Abstract
Background
The recurrence of atherosclerosis is still a global problem. Unfortunately, the drugs currently being used are reported to have resistance.
Objectives
To prevent the recurrence of atherosclerosis disease, this study developed benzoxazinone derivatives as an antiplatelet agent: 2-phenyl-3,1-benzoxazine-4-one (BZ1) and 2-(4′-chlorophenyl)-3,1-benzoxazin-4-one (BZ2).
Materials and Methods
The target compounds were synthesized through acyl nucleophilic substitution from acyl chloride and anthranilic acid using a base catalyst in triethylamine. This compound was then given to mice at three different doses to perform bleeding-time and clotting-time tests, using acetosal as a comparison. In silico, the binding energy between the test compound and the cyclooxygenase 1 enzyme was tested using Molegro Virtual Docker version 5.5 (MVD 5.5). The pharmacokinetic parameters were predicted using pharmacokinetics and chemistry through space mapping (pkCSM).
Results
The synthesis provided 83% yield. The biological assay at a dose of 20 mg/kg body weight (BW) BZ1 showed a higher bleeding time (361 s) than acetosal (324 s), whereas BZ2 was lower (179 s). The molecular analysis revealed that the compound had higher activity than aspirin. The absorption, distribution, metabolism, excretion, and toxicity (ADMET) showed that the compound posed high intestinal absorption.
Conclusion
This research concluded that both of the synthesized compounds have the potential and prospective to be developed as antiplatelet agents.
Keywords
Introduction
Around the globe, cardiovascular disease (CVD) is the number one cause of mortality. In 2016, almost one-third of deaths worldwide were due to CVD. 1 Platelets are one component of blood that play an important role in maintaining cardiovascular integrity. 2 This role is carried out by regulating bleeding processes and clot formation. 3
However, uncontrolled integration of platelets can lead to dangerous and life-threatening arterial blood vessel blockage.4–8 Therefore, antiplatelet compounds are considered a significant tool for treating or preventing thrombotic CVD.9–11 Antiplatelet compounds such as aspirin (acetylsalicylic acid), clopidogrel or ticlopidine and anticoagulants such as warfarin are the two main groups of drugs used in standard protocols for prophylaxis and therapy for venous thrombosis and a reduction in the risk of myocardial infarction.12–15
Aspirin works through the inhibition of the enzyme cyclooxygenase I (COX-1), which is a mediator for the transformation of arachidonic acid into thromboxane A2 (TXA2).16–18 Unfortunately, a study of 10,729 patients showed that aspirin resistance in CVD was 24%. 19 Recent research reports the high expression of multidrug 4 resistance protein or multidrug resistance protein 4 (MRP4) on platelets from aspirin users in a short time.20–23 MRP4 is an adenosine triphosphate (ATP) binding transporter that acts as a unidirectional pump for organic anion compounds such as acetylsalicylate (acetosal), and is associated with acetosal expulsion from platelets, as well as a decrease in pharmacological activity in COX-1 inhibition.20, 24 The lack of optimal inhibition of platelets causes incomplete thromboxane formation suppression, which is at risk of increasing cardiovascular disorders. 25 This is what encourages researchers to develop alternative compounds instead of aspirin.
For the past few years, benzoxazinone has become one of the most attractive compounds for development as a new prospective antiplatelet.26, 27 A series of 2,8-disubstituted benzoxazinones were also evaluated for inhibition of superoxide anion generation and inhibition of neutrophil elastase and suggested that among them possessed aspirin-like activity. 28
In this study, we intend to explore the potential of phenyl benzoxazinone and phenyl chloro benzoxazinone as antiplatelet in vivo by examining bleeding time in mice, evaluating the interaction of compounds with cyclooxygenase 1 enzyme through molecular docking using Molegro Virtual Docker 5.5 (MVD 5.5), as well as investigating the pharmacokinetic profile in silico using the pharmacokinetics and chemistry through space mapping (pKCSM) web tool. The benzoxazinone compounds were prepared by substituting nucleophilic acyl from anthranilic acid and benzoyl chloride. Structural identification was carried out through proton nuclear magnetic resonance (NMR) and Fourier transform infrared (FT-IR) spectra.
Materials and Methods
Chemistry
Materials Used
The materials used in this study included benzoyl chloride, p-chlorobenzoyl chloride, pyridine, n-hexane, ethyl acetate, chloroform, ethanol, natrium carbonate, anthranilic acid, thin-layer chromatography (TLC) plate silica gel 60 GF254 (E. Merck), and gas N2. The equipment included a melt-temperature-electrothermal, Barnstead electrothermal, Spectrophotometer IR M 500 Buck Scientific, Spectrometer FT-NMR Jeol Resonance, 400 MHz. Rotavac Laborta 4000 Heidolph Instrument.
Procedure
0.005 mole of anthranilic acid was added into each 0.01 mole of acyl chloride {benzoyl chloride to synthesize 2-phenyl-3,1-benzoxazine-4-one (BZ1), and p-chlorobenzoyl chloride to synthesize 2-(4′-chlorophenyl)-3,1-benzoxazin-4-one (BZ2)} in a round of bottom flash. 5 mL triethylamine and 0.01 mole of pyridine were added into each flash, covered by a CaCl2 tube, and stirred under nitrogen conditions at room temperature. The completion of the reaction was determined using the TLC method. The product was neutralized using 10% aqueous NaHCO3 and washed using Aquadest. Each benzoxazinone derivative was then recrystallized from ethanol, the purity was determined by TLC and melting point method, and the chemical structure was identified by using FT-IR spectrophotometry and proton nuclear magnetic resonance (H-NMR) spectrometry. 29
Bleeding Time Evaluation
The ethics committee of the Faculty of Veterinary Medicine, Universitas Airlangga, has approved all experiment protocols with letter number 2.KE.144.8.2018. Mice were divided into eight treatment groups: the control positive or aspirin group, the control negative or placebo group, and the BZ1 and BZ2 test groups, each in three doses. Aspirin and benzoxazinone are orally given according to the dose as a suspension in 5% (period/vol) of Arabic gum in saline solution. The tip of the mice’s tail is slashed as long as 2 mm, and the blood that comes out is absorbed on the filter paper every 10–15 s, and the time taken from incisions until the blood stops is recorded as bleeding time. The bleeding time of mice in eight groups was recorded before treatment and after a week of oral administration of the compound. The results obtained were written in seconds as the average bleeding time ± standard deviation (SD) of five mice in each group. 30
Molecular Docking
Tools
Advanced micro devices (AMD) A6 Vision has a specification central processing unit (CPU) @ 1.4 GHz and 4 GB of random access memory (RAM), and the software ChemBioDraw version 11, MVD Ver.5.5.
Receptor
Receptor ovine prostaglandin endoperoxide H synthase-1 (oPGHS-1) downloaded from Protein Data Bank (
Ligand Preparation
The benzoxazinone derivative was prepared as ligand molecules by drawing a two-dimensional structure in ChemBioDraw version 11 software. This two-dimensional ligand was then converted to a three-dimensional ligand in ChemBio3D version 11 software. The most stable minimum energy of this 3D structure was calculated using MMFF94 and saved as a Molecular Design Limited (MDL) Molfile file type (.mol). 31
In Silico Pharmacokinetic Evaluation
Computational pharmacokinetic profile calculation was conducted using the pKCSM web tool. Previously, the structure of the tested compounds, BZ1 and BZ2, was drawn using ChemBioDraw version 11 software. Then, this two-dimensional chemical structure was converted into the SMILEY structure by the SwissADME online tool. Using the pKCSM online tool, the SMILEY structure of BZ1 and BZ2 was then entered, followed by computational calculation of the physicochemical profile, absorbance, distribution, metabolism, excretion, and toxicity. 32
Results
Chemistry
BZ1
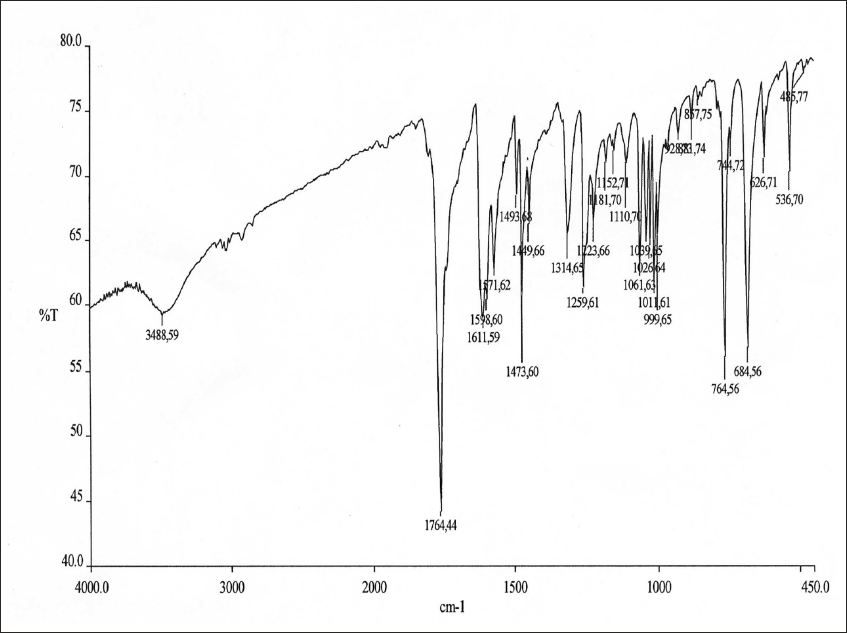
White needle crystal, 122–124°C, yield 83%. Identification using spectroscopy method showed: Spectrum IR (KBr; v cm−1) 1,764 (−C=O lactone); 1,598 (−C=C− sp2); 1,473 (−C=N−); 1,259 (CAr-O-C−). The FT-IR spectra profile of compound BZ1 is presented in Figure 1. Spectrum 1 H-NMR [90 MHz; CDCl3; tetramethylsilane (TMS); δ ppm]: 7.43–7.58 (m, 4H-Ar); 7.71–7.83 (m, 2H-Ar); 8.19–8.36 (m, 3H-Ar).
Fourier Transform Infrared (FT-IR) Spectra of 2-Phenyl-3,1-Benzoxazine-4-One (BZ1).
BZ2
White needle crystal, 188–189°C, yield 83%. Spectrum 1 H-NMR (400 MHz; CDCl3; TMS; δ ppm): 7.46–7.54 (m, 3H-Ar); 7.63–7.67 (d, 2H-Ar, J = 16 Hz); 8.21–8.24 (m, 3H-Ar).
Bleeding Time Evaluation
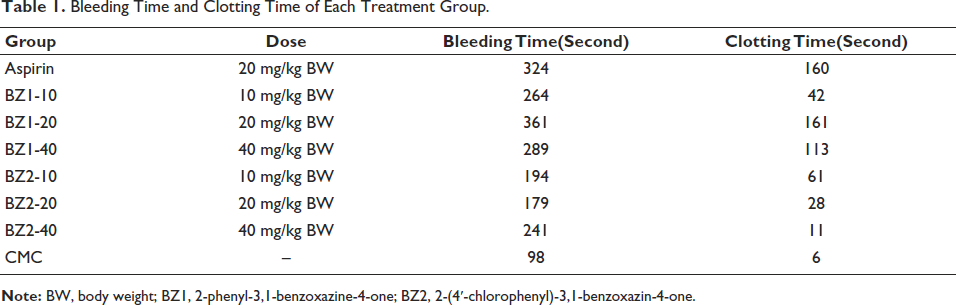
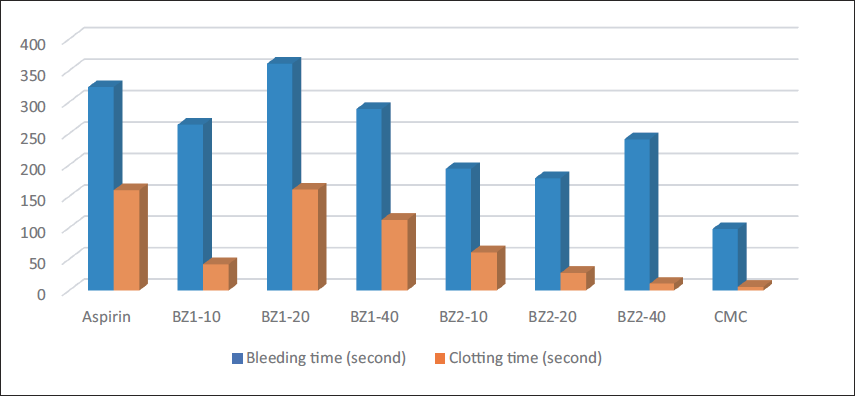
Table 1 summarizes each experimental group’s mean bleeding and clotting times. Both BZ1 and BZ2 have higher bleeding times compared to the negative control. Figure 2 also shows that BZ1 at a dose of 20 mg/kg body weight (BW) shows a longer bleeding time than aspirin (positive control).
Bleeding Time and Clotting Time of Each Treatment Group.
Bleeding Time and Clotting Time of Each Treatment Group.
Furthermore, one-way analysis of variance (ANOVA) statistical analysis using SPSS 22 was conducted to see the differences between treatment groups in bleeding and clotting times. From the test, the results of the significance value were 0.000 for both bleeding time and clotting time; hence, it can be concluded that the difference was significant.
Molecular Docking
Table 2 shows the ranking scores of the compounds BZ1, BZ2, and aspirin against the COX-1 receptor. The BZ1 and BZ2 compounds have a lower rerank score than aspirin.
Reranked Score of the Test Compound Against the Cyclooxygenase I (COX) Receptor 1.
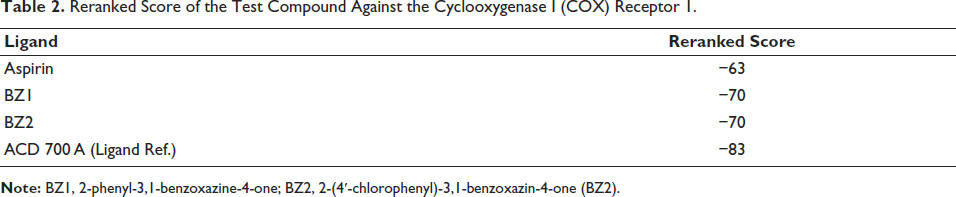
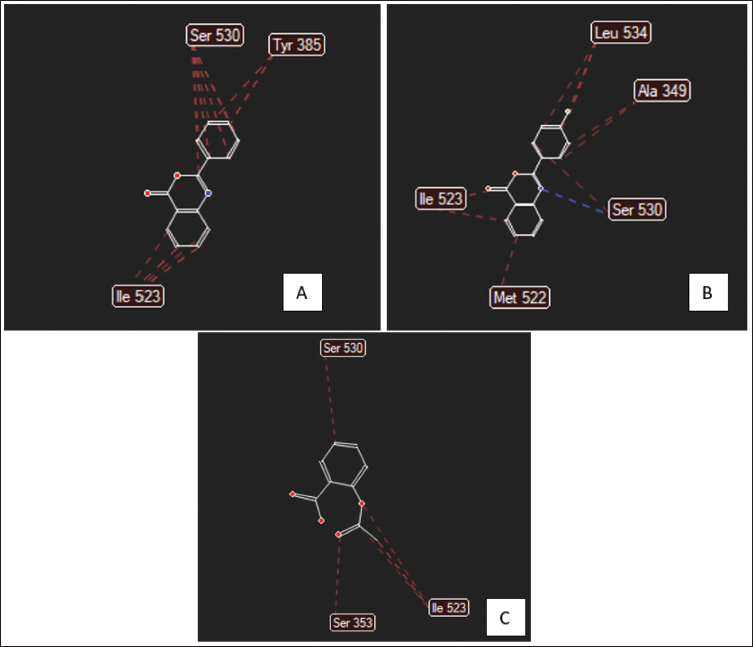
The interaction map between BZ1, BZ2, and aspirin compounds against the COX-1 receptor can be seen in Figure 3. Figure 3(A) shows that BZ1 has steric interactions of both phenyl groups with Ser 530, Tyr 385, and Ile 523. Figure 3(B) states that BZ2 has hydrogen bonds between the nitrogen of the benzoxazinone core and hydrogen of ser 530 and has steric interactions between the phenyl group and Leu 534, Ala 349, Ile 523, and Met 322. Additional steric interaction with Leu 534 was also contributed by the chloro group. In Figure 3(C), it can be seen that there are steric interactions between the phenyl group of aspirin and Ser 353. The acetyl aspirin group also sterically interacted with Ser 530 and Ile 523.
2D Interaction Map of the Test Compound [A = 2-Phenyl-3,1-Benzoxazine-4-One (BZ1)], (B = 2-(4′-Chlorophenyl)-3,1-Benzoxazin-4-One (BZ2)], (C = Aspirin) to the Cyclooxygenase I (COX-1) Receptor.
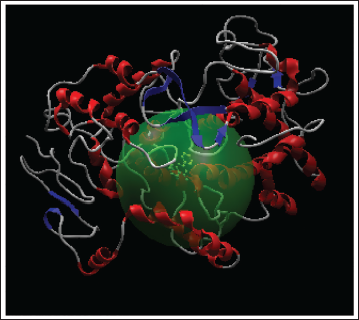
The test compounds (BZ1 and BZ2) interact against the target protein in cavity 3. This is clearly seen in Figure 4.
Interaction of 2-Phenyl-3,1-Benzoxazine-4-One (BZ1) (A) and 2-(4′-Chlorophenyl)-3,1-Benzoxazin-4-One (BZ2) (B) Compounds in Cavity 3 Protein 1U67.

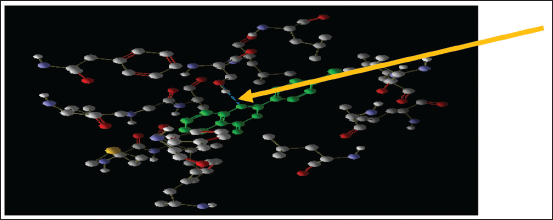
Figure 5. shows how BZ1 interacts with this protein in its secondary structure. Figure 6 shows the target protein’s hydrogen bonds between BZ2 and ser 530.
Interaction of 2-Phenyl-3,1-Benzoxazine-4-One (BZ1) Compounds in Cavity 3 1U67 Protein in the Secondary Structure.
Hydrogen Bonds Between 2-(4′-Chlorophenyl)-3,1-Benzoxazin-4-One (BZ2) and ser 530 in the Target Protein.
In Silico Pharmacokinetic Evaluation
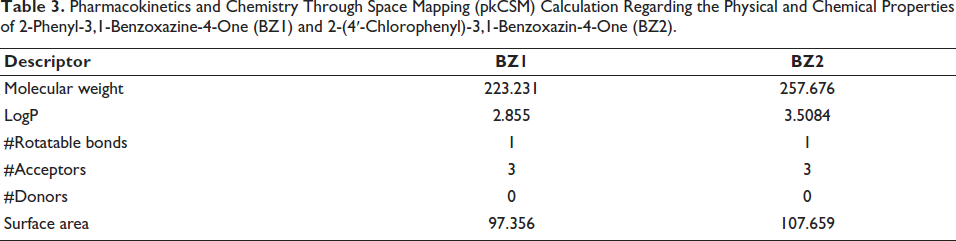
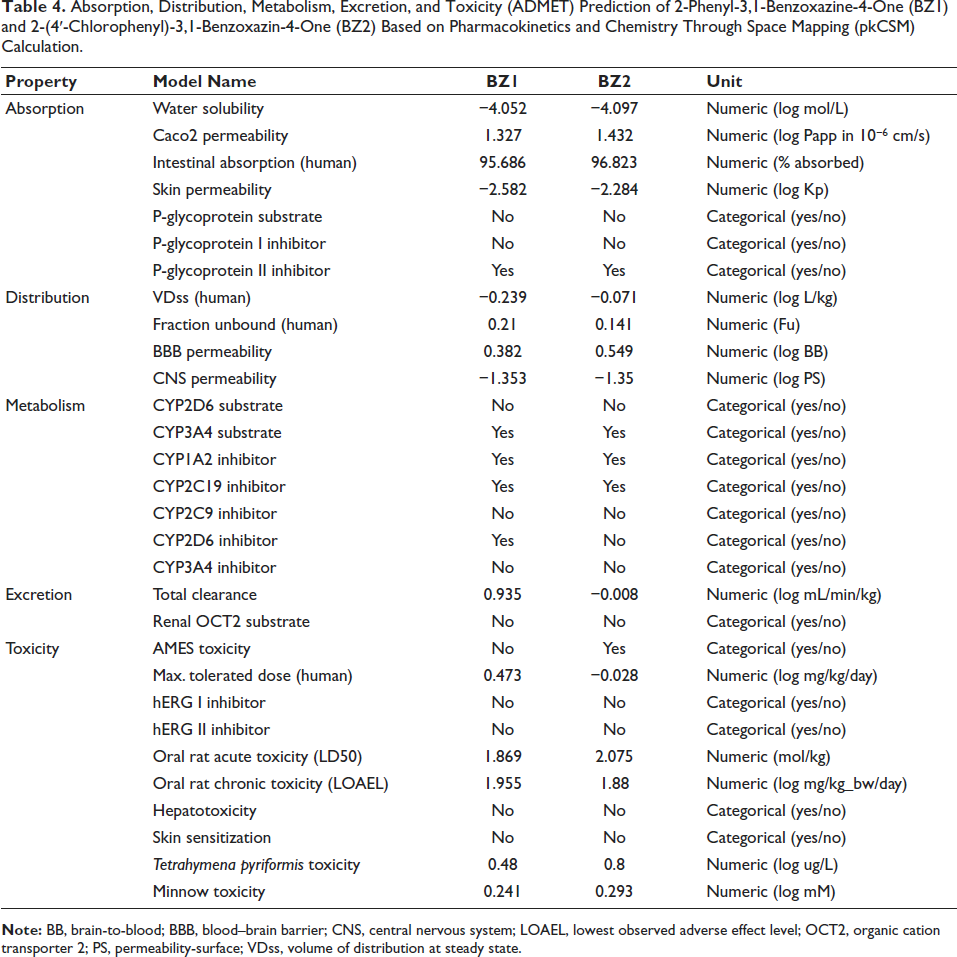
Table 3 shows the results of pkCSM calculations for chemical physics properties, showing that BZ1 and BZ2 meet Lipinsky’s Rule of Five. Table 4 is the absorption, distribution, metabolism, excretion, and toxicity (ADMET) prediction based on the pkCSM calculation estimating that BZ1 and BZ2 have sound absorption.
Pharmacokinetics and Chemistry Through Space Mapping (pkCSM) Calculation Regarding the Physical and Chemical Properties of 2-Phenyl-3,1-Benzoxazine-4-One (BZ1) and 2-(4′-Chlorophenyl)-3,1-Benzoxazin-4-One (BZ2).
Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) Prediction of 2-Phenyl-3,1-Benzoxazine-4-One (BZ1) and 2-(4′-Chlorophenyl)-3,1-Benzoxazin-4-One (BZ2) Based on Pharmacokinetics and Chemistry Through Space Mapping (pkCSM) Calculation.
Discussion
Chemistry
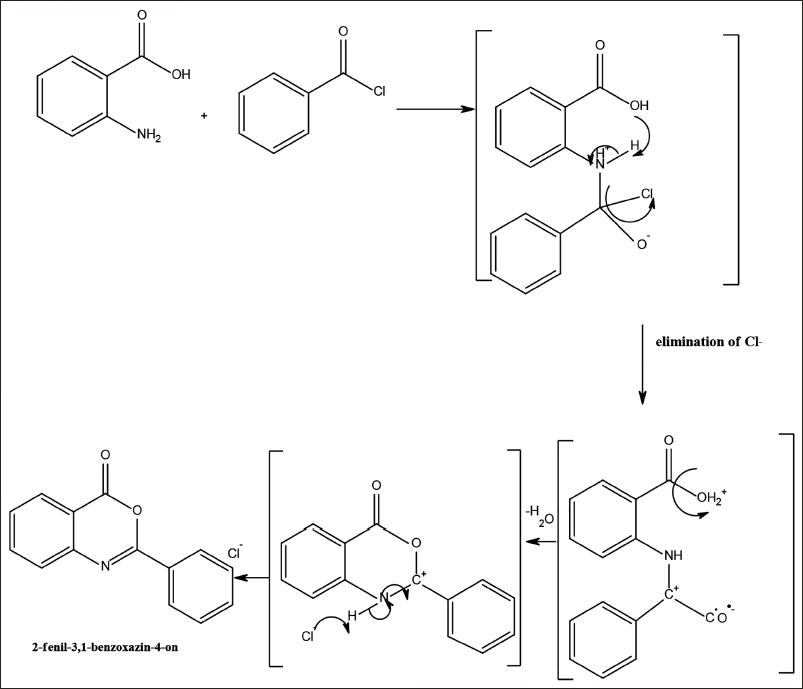
Benzoxzinone is a lactone that can be prepared through acyl nucleophilic substitution of acyl chloride and anthranilic acid. The nucleophile attacks the carbonyl group of acyl chloride, followed by eliminating –Cl as a leaving group. Then, H of the amino group is transferred into –OH of anthranilic acid and leaves as a water molecule. Since it lost the –OH group, the electron of carbonyl of anthranilic acid started to be delocalized and became more vulnerable to the attack from O of acyl chloride. This leads to intramolecular esterification and the formation of benzoxazine (Figure 7).
Reaction Mechanism for the Formation of Benzoxazinone.
From the IR spectrum, 2-phenyl-3,1-benzoxazin-4-on (Figure 1) compound can be seen that compound 2-phenyl-3,1-benzoxazin-4-on has specific groups of functional bonds that give different peaks at wave numbers. The wave numbers obtained are then compared with the wave numbers in the reference. 22 The functional groups and wave numbers include C=O lactone at wave number 1,764 cm−1 (library 1,795–1,760 cm−1), C=C sp2 at wave number 1,598 cm−1 (library 1,600–1,400 cm−1), C=N at wave number 1,473 cm−1 (library 1,600–1,430), and CAr-OC at wave number 1,259 cm−1 (literature 1,275–1,200 cm−1).
The spectrum of 1 H-NMR 2-phenyl-3,1-benzoxazin-4-on compound shows a multiplet peak that cannot be adequately separated because of its adjacent location. The comparison of integration is 4:2:3. In chemical shifts, δ 7.58–7.43 ppm, δ 7.83–7.71 ppm, and δ 8.36–8.19 ppm. This shift value is in accordance with the theoretical chemical shift of the 6.9–9.0 ppm aromatic ring. 33 Correspondingly, the NMR spectra for compound BZ2 also showed three signals with 3:2:3 integration, with a chemical shift and multiplicity corresponding to BZ2.
Bleeding and Clotting Time Evaluation
The bleeding time and clotting time data presented in Table 1 represent the average difference between the data before and after treatment for each group.
The graph above reveals that BZ1 dose 20 mg/kg BW showed the highest activity, which was an increase in bleeding time by 361 s, higher than the positive control group, aspirin 20 mg/kg BW of 324 s. The increase in clotting time of BZ1 at a dose of 20 mg/kg BW was the same as aspirin, which was 161 and 160 s, respectively. The bleeding time and clotting time of BZ1 at a dose of 40 mg/kg BW were smaller compared to the 20 mg/kg BW dose group, which were 289 and 113 s. The smallest dose of BZ1, 10 mg/kg BW, increased in 264 s of bleeding time and 42 s of clotting time, longer than the negative control group, which were 100 and 6 s. Meanwhile, BZ2 compounds have lower activity compared to BZ1 and aspirin at all doses but are higher than the placebo group or negative controls. The significantly lower activity of BZ2 under BZ1 is interesting because, based on its molecular docking data, both test compounds have the same rerank score. This can be caused by the differences in physicochemical properties, where the logP of the BZ1 compound is 2.855, while BZ2 is 3.508 (Table 3). Considering the route of drug administration in this experiment is conducted orally, it can be assumed that the logP value is important in determining. 34
Molecular Docking
Redocking was conducted to validate MVD software Ver.5.5 to be used as a docking method for virtual screening of the tested compounds, benzoxazinone. Validation is done by redocking the cocrystal ligand ACD 700_A (5Z,8Z,11Z)-trideca-5,8,11-trienoate. The redocking acceptance parameter is the root mean square deviation (RMSD) value <2.5 Å. The RMSD value showed the suitability of the ligand coordinates from the results of crystallography compared to the ligand coordinates that are redocking with MVD software Ver.5.5.35, 36
The RMDS value of the ACD 700_A ligand process with the oPGHS-1 receptor is 1.36 Å. The RMSD value from the redocking results proves that the PDB files that were downloaded and the software MVD Ver.5.5 can be used as a docking method for the virtual screening of benzoxazinone compounds compared to antiplatelet (aspirin).
Table 2 showed that the reranked score of the tested compounds, BZ1 and BZ2 approached the reference ligands, which were −70, −70, and −83, respectively. Compared to aspirin, which has a rerank score of −63, it can be predicted that the bond between BZ1 and BZ2 to COX-1 is more stable. A rerank score shows the energy bond between the ligand and the receptor. The lower the reranked score, the more stable the bond between ligand and receptor. 31 This reranked score data state that the test compound has the potential to be developed as a new antiplatelet. This is in line with the results of previous studies, which evaluated the binding of 22 cinnamate derivatives to the COX-1 enzyme to measure its potential as a new antiplatelet. 32
In Silico Pharmacokinetic Evaluation
The identification of the properties of ADMET is an essential initial step in determining the success of a new compound. Testing the ADMET properties can be done in silico using a computer program. Many in silico approaches have been developed to predict pharmacokinetics properties and compound toxicity, including data-based approaches such as quantitative structure and activity relationship (QSAR), similarity searches, and 3D QSAR for structure-based methods such as ligand-protein docking and pharmacophore modeling. 37
Apart from the above methods, another method for determining pharmacokinetic properties is the concept of graphical structural signatures called pkCSM. The pkCSM approach is available in a user-friendly web platform and is freely available. Through testing with the online pkCSM program, it can help find a balance between potency, safety, and pharmacokinetic properties. 37
The first parameter pkCSM tests is chemical physics properties, including molecular weight, log P, donor hydrogen, and acceptor hydrogen. This parameter is the determining factor in whether the drug is predicted to be able to be absorbed through passive diffusion in Lipinsky’s Rule of Five. Drugs with a molecular weight of less than 500 are thought to be able to enter through the gaps between cells. Likewise, the lipophilicity shown by logP is less than 5, allowing the drug compound to penetrate the membrane in the digestive tract.38, 39 Based on the results in Table 3, the BZ1 and BZ2 compounds meet the Lipinsky rule, so it is predicted that they will be well absorbed through passive diffusion.
In line with this, the prediction of absorption of BZ1 and BZ2 compounds in the intestine shows a good profile. This can be seen in Table 4, which shows that absorption in the intestine is above 95%. In addition, BZ1 and BZ2 compounds were not detected as P-glycoprotein (Pgp) substrates. Pgp is a protein that detects xenobiotics and moves them out of cells. 37 In pkCSM, drug distribution is considered low if the value is less than 0.71 L/kg. 37 From Table 4, it is known that the volume of distribution at steady state (VDss) BZ1 and BZ2 are low. This indicates that the two compounds will be distributed in the plasma compared to the tissue.
The Ames test is a method used to assess a potentially mutagenic compound using bacteria. A positive test indicates that the compound is mutagenic and, therefore, can act as a carcinogen. 37 From Table 4, it is estimated that BZ1 is not a carcinogen, while BZ2 has the potential to cause genetic mutations. From this, it can be concluded that the BZ1 compound has a very good ADMET profile and great potential to develop as a new antiplatelet.
Conclusion
In this study, benzoxazinone derivatives can be synthesized through nucleophilic acyl substitution reactions between anthranilic acid and benzoyl chloride derivatives. Virtual screening shows that BZ1 and BZ2 compounds can be bound to COX-1 receptors, so they can potentially be developed as antiplatelets. If tested using the bleeding time and clotting time measurement method, the synthesized compounds have antiplatelet activity. The in silico pharmacokinetic test also showed that BZ1 and BZ2 were well absorbed. Reduced BZ1 does not cause genetic mutations. Unfortunately, BZ2 is a potential carcinogen. For further development, the compound structure can be modified to obtain BZ1 and BZ2 derivatives, and in vitro testing can be carried out on the COX-1 enzyme.
Footnotes
Abbreviations
ADMET: Absorption, distribution, metabolism, excretion, and toxicity; ATP: Adenosine triphosphate; BW: Body weight; BZ1: 2-Phenyl-3,1-benzoxazine-4-one; BZ2: 2-(4′-chlorophenyl)-3,1-benzoxazin-4-one; CVD: Cardiovascular disease; FT-IR: Fourier transform infrared; MRP4: Multidrug resistance protein 4; MVD 5.5: Molegro Virtual Docker version 5.5; NMR: Nuclear magnetic resonance; QSAR: Quantitative structure-activity relationship.
Acknowledgments
The authors acknowledge the Faculty of Pharmacy Universitas Airlangga, Surabaya, Indonesia, for the facilities provided.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The ethical clearance has been obtained from the ethics committee of the Faculty of Veterinary Medicine, Universitas Airlangga.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Airlangga University through research decree number 886/UN3/2018.
