Abstract
Background
Saraca asoca (Roxb.) de Wild has been utilized in various commercial polyherbal preparations to treat gynecological disorders. Adulteration or replacement of similar raw pharmaceuticals is a widespread technique in its manufacture due to shortage.
Objectives
The current study aimed to determine the pharmacological efficiency of Kingiodendron pinnatum and Cynometra travancorica as alternatives for S. asoca in Arishta formulations.
Materials and Methods
Arishta was prepared using S. asoca and with the substitutes as per Ayurvedic Pharmacopoeia. The antiestrogenic activity, anti-inflammatory activity, and antioxidant activity of each Arishtam were evaluated.
Results
After administering each Arishtam of dose 400 µl/kg body weight orally, when compared to normal, the estrogen level of Arishtam prepared with C. travancorica showed the greatest decrease (131.45 ± 10.62 pg/ml) compared to the other two groups. Upon oral administration of each Arishtam of doses 200 and 400 µl/kg body weight, the edema in the animal group administered with the higher dosage of Ashokarishtam was reduced by 65.88% when compared to the control. Arishtam prepared with K. pinnatum and C. travancorica showed 60% and 67.06% of inhibition, respectively. Each Arishtam also improved the antioxidant levels in both blood and liver.
Conclusion
The study supports the use of K. pinnatum and C. travancorica as substitutes for S. asoca in Asokarishta and provides empirical support for Asokarishta in gynecological illnesses.
Keywords
Introduction
The Ayurvedic Materia Medica comprises a vast collection of herbal remedies, encompassing more than 600 medicinal plants and their corresponding therapeutic applications. 1 Prior to therapy, a person’s constitutional type must be assessed in Ayurveda, and there is a direct link between sickness and an individual’s metaphysical condition. 2 Plant formulations and blended plant extracts are preferred over isolated ones in traditional Indian medicine. Herbs are highly potent medicinal components and are referenced in the literature alongside the Vedas and Samhitas. 3 The bulk of Ayurvedic herbal preparations are polyherbal formulations, and they are often made in a variety of dose forms.4, 5
The scarcity of authentic raw materials is a major source of worry in Ayurvedic medication formulations. In most Indian systems of medicine, raw medicament is often associated with a single species as its botanical source. However, the continuous extraction of one particular species results in its scarcity or eventual extinction. As a result, many valuable medicinal plants have been overexploited, becoming reduced or even extinct in their native environments. 6 Because real plant materials are unavailable, a range of ways to increase plant or plant product production have been devised. It is imperative to address the scarcity of therapeutic plants through cultivation. However, increased utilization of artificial fertilizers and pesticides can lead to a reduction in the production of secondary metabolites and the accumulation of harmful chemicals in plant tissues. 7 The adulteration of medicinal preparations poses a substantial risk to the reputation of the Ayurvedic system of medicine, making it a critical concern for the Ayurvedic sector.8, 9 Nevertheless, herb substitution is imperative as over 300 significant medicinal plants have been classified as red-listed in India. Prior to utilizing plants as substitutes, it is imperative to thoroughly examine the chemical composition and pharmacological properties of the plants. These approaches may bring herbal medications up to World Health Organization (WHO) 10 standards and encourage Ayurvedic practice internationally.11, 12
Saraca asoca (S. asoca), a member of the Fabaceae family, is extensively utilized in Ayurvedic remedies. The species referred to as “Asokam” are highly favored in Ayurveda.13, 14 From antiquity to the present, Asoka has been one of the most important plants used. The bark of S. asoca is a crucial ingredient in “Asokaristam” and other medicinal preparations. Ayurveda recognizes this tree as a sacred plant of India, renowned for its efficacy in addressing gynecological issues, specifically menorrhagia. 14 Asoka is a holy tree in India that is well-known for its use in treating gynecological problems, particularly menorrhagia.15–17
Because of the plant’s broad-spectrum qualities, S. asoca has become overexploited, and the number of wild populations in the country has been decreasing over time. In India, the pharmaceutical sector consumes around 5,300 tons of bark every year. The yearly use of Asoka in India’s Ayurvedic medicinal business is estimated to be over 850 tons per year. 18 The International Union for Conservation of Nature and Natural Resources (IUCN) has designated the species as “globally vulnerable.” 19 Because this tree is scarce, the bark of other similar or unrelated species has been used as a substitute. The bark of Polyalthia longifolia is frequently employed for the purpose of adulterating pharmaceuticals, but Kingiodendron pinnatum (KPM) and Cynometra travancorica (CTA) are utilized as alternative options. KPM and CTA are enormous trees found in the evergreen forests of India’s Western Ghats. Although the species has a smaller natural population, it is frequently used as a substitute because of its enormous size and its capacity to acquire substantial amounts of bark in comparison to an Asoka tree. Historically, tribes have used an oleo-gum-resin derived from KPM to remedy gonorrhea, catarrhal ailments affecting the genito-urinary and respiratory systems, as well as wounds in elephants. 20 The toxicological and various pharmacological properties of CTA were also proven. 21 In this regard, the study sought to determine the acceptability of KPM and CTA as substitutes for Asoka in the preparation of Arishta.
Materials and Methods
Preparation of Arishtam
S. asoca, KPM, and CTA were collected from different parts of the Western Ghats, like Nelliyampathy, Thrissur, Wayanad, and Munnar, and the plant voucher specimens are kept at the Kerala Forest Research Institute (KFRI) Herbarium (No. 4295, 4297, and 4299). Arishta with S. asoca (AKA), K. pinnatum (KPA), and C. travancorica (CMA) were prepared as per the Indian Ayurvedic Pharmacopoeia. Briefly, the main component’s chopped bark was combined with the minor elements from the other 14 plants, heated in water until the volume was decreased to a quarter, and then the mixture was removed from heat. After removing the particles, the liquid was left to ferment for 30 days in a sealed China clay container. The container included sugar sweets and Woodfordia fruticosa flowers. After a period of 30 days, the Arishta underwent filtration and was after that utilized for the purpose of research. 22
Animals
Purchases were made at the Small Animal Breeding Station (SABS), College of Veterinary, Kerala Veterinary and Animal Science University (KVASU), Thrissur, Mannuthy, Kerala, for female Swiss albino mice (8–10 weeks old, 25–35 g) and Wistar female rats (14 days old). The standard operational parameters for the animals’ care were a temperature range of 23–25℃, a relative humidity range of 60–70%, and a light/dark cycle of 12 hours. In addition, the rats were provided with a regular rat diet from Sai Durga Feeds and Foods and had unrestricted access to water. The Institutional Animal Ethics Committee approved (IAEC/ACP/076/22) the use of animals in all testing at Al Shifa College of Pharmacy.
Experimental Design for Anti-inflammatory Activity
Swiss albino mice were made into nine groups with six animals in each group (n = 6). Group 1 remained as control, Group 2 received carrageenan (CRNN) (0.02 ml), Group 3 received standard diclofenac (10 mg/kg), and Groups 4, 5, 6, 7, 8, and 9 received 200 and 400 µl/kg body weight of each Arishtam orally for 5 days. On the 6th day, exactly 1 hour after the animals received Arishtam, they were injected with 0.02 ml of freshly prepared 1% CRNN in 0.1% carboxymethyl cellulose for each group (except Group 1) in the sub-plantar region of the right paw 23 to induce inflammation. The thickness of the paw was determined using a digital plethysmometer with a camera (VJDP-01) before (0th hour) and after CRNN injection, and thereafter at the 1st, 2nd, 3rd, 4th, 5th, and 6th hours.
Experimental Design for Antiestrogenic Activity
The study utilized female Wistar rats that were 2 weeks old. A total of 30 animals were divided into 5 groups, with each group containing 6 animals. The estrogen level in all groups, except for the normal and vehicle control groups, was increased by injecting 20 µg/kg body weight of estradiol in 0.05 ml of propanediol intraperitoneally for 5 consecutive days. 24 The animals in the second group were retained as a control group and were not given any drugs. The 3rd group received AKA at 400 µl/kg body weight, and the 4th and 5th groups received KPA (400 µl/kg body weight) and CMA (400 µl/kg body weight) orally for 5 alternative days along with the injection of estradiol. Radioimmunoassay (RIA) was used to assess the blood estrogen level on the 10th day of medication or 24 days after delivery.
Experimental Design for In Vivo Antioxidant Activity
The study used male Swiss albino mice. Animals were euthanized, and their livers were extracted. Blood samples were collected from the heart using heparinized tubes for biochemical and hematological analysis. This was done following the oral administration of AKA, KPA, and CMA for 5 consecutive days. The supernatant of the samples, which were subjected to centrifugation at 1,000 × g for 15 minutes, was discarded. The erythrocytes that settled at the bottom were washed three times with pH 7.4 phosphate buffer saline. Hypotonic phosphate buffer was used to lyse a known quantity of red blood cells. The lysates were retrieved after the solutions were centrifuged at 3,000 × g for 15 minutes to remove the red blood cell debris. The activities of catalase and superoxide dismutase (SOD), 25 as well as the amount of reduced glutathione (GSH) 26 in the blood and liver, were measured.
Statistical Analysis
For the data, we used IBM SPSS Statistics 21 with the Graph Pad Prism trial version and presented in the format of the mean value ± SD. Dunnett’s test was employed to assess disparities among group means following a one-way analysis of variance. Statistical significance was determined when the value of p < 0.05.
Results
Anti-inflammatory Activity
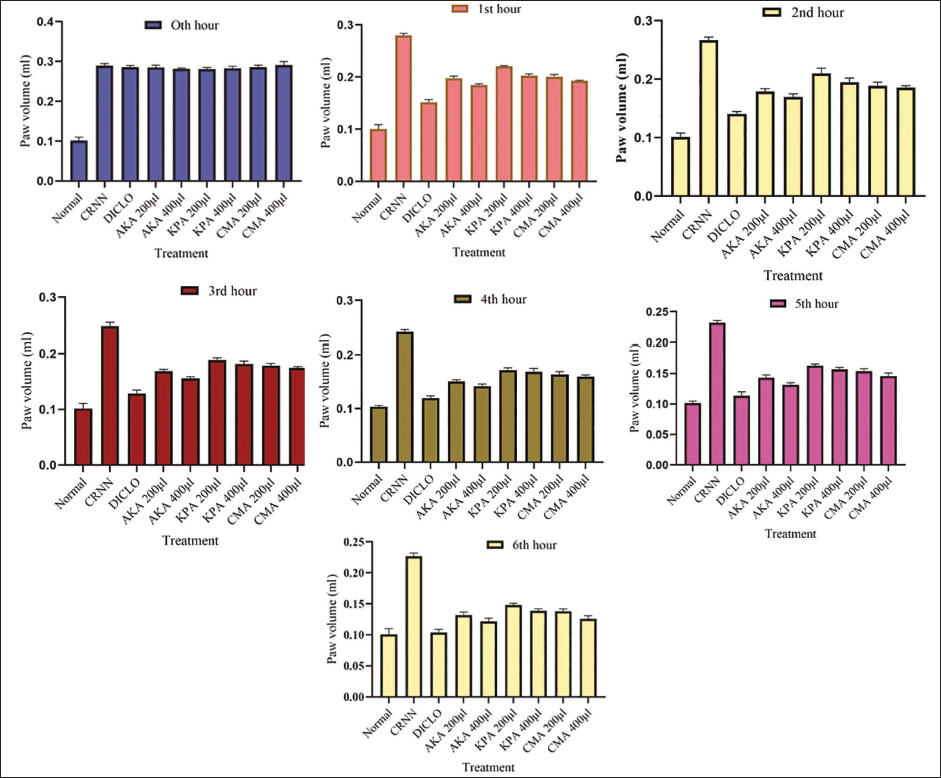
Animals were treated with AKA, KPA, and CMA of doses 200 and 400 µl/kg. In mice, the injection of CRNN into the paw caused a localized inflammatory reaction, reaching its maximum swelling during the 3rd hour. After the 3rd hour, the edema in the animal group administered with the higher dosage of AKA was reduced by 65.88% when compared to the control. KPA and CMA showed 61% and 68.07% of inhibition, respectively (Figure 1).
Effect of Arishtam Prepared with Saraca asoca (AKA), Arishtam Prepared with Kingiodendron pinnatum (KPA), and Arishtam Prepared with Cynometra travancorica (CMA) on Acute Inflammation in Swiss Albino Mice. Values are Mean ± SD (n = 6).
Antiestrogenic Activity
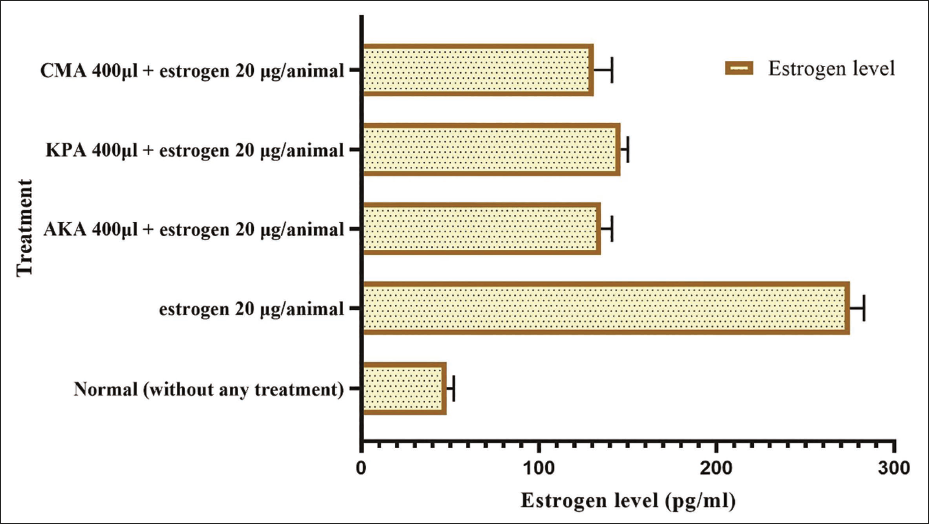
Administering a dose of 400 µl/kg of each Arishtam was reported to effectively prevent elevated estrogen levels in female Wistar rats. On the 10th day of therapy, each animal’s blood was drawn, and serum was collected. Normal rats had an estrogen level of 48.52 ± 4.42 pg/ml. When estradiol was injected, the level jumped to 275.32 ± 8.42 pg/ml. Estrogen levels in Arishtam-treated groups were considerably lower. The CMA-treated group showed the greatest decrease (131.45 ± 10.62 pg/ml) in estrogen level (Figure 2).
Effect of Arishtam Prepared with Saraca asoca (AKA), Arishtam Prepared with Kingiodendron pinnatum (KPA), and Arishtam Prepared with Cynometra travancorica (CMA) on Estrogen Level. Values are Mean ± SD; n = 6, p < .001.
In Vivo Antioxidant Activity
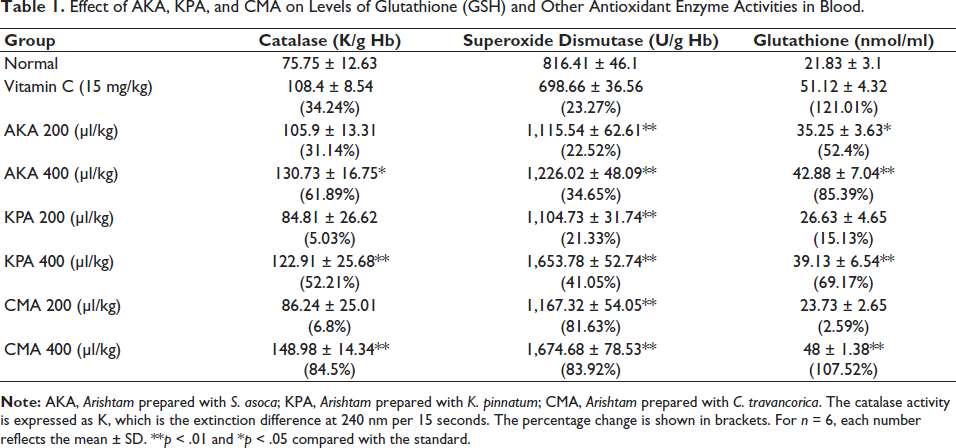
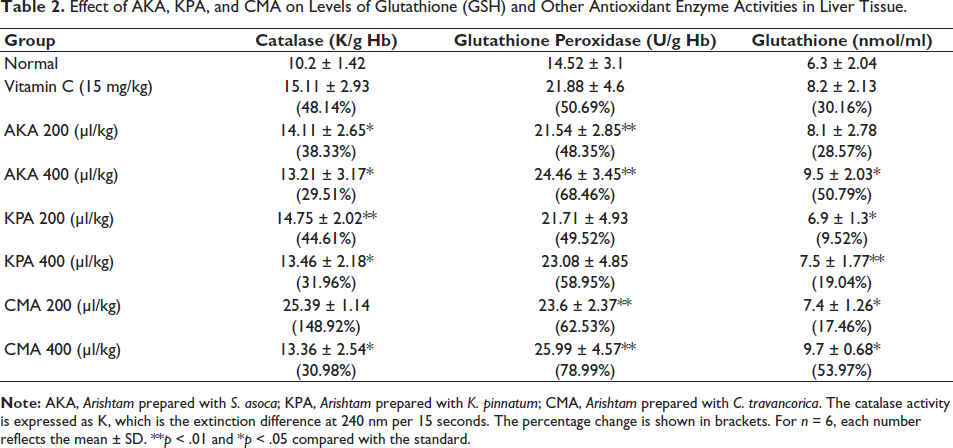
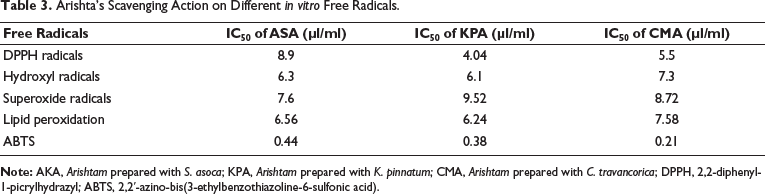
The effects of Arishta on antioxidant molecules in the blood and liver of mice were observed for 30 days after administration. The results are presented in Tables 1 and 2. It was shown that the concentrations of GSH, catalase, glutathione peroxidase (GPx), and SOD were all increasing significantly. The administration of 200 and 400 µl/kg of AKA resulted in a 22.52% and 34.65% rise in the activity of SOD, respectively. In comparison, the same doses of KPA and CMA showed activity increases of 21.33% and 41.05% and 81.63% and 83.92%, respectively. With the administration of 400 µl/kg AKA, KPA, and CMA, the percentage rise of GPx in liver tissue was determined to be 85.39%, 69.17%, and 107.52%, respectively. With 200 and 400 µl/kg, catalase activity in the blood was shown to be considerably enhanced. The percentage increases for AKA, KPA, and CMA catalase activity were 61.89%, 52.21%, and 84.5%, correspondingly. The level of GSH in liver tissue was found to have increased to 48.35% and 68.46% in the animals treated with the respective concentrations of AKA, 21.33% and 58.95% for KPA, and 62.53% and 78.99% for CMA. Table 3 represents Arishta’s scavenging action on different in vitro free radicals.
Effect of AKA, KPA, and CMA on Levels of Glutathione (GSH) and Other Antioxidant Enzyme Activities in Blood.
Effect of AKA, KPA, and CMA on Levels of Glutathione (GSH) and Other Antioxidant Enzyme Activities in Liver Tissue.
Arishta’s Scavenging Action on Different in vitro Free Radicals.
Discussion
The two main gynecological issues that affect women, dysmenorrhea, and abnormal uterine bleeding share comparable physiological symptoms that are brought on by acute inflammatory processes. The pathophysiology is believed to be regulated by local factors such as prostaglandin, inflammatory cytokines, vascular endothelial growth factors, and different matrix metalloproteinases. 27 Hormonal contraceptives have been proposed as a means of managing discomfort and irregular bleeding. Estrogenic supplementation is recommended for dysmenorrhea, while antiestrogenic or progesterone options are advised for abnormal uterine bleeding. Nevertheless, there is purportedly a scarcity of clinical data to substantiate their effectiveness. Consequently, nonsteroidal anti-inflammatory drugs (NSAIDs) remain the primary choice of treatment. 28 Ayurvedic medicine employs AKA to treat several conditions associated with the female reproductive system, such as dysmenorrhea and irregular uterine hemorrhage. 29 A study in 2012 found that AKA is a safe, effective alternative to contemporary medications for psychological and physical issues associated with menopausal syndrome. 30 Our earlier investigations assessed and contrasted the methanolic bark extract of the same plant’s antiestrogenic, anti-inflammatory, antioxidant, and toxicological evaluations.21, 31, 32 In addition to greatly lowering the blood level of estrogen in rats, AKA, KPA, and CMA has also been shown to significantly decrease chronic inflammation in mice.
The body’s level of estrogen and progesterone controls a woman’s menstrual cycle. When progesterone fails to prepare the uterine endometrium for implantation, prostaglandin levels rise and progesterone levels decline. The pathophysiological processes of dysmenorrhea, which are characterized by profuse bleeding, spasm, and discomfort, have been linked to elevated prostaglandin levels. 33 AKA’s function is to inhibit prostaglandin formation or serve as a progesterone antagonist. The major component of Asokarishta, S. asoca bark, has been shown to suppress cyclooxygenase activity and prostaglandin synthesis with no harmful effects. 34 Asokarishta exhibits anti-inflammatory effects in animal models in the current study. This information implies that Asokarishta’s therapeutic effectiveness may be due to the suppression of prostaglandin and cyclooxygenase. KPA and CMA have the same anti-inflammatory properties as Asokarishta. Additionally, progesterone and estrogen have been reported to interact negatively. Despite the fact that several reports back up the phytoestrogenic properties of the plant S. asoca and its preparation Asokarishta, some reports suggest vice versa. 35 There has to be more research to determine whether these phytoestrogens have estrogen-like ability to block progesterone’s action.
S. asoca extract has been shown to have antioxidant properties in the past by a number of researchers.36–38 From these findings, it is anticipated that KPM and CTA, Arishta’s two main constituents, would have the same antioxidant, anti-inflammatory, and antiestrogenic properties as those of S. asoca, which may contribute to its clinical effectiveness.
Conclusion
At some point, the widespread and unregulated use of Asoka bark, along with its unprofessional extraction methods, has resulted in a significant scarcity of authentic raw medication. Consequently, this scarcity has caused increased costs and widespread contamination of the substance. Overall, the research sheds light on the mechanism behind how the Asokarishta treats gynecological problems. The pharmacological properties of KPA and CMA are comparable to those of Asokarishta. As a result, the study offers a rationale for using KPM and CTA in Arishta formulations. Due to the relative affordability of alternatives, this might lower the cost of production.
Footnotes
Abbreviations
Acknowledgments
The authors thank all faculties and nonteaching staff of Al Shifa College of Pharmacy, Department of Pharmacy, Annamalai University and KFRI for their support and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
With CPCSEA approval, all relevant animal studies are conducted at Animal House, Department of Pharmacology, Al Shifa College of Pharmacy (IAEC/ACP/076/22).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
