Abstract
Background
Plants are attractive sources of anti-cancer agents. However, there is limited data about the anti-cancer potential of Ethiopian medicinal plants traditionally used for the treatment of cancer.
Objectives
To screen the cytotoxicity of 26 Ethiopian medicinal plants against breast cancer cells (MCF-7) and to demonstrate the potential consistent cytotoxicity of the six most active extracts against prostate cancer cell line (PC-3) and neuroblastoma (SH-SY5Y). Furthermore, the mode of action of the most active extracts should be investigated.
Materials and Methods
Twenty-six Ethiopian medicinal plants were collected from 12 sites and extracted using 80% methanol through maceration. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was used to test their cytotoxicity. Annexin V/propidium iodide (PI) assay was used to segregate the type of death induced by the extracts. The Western Blot was used to analyze deoxyribonucleic acid (DNA) damaging properties of active extracts, while chromatographic techniques [thin-layer chromatography (TLC) and high-performance liquid chromatography (HPLC)] were employed to provide a fingerprint of the most active extract.
Results
Dovyalis abyssinica (A. Rich.) Warb. extract displayed the most potent cytotoxic activity against MCF-7 and SH-SY5Y cells with the half-maximal inhibitory concentration (IC50) values of 28.28 ± 3.56 and 5.5 ± 3.9 µg/mL, respectively. It also increased apoptotic cell populations from 0.05% to 12.4%, 0.9% to 14.4%, and 1.8% to 64.5% (p < 0.0001 in all cases) in MCF-7, SH-SY5Y, and PC-3 cells. Acokanthera schimperi Benth. & Hook.f., D. abyssinica, Erythrina brucei Schweinf., Pittosporum abyssinicum Delile, Rubia cordifolia L., and Stephania abyssinica var. tomentella Oliv. significantly (p < 0.0001) elevated the expression of phosphohistone H2AX (γH2AX) in at least one cancer cell line. Poly-ADP-ribose polymerase-1 (PARP-1) also significantly increased (p < 0.0001) by D. abyssinica and R. cordifolia in MCF-7 cells.
Conclusion
Some Ethiopian medicinal plant extracts showed marked cytotoxicity against MCF-7, PC-3, and SH-SY5Y cancer cell lines, promoting apoptosis through DNA damage and encouraging phytochemical investigations for potential drug development.
Introduction
Natural products have been used to treat cancer for over half a century and are still an essential source of novel anti-cancer drug discovery. 1 Despite drawbacks and challenges, drugs of plant origin, such as vinca alkaloids, epipodophyllotoxins, taxanes, and camptothecin derivatives, are essential to clinical cancer chemotherapy. 2 Furthermore, thiol-containing maytansinoids, used in anti-body-drug conjugates, are derived from the Ethiopian medicinal plant Maytenus serrata, formerly known as Maytenus ovatus.3–5
The currently available cancer chemotherapeutic agents often cause side effects like alopecia, nausea, vomiting, anemia, and immunosuppression, as they primarily affect rapidly proliferating body cells. 6 In addition, resistance has already developed to both free-drug chemotherapeutic agents 7 and anti-body drug conjugates (ADCs) 8 to different degrees. Reports also show that up to 90% of cancer treatment failure is associated with resistance to chemotherapeutic agents. 9
Previous reports indicated that about 80% of Ethiopians depend on traditional medicine, which primarily involves medicinal plants.
10
To increase the effectiveness of their conventional medication, the majority of Ethiopian cancer patients combine herbal remedies with their medications. For example, the fruit, bark, and whole plant of Dovyalis abyssinica (A. Rich.) Warb. (Flacourtiaceae) are used for the treatment of cancer in some districts in Ethiopia.
11
The root of Stephania abyssinica Oliv. (Menispermaceae) and in some cases, the whole part of the plant is also used for the treatment of cancer.
12
The plant names have been checked with “World Flora Online” (
The present study was designed to determine whether common plants used in Ethiopian traditional medicine for the treatment of cancer or cancer-like diseases are endowed with cytotoxic activity against cancer cell lines. Thus, the lead compound/s from the cytotoxic extracts can be investigated and used to develop novel anti-cancer chemotherapeutic agents.
Materials and Methods
Plant Collection
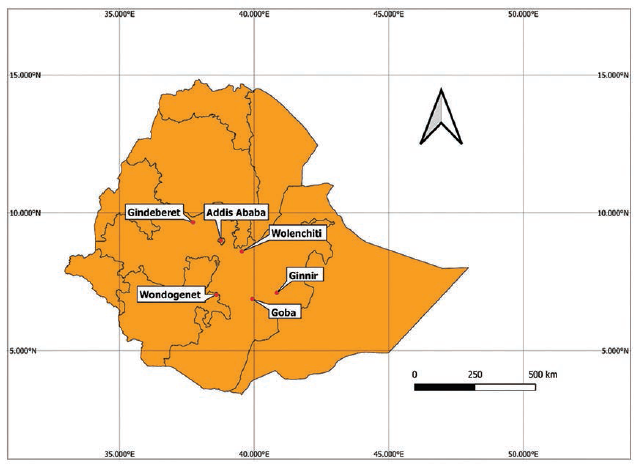
The plants were collected from 12 different localities, namely, Addis Ababa, Sebeta, Holeta, Dukem, Bishoftu, Burayu, Sululta, Goba, Ginnir, Gindeberet, Wondogenet, and Welenchiti. The plant collection sites are indicated in Figure 1, while the specific geolocation of the plant collection sites is summarized in Supplementary 1. The collected plant parts were temporarily stored in a fiber bag and transported to the lab. A botanist, Mr. Melaku Wondafrash, identified the plants, and a specimen of each plant was deposited at the National Herbarium, College of Natural and Computational Sciences, Addis Ababa University. The collected plant materials were washed with distilled water and air-dried at room temperature.
Map Showing Study Areas: Study Areas not Shown in the Map like Sebeta, Holeta, Burayu, Sululta, Dukem, and Bishoftu are Found within a Maximum Distance of 50 km from Addis Ababa, the Capital of Ethiopia.
Plant Extraction
The dried samples were ground into a coarse powder using a Micro-Mill Grinder (Thomas Scientific). Three hundred grams of each powdered plant parts were mixed with 750 mL of 80% methanol and placed on an orbital shaker at 130 rpm for 72 h at room temperature. The extracts were then filtered using Whatman No. 1 filter paper (150 mm diameter) and concentrated at 37°C in a rotary evaporator (Buchi, Switzerland) at a temperature not exceeding 40°C. The concentrates were kept in a dry oven at 37°C for 3–5 days to obtain the dry powder of the extracts. The yields of the extracts were calculated and depicted in Supplementary 1. The powder of the plant extracts was dissolved in dimethyl sulfoxide (DMSO) (Sigma–Aldrich) to obtain the final stock concentration of 2 mg/mL.13, 14
Dilution of the Stock Solution
The stock solution was diluted in Dulbecco’s Modified Eagle Medium (DMEM) (Thermo Fisher Scientific). The final concentration of DMSO was adjusted not to exceed 0.01% for all working crude extract solutions. All media used had 10% fetal bovine serum (FBS), 1%
Cell Culture
Human breast cancer cell line (MCF-7) (HTB-22), human prostate cancer cell line (PC-3) (CVCL-0035), and human neuroblastoma cell line (SH-SY5Y) (CRL-2266) cell lines were bought from the American Type Culture Collection (ATCC) and cultured in a water bath warmed with DMEM containing 10% FBS (Sigma–Aldrich), 1%
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Assay
The MTT assay utilizes the ability of alive cells to convert MTT to purple-colored formazan dye metabolically. 15 The intensity of the purple color can indirectly be used to estimate the number of viable cells in the media.16, 17 The cells were seeded in 24-well plates, at a density of 6 × 104 cells per well, in 500 µL DMEM and incubated overnight before treatment. The cells were then treated with 6.25, 12.5, 25, 50, 100, and 200 µg/mL final concentrations of the crude extracts for 48 h. Following incubation, a mixture of 50 µL MTT solution and 500 µL DMEM without phenol red was added to all the wells, and the plates were then maintained at 37°C for 1 h. The media was aspirated from all wells; 500 µL of DMSO was added to each well, and the plates were shaken gently for 10 min. The absorbance was recorded using a Tecan Ultra Evolution plate reader (Männedorf, Switzerland) at 570 nm. All experiments were performed in triplicates, and the average values were recorded. Cell viabilities were estimated as viable cell percentages compared to controls. Doxorubicin was used as positive control, and IC50 values of the plant extracts in the respective cancer cell lines were calculated using GraphPad Prism 9 software.
Annexin V/Propidium Iodide (PI) Binding Assay
The cells were seeded onto plates, treated with plant extracts at IC50 concentrations, incubated for 24 h, collected, and diluted with phosphate buffered saline (PBS) containing 1% FBS and incubated with Annexin V and PI reagents for 15 min at room temperature in the dark. Live, dead, early, and late apoptotic cell populations were finally analyzed using BD Accuri® C6 flow cytometer (New Jersey, USA). In each assay, at least 40,000 cellular events were recorded.
Western Blot
The cells were plated in 25 cm2 plates, incubated overnight, treated with active extracts, washed, and lysed using lysis buffer [containing 140 mM NaCl, 50 mM ethylenediaminetetraacetic acid (EDTA), 10% glycerol, 1% Nonidet P-40, 20 mM Tris, potential of hydrogen (pH) 7.0; 25 mM β-glycerol phosphate (Sigma), 50 mM sodium fluoride (Sigma), 1 mM sodium orthovanadate (Sigma), 1 µg/mL aprotinin (Sigma), 1 µM pepstatin (Sigma), 1 µg/mL leupeptin (Sigma), and 1 mM phenylmethylsulphonyl fluoride (PMSF) (Sigma)]. Cell lysis was achieved within 10 min of centrifugation at 10,000 rpm at 4℃. The cell lysates were then centrifuged, and the supernatants were collected. The protein amount was measured using a bicinchoninic acid assay (BCA kit, Thermo Fisher). Equal protein amounts were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to Immobilone®-P polyvinylidene fluoride (PVDF) transfer membranes (Millipore, Germany) using a semi-dry transfer method. The membranes were probed with primary anti-bodies against phosphohistone H2AX (γH2AX0) (sc-517336) and poly-ADP-ribose polymerase-1 (PARP-1) (sc-8007), (Santa Cruz Biotechnology, Inc., Dallas, TX, USA). The membranes were blocked in Tris-Buffered Saline with Tween 20 (TBST) (Tris, pH 7.5, 100 mM; NaCl, 150 mM; Tween 20, 0.05%) containing 1% bovine serum albumin (BSA) or 5% skimmed milk for 2 h and then incubated with the corresponding anti-body overnight. After washing three times with TBST for 10 min, membranes were incubated with anti-mouse (GE HealthCare Technologies Inc.) or anti-rabbit (Bio-Rad laboratories) secondary anti-bodies for 30 min. After incubation with the secondary anti-body, the membranes were washed three times with TBST, and the bands were visualized using enhanced chemiluminescence. 18
Thin-layer Chromatography (TLC)
TLC analysis was performed for the most active extract, D. abyssinica extract (DAE). DAE (10 mg/mL) was dissolved in methanol, and 15 µL of the sample was applied to analytical POLYGRAM® SILG/UV254 TLC plates (Macherey-Nagel, 20 × 5 cm plates). The solvent system was chloroform: MeOH: ammonia (60:40:5 v/v) and was allowed to migrate 18 cm from the starting line. The TLC plate was sprayed using vanillin-H2SO4 and then heated at 110℃ for 5 min. Another plate was sprayed with Dragendorff’s reagent to analyze the presence of alkaloids. The bands were observed under short-wave and long-wave ultraviolet (UV) light. Moreover, pictures of the sprayed TLC plates were taken under visible light.
Ultra-High-pressure Chromatography-PDA (UHPLC-PDA)
The UHPLC fingerprint for DAE was performed using the Waters® e2695 family of Separations Module (Waters Corporation, Massachusetts, USA). Column: Agilent ZOBRAX Eclipse C18, 4.6 × 150 mm, 3.5 m. Mobile phase: solvent (A): methanol; Solvent (B): aqueous solution of ammonium citrate (10 mM) adjusted to pH = 4 with acetic acid. The gradient was: 0–20 min solvent A (10%); 40 min solvent A (20%); 50 min solvent A (30%); and 60 min solvent A (10%). The flow rate was 0.65 mL/min; the column temperature was 25°C; the system pressure was 1,300 psi. The concentration of the crude extract was 0.5 mg/mL, and the injection volume was 20 µL.
Statistics
Data are expressed as mean ± standard deviation. All experiments were performed in triplicates, and the average values were recorded. Analysis was performed using one-way analysis of variance (ANOVA) followed by a post hoc Tukey’s test. A p value less than 0.05 was considered for statistical significance.
Results
A total of 26 plant parts were collected from 12 different study sites. The collected plant parts include leaves, roots, stembark, areal part, whole-plant, gum, and rhizome. Names of the plants, family, vernacular names, places of collection, specific geolocations, and percent yield after macerations were summarized in Supplementary 1.
Cell Viability
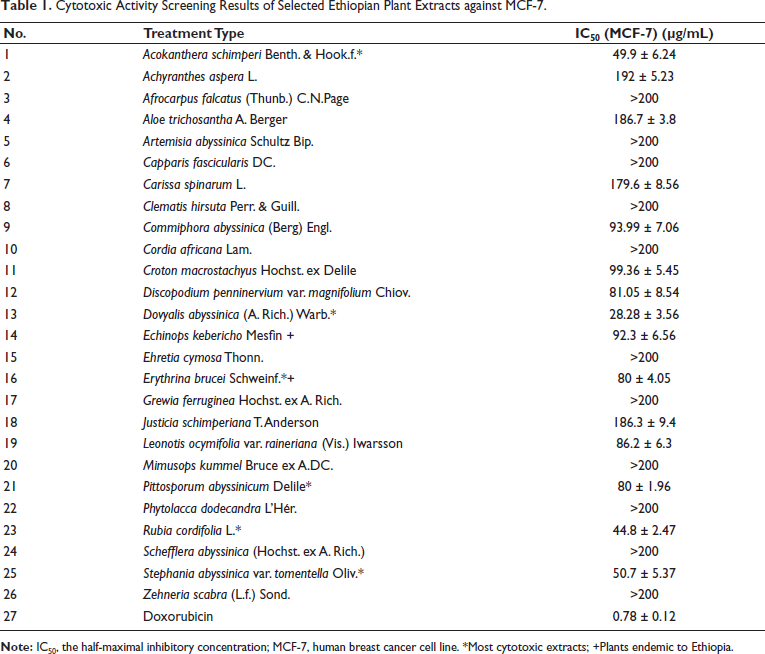
The plant extracts displayed different degrees of cytotoxic activity, with six of them exhibiting cytotoxicity with an IC50 of <80 µg/mL against MCF-7 cells. These were A. schimperi extract (ASE), DAE, E. brucei extract (EBE), P. abyssinicum extract (PAE), R. cordifolia extract (RCE), and S. abyssinica var. tomentella Oliv extract (SAE). Among these extracts, DAE (28.28 ± 3.56 µg/mL) was the only extract having an IC50 of <30 µg/mL. Thus, these six extracts were further screened against the two other cell lines (PC-3 and SH-SY5Y). Accordingly, while DAE exhibited a better activity against both PC-3 (14.8 ± 4.01 µg/mL) and SH-SY5Y (5.5 ± 3.9 µg/mL) than MCF-7 cells, the rest of the extracts had a variable activity with ASE (14.42 ± 5.14 µg/mL) and PAE (32.8 ± 2 µg/mL) showing the most notable activity against PC-3 cell line. The cytotoxicity profile expressed with IC50 values of all screened extracts in the MCF-7 cell line is presented in Table 1.
Cytotoxic Activity Screening Results of Selected Ethiopian Plant Extracts against MCF-7.
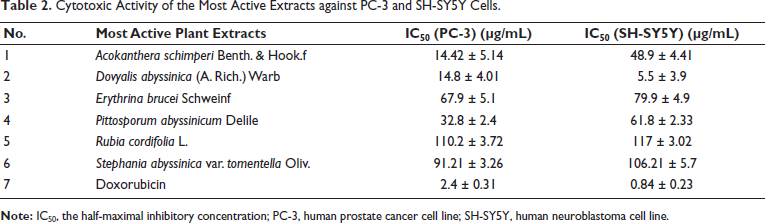
All the six most active extracts that displayed cytotoxicity with IC50 ≤ 80 µg/mL were tested against PC-3 prostate cancer and SH-SY5Y neuroblastoma cell lines. All except RCE and SAE displayed consistently marked cytotoxicity against all the tested cell lines. The extracts that showed consistently increased cytotoxic activity against PC-3 and SH-SY5Y cells exhibited marked cytotoxicity compared to MCF-7 cells (Table 2). DAE displayed the most cytotoxic activity against SH-SY5Y cells with an IC50 value of 5.5 ± 3.9 µg/mL.
Cytotoxic Activity of the Most Active Extracts against PC-3 and SH-SY5Y Cells.
Cell Death Mechanisms
The potential apoptotic effect of the hydroalcoholic extract of the six plants was explored by double staining with Annexin V/PI (Figures 2 and 3). The results suggested that the observed cytotoxicity could be linked with apoptosis-associated mechanisms. Indeed, the Annexin V/PI binding assay results showed that all crude extracts significantly increased the early and late apoptosis cell populations compared to the controls. Accordingly, ASE increased the early apoptosis in MCF-7 cells from 0.0% to 48.7% (p < 0.0001). ASE also increased the late apoptotic MCF-7 cell population from 0.0% to 20% (p < 0.0001) after 24 h of treatment. On the other hand, MCF-7 cells treated with IC50 concentrations of PAE, DAE, SAE, and EBE significantly increased (p < 0.0001) the early apoptotic cell population from 0.0% to 34.7%, 5.4%, 40.3%, 38.3%, respectively. Additionally, in MCF-7 cells treated with IC50 concentrations of PAE, DAE, SEA, and EBE, the late apoptotic cell populations were significantly increased (p < 0.001) from 0.0% to 5.3%, 7.0%, 38.5%, and 26.1%, respectively.
Representative Flow Cytometry Scattergram for Dovyalis abyssinica Extract (DAE) against Human Breast Cancer Cell Line (MCF-7), Human Neuroblastoma Cell Line (SH-SY5Y), and Human Prostate Cancer Cell Line (PC-3) Cells: Cells were Treated with the Respective the Half-Maximal Inhibitory Concentration (IC50) of DAE and Incubated for 24 h, and the Cell Death Profile was Evaluated using the BD Accuri C6 Flow Cytometry. DAE, Dovyalis abyssinica extract.
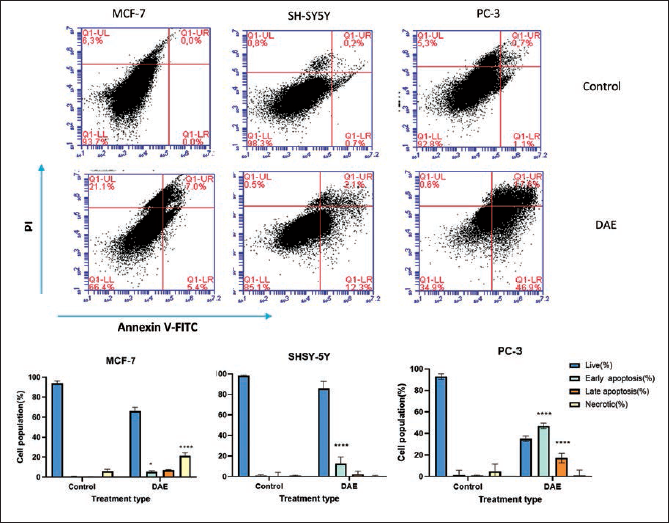
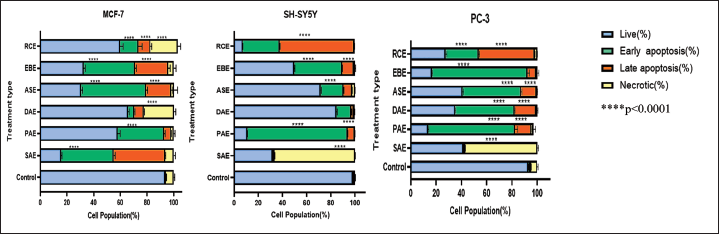
PAE increased the early apoptotic cell population from 0.7% to 82.0% (p < 0.0001) in SHSY-5Y cells. Additionally, PAE increased the late apoptotic cell population from 0.2% to 6.8% (p < 0.0001) 24 h following treatment. Furthermore, DAE, ASE, and EBE increased the early apoptotic cell population from 0.7% to 12.3%, 19.0%, and 39.6% (p < 0.0001), respectively. On the other hand, SH-SY5Y cells treated with SAE were observed to undergo a significant increase in necrotic cells from 0.8% to 66.4% (p < 0.0001).
EBE showed the highest rate of increase in early apoptotic cell population in PC-3 cells treated with the extract’s IC50 concentration. Accordingly, EBE increased the early apoptotic cell population from 1.1% to 76.5% (p < 0.0001). EBE also increased the late apoptotic cell population from 0.7% to 6.6% (p < 0.0001). PAE, ASE, and DAE also increased the early apoptotic cell population from 1.1% to 67.3%, 45.8%, and 46.9% (p < 0.0001), respectively.
Deoxyribonucleic Acid (DNA) Damage Assay
The Western blot data showed that cells treated with IC50 concentrations of DAE, SAE, and EBE had a significantly increased (p < 0.0001) expression of γH2AX than the controls in MCF-7 cells (Figure 4A and B). The rank order of effectiveness is DAE>>SAE>>EBE. These findings indicate that EBE, DAE, and SAE exert cell death activity through double-strand DNA damage in MCF-7 cells. Furthermore, DAE and RCE significantly (p < 0.0001) increased the expression of PARP-1 protein in MCF-7 cells by 53.8% and 41.6%, respectively, compared to the controls. PARP-1 expression was assessed in SH-SY5Y and PC-3 cells as well. However, no reportable data were obtained. In SH-SY5Y cells, ASE, DAE, and SAE significantly (p < 0.0001) increased the expression of γH2AX by 59.9%, 23.4%, and 7 folds, respectively, compared to the controls. ASE (21-fold) and SAE (7-fold) demonstrated a marked (p < 0.0001) increase in the expression of γH2AX in PC-3 cells.
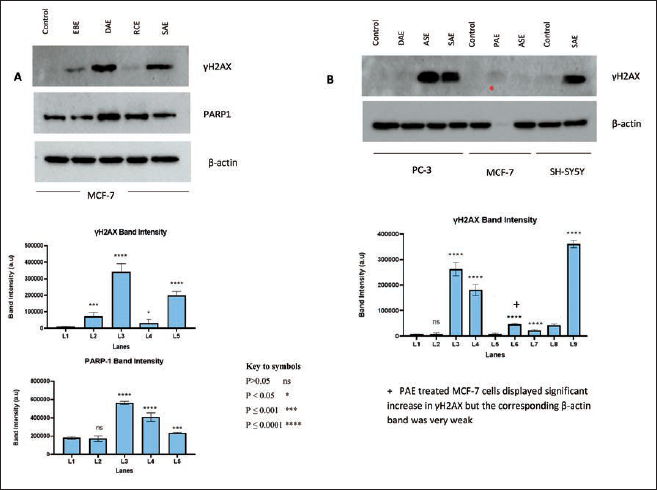
In addition to significant variation from the control, a substantial difference in the expression of γH2AX was observed among the treated groups. Accordingly, DAE and SAE-treated MCF-7 cells exhibited an increased expression of γH2AX, approximately by 2 folds (p < 0.0001) compared to EBE and RCE-treated cells. Likewise, ASE-treated cells increased the expression of γH2AX significantly (p < 0.0001) by about 30 folds compared to DAE-treated PC-3 cells. SAE-treated PC-3 cells also expressed γH2AX 20 times (p < 0.0001) greater compared to DAE-treated ones.
Microscopy
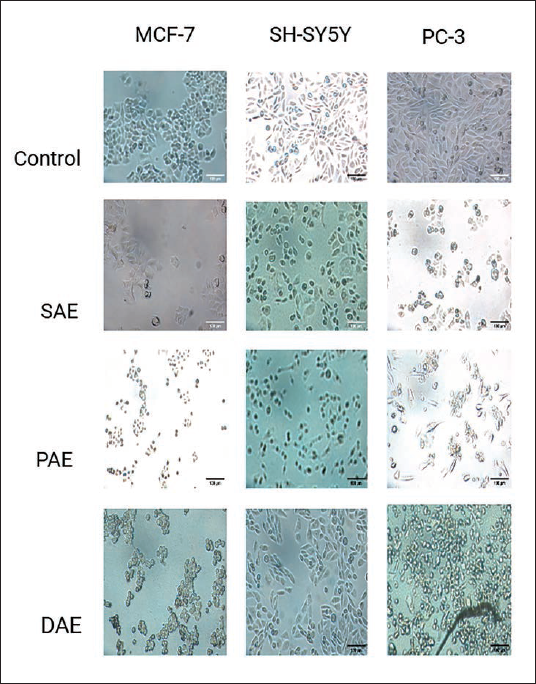
Microscopic images of the cancer cells treated with the most active extracts go in line with the MTT assay and mode of action study results. The images showed depleted, morphologically disfigured, and in some cases floating cellular debris (Figure 5). The decrease in viable cells and changes in cell size and shape are an indicator of cytotoxicity.
Microscopy of Human Breast Cancer Cell Line (MCF-7), Human Neuroblastoma Cell Line (SH-SY5Y), and Human Prostate Cancer Cell Line (PC-3) cells after (24 h) Treatment with Respective the Half-Maximal Inhibitory Concentrations (IC50) of the Three Most Active Crude Extracts Compared to (–) Control.
TLC
TLC was performed for the most active extract DAE, and the result showed that DAE has at least 10 different compounds (Figure 6). Moreover, the TLC plate sprayed with Dragendorff’s reagent indicated that at least five of these compounds are alkaloids.
Thin-layer Chromatography (TLC) Fingerprint of Crude 80% Methanol Bark Extract of Dovyalis abyssinica: using Shortwave Ultraviolet (UV) (A), Longwave UV (B), after Spraying with Dragendorff’s Reagent (C), and Spraying with Vanillin- H2SO4 (D).

UHPLC-PDA Results
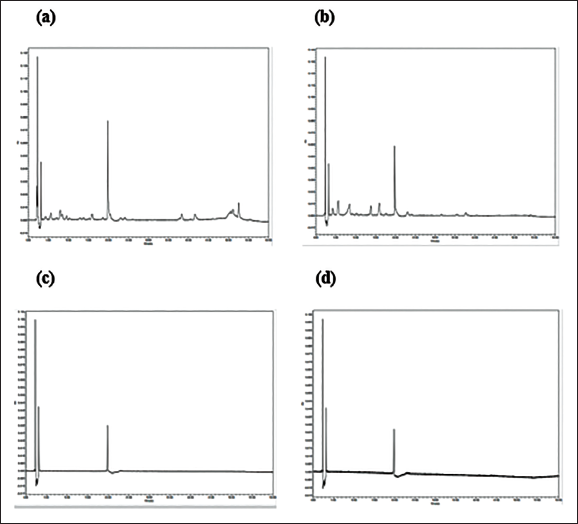
The HPLC analysis showed that DAE has one dominant compound eluted at 20 min (Figure 7). There were also many notable peaks justifying the presence of many compounds in the extract. The dominant compound appeared visible at all observed wavelengths (254, 300, 400, 500, 600, and 798 nm).
Ultra-high-pressure Chromatography (UHPLC) Fingerprint of Dovyalis abyssinica Extract (DAE) at 254 nm (a); 300 nm (b); 400 nm (c); 798 nm (d).
Discussion
The present study screened the cytotoxic activity of 26 Ethiopian medicinal plant extracts against the MCF-7 breast cancer cell line. Furthermore, we studied the consistency of cytotoxicity of the six most active extracts against PC-3 prostate cancer and SH-SY5Y neuroblastoma cells. Additionally, the mode(s) of action of cytotoxicity of the six most active extracts were investigated. The initial screening with MCF-7 cells revealed that DAE was the only extract with an IC50 of <30 µg/mL. Subsequent screening with PC-3 and SH-SY-5Y added ASE and PAE to the list, with EBE, RCE, and SAE exhibiting a noteworthy activity. Interestingly, DAE had an IC50 of <30 µg/mL against all the cell lines used in the present study, suggesting the extract is endowed with notable anti-cancer activity. Indeed, the leaf extract of D. abyssinica was reported to be cytotoxic to PC-3 cells with an IC50 of 68.40 µg/mL. 19 The higher IC50 value of the leaf extract compared to the bark extract reported in this study (14.8 ± 4.01) may suggest that the cytotoxic principles are more concentrated in the bark than in the leaf. ASE has also been shown to have an IC50 ranging from 7.1 to 10.31 µg/mL, which is lower than the one we reported in this study (14.42–49.9 µg/mL).20, 21 Although the difference among the findings may be attributed to various factors, the findings collectively indicate that ASE is another important cytotoxic extract.
To the best of our knowledge, there is no previous report of cytotoxicity for EBE. However, compounds isolated from the genus Erythrina, including Erythrina senegalensis 22 and Erythrina abyssinica 23 were reported to possess cytotoxic activity against different cell lines. Moreover, some evidence indicated that the extract of E. abyssinica has anti-microbial, 24 anti-viral, 25 and radical scavenging activity. 26 These findings, coupled with ours, strongly suggest that plants belonging to the genus Erythrina could be associated with potential anti-cancer activity.
There is no previous cytotoxicity report for PAE. Nevertheless, there are reports of cytotoxicity, anti-oxidant, and anti-inflammatory activities for other Pittosporum species.27–31 Madikizela et al. reported that the acetone extract of Pittosporum viridiflorum induced significant cytotoxicity with IC50 ranging from 3.16 to 26.87 µg/mL against MCF-7, Caco-2, A549, and Hela cells. 29 Noble cytotoxic triterpene glycosides with IC50 values within a range of 1.74–34.1 µM were also isolated from the seed extract of P. angustifolium. 27 These results align with our findings and indicate that the genus Pittosporaceae is a potential source of noble cytotoxic phytochemicals.
There is evidence for the cytotoxicity of RCE. A bicyclic hexapeptide with cytotoxic characteristics was elucidated from the root of this plant, with an IC50 of 0.012 µg/mL against P-388 leukemia cells. 32 Moreover, 11 cytotoxic anthraquinones and naphthohydroquinones were isolated from the root extract of the plant, and IC50 values of the most active compounds ranged from 0.12 to 9.7 µg/mL against V-79, P388, and KB cells. 33 Our findings lend further evidence for the broad-spectrum cytotoxic activity of RCE. Several studies have demonstrated the cytotoxic activities of plants of the genus Stephania against cancer cells34–37 and pathogens.38–40 Despite a thorough literature search, we could not find evidence for the cytotoxic activity of SAE against cancer cell lines. The isolation of cytotoxic bis-benzylisoquinoline alkaloids from other related plant species,35, 37 however, suggests that SAE could have a cytotoxic activity as reported in the present study.
The data regarding the apoptosis-inducing capacity of the crude plant extracts are scarce, and some findings align with the current study. A report indicates that crebanine, a compound isolated from Stephania venosa, induces apoptosis by increasing the cleavage of PARP in HL-60 and U937 cells. 41 Additionally, tetrandrine, an alkaloid isolated from the root of Stephania tetrandra was reported to have an apoptotic effect on human hepatoma cell lines. 42 These results are supportive of our findings, as SAE was observed to induce apoptosis as well as increase the expression of PARP-1. Furthermore, some lines of evidence support an apoptosis-inducing potential of chemicals isolated from the genus Erythrina. Flavonoids, flavanones, and isoflavones, which were isolated from different species of the genus were reported to induce apoptosis in human leukemia HL-60,43–45 and breast cancer MDA-MB231 cells. 46 To the best of our knowledge, this work is the first to report the induction of apoptosis by extracts from plants belonging to the genera Pittosporum, Acokanthera, Dovyalis, or Rubia.
As DNA damage is the main driving factor for cells to commit suicide, 47 we focused on determining proteins involved in DNA repair mechanisms, including γH2AX and PARP. Phosphorylation of H2AX is one of the most immediate cellular responses to double-strand DNA break (DSB) and is required to assemble DNA repair proteins at the damage site. 48 Thus, γH2AX is thought to be the most sensitive marker that may be used to analyze the DNA damage caused by various physical and chemical agents since it correlates well with DSB. 49 On the other hand, PARP1 is widely recognized for being activated upon binding to DNA single-strand and DSB. 50
Treatment of the cell lines with the extracts resulted in an enhanced expression of these DNA repair proteins. The extracts appear to produce this effect in a protein- as well as cell-line-dependent manner. In MCF-7 cells, DAE and SAE induced γH2AX, but PARP was induced by DAE and RCE. γH2AX expression was preferentially increased by ASE and SAE in PC-3 cells but not by ASE and DAE in SH-SY-5Y cells. The reason why an extract prefers a given cell line is a subject of interest for future research. However, the findings indicate that the extracts induce apoptosis by DSB of DNA. Indeed, there are reports of DNA damage for components of ASE,51, 52 the genus Erythrina, 53 and the genus Rubia.33, 54–56 This report is the first to demonstrate the DNA-damaging activity of SAE and DAE, calling for further work on this topic.
Conclusion
The study revealed that six extracts showed significant cytotoxicity in cell lines, with DAE having the most consistent activity. The mode of action studies suggested that the cytotoxicity induced was due to DNA double-strand breaks leading to apoptosis. The TLC analysis elicited that alkaloids may be potential candidates. These findings are crucial for developing countries like Ethiopia, where cancer is becoming a major health issue. Bioassay-guided phytochemical investigations and mechanistic characterizations are recommended to explore potential cancer chemotherapeutic agents from the most active plant extracts.
Footnotes
Abbreviations
ADC: Anti-body drug conjugate; ANOVA: Analysis of variance; ASE: Acokanthera schimperi extract; AS: Acokanthera schimperi; ATCC: American type culture collection; BCA: Bicinchoninic acid; DA: Dovyalis abyssinica; DAE: Dovyalis abyssinica extract; DMEM: Dulbecco’s Modified Eagle Medium; DMSO: Dimethyl sulfoxide; DNA: Deoxyribonucleic acid; DSB: Double strand break; EA: Erythrina abyssinica; EAE: Erythrina abyssinica extract; EB: Erythrina brucei; EBE: Erythrina brucei extract; FBS: Fetal bovine serum; IC50: The half-maximal inhibitory concentration; MCF-7: Human breast cancer cell line; PA: Pittosporum abyssinicum; PAE: Pittosporum abyssinicum extract; PARP-1: Poly-ADP-ribose polymerase-1; PBS: Phosphate buffered saline; PC-3: Human prostate cancer cell line; PI: Propidium iodide; RC: Rubia cordifolia; SA: Stephania abyssinica; SAE: Stephania abyssinica extract; SDS-PAGE: Sodium dodecyl sulfate-polyacrylamide gel electrophoresis; SH-SY5Y: Human neuroblastoma cell line; TLC: Thin-layer chromatography; TM: Traditional medicine; γH2AX: Phosphohistone H2AX.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The research was conducted after ethical approval was granted from the IRB of the College of Health Sciences, Addis Ababa University (Protocol no. 089/20 SoP). In this study, no humans or animals were recruited as test individuals. The human cell lines were not utilized for other assays than those described above. After each assay, the cell lines were discarded according to the national legislative requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is sponsored by Addis Ababa University, the University of Salamanca, and Madda Walabu University.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
