Abstract
Background
In recent years, ellagic acid has emerged as a focal point in pharmacological research, showcasing promising developments and potential therapeutic applications as a competitive inhibitor of Src homology phosphotyrosyl phosphatase 2 for the treatment of cancer, as a shielding impact that stimulates sirtuin 6 (SIRT 6), against nephrotoxicity induced by cisplatin, as adjuvant treatment in sickle cell anemia, as antiaging and to avoid mild cognitive impairment.
Purpose
This review provides a concise overview of the latest advancements in ellagic acid research, highlighting novel pharmacological findings and emerging trends.
Methodology
A comprehensive search of relevant literature was conducted to gather information on the pharmacological properties and therapeutic applications of ellagic acid. Studies investigating its antioxidant properties, anti-inflammatory effects, molecular interactions, and therapeutic implications were included in the analysis.
Results
The review summarizes the multifaceted pharmacological landscape of ellagic acid, encompassing its antioxidant properties, anti-inflammatory effects, and potential therapeutic applications. It sheds light on its evolving role in modern medicine and underscores its significance as a promising avenue for future pharmaceutical exploration.
Conclusion
The recent strides in ellagic acid development highlight its potential as a valuable therapeutic agent in various health conditions. Further research into its molecular interactions and clinical applications is warranted to fully harness its therapeutic potential and improve patient outcomes.
Introduction
Phytochemicals derived from diverse plants and crude plant extracts have found applications in both contemporary and traditional medical practices. These bioactive compounds serve as medications and nutraceuticals, playing a crucial role in the treatment and management of various chronic conditions. From addressing obesity and cardiovascular diseases (CVDs) to tackling diabetes, infectious diseases, neoplasms, neurological disorders, and inflammatory diseases, these botanical derivatives contribute to the expansive landscape of therapeutic interventions.1–4 In contemporary contexts, the term “polyphenol” encompasses a broad category, encompassing chemically altered or polymerized derivatives such as tannins, chalcones, phenolic acids, and flavonoids. Recent research strongly supports the role of polyphenols in safeguarding against degenerative illnesses, including CVD, cancer, and neurological disorders.5, 6 These compounds serve as potent antioxidants, reinforcing the functions of antioxidant vitamins and enzymes, and acting as a front-line defense against oxidative stress induced by an excess of Reactive Oxygen Species (ROS). While much of the evidence supporting their antioxidant activity is derived from in vitro studies, a growing body of data suggests that in vivo, polyphenols may exert effects beyond their role as antioxidants. Their capacity to influence cell signaling pathways emerges as a key factor in elucidating the mechanisms underlying the impacts of dietary regimens rich in polyphenols.7, 8
Tannins, polyphenolic compounds with molecular weights ranging from 500 to 20,000 Da, are widely distributed across various plant species, residing in stalks, fruits, leaves, roots, and peels. 9 The concentration and specific types of tannins are regulated by the respective plant species. Within the realm of plant physiology, tannins are categorized as secondary metabolites and can be classified into four groups: ellagitannins (ETs), gallotannins, hydrolyzable tannins, and condensed tannins.10, 11
ETs represent quinic acid or glucose esters of ellagic acid (EA), originating from the oxidative interaction of hexahydroxydyphenic acid (HHDP)-derived gallotannins with at least two galoyl units. Upon hydrolysis, this group is released and subsequently undergoes either spontaneous lactonization or rearrangement to form EA. The catalog of ET structures is extensive, with claims of more than 500 distinct structures. 12 Oaks’ bark, primarily sourced from Quercus spp., stands out as the main reservoir of ETs. As outlined by Bianco et al., the classification of ETs into polymeric, oligomeric, or monomeric forms (degree of polymerization) is contingent upon the quantity of HHDP groups present in the molecule. 13
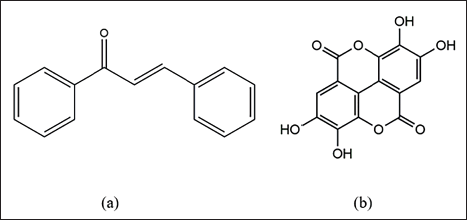
Chalcones, a significant dietary polyphenol, belong to the class of polyphenolic substances derived from flavonoids in plants. As a primary family of natural compounds, chalcones exhibit widespread distribution in tea, vegetables, soy-based foods, fruits, and spices and are considered predecessors of isoflavonoids and flavonoids. The term “chalcone” finds its origin in the Greek word “chalcos,” meaning bronze. Natural chalcones, present in flower petals and various plant parts such as roots, botanicals, heartwood, bark, leaves, and fruits, as well as their synthetic counterparts, showcase diverse biological activities. 14 This versatility has garnered continuous attention from both the business and academic realms. Structurally, chalcones, also known as 1,3-diaryl-2-propen-1-ones, feature two aromatic rings connected by a three-carbon-unsaturated carbonyl system, representing a type of open-chain flavonoid (Figure 1a).
(a) Chalcone and (b) Ellagic Acid.
Several pure chalcones have successfully transitioned into clinical trials or human experiments. Licochalcones, derived from the licorice plant, exhibits a spectrum of biological actions, including anti-malarial, anti-inflammatory, chemopreventive, anti-fungal, anti-bacterial, anti-spasmodic, anti-tumor, and anti-oxidant effects.15, 16 Both chalcones and dihydrochalcones are abundant in nutritional sources found in acidic and apple fruits. Interestingly, these compounds may contribute more substantially to the daily intake of unprocessed or organic polyphenolic components when compared to other extensively studied flavonoids. 17 Chalcone glucoside, a primary pigment responsible for the yellow hue in flowers, is a noteworthy compound.
This study is designed to deepen our understanding of EA’s potential as a future therapeutic agent. The emphasis will be on elucidating its natural sources, and synthesis, accompanied by a comprehensive summary of its most significant pharmacological properties. Through this exploration, we aim to uncover the potential avenues for harnessing EA in future therapeutic interventions.
EA
EA belongs to the polyphenol extractives, specifically the tannins, and is widely distributed in dicotyledons. 18 Structurally, EA is a derivative of chromene-dione (2,3,7,8-tetrahydroxy-chromeno[5,4,3-cde] chromene-5,10-dione; C14H6O8; molecular weight: 302.194 g/mol), featuring a hydrophilic moiety with four hydroxyl groups and a lipophilic moiety with two hydrocarbon rings. 19 This unique structure imparts to EA the capacity to participate in antioxidant redox processes and accept electrons from various substrates.20, 21 The chemical structure of EA is shown in Figure 1b. 22
Synthesis of EA
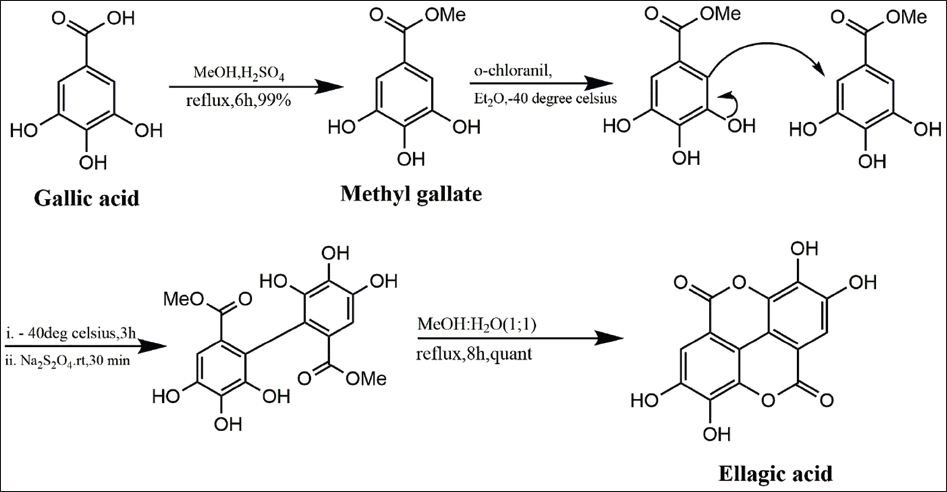
EA synthesis is achievable through a variety of methods, such as oxidative coupling reactions, Ullmann coupling, and lactonization (Figure 2). The synthesis of EA and its 4,4’-di-O-alkyl derivatives from gallic acid was detailed by Alam et al. 23 The process involved an oxidative reaction between gallic acid and o-chloranil, resulting in the synthesis of EA through Ullmann coupling. The functionalized methyl bormogallate was transformed into a biphenyl, which underwent lactonization to yield EA and its alkoxy derivatives. 23
Oxidative Coupling of Gallic Acid to Yield Ellagic Acid.
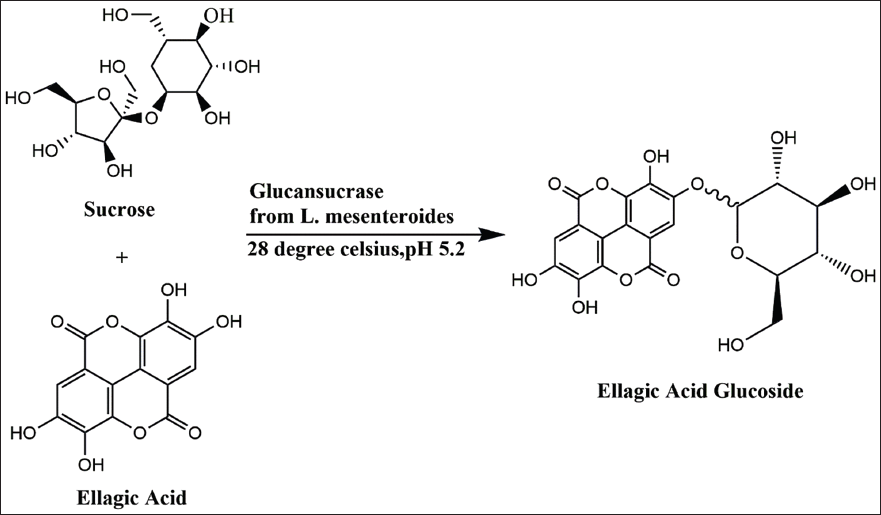
Yu et al. 24 utilized glucansucrase derived from Leuconostoc mesenteroides B-512 FMCM and sucrose for the transglucosylation of EA glucoside (Figure 3). The enzymatically produced product underwent purification through C18 column chromatography and a 50% ethyl acetate fraction. Employing response surface methods with 150 mM sucrose, 300 mU/mL glucansucrase, and 5 mM EA, the yield of EA glucoside reached 69% (3.47 mM). Remarkably, synthetic EA glucoside exhibited superior water solubility, increased protection of brain nerve cells (SH-SY5Y) by up to 58%, a threefold reduction in cortisol, and a fourfold inhibition of acetylcholinesterase (AChE) compared to EA. These results suggest the potential employment of the neuroprotective drug EA glucoside. 24
Optimization of Ellagic Acid Glucoside.
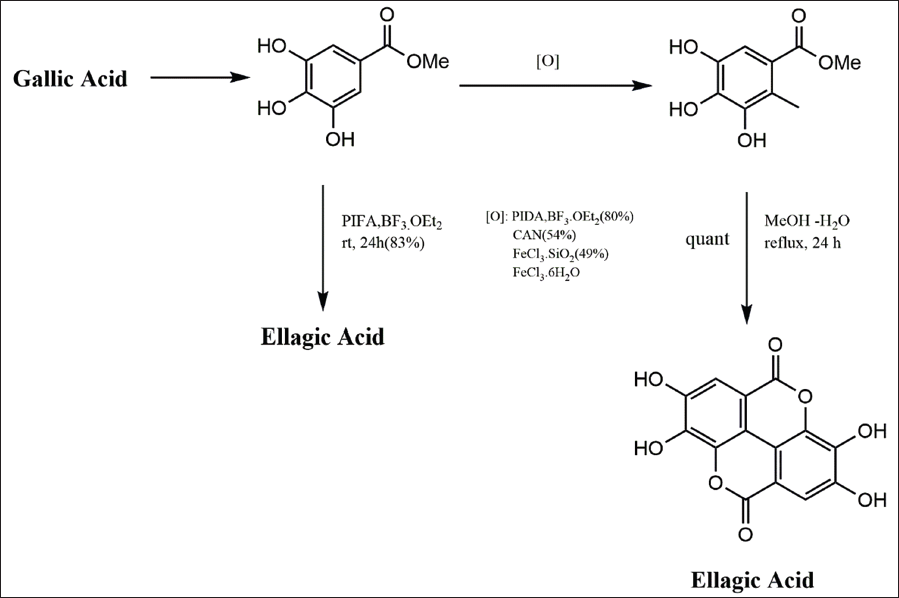
In a study by Alam et al., EA synthesis was accomplished with a 48% yield over four steps, starting from methyl gallate. 25 However, there was an expectation of achieving a more concise and efficient synthesis. Methyl gallate, obtainable commercially or through the methylation of gallic acid, was subjected to various oxidants, including cerium ammonium nitrate, phenyliodine diacetate (PIDA), ferric chloride hexahydrate, and ferric chloride silica. The treatment with PIDA (1.5 equivalent) in boron trifluoride etherate (1.5 mL) at room temperature proved to be the most successful dimerization protocol, yielding the dimeric precursor in 80% after 16 hours. The biaryl was then quantitatively converted to the natural product when heated at reflux in aqueous methanol (1:1) for 24 hours. Remarkably, allowing the phenyliodine bis (trifluoroacetate) reaction mixture to stir at room temperature for 24 hours led to complete cyclization, providing the natural product in 83% yield directly from methyl gallate (Scheme 3; Figure 4). This approach represents an improvement in the synthesis of the bioactive natural product EA. 25
Sources and Pharmacological Effects
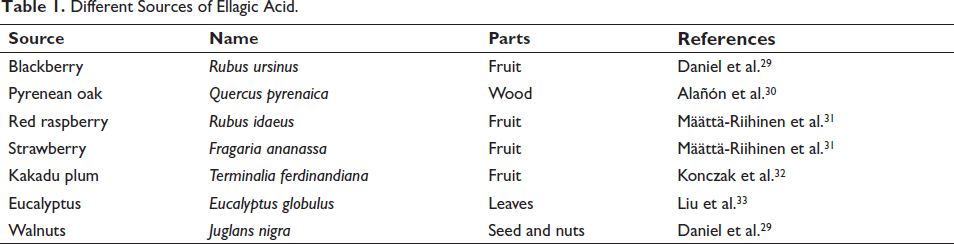
EA, a naturally occurring phenol antioxidant that is present in vegetables and fruits, has a high correlation with a lower chance of contracting chronic illnesses, including CVD and cancer. EA is found in a variety of vegetables and fruits, including persimmons, raspberries, pomegranates, black plums, peaches, and wild strawberries (Table 1). 26 It may exist as derivatives or in free form, mostly as complex polymers referred to as ETs, which can be degraded by gut flora and physiological pH, levels of plasma boost of the acid following the consumption of vegetables as well as fruits. 27 A thorough and current assessment of the biological activities of EA and prospective therapeutic uses was the goal of the Shakeri et al.’s study. EA is found in nuts and fruits in two different forms: bound (ETs) and coupled with hexahydroxydiphenic acid. Many biological characteristics, including antidiabetic, apoptosis-inducing effects, anticancer, and antioxidant, are shown by EA. These pharmacological and biological characteristics are important for treating a number of human disorders. EA is a potential therapeutic agent against human illnesses, including cancer, due to its numerous modes of action. 28
Different Sources of Ellagic Acid.
Biological Activity of EA
EA is still being used as a test substance, and the findings are optimistic. Still, ETs and EA derivatives have also been extensively studied, and it appears that they have more potential to develop. Infectious agents including viruses, fungi, bacteria, and even protozoa have been the subject of reports of their action on diseases.34–37 Furthermore, the GABAergic system involvement of EA/ETs, inhibition of important enzymes like AChE, protein tyrosine phosphatases, and aldose reductase, interaction with serotonergic as well as adrenergic systems and suppression of proinflammatory markers, provide a strong foundation for potential breakthroughs in the treatment and/or prevention of many illnesses and associated clinical complications.38, 39 For instance, some recent research indicated that EA may function as an AChE inhibitor, raising levels of acetylcholine in the brain. As a consequence, cognitive dysfunctions in neurodegenerative diseases like Alzheimer’s disease may be partially reduced or even reversed.40, 41 Last, but not least, it has been revealed that one of EA’s most well-known effects, the prevention of melanogenesis, is at least largely a result of its antioxidant strength. 42
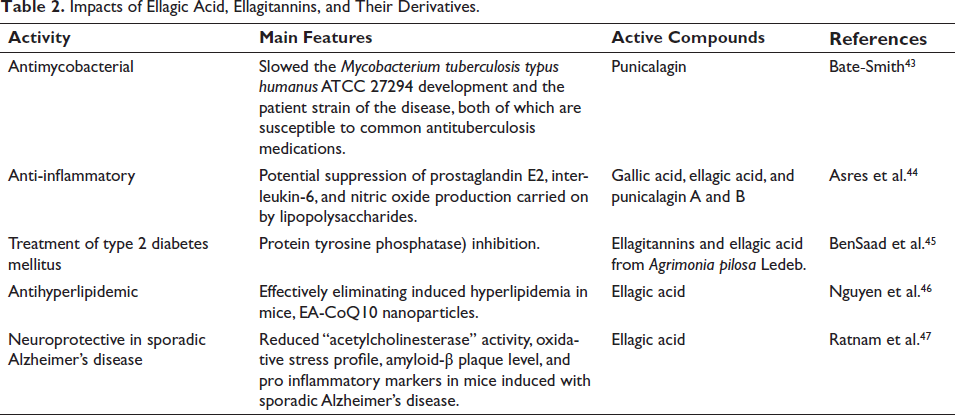
Some possible biological uses of EA, ETs, and derivatives are summarized in Table 2 and Figure 5. It is evident from research conducted in the last 10 years that there are several potential uses for EA and ETs in the pharmaceutical and biomedical industries.
Impacts of Ellagic Acid, Ellagitannins and their Derivatives.
Overview of Ellagic Acid Activity.
Anti-inflammatory
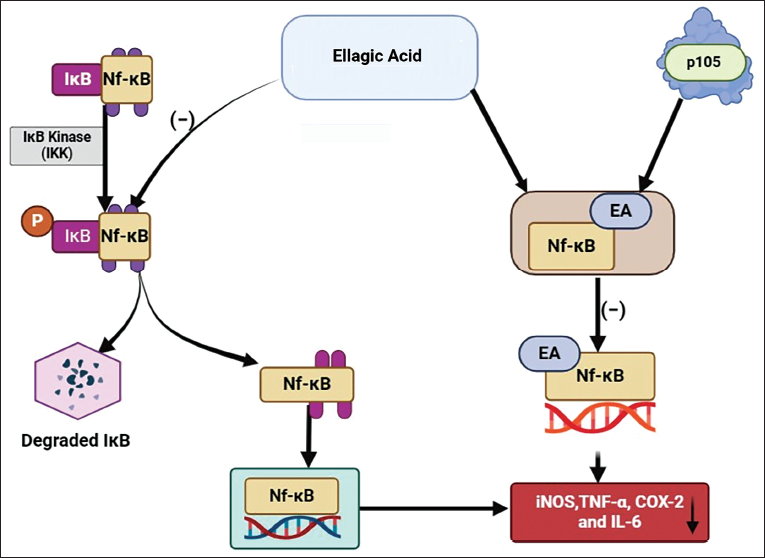
EA has been discovered to form a bond with one of the precursor proteins, p50, of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), specifically p105 (Figure 6). This bonding impedes the ability of NF-κB to bind to a specific DNA domain, thereby preventing the expression of genes that are typically regulated by NF-κB. 48 EA has an additional function regarding NF-κB, which involves inhibiting the phosphorylation of inhibitor of nuclear factor kappa B. This inhibition further prevents the subsequent translocation of NF-κB into the nucleus. 49 Various pieces of evidence indicate that EA can inhibit the NF-κB pathway, leading to the downregulation of the expression of inducible nitric oxide synthase (iNOS), tumor necrosis factor-α (TNF-α), cyclooxygenase-2, and IL-6.50–52
A recent study states that EA, a natural polyphenol compound known for activating sirtuin 6 (SIRT6), exhibits protective effects against cisplatin-induced nephrotoxicity. Pretreatment with EA effectively reduces the cytotoxicity of cisplatin in both primary renal cells and transformed C3H mouse kidney-1 cells. Furthermore, EA demonstrated the ability to improve renal function and alleviate apoptosis and fibrosis induced by cisplatin in mice. Additionally, EA reduced the expression of nephrotoxicity-associated inflammatory factors, IL-1β and IL-6, both in vitro as well as in vivo. In terms of mechanisms, EA reversed the reduction in SIRT6 expression caused by cisplatin and mitigated the increased expression of TNF-α induced by cisplatin. The findings offer a preclinical foundation for considering EA as a viable and promising mediator to mitigate cisplatin-induced acute kidney injury. Moreover, they support the potential role of EA as an adjunctive therapy in future cancer treatments. 53
Antioxidants
EA is one of the key antioxidants, along with well-known vitamins like ascorbic acid and α-tocopherol. 54 Its free radical scavenging action, which has been suggested to be equivalent to that of important vitamins, has been linked to its inherent antioxidant characteristics. EA has the capacity to scavenge a broad range of reactive nitrogen and oxygen species due to the presence of four hydroxyl and two lactone functional groups. 55 In fact, studies have demonstrated that at physiological pH, EA in an aqueous solution may inactivate peroxyl radicals, nitrogen dioxide (NO2), and peroxynitrite, in addition to hydroxyl radicals.56, 57
In decreasing order of scavenging rates, hydroxyl (OH)>methoxyl (OCH3)>nitrogen dioxide (NO2), Tiwari and Mishra showed that EA is an effective radical scavenger, notably against NO2, OH, and OCH3. 58 According to these scientists, EA should generally be a more effective scavenger of ROS than reactive nitrogen species. The metabolites of EA are also capable of effectively scavenging a variety of free radicals, sometimes even quicker than EA itself; therefore, its antiradical activity is not diminished throughout metabolism.
Cancer Prevention and Treatment
Several studies have presented evidence supporting the chemo-preventive and therapeutic properties of EA, whether obtained from the diet or administered through optimized matrices to enhance bioavailability. 59 EA’s potential lies in its ability to counteract oxidative stress-induced DNA damage, preventing mutations in proto-oncogenes and tumor suppressor genes. In a study involving Dalton’s lymphoma-bearing mice, various doses of EA (40, 60, and 80 mg/kg body weight) effectively reduced Protein C signaling, associated with cell proliferation and tumor growth, through NF-κB. Additionally, EA enhanced antioxidant defense mechanisms, providing insights into its role in cancer prevention. 60 In a recent study, Ma et al., showed that EA was first recognized as a competitive inhibitor of Src homology phosphotyrosyl phosphatase 2 (SHP2). Moreover, it exhibited substantial inhibitory effects on SHP2-E76K and SHP2-E76A mutants. Cell proliferation experiments further validated the capacity of EA to impede the proliferation of diverse cancer cells. 61
Glucose and Lipid Lowering
Increased lipid and glucose levels, along with abdominal obesity, high blood pressure, and low high-density lipoprotein cholesterol, are factors that heighten the risks of heart disease, diabetes, and stroke, constituting the primary features of metabolic syndrome. Oxidative stress plays a role in the development of type 2 diabetes and the accompanying vascular complications.62, 63 EA exhibits favorable effects on glucose metabolism and diabetes in murine models, including the reduction of glucose levels, antioxidant, antiglycation, and anti-inflammatory effects, as well as the prevention of microvascular and macrovascular diabetic complications. 64 In a recent investigation, diabetic rats on a high-fat diet were administered both free EA and EA nanoparticles. The findings indicated that EA nanoparticles exhibited prolonged improvements in glucose levels and body weight compared to conventional EA. 65
Hepatoprotective
EA is credited with hepatoprotective effects due to its enhancement of hepatic functions under toxic and pathological conditions. This is attributed to its array of properties, including antioxidant, anti-hepatotoxic, anti-steatotic, anti-cholestatic, anti-fibrogenic, anti-hepatocarcinogenic, and anti-viral attributes. 66 Experiments conducted on murine models have elucidated the molecular mechanisms through which EA exerts its hepatoprotective role. These mechanisms involve scavenging free radicals, regulating antioxidant enzymes, modulating the synthesis of proinflammatory cytokines, and controlling lipid synthesis and degradation, among other factors. 66
Notably, EA demonstrates potential anti-viral properties against hepatitis B 67 and hepatitis C viruses, 68 both of which can lead to long-term complications, such as fibrosis, cirrhosis, and hepatocellular carcinoma. 69
Skin Diseases and Cosmetics
To assess the potential protective impact of EA on keratinocytes and human dermal fibroblasts, Bae et al. subjected these cells to ultraviolet-B (UV-B) radiation and administered the phenolic compound at concentrations ranging from 1 to 10 µM. The results showed a reduction in UV-B damage in both cell types, accompanied by a decrease in collagen levels due to decreased production of metalloproteinase. 70 Furthermore, the authors discovered that the topical application of EA at a concentration of 10 µM shielded hairless mice from skin wrinkle formation upon UV-B exposure. This favorable outcome was linked to a decreased production of proinflammatory cytokines (IL-1β and IL-6) and the alleviation of intercellular adhesion molecule-1 expression. 70
The incorporation of EA as a functional ingredient in cosmetic products has been suggested owing to its antioxidant, skin-lightening, and sunscreen properties. To enhance EA’s solubility and stability, attempts have been made to prepare gold nanoparticles enveloped with chitosan for EA absorption. 71 Favorable and promising outcomes were achieved in the evaluation of the mentioned properties, emphasizing that the nanoparticles were produced in accordance with green chemistry principles. 72
Clinical Investigations
In this article, we have incorporated clinical trials specifically assessing the effectiveness of EA as outlined in Table 3.
Ellagic Acid in Clinical Research.

Intellectual Capability
Liu et al. conducted a randomized clinical trial (RCT) to assess the efficacy of EA in enhancing cognitive performance in middle-aged men, utilizing the Wechsler Adult Intelligence Scale-Revised and Montreal Cognitive Assessment test. Over 12 weeks, patients were randomly allocated to receive a daily dose of 50 mg EA or a placebo. While the EA group exhibited statistically significant improvements in scores among overweight men, no differences were observed in patients with a normal body mass index. 73
Insomnia and Fatigue
In a small non-randomized controlled trial, the efficacy of Robuvit (an extract from the wood of Quercus robur L. containing ETs roburin A–E, EA, and gallic acid) in alleviating fatigue and insomnia was examined. Patients had the autonomy to select their respective groups, and the Fatigue Severity Scale gauged fatigue levels, while the Regensburg Insomnia Scale and Pittsburgh Sleep Quality Index assessed insomnia. Although there was a statistically significant improvement in scores related to fatigue and insomnia questionnaires after 8 weeks of Robuvit supplementation, the study does not provide comparisons between the intervention and control groups. 74
Male Fertility
In a 2014, Danish RCT, the impact of consuming tablets containing Punica granatum extract and standardized Alpinia galanga powder on the total number of motile spermatozoa (TMSC) and sperm morphology, defined by strict criteria, was assessed in adult men with diminished semen quality. Following 3 months of treatment, a statistically significant increase in TMSC was observed (p = .026). However, the difference in follow-up TMSC between groups did not reach statistical significance. No notable differences in sperm morphology were detected post-treatment. The study’s limited size (n = 66) might explain the absence of statistically significant differences. Larger RCTs are warranted, preferably focusing on evaluating EA in isolation without combination interventions. 75
Colorectal Cancer
Núñez-Sánchez et al. conducted a study to explore the potential impact of these metabolites on colorectal cancer patients (n = 52) following the administration of pomegranate extract (900 mg/day, 15 days). They examined the presence of these metabolites in the urine or tissue of both normal and malignant colons. EA was identified in colon tissue, present in both free form and as conjugates. Specifically, samples from colorectal cancer patients who received 291 mg/g of free EA demonstrated 649 ng/g in normal tissue and 195 ng/g in malignant tissue. 76
Ischemia
In a study, individuals with stable coronary heart disease were randomly assigned to either a pomegranate juice (PJ) group or a placebo group. Participants underwent electrocardiographic-gated myocardial perfusion single-photon emission computed tomographic technetium-99m tetrofosmin scintigraphy at rest and during stress at baseline and three months later. A statistically significant reduction (p < .05) in stress-induced ischemia was observed in the PJ group (summed difference score (SDS) −0.8 ± 2.7) compared to the placebo group (SDS 1.2 ± 3.1). 77
Multiple Sclerosis
In a triple-blind study, eligible patients were randomly assigned to two groups: one receiving EA (25 subjects) at a dose of 180 mg for 12 weeks and the control group (25 subjects) receiving a placebo before main meals. The assessment involved gathering data on general information, food consumption, physical action, the expanded disability status scale, anthropometric data, the pain rating index, a health questionnaire, and the fatigue severity scale before and after the 12 weeks. Additionally, serum levels of interferon-gamma, IL-17, IL-4, and transforming growth factor-beta, as well as nitric oxide (NO), were assessed using the enzyme-linked immunoassay method. The addition of EA resulted in a decrease in levels of IFNγ, IL-17, and NO while elevating IL-4 in the EA group. Conversely, no such alterations were noted in the placebo group. 78
Conclusion
The pharmacological properties of EA have been extensively explored in various preclinical and clinical studies, demonstrating its potential as a promising therapeutic agent for a wide range of health conditions. From its anti-inflammatory and antioxidant effects to its role in cancer prevention and treatment, glucose and lipid regulation, hepatoprotection, skin health, and cosmetic applications, EA has shown remarkable versatility in its pharmacological actions.
Clinical investigations have provided valuable insights into the efficacy of EA in addressing conditions such as intellectual capability, insomnia, fatigue, male fertility, colorectal cancer, ischemia, and multiple sclerosis. While many of these studies have shown promising results, further research is warranted to elucidate the underlying mechanisms of action, optimize dosage regimens, and assess long-term safety profiles.
Future Perspectives
Looking ahead, the exploration of EA’s pharmacological potential opens exciting avenues for future research and development. One promising direction involves investigating its synergistic effects with other natural compounds or conventional therapeutics to enhance efficacy and minimize adverse effects. The combination therapies may offer novel approaches for addressing complex diseases and improving patient outcomes.
Furthermore, the development of innovative delivery systems and formulations could enhance the bioavailability and stability of EA, overcoming the limitations associated with its poor solubility and rapid metabolism. Nanotechnology-based approaches, liposomal formulations, and targeted delivery systems represent promising strategies for maximizing the therapeutic benefits of EA in clinical settings.
In addition, the elucidation of specific molecular targets and signaling pathways modulated by EA will provide valuable insights into its mechanisms of action and help identify novel therapeutic targets for drug discovery and development. Advances in systems biology, high-throughput screening, and computational modeling hold great promise for accelerating the pace of discovery in this field.
Moreover, expanding our understanding of the pharmacokinetics and pharmacodynamics of EA across different populations and disease states will facilitate personalized approaches to treatment and optimize therapeutic outcomes. Population-based studies and clinical trials incorporating diverse patient cohorts will be essential for evaluating the efficacy and safety of EA in real-world settings.
The continued investigation of EA’s pharmacological effects offers tremendous potential for addressing unmet medical needs and improving public health outcomes. By leveraging interdisciplinary approaches and harnessing cutting-edge technologies, we can unlock the full therapeutic potential of EA and pave the way for the development of innovative therapies for diverse health conditions.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
