4,000 cm–¹ and compared. 32
X-ray Diffractometer
The crystalline and amorphous characteristics of samples were analyzed using a powder X-ray diffractometer at a 2θ angle, with an average step time of 0.029 s and an acquisition duration of 1 h. 33
Scanning Electron Microscopy
The samples’ shape and surface morphology were analyzed using scanning electron microscopy, and the gold coating on nanoparticles was applied, producing electrical conductivity with operational distance at 8.6–8.8 mm and 15.0 kV voltage. 34
Transmission Electron Microscopy
The surface texture formulation was examined using TEM equipment, with a small amount placed on a copper grid, stained with a 2% uranyl acetate solution, and evaluated. 35
Preparation of Halobetasol–Phosphatidylcholine Complex Gel
Carbopol 940 was dissolved in demineralized water, preservatives were added, and the Carbopol mixture was added to the solution. Triethanolamine was added for gelling properties and pH, and a blank gel (B-GE) was formed. The halobetasol–phosphatidylcholine complex was infused. 36
Visual Inspection
The gel formulation’s homogeneity, color, and lumpy appearance were physically evaluated after being placed in the respective containers. 37
pH
The gel formulation’s pH was ascertained by incorporating a glass electrode into the gel using a digital pH meter with triplicate measurements. 38
Viscosity
The viscosity was measured using a Brookfield viscometer at 25°C and 35°C, with a 0.5 mL gel sample placed in a spindle rotating at 100 rpm for three distinct observations. 39
Spreadability
Glass slides and wooden block-based measuring devices were used to evaluate spreadability, with a 20 g weight pan filled, and the time required for movable upper slide separation from fixed slides recorded. 40
In Vitro Study
A modified Franz diffusion cell was utilized for an in vitro release study of the produced gel, with cellophane membrane activation in saline phosphate buffer. A phosphate buffer solution was used as the acceptor and release medium. The investigation involved collecting 1 mL of the released medium at 48 h, replacing it with fresh medium, and quantifying it at a 235.5 nm absorption maximum. 41
Experimental Groups
Group I: The normal control group received a topical gel without a drug.
Group II: Positive control group induced psoriasis by 5% w/w IMQ cream equivalent to 62.5 mg.
Group III: B-GE-treated group without drug using Carbopol 940.
Group IV: Drug-treated group without complex (halobetasol gel formulation) (equivalent to 0.01% w/w of halobetasol).
Group V: Complex drug gel formulation treated group (halobetasol–phosphatidylcholine complex formulation) (equivalent to 0.01% w/w of halobetasol and 70% phosphatidylcholine complex).
In Vivo Study
The study used BALB/c mice aged 8–10 weeks to evaluate their anti-psoriatic activity, approved by the Institutional Animal Ethics Committee and acquired from NIPER, India (MMCP-194 IAEC-133). Animals were kept in a controlled environment with unrestricted access to food and water, and a week of acclimatization was conducted. Five groups of animals were randomly established (n = 6). All groups, except the normal control group, induced psoriasis in mice through daily topical application of 3.125 mg/2M mL of commercial IMQ cream for 10 days.
Evaluation of Body Weight
Body weight variation was measured and recorded in all mice before and after administration of IMQ with a digital weighing balance. 42
Evaluation of Scratching Behavior
Scratching behavior was assessed on the baseline and the 10th day. On each testing day, mice were individually placed in an observation chamber for 60 min to acclimate to the testing environment, and their monitoring via video to evaluate the extent of scratching behavior was done.43, 44
Evaluation of Skin Thickness
Skin thickness was observed in all mice initially, as well as after IMQ administration, with a vernier caliper. 45
Psoriasis Area Score Index (PASI) Score
PASI scores were determined by analyzing the degree of erythema, thickness, and scaling of the infected mice’s skin surface in the dorsal region. The PASI score was categorized as follows: 0: absent, 1: minimal, 2: mild, 3: moderate, or 4: severe. 46
Histopathology
The skin tissue samples were fixed in 10% formalin, dehydrated in acetone, embedded in paraffin wax, and processed into 4 µm sections using a microtome. Further, stained with hematoxylin-eosin (1:1) and subsequently analyzed histologically with a digital camera. 47
Stability Study
The durability of the halobetasol–phosphatidylcholine complex gel was evaluated by storing the samples in hermetically sealed polyethylene bags under three different temperature settings for 6 months: at 30°C ± 2°C, 65% RH ± 5% RH, and in a refrigerator at 5°C ± 3°C, by ICH recommendations. 48
Statistical Analysis
The data presented in the tables/figures are the mean ± standard deviation (SD). The statistical difference between means was analyzed using analysis of variance (ANOVA) and Tukey’s multiple comparison test. A p value of less than .05 was considered significant.
Results
In Silico Studies
Molecular Docking Analysis
Molecular docking studies of the drug halobetasol and the polymeric carrier phosphatidylcholine showed that the ligand and the phosphatidylcholine polymer were very compatible with each other based on the docking score of the ligand. Table 1 presents the docking score of halobetasol with the polymeric vehicle phosphatidylcholine, while Figure S3 in the supplemental material illustrates the binding conformation graphically.
Binding Score of the Halobetasol Against Polymeric Vehicle Phosphatidylcholine Obtained After Docking Analysis.

Screening of Phospholipids
Equilibrium solubility evaluations were performed for halobetasol. The phospholipid solubility sequence was as follows: phosphatidylcholine > sphingophospholipids > glycerophospholipids > phosphatidylserine. Among these phospholipids, phosphatidylcholine displayed the greatest solubility for halobetasol, whereas phosphatidylserine showed the least solubility.
Solubility of Halobetasol–Phosphatidylcholine Complex
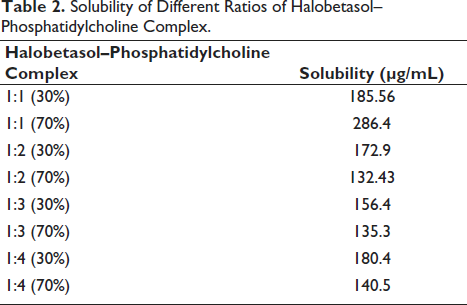
Different molar ratios and percentages of halobetasol–phosphatidylcholine complexes were prepared, in which a 1:1 molar ratio (70% of the total) was better at dissolving in methanol, which depicts that the halobetasol–phosphatidylcholine complex formation is optimal at a 1:1 (70%) molar ratio, as represented in Table 2.
Solubility of Different Ratios of Halobetasol–Phosphatidylcholine Complex.
Particle Size and Polydispersity Index
This phospholipid complex-loaded halobetasol 1:1 (70%) exhibited a particle size of 200 ± 2 nm, and polydispersity index of 0.224 ± 0.005 PDI, as shown in Figure S4 in the supplemental material.
Drug Content
The amount of halobetasol in the phosphatidylcholine complex was found to be 84% after the UV spectrophotometer analysis at 235.5 nm.
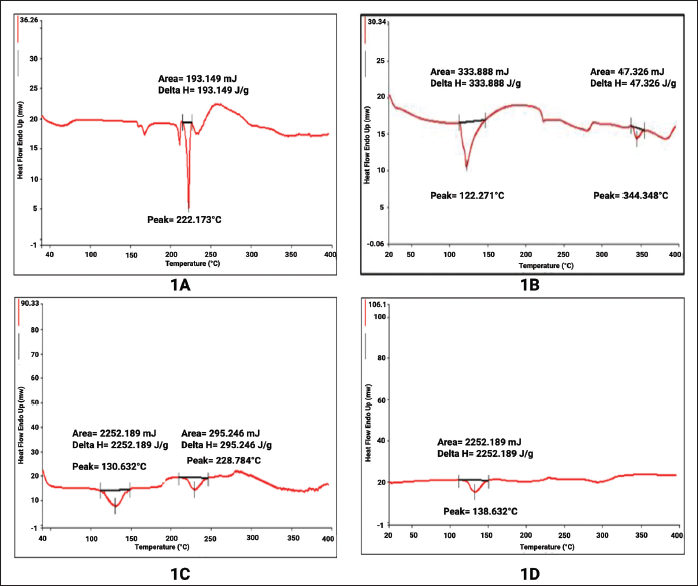
Differential Scanning Calorimetry
The DSC thermograms showed that phosphatidylcholine and halobetasol have sharp melting endotherms at 122.271°C and 222.173°C, respectively, which showed crystallinity.
The melting points of halobetasol and phosphatidylcholine were different and rounded, measuring 228.784°C and 130.632°C in a physical mixture. Figure 1 also shows the melting endotherms 138.532°C, which strongly suggests that halobetasol mixed with the melted phospholipid matrix and changed to an amorphous form, confirming complex formation.
Differential Scanning Calorimetry (DSC) Thermograph of Halobetasol, Phosphatidylcholine, Halobetasol–Phosphatidylcholine Physical Mixture, and Halobetasol–Phosphatidylcholine Complex; (A) Halobetasol, (B) Phosphatidylcholine, (C) Halobetasol–Phosphatidylcholine Physical Mixture, and (D) Halobetasol–Phosphatidylcholine Complex.
Fourier Transform Infrared Spectroscopy
The FTIR test is used to make sure that halobetasol can mix with the phospholipids that are needed for the halobetasol–phospholipid complex to form.
Figure S5 in the supplemental material displays the FTIR spectra of pure halobetasol, phosphatidylcholine, the physical combination, and the phospholipid complex. A peak at 1,406.11 cm– 1 and 3,380.18 cm– 1 was found for the pure drug halobetasol, showing its C=C (aromatic) and OH groups.
FTIR peaks for phosphatidylcholine show the PO4 peak of 569.03 cm– 1 . The physical mixture displayed peaks of PO4 (638.44 cm– 1 ) and OH groups (3,390.66 cm– 1 ).
The phospholipid complex exhibits characteristics like peak broadening and shifting like 578.20 cm for the PO4 group and 3,361.84 cm for the OH group, which were absent in the physical mixture. This shows physical interaction and stable complex formation between halobetasol and phosphatidylcholine.
X-ray Diffraction (XRD)
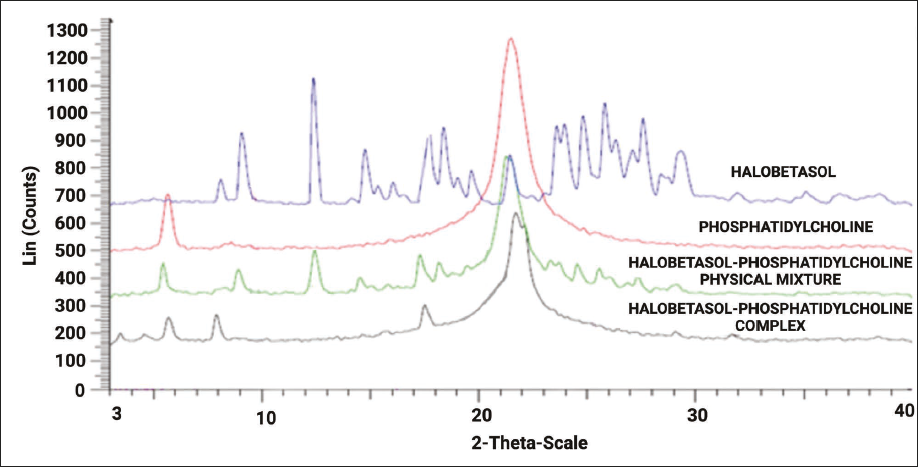
XRD analysis was utilized to determine the properties of a substance formed through precipitation, which could be either crystalline or amorphous. The XRD pattern of the halobetasol drug closely matched that of its crystalline form, with clear peaks at certain diffraction angles, such as 13.5-14, 15-19, 22-23, and 24-31. In contrast, phosphatidylcholine displays broad peaks that characterize its amorphous nature.
There are clear and broad peaks in the physical mixture, displaying the presence of halobetasol and phosphatidylcholine. XRD patterns of the phospholipid complex formulation strongly suggest the transition of crystalline to amorphous state of halobetasol drug, as illustrated in Figure 2.
Powder X-ray Diffraction (PXRD) of Halobetasol, Phosphatidylcholine, Halobetasol–Phosphatidylcholine Physical Mixture, and Halobetasol–Phosphatidylcholine Complex.
Scanning Electron Microscopy
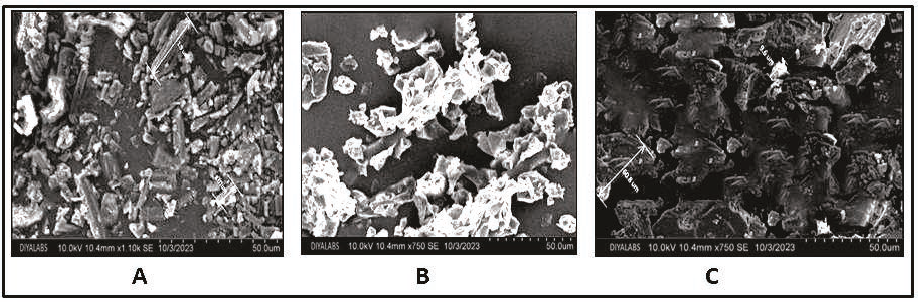
The shape and morphology of phospholipid complexes were studied, and the analyzed images revealed that the halobetasol–phosphatidylcholine complex exhibited irregular shapes with consistent sizes, highlighting a fusion of the phospholipid and the drug. In contrast, pure halobetasol exhibited a crystalline form, as illustrated in Figure 3.
Scanning Electronic Microscopy of Halobetasol (A), Phosphatidylcholine (B), and Halobetasol–Phosphatidylcholine Complex (C).
Transmission Electron Microscopy (TEM)
The TEM images of the halobetasol–phosphatidylcholine complex revealed the formation of a phospholipid complex, characterized by a uniform pattern of spherical micelles featuring an inert dark core with lighter striations, as illustrated in Figure S6 in the supplemental material.
Visual Inspection and pH of the Gel
The gel formulations were visually examined and evaluated for their physical appearance and homogeneity, including clarity, uniformity, and lumps.
The prepared gels underwent pH measurement utilizing a pH meter, with all measurements conducted in triplicate and values ranging from 6.51 to 6.85.
Viscosity
Both halobetasol gel and halobetasol–phosphatidylcholine complex gel had low viscosity (Table 3) at lower temperatures because they are sensitive to temperature. The viscosity of halobetasol gel was 1.629 ± 39 mPas, while the halobetasol–phosphatidylcholine complex gel had a viscosity of 1.758 ± 29 mPas.
Viscosity of Halobetasol Gel and Halobetasol–Phosphatidylcholine Complex Gel.

Spreadability
The spreadability values (Table 4) indicated that all the polymers used produced gels that exhibited minimal shear force during spreading.
Spreadability of Halobetasol Gel and Halobetasol–Phosphatidylcholine Complex Gel.
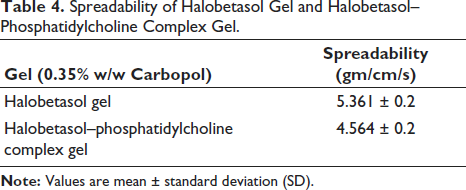
In Vitro Study
The in vitro release profile of the halobetasol gel and the halobetasol–phosphatidylcholine complex gel has been finalized, as illustrated in Figure 4. Over 48 h, the halobetasol gel released the drug quickly (90.02% ± 0.2%), while the halobetasol–phosphatidylcholine complex gel released the drug slowly (52.34% ± 0.2%). The quick release of halobetasol gel is due to better solubilization, which lets free halobetasol move around in the carboplatin-hydrated medium matrix. The halobetasol–phosphatidylcholine complex gel takes longer to break down and spread through the Carbopol matrix. This is because the phospholipid takes longer to break down and spread. This, along with the sustained release mechanism, helps keep the systemic circulation from being toxic. In addition, this consistent behavior stops halobetasol from being lost while also showing beneficial occlusive properties and making it easier to load a lot of drugs into phospholipids.
In Vitro Release Profile of Halobetasol Gel and Halobetasol–Phosphatidylcholine Complex Gel Saline Phosphate Buffer (pH 7.4).
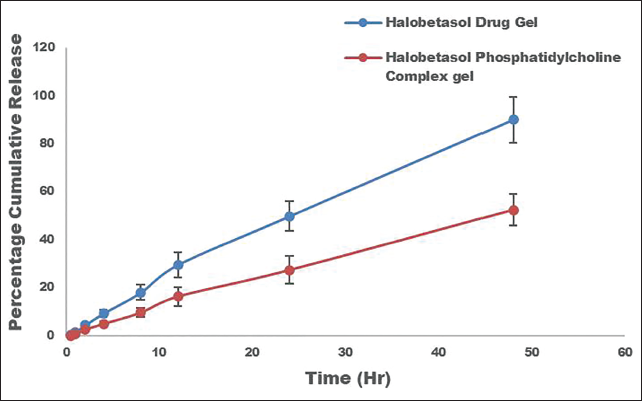
Body Weight, Skin Thickness, and Scratching Behavior
Administration of IMQ to all mice-treated groups resulted in a reduction of body weight. Besides that, the halobetasol–phosphatidylcholine complex gel reduced the body weight faster than IMQ, allowing body weights to return to normal after treatment, as shown in Figure 5. The scratching behavior was assessed in mice by analyzing scratching bouts.
Different Parameters of Treated Groups. Body Weight (A), Scratching Behavior (B), and Skin Thickness (C).
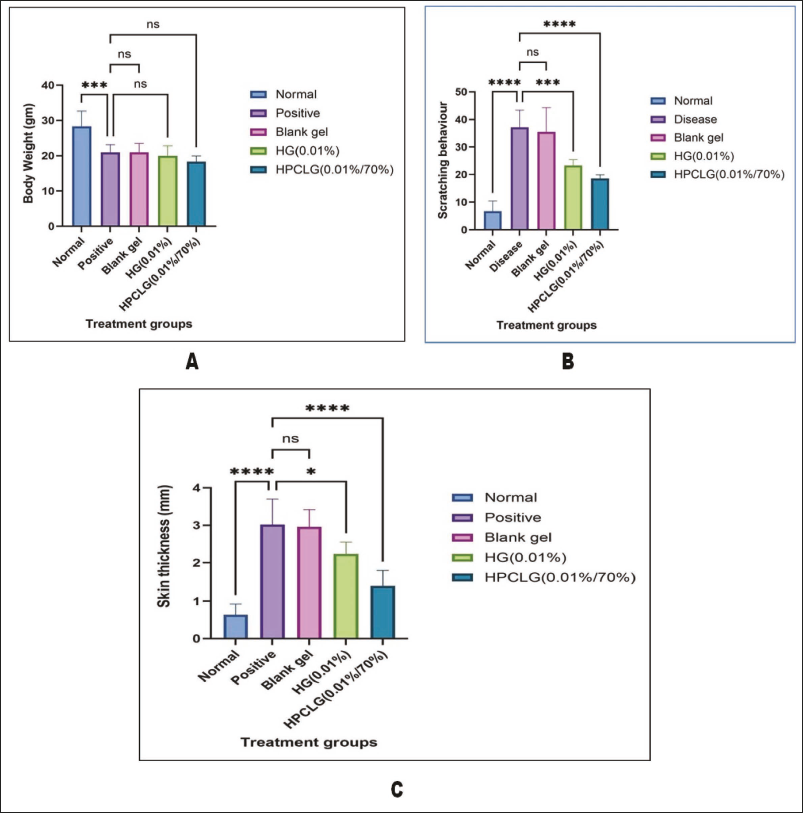
The positive group exhibited a statistically significant (p < .01) increase in the number of scratches, recorded at 37.1 ± 5.61, compared to the normal control group. By making the halobetasol–phosphatidylcholine complex gel, the number of scratching episodes dropped by almost 35%, from 37.1 ± 5.61 to 18.5 ± 1.88, compared to the positive control group.
The sick mice’s skin was measured with a vernier caliper on the 10th day of the IMQ agent. It was 3.01 ± 0.62 mm thick, which was much thicker than the skin of the control group. The halobetasol–phosphatidylcholine complex significantly improved skin thickness therapy that displayed an average thickness of 1.37 ± 0.38 mm throughout the treatment.
PASI Score
The application period of 10 days on the shaved backs of mice demonstrated various symptoms of psoriasis, including scaling, thickness, and erythema, following the application of IMQ cream. The average PASI) in the first phase was 2.3. It went down to 1.6 with the halobetasol gel, 2 with the B-GEL, and 1 with the halobetasol–phosphatidylcholine complex gel (Table 5) when the therapy was over, which displayed the halobetasol–phosphatidylcholine complex gel for reducing PASI score.
Mean Psoriasis Area Score Index (PASI) Score for Different Groups.
Histopathology Studies
Additionally, Figure 6 illustrates the various skin samples and their histopathological characteristics for the treated groups. Skin affected by psoriasis exhibited characteristics of keratosis and hyperkeratosis. Rough and red patches on the skin’s surface indicated keratosis, while the thickening of the epidermal granular layer indicated hyperkeratosis.
Images and Histopathology of Skin Samples; Group 1: Normal Control (A and F), Group 2: Positive Control (B and G), Group 3: Blank (Only Polymer) (C and H), Group 4: Halobetasol Gel (Pure Drug) (D and I), and Group 5: Halobetasol–Phosphatidylcholine Complex Loaded Gel (E and J).
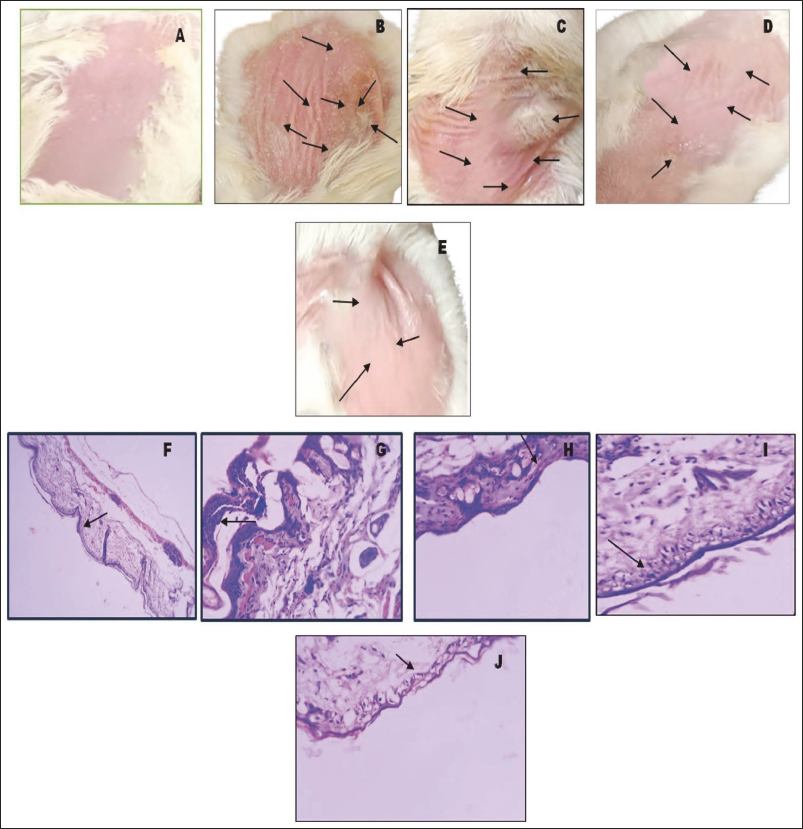
The normal control exhibited both normal and intact skin in mice. Additionally, the positive control demonstrated that disease induction resulted in keratosis and hyperkeratosis. The B-GEL demonstrated significant hyperkeratosis, primarily indicating skin sensitivities. The halobetasol gel showed both hyperkeratosis and minimal keratosis. The halobetasol–phosphatidylcholine complex gel exhibits normal skin with minimal keratosis due to its better permeation and sustained release, demonstrating its effectiveness for topical application in psoriasis.
Stability Analysis
According to the ICH guidelines, the prepared halobetasol–phosphatidylcholine complex gel stored at 30°C ± 2°C, 65% RH ± 5% RH and refrigeration at (5°C ± 3°C) for 6 months showed no significant difference in the drug content as well as physicochemical properties.
Discussion
Psoriasis is an autoimmune disease causing redness, swelling, and keratinocyte overgrowth, triggered by dendritic cells releasing cytokines, T-cells infiltrating, and the innate immune system interacting with keratinocytes. 49 Cytokines, particularly interleukin (IL)-17, stimulate keratinocytes, producing chemokines like CXCL 1/2/3, CXCL8, and CCL20, which worsen psoriatic lesions. 50 The IMQ model, which binds toll-like receptors, induces psoriatic lesions on mice’s skin surface when applied topically, similar to human psoriasis. 51 The application of IMQ was found to enhance dendritic cell proliferation, stimulate keratinocytes, and increase cytokine production.52, 53
Psoriasis, due to its increased prevalence, requires significant attention in clinical practice and investigative studies. Thus, a phospholipid complex formulation with anti-psoriatic drugs is needed, especially with the popularity of halobetasol, a strong corticosteroid known for its ability to stop cell growth and reduce inflammation.
This study explores the use of the halobetasol–phosphatidylcholine complex in gel formulation for psoriasis topical therapy, aiming to improve skin permeation and therapeutic efficacy and minimize adverse effects.
The selection and choice of phospholipids are crucial parameters for the preparation of drug–phospholipid complexes. This study chose 70% phosphatidylcholine as the most solubilizing phospholipid among various phospholipids based on solubility screening, as it demonstrated the highest solubility for halobetasol. 17 The halobetasol–phosphatidylcholine complex was incorporated into the Carbopol 940 gel formulation to enhance its properties of thickening, binding, and mucoadhesion. 54
The particle size significantly impacts therapeutic efficacy, as smaller sizes reduce irritation and enhance epidermal absorption. 55 The halobetasol–phosphatidylcholine complex, with a polydispersity index of 0.224 ± 0.05, produced uniform particles with a size of 200 ± 2 nm, confirming the phospholipid complex formulation. 56 The drug content was determined to be 84%, regarded as a good yield for drugs complexed with phosphatidylcholine. 57 Molecular docking was utilized to analyze the binding affinities of halobetasol and phosphatidylcholine, revealing a strong attraction with a CDockerScore of 27.0829. 58
The compatibility studies of halobetasol with phosphatidylcholine were conducted using various analytical methods. Differential scanning calorimetry studies confirmed halobetasol inclusion within a melted phospholipid matrix, transitioning from crystalline to amorphous form, and resembling glibenclamide phospholipid complex formation. 59 FTIR in Figure S5 in the supplemental material compares halobetasol and its complex, revealing broadening and shifting peaks in the halobetasol–phosphatidylcholine complex, indicating hydrophobic interactions, and similar alterations in the rifampicin–phospholipid complex. 32
XRD studies elucidate the crystalline nature of the samples, revealing larger peaks and molecular dispersion conversion into a phospholipid amorphous state for halobetasol–phosphatidylcholine complex formation, 60 which shows alignment with capsaicin investigation. 61 Scanning electron microscopy (SEM) images (Figure 3) of the halobetasol–phosphatidylcholine complex showed rough, amorphous structures, similar to glibenclamide–phospholipid complex formation, contrasting with crystalline non-uniform dimensions. 59
The study of the halobetasol–phosphatidylcholine complex using TEM revealed a uniform pattern of micelles with dark centers and lighter striations, displaying similarities with the erlotinib–phospholipid complex. 62
The halobetasol gel and halobetasol–phosphatidylcholine complex have a consistent texture, pH, viscosity, and spreadability on psoriatic skin, as per research studies. The study found that halobetasol–phosphatidylcholine released 52.34% ± 2% of the drug slowly over 48 h, while halobetasol gel released 90.02% ± 2% quickly, emphasizing the importance of sustained drug release. Langasco et al.’s study on corticosteroid emulsion phosphatidylcholine revealed sustained release as a key factor in its therapeutic efficacy and reduced drug toxicity. 63
This study utilized the IMQ-induced mice model to investigate the impact of halobetasol gel and halobetasol–phosphatidylcholine complex gel on PASI symptoms. Histopathological studies confirmed epidermal thickness in both therapies, with the halobetasol–phosphatidylcholine complex gel showing significantly more effects. A reported study evaluated halobetasol therapeutic efficacy by comparing various halobetasol formulations in terms of permeation, efficacy, and safety. 64 The investigation found that IMQ, a corticosteroid, caused a decrease in body weight in mice due to its toxic metabolic effects, leading to reduced food intake. The halobetasol–phosphatidylcholine complex gel significantly reduced scratching behavior by 35% compared to the positive control group due to its anti-inflammatory properties and effective permeation. 65
The positive group’s skin thickness decreased from 3.8 ± 0.62 mm during IMQ therapy to 1.8 ± 0.38 mm after applying halobetasol–phospholipid complex gel. The PASI score was evaluated on shaved back mice, which showed psoriasis symptoms upon IMQ treatment. The halobetasol–phosphatidylcholine complex gel effectively reduced psoriasis area and improved the PASI score from 2.3 to 1, indicating better permeation potential. Nourbakhsh et al.’s study highlights the significance of phosphatidylcholine in standard therapy in terms of sustained release and PASI score. 66 Gel stability studies followed ICH storage guidelines, revealing no significant changes. In vitro and in vivo assessments and histopathological evaluations determine the long-term efficacy of halobetasol–phosphatidylcholine complex gel in managing psoriasis.
Conclusion
The investigation assessed the possible application of halobetasol–phosphatidylcholine in the treatment of psoriasis. The halobetasol–phosphatidylcholine complex had important physicochemical properties that made it safe for use on the skin, as found through formulation, characterization, and evaluation. DSC as well as TEM tests confirm the complex formation. The new halobetasol–phosphatidylcholine complex gel has 200 ± 2 nm particles, a PDI of 0.224 ± 0.05, and 84% of the gel is drug. The new halobetasol–phosphatidylcholine complex releases over a longer period than halobetasol gel, with results of 52.34% ± 02% and 90.02% ± 02%, respectively. After extensive testing and evaluation in living organisms, it was determined that the gel containing the halobetasol–phosphatidylcholine complex was more effective in treating psoriasis. The PASI index and histopathological evaluation indicate improvement in psoriasis, while halobetasol gel reveals signs of hyperkeratosis and minimal keratosis. The article provides a detailed review of the application of halobetasol–phosphatidylcholine complex gel topically for treating psoriasis.
Footnotes
Abbreviations
ANOVA: Analysis of variance; IMQ: Imiquimod; MM2: Molecular mechanics; PASI: Psoriasis Area Score Index; USFDA: US Food and Drug Administration.
Acknowledgments
The authors would like to acknowledge all authors for idea, execution, and implementation. They also appreciate management for giving the opportunity to writing this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study (MMCP/IAEC/133) was approved by the Institutional Animal Ethics Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
