Abstract
Background
Glabridin is one of the main components of the isoflavonoids in Glycyrrhiza glabra and possesses anti-inflammatory, anti-bacterial, and anti-cancer effects.
Objectives
Herein, the therapeutic effects and mechanisms of action of the glabridin liposome (GL) complex were studied in mice with imiquimod-induced psoriasis.
Materials and Methods
After treatment with GLs, their effectiveness was assessed using parameters, such as Psoriasis Area and Severity Index (PASI) score, histopathology, inflammatory cytokines, and psoriasis-associated proteins.
Results
The results demonstrated that GLs could significantly improve psoriatic symptoms, downregulate mast cell infiltration, and significantly reduce the PASI score of psoriatic mice. Quantitative reverse transcription polymerase chain reaction (RT-qPCR) results showed that GLs significantly decreased IL-23 and STAT3 mRNA expression. The results of immunohistochemistry and the enzyme-linked immunosorbent assay indicated that GLs decreased the expression of tumor necrosis factor-alpha (TNF-α), IL-17, and IL-22.
Conclusion
GLs can alleviate psoriasis by inhibiting the expression of keratinocyte-activating proteins. These results suggest that GLs have great potential for the clinical treatment of psoriasis.
Introduction
With intensive research into the efficacy of medicinal plants, there has been a strong rise in interest in botanicals and plant extracts in recent years because of their multiple biological effects, such as antioxidant, anti-inflammatory, antibacterial, antifungal, and antitumor properties, and their ability to combat the development of chronic diseases (Lu et al., 2022; Lenzi et al., 2018; Mitra et al., 2022). Psoriasis is a skin disease determined by many factors, such as environmental stimulation, multigene control, and immune mediation. It is characterized by inflammatory cell infiltration, epidermal hyperplasia, and excessive proliferation of keratinocytes (Boehncke & Schön, 2015; Greb et al., 2016; Griffiths & Barker, 2007). A typical clinical manifestation of psoriasis is squamous skin erythema or the presence of skin debris, which is often distributed locally on the skin and is also present systemically (Hung et al., 2018; Nestle et al., 2009). With an incidence of 1%–3% of the population, psoriasis poses an immense psychological and social burden on patients and their relatives (Griffiths & Barker, 2007; Shrivastava et al., 2015).
The exact pathogenesis of psoriasis is complex. It is generally believed that the abnormal synergy between immune cells and keratinocytes plays a critical role in the pathological course of psoriasis (Harden et al., 2015; Kim & Krueger, 2015). Stimulation of the secretion of various cytokines by immune cells, such as IL-17, IL-22, IL-23, and tumor necrosis factor-alpha (TNF-α) may promote excessive proliferation and activation of keratinocytes, thereby causing psoriatic symptoms (Albanesi, et al., 2007; van der Fits et al., 2009; Zhang et al., 2016). Furthermore, abnormal activation of cytokeratin, signal transduction, and transcription activator proteins is also considered to mediate the development of psoriasis.
In recent years, an increase in therapeutic biological agents has greatly alleviated the condition of patients with moderate and severe psoriasis and enhanced their quality of life. The treatment of psoriasis has changed from traditional therapy to an era of biological agents (Shrivastava et al., 2015). However, drug resistance and adverse effects caused by the long-term use of synthetic drugs and biological agents are problematic for patients (Zhang et al., 2016). Therefore, it is important to develop anti-psoriasis drugs that are safer, have better therapeutic effects, and have fewer side effects.
Glycyrrhiza glabra L. (licorice) is widely used as an herbal medicine in China. It has many functions, such as clearing heat, detoxifying and invigorating the spleen, tonifying the stomach, and promoting anti-inflammatory, antibacterial, and anticancer effects (Fujita et al., 2016). Glabridin (Glab) is a major hydrophobic flavonoid isolated from G. glabra. It has antioxidant, antibacterial, antitumor, anti-inflammatory, and other pharmacological activities (Dogra et al., 2021; Goel et al., 2021; Vaillancourt et al., 2021). Glab regulates immune reactions and reduces inflammation by inhibiting the release of inflammatory factors. However, Glab is not as effective when used for the treatment of skin diseases due to its low water solubility, which greatly hinders its transdermal penetration and active absorption.
Liposomes are drug carriers that can increase the solubility, bioavailability, and stability of insoluble drugs (Parlar et al., 2020; Rattanapak et al., 2012). Hence, the use of liposomes can overcome the poor water solubility of Glab. A formulation containing Glab complexed with liposomes, designated as glabridin liposomes (GLs), may be effective in promoting skin penetration and absorption of Glab, thus leading to better efficacy. To evaluate the potential alleviatory effect of GLs on psoriasis and explore its mechanism of action, a mouse psoriasis model induced by imiquimod was established to assess the expression of inflammatory cell infiltration and proinflammatory factors, which were detected through histopathological and immunohistochemical analyses, quantitative reverse transcription polymerase chain reaction (RT-qPCR), and enzyme-linked immunosorbent assay (ELISA).
Materials and Methods
Materials
GL (0.1%, 0.5%, and 1.0%) was obtained from Guangdong He Ji Biotech Co., Ltd. (Guangdong, China); Hematoxylin & Eosin (H&E) staining kit, 3,3′-diaminobenzidine (DAB), anti-TNF-α rabbit pAb, anti-IL-17 rabbit pAb, anti-filaggrin rabbit pAb, and horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (H + L) were purchased from Wuhan ServiceBio Technology Co., Ltd. (Wuhan, China); The ELISA kit was obtained from Jiangsu Meibiao Biotechnology Co., Ltd. (Jiangsu, China); The imiquimod ointment was from Sichuan Mingxin Pharmaceutical Co., Ltd. (Sichuan, China); The tacrolimus ointment (0.1%) was purchased from Astellas Pharma Tech Co., Ltd. (Japan).
Size and Morphology of Particle
The particle size and zeta potential of GL were analyzed by the Zetasizer NANO ZS (Malvern Instruments, UK). The morphology of GL was observed using a scanning electron microscope (SEM) (ZEISS Gemini SEM 300, JEOL, Japan).
Determination of Glab Content
Quantitation analysis of Glab was performed using a C18 chromatographic column (250 mm × 4.6 mm × 5 mm). The mobile phase comprised 70% acetonitrile and 30% water (v/v) at 30°C; the flow rate was 1.0 mL/min; the wavelength was 280 nm; and the injection volume was 10 µL. A linear regression curve of Glab content was obtained based on the pre-distributed concentrations and the absorption peak areas using high-performance liquid chromatography (HPLC).
Animals
Kunming mice were provided by the Guangdong Experimental Animal Center (approval document: SCXK/20130002). During the experiments, the following environmental conditions were maintained: ambient temperature 25 ± 2°C; humidity 55%–65%, and circadian rhythm of 12-h light/dark cycle. All the animal experiments were performed according to the guidelines laid down by the Animal Experimental Ethics Committee of the Guangdong University of Technology.
Male KM mice weighing 38 g each were stochastically divided into six groups (n = 10) as follows: tacrolimus, imiquimod, control, GL-L (0.1% GL), GL-M (0.5% GL), and GL-H (1.0% GL). Except for the control group, all the other groups of mice were topically treated with imiquimod (62.5 mg) daily for 1 week. During the second week, the GL groups were topically treated with different doses of GL (0.1%, 0.5%, 1.0%, or 200 µL per mouse) every day for a week, The tacrolimus group was treated with tacrolimus ointment (200 µL/mouse) for a week, whereas the imiquimod-treated and control groups were left untreated. Finally, the mice were euthanized via cervical dislocation, and their back skin tissues were fixed and frozen in 4% paraformaldehyde for further analysis.
Apparent Comparison and Psoriasis Area and Severity Index Score
After treating all the mouse groups, the mice were photographed to record the external appearance of the back skin, and Psoriasis Area and Severity Index (PASI) evaluated the severity of the back skin lesions. The degree of skin erythema and severity of skin damage were scored from 0 to 4 points as follows: 0 = none; 1 = mild; 2 = moderate; 3 = severe; and 4 = very severe.
Pathology Section Analysis
Dorsal skin tissues of the mice were fixed in 4% paraformaldehyde, embedded in paraffin, and cut into 4 µm sections. H&E and toluidine blue were used for staining after dewaxing and rehydration. H&E staining characterizes epidermal hyperplasia, while toluidine blue staining characterizes the degree of mast cell infiltration.
Immunohistochemistry
The tissues were deparaffinized and rehydrated in different concentrations of xylene and ethanol, respectively. Antigens were recovered using EDTA, and 3% hydrogen peroxide was added to block endogenous peroxidase. The tissue slides were then incubated overnight with anti-TNF-α rabbit pAb and anti-IL-17 rabbit pAb (1:800) at 4°C. The samples were then incubated with HRP-conjugated goat anti-rabbit IgG (H + L) (1:2000) for 1 h. DAB was used for the development of color, and hematoxylin was employed to restain the nuclei. The tissue sections were photographed using an automatic digital glass scanning system (Z1/AxioScan, Z1, ZEISS, Germany), and optical density (OD) values were measured using ImageJ software (NIH, Bethesda, MD, USA) (Gupta et al., 2012).
RT-qPCR
Samples were first mixed with mouse skin tissue, homogenized, the supernatant obtained, and RNA extracted with chloroform. RNA was quantitated using a NanoDrop 2000 spectrophotometer, and cDNA was then synthesized using the RevertAid First Strand cDNA Synthesis Kit.
Next, the expression of the genes of interest was measured employing the ABI StepOne Plus Fast RT-PCR system using the SYBR green method. In the first cycle of PCR amplification, the conditions used were as follows: 95°C for 10 min, followed by 45 cycles of 95°C for 15 s and 60°C for 60 s. The primers used for RT-qPCR are listed in Table 1.
The Information of Primer.

ELISA
Mouse skin tissues from each group were homogenized, and the supernatants were collected after centrifugation. interferon-γ (INF-γ) and IL-22 levels were detected using ELISA according to the manufacturer’s instructions (Jiangsu Meibiao Biotechnology Co., Ltd., Jiangsu, China).
Statistical Analysis
One-way ANOVA was used to analyze statistical significance; the experiments were repeated three times. p ≤ 0.05 (*) and p ≤ 0.01 (**) were considered to be significant.
Results
Morphology and Size of GLs
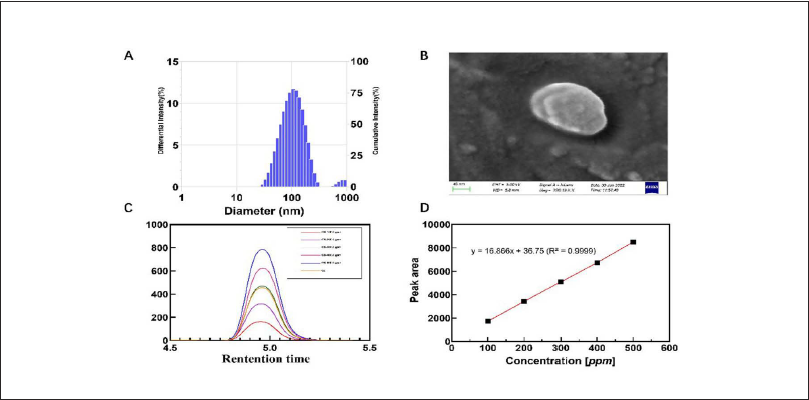
Figure 1A shows that the average particle size of GL was approximately 100 nm, with a polydispersity index of 0.2, indicating that the liposomes had a homogeneous particle size distribution. As shown by the SEM (Figure 1B), the liposomes displayed a spherical morphology of 150–200 nm. HPLC was used to identify glycyrrhizin in the liposomes (Figure 1C); the concentration (9401.89 ppm) was calculated using linear regression (Figure 1D).
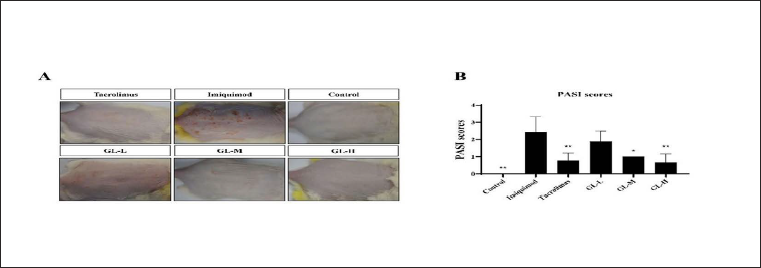
GL Reduced Psoriasis-like Skin Symptoms Induced by Imiquimod
In the present study, imiquimod was used to induce psoriasis. As shown in Figure 2A, imiquimod significantly increased erythema and squamous psoriasis on the back skin of the mice. However, after GL treatment, these symptoms were significantly reduced; increased alleviation was observed by treatment with higher doses of GLs. Psoriasis-like symptoms such as erythema, scaling, and hypertrophy are usually evaluated using PASI (Zhang et al., 2020). The score increased from 0 to 4, corresponding to the degree of deterioration caused by the psoriatic symptoms. As shown in Figure 2B, treatment with tacrolimus and GLs significantly reduced psoriasis-like lesions; moreover, GL treatment decreased the PASI score in a dose-dependent manner.
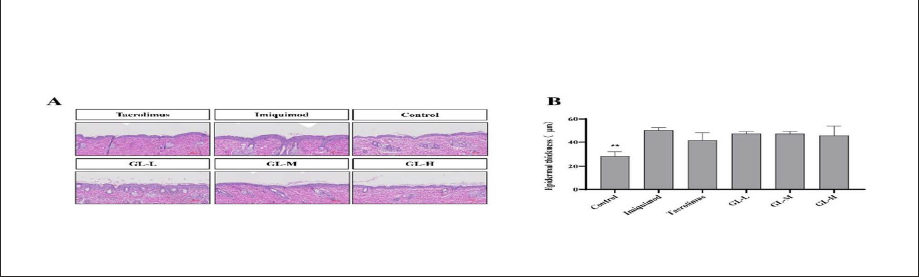
Epidermal hyperplasia is a characteristic feature of psoriasis-like skin injuries. H&E staining was used to evaluate the inhibitory effect of GLs on imiquimod-induced epidermal hyperplasia (Xiang et al., 2018). As depicted in Figure 3, histological images of imiquimod-treated mice show more obvious epidermal keratinocyte hyperplasia than that of the control group, which is accompanied by keratosis and micro-abscesses. The degree of epidermal hyperplasia observed in the imiquimod-treated model mouse group was higher than that seen in tacrolimus-treated or GL-treated groups, indicating that local application of GLs can effectively inhibit psoriasis-like epidermal hyperplasia and keratinization.
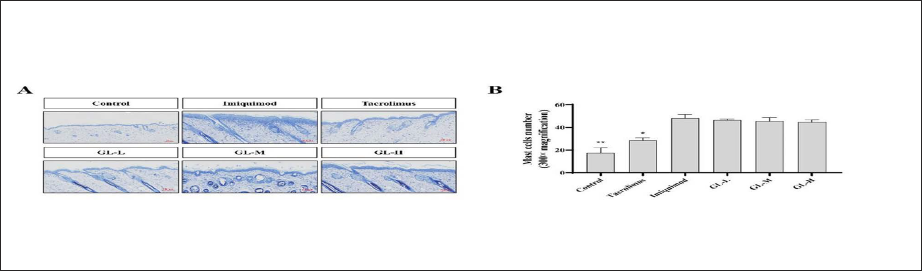
GL Treatment Reduces Mast Cell Accumulation and Inflammation
Activation and accumulation of mast cells and inflammatory responses are characteristic of psoriasis-like skin inflammation (Bożek & Reich, 2017). Therefore, toluidine blue staining was used to show the inflammatory activation and degranulation of mast cells in the skin tissues of GL-treated psoriatic mice. As shown in Figure 4, the proliferation of mast cells in the skin tissue of the imiquimod-induced group was significantly increased in the control and tacrolimus-treated groups. After treatment with different doses of GLs, mast cell activation and accumulation were alleviated compared to those in the imiquimod-induced group.
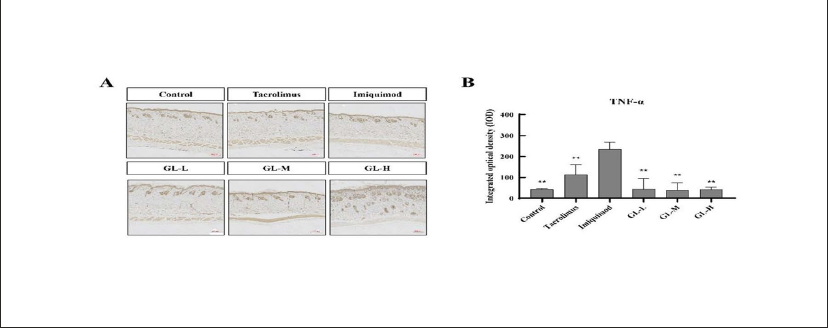
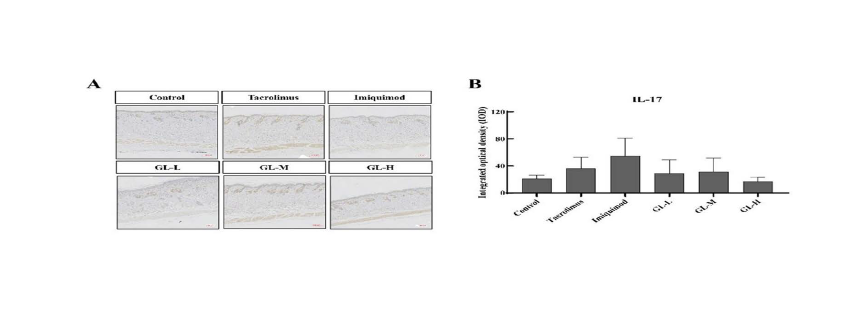
GLs Inhibiting the Proinflammatory Cytokines Expression
The expression of several proinflammatory cytokines, such as IL-22, IL-23, and IL-17, in cells is mediated by TNF-α (Albanesi et al., 2007; Hsu et al., 2013). The IL-23 and IL-17 axes mediate imiquimod-induced psoriasis-like skin lesions. The effect of GLs on the expression of TNF-α and IL-17 in imiquimod-induced psoriasis-like skin lesions was evaluated using immunohistochemical staining. The IL-22 protein level in the skin was determined by ELISA. The expression of IL-23 mRNA in the skin was determined using RT-qPCR.
As shown in Figures. 5A, B–7A, B, the expression of IL-22, IL-17, TNF-α proteins, and IL-23 mRNA in mouse skin increased after imiquimod induction (IL-23 and TNF-α had obvious aboriginality). At the same time, treatment with tacrolimus and GLs significantly inhibited the overexpression of these proinflammatory cytokines. The expression of skin proinflammatory cytokines was significantly lower in the tacrolimus- and GL-treated groups than in the imiquimod group. Moreover, treatment with a high dose of GL (GL-H) was more effective than that with tacrolimus. The results showed that GLs can reduce the inflammatory response in psoriasis by downregulating the expression of proinflammatory cytokines and their mRNAs, thereby attenuating imiquimod-induced psoriasis-like skin lesions.
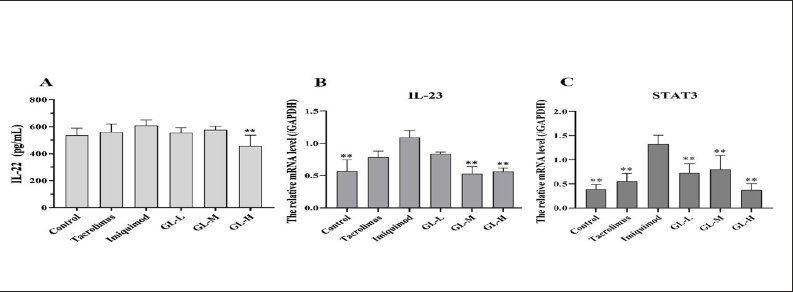
GLs Downregulate the Expression of STAT3 mRNA
STAT3 is a transcription factor that causes inflammation and regulates cell differentiation. Abnormal activation of STAT3 leads to psoriasis-like changes, such as inflammatory cell infiltration, neutrophil aggregation, and epidermal hyperplasia, which play an important regulatory role in the progression of psoriasis. We used RT-qPCR to determine the expression of STAT3 mRNA in the skin after GL treatment. As shown in Figure 7C, STAT3 mRNA expression in the skin was significantly increased under imiquimod induction but significantly decreased upon treatment with tacrolimus and medium (GL-M) and high (GL-H) doses of GL. Taken together, GLs can effectively improve psoriatic skin lesions by inhibiting STAT3 activation.
Discussion
Psoriasis is a common type of squamous dermatosis that exhibits abnormal keratinocyte proliferation. The incidence of psoriasis worldwide is 1%–3%; consequently, it poses a huge psychological and social burden on patients and their relatives (Shrivastava et al., 2015). Therefore, in recent years, the development of safer and more effective anti-psoriasis drugs has presented a great challenge worldwide.
The imiquimod-induced mouse psoriasis model is widely used for screening psoriasis drugs (Gao et al., 2020; Hung et al., 2018). The present study investigated the therapeutic effects and mechanisms of action of GLs on imiquimod-induced psoriasis in mice. Experiments showed that the PASI scores of mice were significantly reduced after GL treatment in a dose-dependent manner. Thus, GLs are implicated in having a significant therapeutic effect on imiquimod-induced psoriasis-like skin injury. In addition, analysis of pathological skin tissue sections revealed that GLs alleviated several imiquimod-induced psoriatic symptoms, such as epidermal dysplasia, mast cell infiltration, and degranulation. Proinflammatory cytokines such as IL-17, IL-23, and TNF-α play important roles in the pathological mechanisms of psoriasis (Albanesi et al., 2005; Griffiths & Barker, 2007; Hsu et al., 2013). Therefore, we measured the levels of these proinflammatory factors in the mouse skin tissues after GL treatment. The results showed that GLs can treat psoriasis by significantly decreasing the levels of these proinflammatory factors. STAT3 is a known transcription factor that transmits signals to the nucleus upon stimulation by cytokines or growth factors, such as IL-6 and IL-22, leading to the activation of downstream signals to induce inflammation and regulate cell differentiation. Abnormal activation of signals is one of the most important factors in the occurrence and development of psoriasis; therefore, the pathological symptoms of psoriasis can be effectively alleviated by inhibiting the activation of STAT3 (Zhang et al., 2016). The expression of STAT3 mRNA in mouse skin was decreased after GL treatment, suggesting that GLs may alleviate psoriasis injury by inhibiting the abnormal activation of STAT3.
In summary, we found that GLs could improve the symptoms of psoriasis by reducing epidermal hyperplasia and mast cell infiltration while simultaneously downregulating the expression of a variety of proinflammatory factors. Therefore, it has excellent potential to become a safe, effective, and well-tolerated treatment for psoriasis patients.
Summary
GLs could improve the symptoms of psoriasis by reducing epidermal hyperplasia and mast cell infiltration, while simultaneously downregulating the expression of a variety of proinflammatory factors.
Abbreviations
DAB: 3,3′-diaminobenzidine; GL: Glabridin liposome, Glab: Glabridin; HE: Hematoxylin-eosin; HPLC: High-performance liquid chromatography; HRP: Horseradish peroxidase; PASI: Psoriasis area and severity index; PDI: Polydispersity index; SEM: Scanning electron microscope.
Footnotes
Acknowledgments
The authors are grateful to the Innovation and Entrepreneurship Leading Team Project of Panyu District (Grant No. 2019-R01-6) for providing financial support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
This animal study protocol was approved by Ethics Committee of Guangdong University of Technology.
Funding
Innovation and Entrepreneurship Leading Team Project of the Panyu District, Guangzhou, Guangdong, China (Grant No. 2019-R01-6).
