Abstract
Introduction
TYK2 inhibitors and traditional natural drugs as promising drugs for psoriasis therapy are receiving increasing attention. They both affect different molecules of JAK/STAT pathway, but it is currently unclear whether their combination will enhance the effect on psoriasis. In this study, we used imiquimod (IMQ)-induced psoriasis mouse model to investigate the therapeutic effects of the combined administration of deucravacitinib (TYK2 inhibitor) and shikonin.
Methods
Aldara cream containing 5% IMQ was used to topically treat the dorsal skin of each mouse for a total of six consecutive days to induce psoriasis. The psoriasis area and severity index (PASI) scores were recorded every day. On the 7th day, skin tissues were taken for histopathological examination and the content of cytokines in skin were evaluated. The frequency of immune cells in peripheral blood, spleen and skin were detected through flow cytometry.
Results
Compared to the vehicle control group, the psoriasis symptoms and immune disorder improved significantly in the combination therapy group and deucravacitinib treatment group on the 7th day, and the expressions of p-STAT3 and Ki67 in skin were reduced as well. Moreover, the combined treatment of deucravacitinib and shikonin for psoriasis was superior to the monotherapy group, especially in inhibiting abnormal capillaries proliferation, reducing immune cells infiltration and decreasing the concentration of IL-12p70 in skin.
Conclusion
The combination of deucravacitinib and shikonin is a promising clinical application.
Introduction
As a chronic inflammatory skin disease, psoriasis has diverse clinical manifestations, often accompanied by comorbidities such as cardiovascular disease and inflammatory bowel disease, which greatly affects the physical and mental health of patients. As reported, the post-treatment improvement rate of psoriatic patients is not ideal and 36% of patients had no significant improvement. 1 Therefore, it is urgent to explore more effective treatment methods to improve the life quality of psoriasis patients.
Psoriatic skin lesions contain a large amount of lymphocytes infiltration, including dendritic cells (DC), T cells and neutrophils. It has been found that IL-17A level and γ/δ T cells frequency in lesion skin are significantly decreased after anti-psoriasis treatment and the upregulation of CD11c+ DC can also be reversed. 2 DC, as an antigen-presenting cell, secretes pro-inflammatory cytokines such as TNF- α, IL-6, IL-12, and IL-23 involving in innate and adaptive immune signaling cascades. IL-23/IL-6 derived from DC plays an indispensable role in the polarization of Th17 cells, which play a fundamental role in the pathogenesis of psoriasis in humans and mice. Alzahrani et al found that inhibiting the function of DC can alleviate psoriasis. 3 In the skin of IMQ-induced psoriasis mice the infiltration of neutrophils, Th17, and Th1 cells are related to the level of phosphorylated STAT3 (p-STAT3) as well. 4 Therefore, it is thought that the IL-23/IL-17/STAT3 axis is closely related to the psoriasis pathogenesis.5,6 It is extensively known that IL-23 is considered a “master regulator” of Th17 cell development and IL-17 production, 7 which accelerates the differentiation of CD4+ and γδ T cells,8,9 and stimulates chemokine production by phosphorylating STAT3, leading to inflammation and tissue damage. 10 In psoriasis, p-STAT3modulates the corresponding genes in nucleus to trigger abnormal proliferation of keratinocytes.11,12 Additionally, the inflammatory environment induces and activates pro-angiogenic factors to exert a promoting effect on vascular proliferation in skin.
Due to the Janus kinase-signal transducer and activator of transcription (JAK/STAT) pathway tightly correlates to the psoriasis pathogenesis, 13 JAK inhibitors have been developed and applied in psoriasis therapy. Traditional JAK inhibitors competitive bind to the JH1 (catalytic kinase) domain. Due to the high similarity of the JH1 domain in the JAK family, these inhibitors not only affect the specificity of drug therapy, but may also produce off-target effects, reducing treatment selectivity. In 2022, deucravacitinib developed by BMS has been approved for the treatment of adult patients with moderate to severe plaque psoriasis. 14 Deucravacitinib selectively binds to the JH2 (regulatory kinase) domain of TYK2, causing a conformational change in the JH1 domain that prevents it binding to substrates, leading to TYK2 inactivation and blocking the TYK2/STAT pathway, thereby inhibiting the differentiation of Th17 and Th1, decreasing the secretion of IL-23, IL-12, IL-17 and TNF- α, and preventing psoriasis skin lesions. 15 In the two global large phase III double-blind trials of POETYK PSO-1 and PSO-2, both of the two main efficacy endpoints of deucravacitinib in the clinic trials, PASI-75 (75% reduction in psoriasis area and severity index) and sPGA0/1 (complete/almost complete clearance of skin symptoms according to static physician assessment), are superior to them of placebo or the control drug apremilast. 16 TYK2 inhibitors are becoming an encouraging class of drugs in psoriasis. Therefore, intensive research on the selectivity, efficacy, and safety of TYK2 inhibitors is expected to benefit a larger population of psoriasis patients.
With the development of molecular biology, the research on the effects of traditional natural medicines in psoriasis therapy has been increasing in recent years. Especially the research on Traditional Chinese medicine (TCM) term from different levels, such as immunity and meridian circulation, has strengthened the understanding of the treatment of psoriasis. At present, TCM holds great promise in the treatment of psoriasis with its advantages of syndrome differentiation and treatment, flexible medication, and few side effects. 17 Shikonin is an active ingredient of the commonly used TCM lithospermi radix (LR), which has the effects of cooling blood, activating blood, clearing heat, and detoxifying. 18 Wang et al reviewed the efficacy and safety of LR and shikonin in psoriasis treatment and showed that the TCM prescription that incorporates LR as the sovereign herb has better efficacy in the treatment of psoriasis. Shikonin effectively inhibited the proliferation of human immortalized epidermal cells and promoted the apoptosis by downregulating the protein levels of STAT1, STAT3, and JAK2. 19 The combination therapy of shikonin can significantly improve the curative effect. 20 Shikonin, as a natural antioxidant, 21 has good anti-inflammatory effects. 22 Compared to deucravacitinib, shikonin not only regulates more immune related signaling pathways, such as p38 MAPK 23 and NF-κB pathway, 24 but also has antioxidant and anti-inflammatory effects. Due to the multiple targets and mild effects of shikonin, its auxiliary effect on long-term treatment of psoriasis deserves attention. Therefore, we believe that for psoriasis treatment, integrating skikonin and TYK2 inhibitors may draw upon strong points of each other. Considering that there are no literature reporting the results on the combination of deucravacitinib and shikonin on psoriasis therapy in clinical or preclinical research so far, in this study we used mice model to investigate whether the combination of shikonin and deucravacitinib has a synergistic effect on psoriasis therapy and identify the main targets of their combined effects.
Materials and methods
Materials
Animals: Female BALB/C mice aged 8 weeks were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd., Beijing, China. The animals were housed under controlled conditions with ad libitum access to food and water. The environment was maintained at a temperature of 20°C–26°C with a relative humidity of 40%-70% and a 12-h light-dark cycle. All animal experiments were conducted in accordance with the guidelines and approved by the Ethics Committee of Crown Bioscience, Inc.
Main Reagents: Imiquimod (IMQ) (Aldara, GWA051 A), deucravacitinib (MCE, 59262), shikonin (MCE, 256116), Purified Rat Anti-Mouse CD16/CD32 (Mouse BD Fc Block™) (BD Biosciences, 553141), eBioscience™ IC Fixation Buffer (Invitrogen, 00-8222-49), p-STAT3 (Cell Signaling, 9145), CD31 (Cell Signaling, 77699), Ki67 (Abcam, ab16667), V-PLEX Proinflammatory Panel 1 Mouse Kit (MSD, K15048D), U-PLEX TH17 Combo 3 (ms) (MSD, K15354 K). The antibodies used for flow cytometry analysis are listed in Table S1.
Methods
Animal treatment
According to Resource Equation Approach, 25 we doubled the maximum number of the total animals, from 25 to 50, as our experiment required collecting skin tissues to evaluate cytokine levels, lymphocytes infiltration and histopathological damage. 50 mice were randomized into five groups: blank control, vehicle control (IMQ + vehicle), deucravacitinib (IMQ + Deucravacitinib), shikonin (IMQ + Shikonin), and deucravacitinib and shikonin co-administration (IMQ + Deucravacitinib + Shikonin) groups, with 10 mice in each group. On the first day, the backs of the mice were shaved under anesthesia with 3%–5% isoflurane. Subsequently, 62.5 mg of Aldara cream containing 5% IMQ 26 was uniformly applied to the exposed dorsal skin of each mouse, except for blank control mice, for six consecutive days, whereas Vaseline was uniformly applied to the exposed back skin of the mice in the blank group.
Treatment Schedule of each group of mice.

Pathological histological analysis
Gross pathology of lesioned skin
The thickness of the mouse dorsal skin was measured daily with vernier calipers, and all dorsal lesions (skin redness and scales) of the mice were assessed with the Psoriasis Area and Severity Index (PASI) scoring system: both erythema and scales were scored on a scale of 0–4 (0, no; 1, mild; 2, moderate; 3, significant; and 4, severe). Mean skin thickness was calculated from two measurements at a distance from each other, and the percentage of changes in skin thickness was calculated and scored accordingly. The final score was the sum of the scores for erythema, scales, and skin thickness. 29
Hematoxylin and eosin (HE) staining and immunohistochemistry staining
The lesioned skin tissues from the back of mice were subjected to fixing with 10% formaldehyde solutions for 18–24 h, dehydration, and paraffin-embedding, and then sectioned for HE staining and immunohistochemistry staining of p-STAT3 (primary antibody 1:200), CD31 (primary antibody 1:100) and Ki67 (primary antibody 1:400). 30 Immunohistochemistry staining was conducted with a Leica Bond RX immunohistochemistry and in-situ hybridization staining machine, and the sections were scanned with a digital pathology slide scanner. The entire section was analyzed with Halo digital pathology image analysis software.
Baker scoring criteria.

Histopathology score = Keratinocyte score + Epidermis score + Dermis score.
Data analysis was conducted for CD31 staining images. Specifically, the number of CD31 positive cells was counted in the dermal region of the entire section and then divided by the area of the dermal region to calculate the density of CD31 positive cells for statistical analysis.
Data analysis was performed for images of Phospho-STAT3 and Ki67 staining according to the following procedures: necrotic and interstitial regions were excluded from the whole section, and fat, sebaceous glands, and hair follicles were removed. Then, the number of positive cells and the total number of cells in the dermis were counted. Cells in each target site were scored at 4 levels: 0, negative; 1, weakly positive; 2, moderately positive; and 3, strongly positive. The H-Score was calculated with the percentage of cells with different intensities to all cells.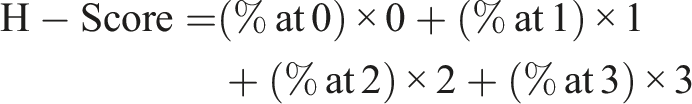
Flow cytometry analysis
The skin lesion tissues were obtained from the back of mice, clipped, and placed in 3.5 mL of Dulbecco’s Modified Eagle Medium containing 1 mg/mL collagenase P and 100 μg/mL DNase I, and digested at 37°C for 1.5 h. Single cells were acquired, sealed with Mouse BD Fc Block™ at 4°C for 5 min, and surface-labeled according to the staining protocol in Table S2. 32 After 30-min culture at 4°C, the cells were washed with phosphate-buffered saline (PBS) and fixed with IC Fixation Buffer (00-8222-49; Invitrogen, Carlsbad, California, USA) at room temperature for 30 min. After washing with PBS, the cells were detected with a flow cytometer, and the results of flow cytometry were analyzed with Kaluza software.
Spleens were attained from all mice, weighed and photographed, and then placed individually into gentleMACS C tubes (130-096-334; Miltenyi Biotec, Bergisch Gladbach, Germany) containing 5 mL of Roswell Park Memorial Institute 1640 medium. Tissues were dissociated using the gentleMACS™ Octo Dissociator with Heaters (130 -096-427; Miltenyi Biotec) to obtain single cells, which were sealed with Mouse BD Fc Block™ (553141; BD Biosciences, Franklin Lakes, NJ, USA) at 4°C and 3 million cells/100 μL for 5 min and surface-labeled based on the staining protocol in Table S2. 33 The cells were cultured at 4°C for 30 min and washed with PBS, followed by 30 min of fixing with IC Fixation Buffer (00-8222-49; Invitrogen) at room temperature. After PBS washing, the cells were detected with a flow cytometer, 34 and the results of flow cytometry were analyzed with Kaluza software. The gating strategy for analysis is shown in the supplementary file (Figure S1-S3).
Whole blood (150 μL) was added to a flow cytometric tube and sealed with Mouse BD Fc Block™ (553141; BD Biosciences) at 4°C for 5 min, followed by surface-labeling according to the staining protocol in Table S2. After 30-min culture at 4°C, the cells were lysed with Red Blood Cell Lysing Buffer (64010-00-100; Bio-gems Westlake Village, California, USA), washed with PBS, and fixed with IC Fixation Buffer (00-8222-49; Invitrogen) at room temperature for 30 min. After PBS washing, the cells were detected by a flow cytometer, and the results of flow cytometry were analyzed with Kaluza software.
Cytokine measurement
Skin tissues were pulverized with a freezing grinder, lysed, cultured at 4°C for 30 min, and then centrifuged at 12,000 rpm for 10 min, followed by the collection of the supernatant.
MSD plates (MSD Company, Rahway, NJ, USA) coated with the capture antibody were prepared and added with the standard and the skin supernatant for 2 h of incubation at room temperature. After washing, the samples were incubated with the detection antibody mixture labeled with SULFO-TAG at room temperature for 2 h. After washing, the plates were added with 150 μL of plate reading solutions and read with the MSD instrument. Specific procedures and preparation of related reagents were performed in strict accordance with the instructions of the V-PLEX Proinflammatory Panel 1 Mouse Kit (K15048D; MSD Company) and U-PLEX TH17 Combo 3 (ms) Kit (K15354 K; MSD Company). The obtained data were analyzed with the MSD Discovery Workbench software on the instrument.
Statistical analysis
Data were analyzed with GraphPad Prism 9 software and expressed as mean ± SEM. One-way analysis of variance (ANOVA) was used for comparisons among three or more groups, and the
Results
Co-administration of deucravacitinib and shikonin alleviates skin lesions in psoriatic mice
As the results shown in Figure 1, the skin of IMQ-treated mice showed erythema and scales since the second day, accompanied with skin thickening. The erythema and scales in the vehicle control group were progressively obvious over time, while no obvious abnormalities were observed in the blank group. Erythema and scale scores (from Day 2), as well as skin thickness scores (from Day 3), were significantly higher in IMQ-treated groups than in the blank control group ( Co-administration of deucravacitinib and shikonin alleviates skin lesions in psoriatic mice. Mice were randomly divided into Blank, IMQ + vehicle, IMQ + Deucravacitinib (30 mg/kg), IMQ + Shikonin (10 mg/kg), and IMQ + Deucravacitinib (30 mg/kg) + Shikonin (10 mg/kg) groups, with 10 mice in each group. On the first day, the backs of the mice were shaved under anesthesia with 3%–5% isoflurane. Subsequently, 62.5 mg of Aldara cream containing 5% IMQ was uniformly applied to the exposed dorsal skin of each mouse for six consecutive days, except for blank control mice, which were given Vaseline. Each group of mice was anesthetized with 3%–5% isoflurane on Day 3, 5, and 7, and their back skin photos were taken (A). The thickness of mice back skins were measured by a vernier caliper every day and the skin erythema (B), scale score (C), skin thickness score (D) and the PASI score (E) were observed and evaluated. The data is expressed as mean ± SEM, 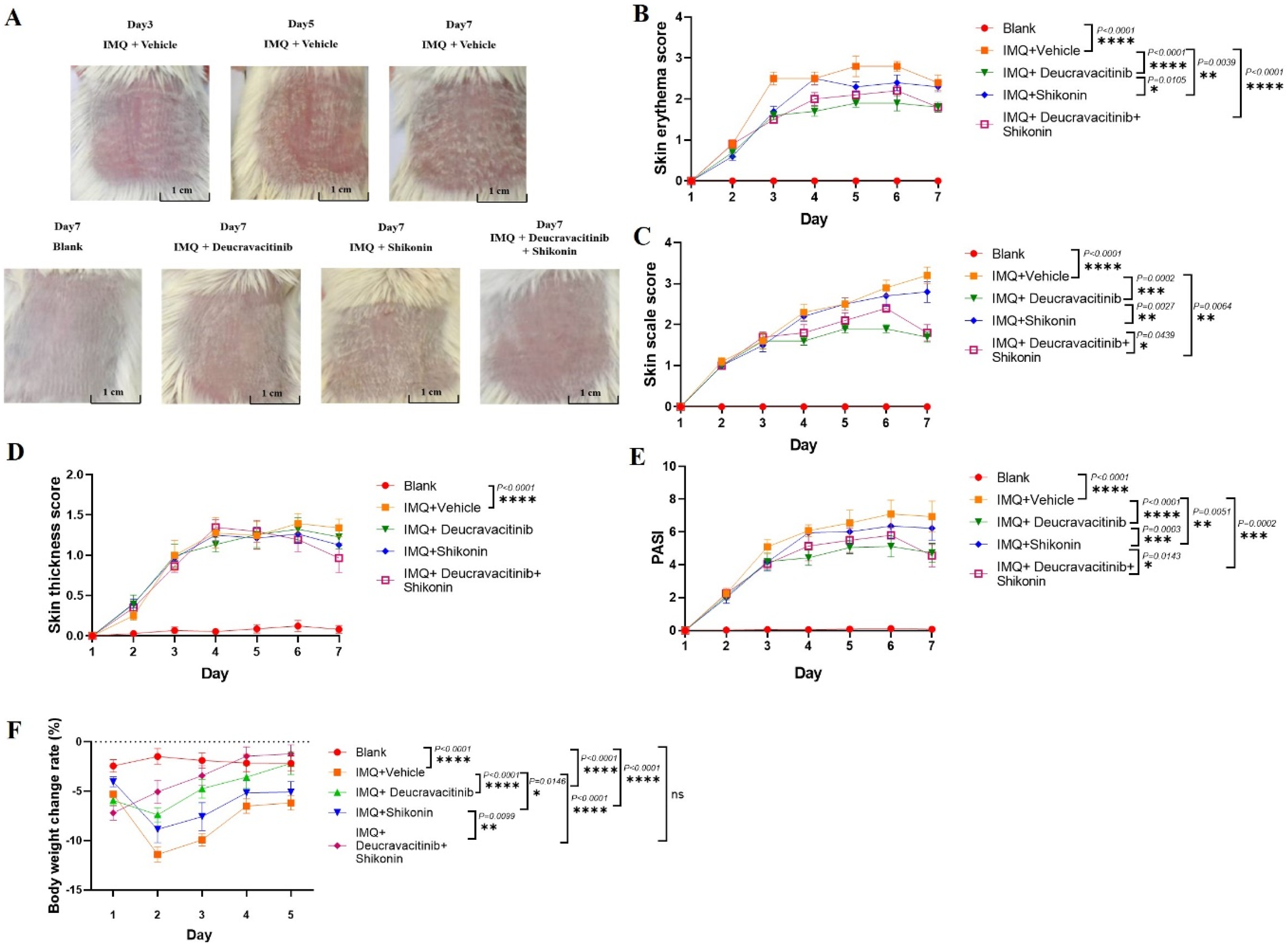
In addition, during daily observations, it was noted that some mice in the deucravacitinib alone treatment group exhibited symptoms such as loss of appetite. According to the rate of change in mice body weight during the experimental period, all mice showed a decrease in weight. However, compared to the blank group, the decrease in weight change rate in the deucravacitinib and shikonin co-administration group was moderate compared to the monotherapy groups (F(2.382,21.44) = 9.381,
Co-administration of deucravacitinib and shikonin improves skin histopathology, CD31, Ki67 and p-STAT3 expression in psoriatic mice
Mice in the blank control group had thin dorsal skin, with clear layers and no obvious infiltration of granulocytes or mononuclear cells. The mice epidermis of the dorsal skin in the administration groups was observed significantly thickened, with different degrees of hyperkeratosis, microabscess in the cuticle, acanthosis in epidermis, and rete ridges elongation or undulation, accompanied by inflammatory cell infiltration, capillaries dilation and dermal papillae hyperemia (Figure 2A). The average vertical thickness of the epithelium showed that IMQ induced the skin thickening. Compared to the vehicle control group, the skin thickness significantly reduced in the deucravacitinib, and the combination therapy groups (Figure 2B). The trend of Baker score results was consistent with that of epithelial thickness (Figure 2C) (Figure 2B: F(4,20) = 41.07, Co-administration of deucravacitinib and shikonin improves skin histopathology in psoriatic mice. The skin tissue on the back of mice was fixed, dehydrated, paraffin embedded, and sliced for HE staining. A shows HE staining images (10×) of Blank, IMQ + vehicle, IMQ + Deucravacitinib, IMQ + Shikonin, and IMQ + Deucravacitinib + Shikonin groups. Black arrow: the munro abscess; Red arrow: the hyperkeratosis; Green arrow: the acanthosis; Black triangle: the lengthening and clubbing of rete ridges; Black star: the inflammatory cell infiltration; Yellow arrow: the angiotelectasis/papillary congestion. B is the measurement of epithelial thickness by randomly selecting 10 intact and flat areas of the skin from the entire slice by Halo digital pathological image analysis software. C is the HE scores for the entire slice according to the Baker scoring criteria (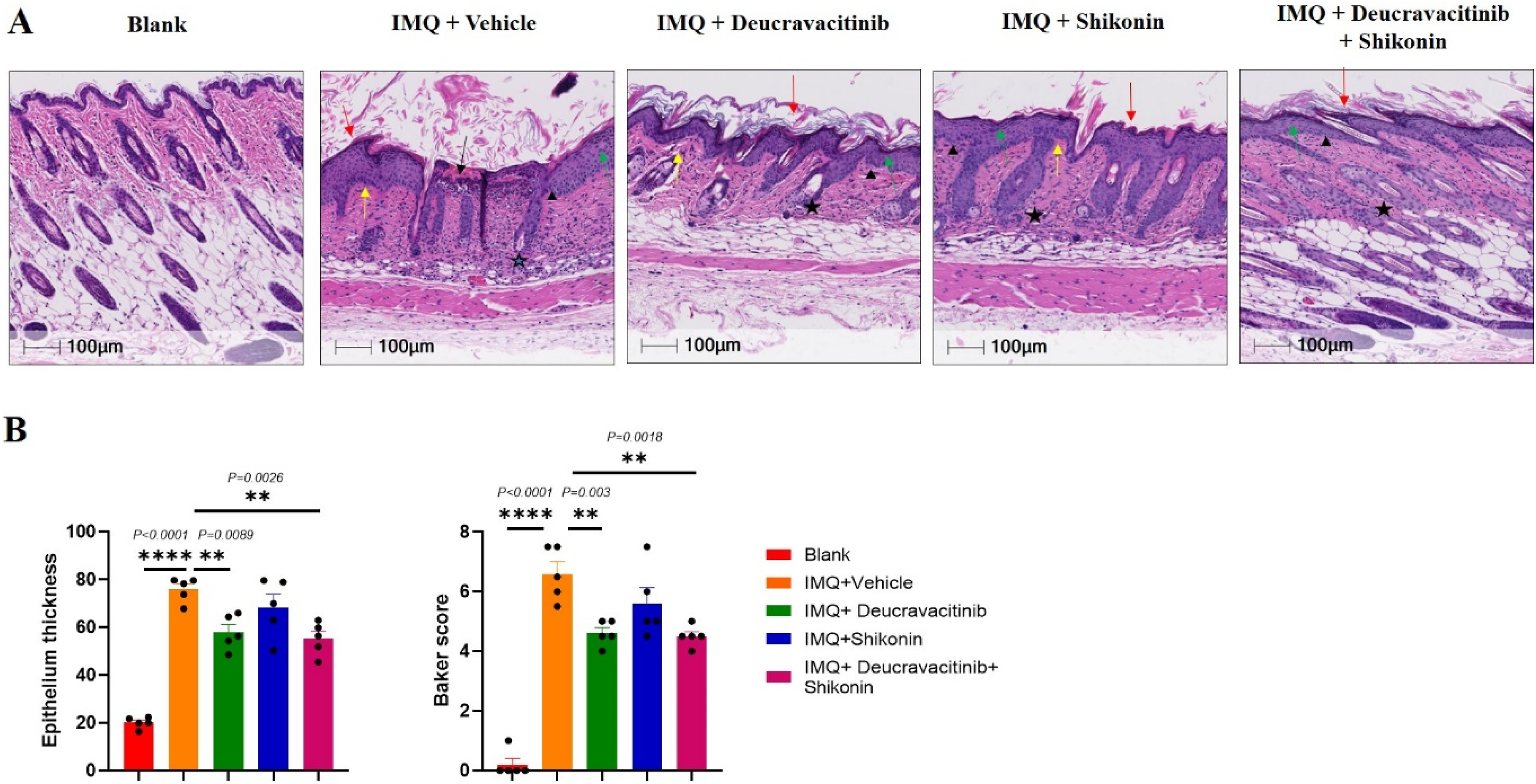
With regard to immunohistochemistry results, the density of CD31 and Ki67 positive cells in the dermis of IMQ-treated groups was higher than that of the blank group. But the increasing trends of CD31 and Ki67 expressions were inhibited after the treatment of deucravacitinib or the co-administration of deucravacitinib and shikonin. Moreover, the inhibitory effects of these drug treatment group on cell proliferation was more significant than that of the shikonin group (Figure 3B: F(4,20) = 40.89, Co-administration of deucravacitinib and shikonin improves CD31, Ki67 and p-STAT3 expression in psoriatic mice. (A), (C) and (E show CD31, Ki67 and p-STAT3 staining images (20×) of Blank, IM)Q + Vehicle, IMQ + Deucravacitinib, IMQ + Shikonin, and IMQ + Deucravacitinib + Shikonin groups in sequence. The density of CD31 positive cells was analyzed by Halo digital pathological image analysis software (B). The number of Ki67 (D) or p-STAT3 (F) positive cells in the dermis were calculated. The H-Score was calculated with the percentage of positive cells with different intensities to all cells, while the necrotic and interstitial regions, fat, sebaceous glands, hair follicles were excluded from the whole section. The data is expressed as mean ± SEM, 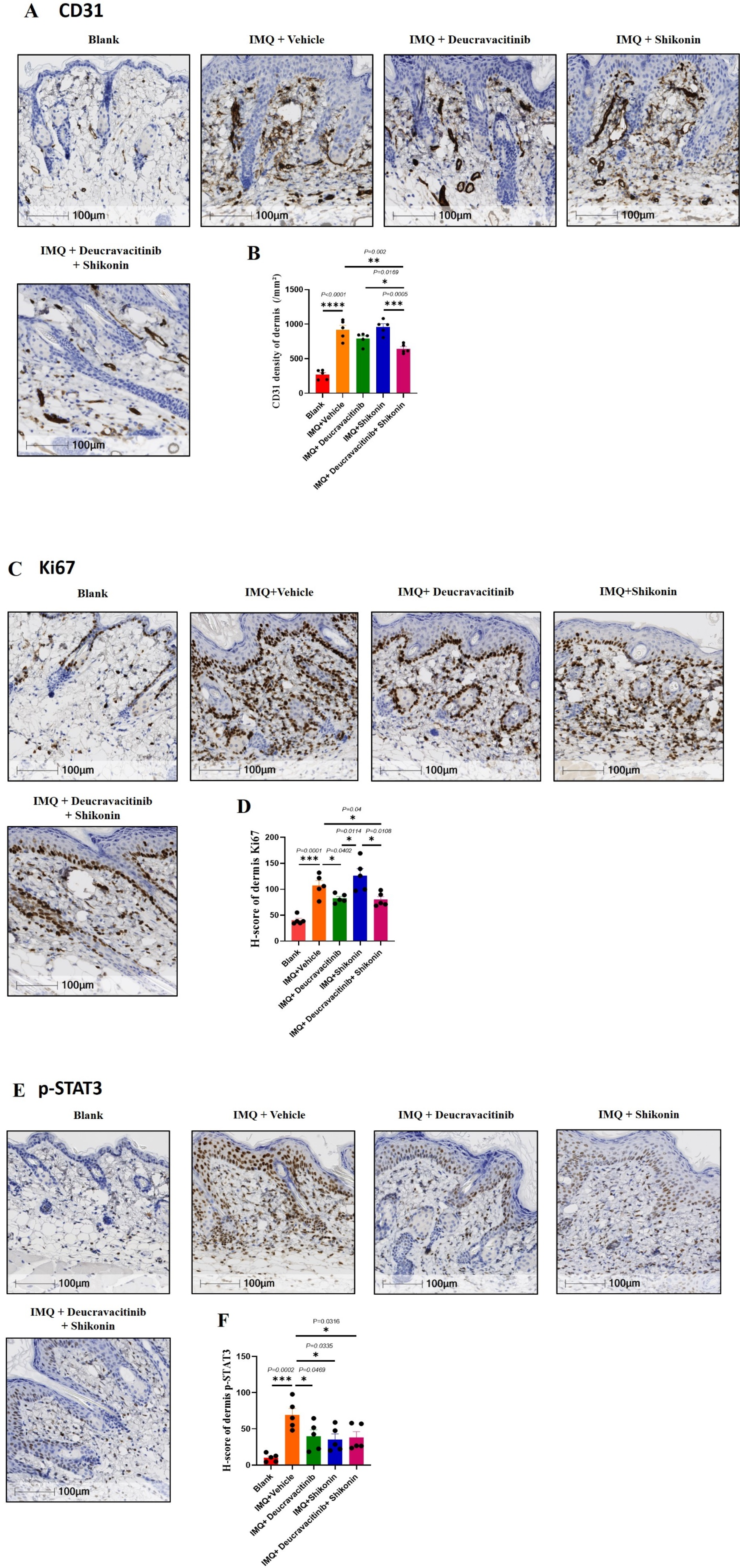
After IMQ induction, there was a significant increase in dermal p-STAT3 expression. However, after treatment with deucravacitinib, shikonin, or the co-administration of deucravacitinib and shikonin, the enhanced p-STAT3 expression was partially reversed (Figure 3F F(4,20) = 7.835,
These data suggested that both deucravacitinib and shikonin can alleviate IMQ-induced psoriatic symptoms through reducing the dermal levels of Ki67 and p-STAT3. The combiantion therapy of the two drugs has a good effect on blocking the psoriatic vascular proliferation in skin, and the combined application has the similar effects on the psoriatic skin symptoms as deucravacitinib.
Co-administration of deucravacitinib and shikonin improves spleen weight and cytokines expression in the skin of psoriasis mice
As shown in Figure 4A and B, IMQ induction substantially increased the spleen weight of mice. The spleen weight of IMQ-induced mice was diminished after treatment with deucravacitinib or co-administration with deucravacitinib and shikonin versus that of the vehicle control group. The spleen weight in the deucravacitinib and shikonin co-administration group was restored to the similar level of the blank group. Additionally, there was no significant changes in spleen weight between the shikonin-treated group and the vehicle control group (Figure 4A and B: F(4,20) = 34.36, Co-administration of deucravacitinib and shikonin improves spleen size and cytokines expression in the skin of psoriasis mice. (A) is a representative photo of the mice spleen of each group. (B) is the spleen weight (
Cytokine expression in the skin of mice is presented in Figure 4C. IL-12p70, tumor necrosis factor-alpha (TNF-α), IL-1β, IL-6, and IL-17A were significantly upregulated in the skin of IMQ-treated mice Compared to IMQ-induced mice, TNF-α, IL-1β, IL-6 and IL-17A levels were decreased in the deucravacitinib alone group and the combination group. Notably, IL-12p70 and IL-17A levels were greatly lower in the deucravacitinib and shikonin co-administration group than in deucravacitinib group (IL-12p70: F(4,20) = 7.936,
Co-administration of deucravacitinib and shikonin inhibits the frequency of γ/δ T cells, skin neutrophils and splenic DCs in psoriatic mice
Various immune cell subpopulations in peripheral blood, spleen, and skin were examined with flow cytometry, and the gating strategy of flow cytometry was shown in Figure S1-S3. Statistical results demonstrated that γδ T cell infiltration was markedly elevated in mice skin, peripheral blood, and spleen after IMQ induction ( Co-administration of deucravacitinib and shikonin inhibits the infiltration of γ/δ T cells, skin neutrophils and splenic DCs in psoriatic mice. Immune cells were isolated from mice skin, spleen and peripheral blood. The frequencies of γ/δ T cells (A), (B), neutrophils (C), (D) and DC (E), (F) were analyzed by flow cytometry. The data is shown as mean ± SEM, 
IMQ induction significantly enhanced neutrophil infiltration in mice skin and peripheral blood (
Compared to the blank group, dendritic cell (DC) infiltration in the skin was not markedly altered in the vehicle control group but in peripheral blood and spleen the DC infiltration incresed significantly (F(4,20) = 2.874,
The analysis of plasmacytoid (pDC) and classical DC (cDC1 and cDC2) in peripheral blood and spleen showed that the frequency of pDC in peripheral blood was significantly higher in the blank group than in IMQ-treated groups ( Co-administration of deucravacitinib and shikonin inhibits the infiltration of pDC and cDC in psoriatic mice. Immune cells were isolated from mice spleen and whole blood of mice. The pDC (A), (B) and cDC (C), (D) were analyzed by flow cytometry. The data is shown as mean ± SEM, 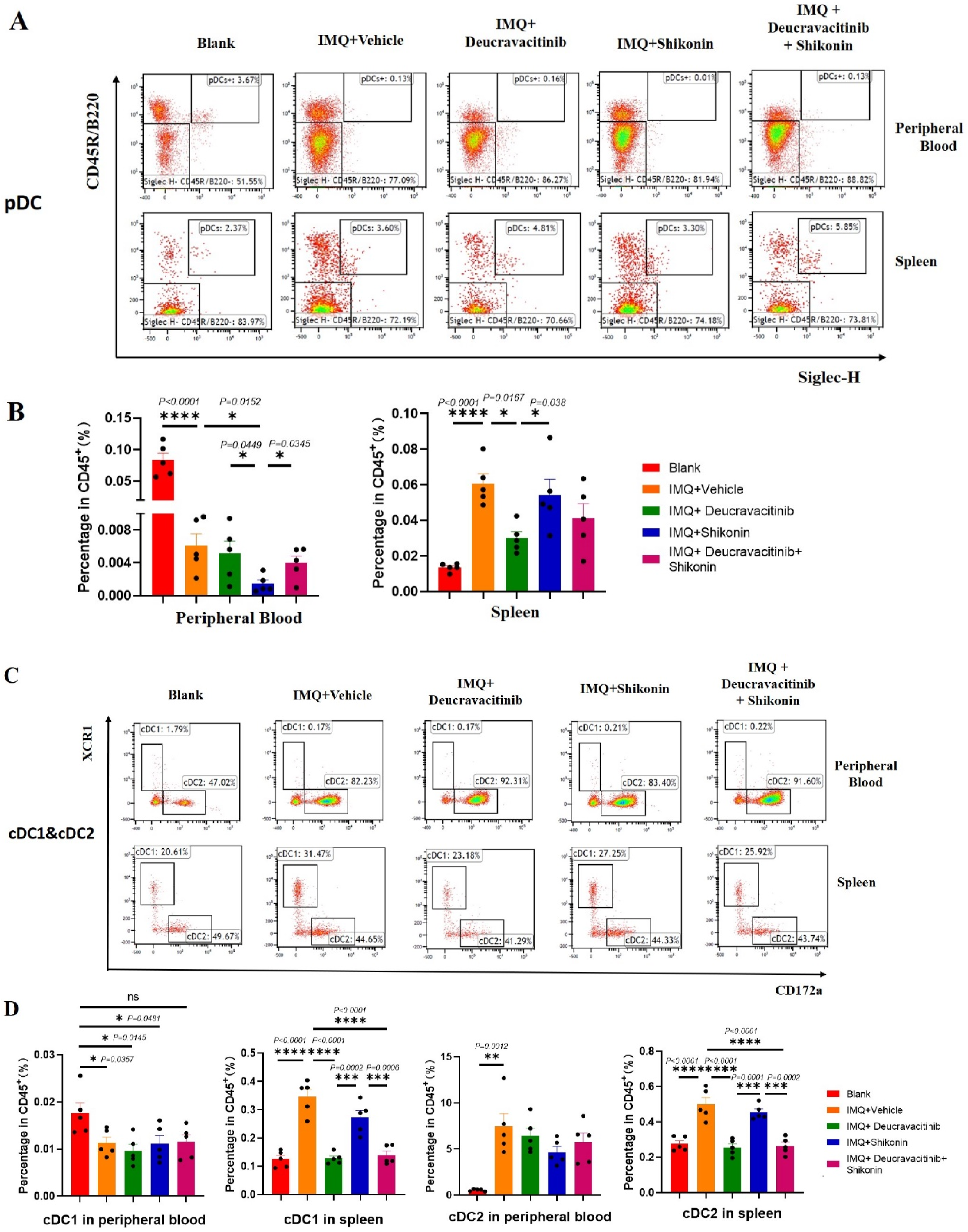
The expression of cDC1 cell populations in peripheral blood was extremely low and there were no statistically significant differences in all groups because of the few of scatters in the gate. The splenic cDC1 infiltration and the frequency of cDC2 in periperal blood and spleen were substantially higher in IMQ-treated mice than in the blank group (
Discussion
Psoriasis often damages the skin, scalp, joints, etc. of the whole body, accompanied by various complications including nephritis and hepatitis, or trigger systemic inflammation and oxidative stress reactions.35,36 It has been found that the incidence of psoriasis inflammation increased when DC induced the immune response are enhanced, such as TLR7/IL-23/IL-6 and Th17 related signaling pathways activated. 37 Therefore, therapies that reduce immune activation and antioxidant therapy can serve as adjunctive treatments for psoriasis. To date, despite the appearing of promising drugs such as JAK inhibitors, the treatment outcomes for psoriasis are still unsatisfactory. 38 In this study, we found that the combination of the TYK2 inhibitor deucravacitinib and the TCM preparation shikonin has better therapeutic effects on IMQ- induced psoriasis in mice than monotherapy controls and the mechanisms related to inhibit vascular proliferation and cellular immune response in the skin lesion area.
The common symptoms are the appearance of red patches on the skin, 39 which usually accompanied by silvery-white scales. In the present study, we used psoriasis-like erythema and scales on the dorsal skin to evaluate the psoriatic symptoms in IMQ-treated mice. 40 The results showed that through visual observation and PASI scores, the combination therapy of deucravacitinib and shikonin has the similar effects as deucravacitinib on alleviating the skin psoriatic symptoms, which was demonstrated by the skin histological data as well. Moreover, from the perspective of the body weight change rate, the incidence of adverse effects in the combination therapy should be less than that of monotherapy of deucravacitinib or shikonin.
In the present study, p-STAT3, CD31, and Ki67 expression in the psoriatic dermal tissue increased after IMQ induction. CD31, mainly expressed on the surface of endothelial cells, reflects changes in vascular proliferation and tissue morphology. 41 Ki67 is a cell proliferation-related antigen and related to mitosis and cell proliferation. STAT3 protein was phosphorylated by JAK and p-STAT3 can translocate into the nucleus and bind to DNA to exert multiple regulatory effects. 42 In the process of psoriasis, p-STAT3 participates in the abnormal proliferation of keratinocytes and capillary vessels. After the combination therapy of deucravacitinib and shikonin, CD31, Ki67 and p-STAT3 protein in the dermis all decreased, especially the level of CD31 had more significantly than in the monotherapy of deucravacitinib. These findings illustrate that the co-administration of deucravacitinib and shikonin has synergistic effects on reducing the abnormal proliferation of keratinocytes and capillary vessels in psoriasis.
It has been recently reported that interactions between cytokines, including IL-17, IL-12, and IL-1, and TNF-α, can explain most of the clinical features of psoriasis, such as keratinocyte hyperproliferation, neovascularization and inflammation. 43 Repeated application of IMQ cream induces and exacerbates inflammatory lesions of skin, stimulates dermal DC to secrete IL-23, and activates STAT3 to promote IL-17A secretion from T cells. 44 IL-12p70 is the biologically active form of IL-12, and the IL-12 levels in psoriatic patients are significantly higher than those in healthy individuals. 45 In the process of IMQ-induced psoriasis, activated T cells, keratinocytes, and DC can synergistically secrete a large number of cytokines to constitute a cytokine network, 46 inducing and activating each other, forming a self-sustaining inflammatory vicious cycle. Our results presented that IL-12p70, TNF-α, IL-1β, IL-6, and IL-17A are all key mediators in the pathogenesis of psoriasis. The secretion of these cytokines significantly increased in the skin lesions of IMQ-treated mice and decreased after monotherapy of deucravacitinib alone or the combination therapy of deucravacitinib and shikonin. Furthermore, the efficacy of the combination therapy was better than that of the monotherapy, which is related to the stronger inhibition of IL-12p70, IL-17A and IL-1 β.
Consistent with the immune disorder in psoriatic patients, the IMQ-treated mice also exhibit systemic inflammatory responses, including enlargement of the spleen and draining lymph nodes. 43 Both deucravacitinib monotherapy and combined administration effectively ameliorated splenomegaly in mice. It is known that the concentration of various chemokines in peripheral blood of psoriatic patients increases, which promotes the migration and aggregation of neutrophils and activated T cells to the lesions, increases the release of inflammatory cytokines and aggravates skin lesions. 47 According to the statistical analysis of various immune cell populations in peripheral blood, spleen and skin, we found IMQ stimulated the activation of γδ T cells to increase IL-17A secretion and play a key role in initiating inflammatory responses, regulating neutrophil expansion and aggregation. Our results showed the trend of IL-17A concentration in skin was consistent with the frequency of γδ T cells in all experimental groups, especially in deucravacitinib monotherapy group and the combination group. Meanwhile, we noted that IMQ increased the neutrophils and DC infiltration in skin and peripheral blood but there were basically no significant differences in spleen. Moreover, the deucravacitinib and shikonin co-administration group had few neutrophils and DC infiltration in the skin as compared to the vehicle control and deucravacitinib monotherapy group. It suggests that combination therapy has a significant inhibitory effect on the accumulation of neutrophils and DC in psoriasis lesions, thus reducing the regulation of cytokines on local immune responses and improving disease symptoms of psoriasis.
Multiple transcription factors are expressed in unique combinations to influence the development of cDC and pDC, which are involved in the onset and progression of psoriasis. pDC develop in the bone marrow and are potent producers of type I interferons. cDC comprise two major subpopulations, cDC1 and cDC2, which differentiate in the periphery and drive the activation of T cells to produce effector cytokines that induce proliferation and aberrant differentiation of keratinocytes. 48 In this work, pDC, cDC1 and cDC2, showed the same trend in the spleen as in the psoriatic skin symptoms and compared to the vehicle control group, the frequency of these populations was substantially decreased in the deucravacitinib group or the deucravacitinib and shikonin co-administration group. The populations of pDC and cDC1 in peripheral blood were fewer in IMQ-treated mice than in the blank control mice on flow charts. It hints that pDC acquire the migratory capacity by upregulating chemokines and aggregate towards the inflammatory site or target organ, leading to a decrease in the number of pDC in peripheral blood. Nevertheless, each DC subpopulation in mice skin was not examined in the current study, which needs further research to confirm this speculation.
In present work, IMQ-induced psoriasis in mice can only last for about a week, which is inconsistent with the chronic course of human psoriasis, and there are no significant complications similar to human psoriasis. Therefore, this animal model has limitations in studying the effects of drugs on the chronic course and complications of diseases. Next, we will use other models to further observe the long-term efficacy and mechanisms of combination therapy for psoriasis, including cellular immune and molecular signaling pathways. In addition, our data shows shikonin has good auxiliary effects on inhibiting skin CD31 expression, reducing neutrophil and DC infiltration, and lowering skin IL-12p70 concentrations. It provides the main targets and research directions for further in-depth research on the combination therapy of deucravacitinib and shikonin.
Conclusion
In summary, the combination of deucravacitinib and shikonin reduce the phosphorylation of STAT3, downregulate various inflammation-associated cytokines, and depress the migration and aggregation of various types of immune cells, leading to alleviate psoriasis symptoms. The combined treatment of deucravacitinib and shikonin for psoriasis was superior to the deucravacitinib monotherapy group, especially in inhibiting capillaries proliferation, reducing the infiltration of neutrophils and DC, and decreasing the concentration of IL-12p70 in skin. Furthermore, the combination therapy effectively prevents weight loss caused by psoriasis, and it is more effective than deucravacitinib monotherapy. It suggests that the combined therapy has good clinical application prospects.
Supplemental Material
Supplemental Material - Deucravacitinib and shikonin combination therapy ameliorates imiquimod-induced psoriasis in mice
Supplemental Material for Deucravacitinib and shikonin combination therapy ameliorates imiquimod-induced psoriasis in mice by Ling Xu, Jun Liu, Ying Jiang, Fuping Xu, Hongjiang Yu, Fang Fang and Lin Luo in International Journal of Immunopathology and Pharmacology.
Supplemental Material
Supplemental Material - Deucravacitinib and shikonin combination therapy ameliorates imiquimod-induced psoriasis in mice
Supplemental Material for Deucravacitinib and shikonin combination therapy ameliorates imiquimod-induced psoriasis in mice by Ling Xu, Ying Jiang, Fuping Xu, Jun Liu, Yuhong Jiang, Fang Fang and Lin Luo in International Journal of Immunopathology and Pharmacology.
Footnotes
Author contributions
Ling Xu: Investigation, Methodology, Conceptualization, Writing-original draft. Ying Jiang: Interpretation of data for the work, Writing-original draft, Revising manuscript. Jun Liu: Investigation, Data curation, Revising manuscript. Fuping Xu: flow experiments. Hongjiang Yu: pathological experiments. Fang Fang: immunohistochemical analysis. Lin Luo: Conceptualization, Interpretation of data for the work, Writing-review & editing, Supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Animal welfare
The present study followed the guidelines of the Declaration of Helsinki, and all animal protocols and procedures were reviewed and approved by the Ethics Committee of Crown Bioscience, Inc. for animal treatment and complied with relevant legislation.
Statement of ethics
This study was conducted according to the guidelines of the Declaration of Helsinki, and all animal protocols and procedures were reviewed and approved by the Ethics Committee of Crown Bioscience, Inc. (AN-2204-05-1942).
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
