Abstract
Objectives
To evaluate the hepatoprotective activity of fractions of Jasminum elongatum methanolic extract against acetaminophen-induced hepatic damage.
Materials and Methods
Fractions obtained from methanolic extract of J. elongatum (petroleum ether, chloroform and hydroalcoholic) were investigated for hepatoprotective activity. Doses of 100 mg/kg and 200 mg/kg of different fractions were administered for 7 days, and acetaminophen toxicity was induced on the eighth day. After 24 hr of toxicity induction, biochemical markers and histological alterations were measured to determine the amount of hepatoprotection.
Results
The chloroform and hydroalcoholic fractions showed a significant decrease in biochemical parameters when compared to the toxic group, which was confirmed by histopathological changes and antioxidant parameters.
Conclusion
The results of this study revealed that the chloroform and hydroalcoholic fraction of J. elongatum have significant hepatoprotective activity against acetaminophen-induced toxicity.
Introduction
The liver, the largest solid organ in the human body, plays a vital role in the metabolic, secretory, immunological and excretory functions, and in addition, maintains the body’s homeostasis. 1 As a result, the liver and its hepatocytes are prime targets for various toxicants like chemicals, drugs, heavy metals, microbes and viral infections. 2 The above-mentioned factors generate many free radicals which are neutralised by endogenous antioxidants, but when natural protective mechanisms are overpowered, frequent injuries occur and thus result in inflammatory (hepatitis) and non-inflammatory liver diseases (alcoholic liver diseases). 3 Drugs currently available for the treatment of liver diseases like corticosteroids and immunosuppressants have major drawbacks, including side effects, poor bioavailability, selectivity and stability. As a result, herbal drugs have grown in importance and popularity in recent years due to their efficacy, cost-effectiveness and less than one side effect as compared to synthetic drugs. 4
Jasminum species belonging to the family Oleaceae have been used traditionally to cure various ailments such as gastritis, diarrhoea, arthritis, hepatitis and dysmenorrhoea. Scientifically, the leaf, stem, bark and roots of Jasminum species are key sources of chemicals reported for pharmacological actions such as antibacterial, antioxidant, 5 antiarthritic, hepatoprotective and antidiarrhoeal. 6 Jasmine oil is one of the main elements of aromatherapy, and it promotes a feeling of well-being and mental relaxation. It is also utilised as a flavouring agent in products such as chewing gum and confectionery. 7 Jasminum sambac, Jasminum auriculatum, Jasminum grandiflorum, Jasminum flexile and Jasminum elongatum are the most widely grown Jasminum species worldwide, including India. 8
The J. elongatum shrub is native to India and can be found in the Sikkim, Assam and Khasi hills at elevations of 300 to 1700 m. In China, powdered twigs and leaves have traditionally been used as a hydragogue, febrifuge, and for the treatment of diarrhoea and bellyache. The plant’s leaves and stems include secoiridoid glucosides such as jasamplexosides A, B and C, as well as 10-hydroxy ligstroside and jasminoside 9 with the leaves also containing jaslanceosides B and E, jasminoside and isojasminoside. 10 The plant’s methanolic extract was found to have analgesic and antidiarrhoeal properties. 11 The species of the genus Jasminum are reported for hepatic disorders, 12 and also in our previous investigation, we have reported the hepatoprotective activity of methanolic extract of J. elongatum (MEJE). 13 So, the current research is intended to isolate the active fraction(s) of J. elongatum through fractionation and screen the various fractions for hepatoprotective activity against paracetamol-induced hepatotoxicity in rats.
Materials and Methods
Preparation of Extract and Fractionation
The twigs (3 kg) of the plant J. elongatum were collected from Pune District (Maharashtra) during the flowering season. The plant was authenticated by the Botanical Survey of India, Pune. Twigs were air-dried in shade and then powdered. Weighed quantity of powder drug (600 g) extracted with methanol (1.5 L) by cold maceration technique at room temperature for 7 days with intermittent shaking. The yield of the methanolic extract was 6.6% and the extract obtained was labelled as MEJE.
The methanolic extract (40 g) was fractionated sequentially into petroleum ether, chloroform and double-distilled water (each 300 mL × 4) fractions. The fractions obtained are labelled as petroleum ether fraction of methanolic extract of J. elongatum (PE-MEJE), chloroform fraction of methanolic extract of J. elongatum (CF-MEJE) and hydroalcoholic fraction of methanolic extract of J. elongatum HA-MEJE and the respective yield was found to be 4.08%, 2.78%, and 38.72%.
Chemicals and Reagents
Silymarin was obtained from Yucca enterprises, Mumbai. Acetaminophen (APAP) was obtained as a gift sample from Cipla, Mumbai. Assay kits for the determination of aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP) and total bilirubin (TB) were obtained from Accurex Biomedical Private Limited., Mumbai. All other chemicals were of the highest analytical grade.
Experimental Animals
In this investigation, male Wister rats (170−200 g) were used. Animals were housed in clean polypropylene cages and fed standard feed (Pranav Agro Pvt Ltd, Sangli, India) after being obtained from the National Institute of Biosciences in Pune, India. Water was provided ad libitum. The animals were kept in a controlled environment with a temperature of 22 ± 2°C and relative humidity of 45% to 55% under a 12-hr light/12-hr dark cycle. Research protocol approval was approved by the Institutional Animal Ethics Committee (IAEC) of Poona College of Pharmacy, Pune, and it is constituted in accordance with the rules and guidelines of the Committee for Control and Supervision on Experimental Animals (CPCSEA), India (Protocol approval number CPCSEA/59/14).
Preliminary Phytochemical Screening
Phytochemical screening for the fractions of J. elongatum was performed to identify the phytoconstituents present in individual fractions. 14
Pharmacological Study of Fractions of MEJE
Acute Oral Toxicity Study
The acute oral toxicity test was carried out in accordance with OECD-423 guidelines. Wister rats were fasted overnight and divided into three groups of six animals each. Fractions of MEJE (PEJE, CFJE and HAJE) were administered orally at a single dose level of 2,000 mg/kg b.w. Animals were observed for toxicity symptoms and mortality at least once during the first 30 min, periodically during 24 hr with special attention during the first 4 hours and daily thereafter for a total of 14 days. 15
Assessment of Hepatoprotective Activity
Hepatoprotective activity of fractions of MEJE was performed according to Eesha et al. 16 A total of 54 experimental rats were divided into nine groups, each with six animals. The drugs and extracts were suspended in a 5% gum acacia vehicle.
Experimental design: Group I (Normal control) received the vehicle p.o (1 mL/kg b.w of 5% gum acacia) for 8 days. Group II (Toxic control) received 5% gum acacia p.o. for 7 days. Group III (Standard) administered silymarin (50 mg/kg) for 7 days. Group IV, V, VI, VII, VIII and IX were given orally a single-dose PE-MEJE (100 mg/kg), PE-MEJE (200 mg/kg), CF-MEJE (100 mg/kg), CF-MEJE (200 mg/kg), HA-MEJE (100 mg/kg), HA-MEJE (200 mg/kg), respectively, daily for 7 days. Except for Group I, all other groups received acetaminophen (2 g/kg) after 24 hr of the last treatment, that is, on the eighth day.
Blood samples were taken from the retro-orbital plexus after 24 hr of toxicity induction and centrifuged at 2,500 rpm for 15 min to separate serum for examination of several biochemical markers, such as AST, ALT, ALP and TB.
Histopathology
The livers of all rats were isolated after they were sacrificed via cervical dislocation. To determine the effect of extracts on toxicity-induced rats histologically, the liver of one animal from higher dose groups, along with normal, toxic and standard groups were fixed in 10% buffered formalin solution and sections of 5 µm thickness were prepared by paraffin embedding and subsequently stained with haematoxylin-eosin. The remaining livers were used for the estimation of antioxidant parameters.
Measurement of Malondialdehyde (MDA), Glutathione (GSH) and Superoxide Dismutase (SOD) in Liver Homogenate
Livers were homogenised in 0.05 M sodium phosphate buffer after being rinsed with ice-cold saline (pH 7.0). The homogenate was centrifuged for 10 min at 4°C at 4000 rpm, and the supernatant was utilised to calculate oxidative stress markers such as MDA, 17 GSH 18 and SOD. 19
Statistical Analysis
The results were expressed as mean ± SEM. GraphPad Prism-5 software was used for the statistical analysis, which included a one-way analysis of variance (ANOVA) followed by a Bonferroni post-hoc test. p < .05 was regarded as significant.
Results
Phytochemical Screening of Fractions of J. elongatum
Phytochemical screening of fractions of PE-MEJE showed the presence of steroids and saponins, whereas screening of fractions of CE-MEJE revealed the presence of steroids, saponins and flavonoids. Phytoconstituents such as carbohydrates, saponins, flavonoids and phenols were detected in hydroalcoholic fraction, HA-MEJE (Table 1).
Preliminary Phytochemical Screening of MEJE Leaf Fractions.

Pharmacological Studies of Fractions of MEJE
Acute Oral Toxicity
In an acute oral toxicity study, animals administered with MEJE fractions (PE-MEJE, CF-MEJE and HA-MEJE) at a single dose of 2,000 mg/kg, showed no significant behavioural changes. There was no mortality in any of the groups treated. Therefore, considering fractions as safe and non-toxic, further pharmacological screening was performed using one-tenth of the maximum dose.
Assessment of Hepatoprotective Activity
The results of the fractions of MEJE against acetaminophen-induced toxicity are shown in Table 2. In the toxic group, the significant hepatic injury was manifested by increased levels of serum markers, AST, ALT, ALP and TB when compared to the control group. But oral administration of fractions of CF-MEJE (200 mg/kg), HA-MEJE (100 mg/kg) and HA-MEJE (200 mg/kg) reduced the elevated levels of serum parameters significantly (p < .001) when compared to the toxic group, whereas CF-MEJE (100 mg/kg) reduced the level at a significance of p < .01, indicating regeneration of damaged lived cells or enhancement of structural integrity of hepatocytic cell membrane by the fractions. The petroleum ether extract did not exhibit any significant reduction in parameters except PE-MEJE (200 mg/kg), which showed significance in one parameter.
Results of the Fraction of MEJE on Rat Serum Parameters after APAP Intoxication.
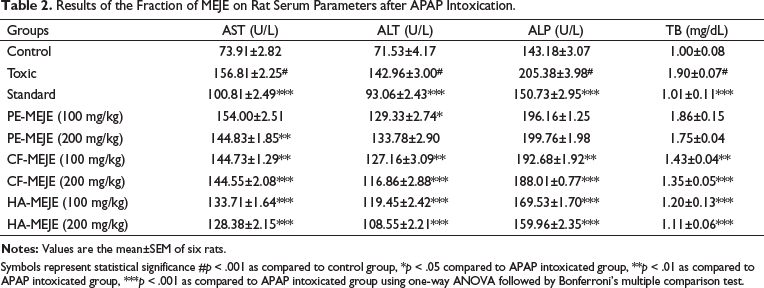
Symbols represent statistical significance #p < 0.001 as compared to control group, *p < 0.05 compared to APAP intoxicated group, **p < 0.01 as compared to APAP intoxicated group, ***p < 0.001 as compared to APAP intoxicated group using one-way ANOVA followed by Bonferroni’s multiple comparison test.
Histopathological Findings
Figure 1 depicts a magnification of changes in liver architecture between the normal and model groups by HE stains. Based on the stained tissue sections, HA-MEJE (200 mg/kg) and CF-MEJE (200 mg/kg) caused no appreciable changes in the liver histology as compared to the standard treated group. In contrast, acetaminophen alone intoxicated rats produced significant changes in liver morphology like necrosis, inflammation and ballooning degeneration in comparison to the control group. The histopathological observations support the results of the biochemical study.
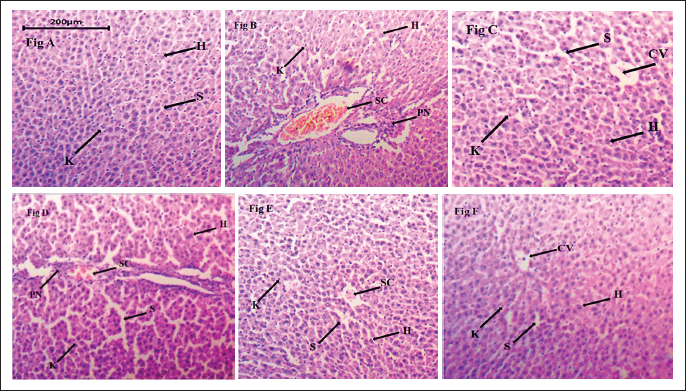
(A) Section of Normal Rat.
Estimation of Antioxidant Parameters
Table 3 shows the extent of antioxidant status in APAP-intoxicated rats and treated groups. In comparison to the control group, APAP-intoxicated rats had significantly higher levels of MDA, an indication of oxidative damage and one of the main products of peroxidised polyunsaturated fatty acids. Treatment of fractions particularly CF-MEJE-200 mg/kg (p < .01) and HA-MEJE-200 mg/kg (p < .001) attenuated these levels. The animals treated with fractions of CF-MEJE and HA-MEJE also increased the antioxidant status significantly by increasing the levels of GSH and SOD compared to the intoxicated group.
Effects of MEJE Fractions on MDA (nmole/mg of Protein), GSH (nmole/mg of Protein), SOD (unit/mg of Protein).
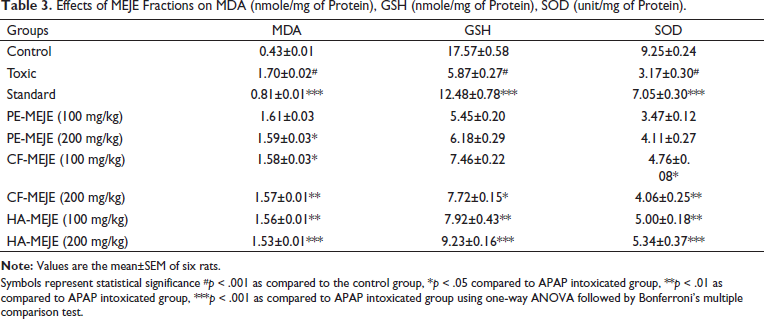
Symbols represent statistical significance #p < 0.001 as compared to the control group, *p < 0.05 compared to APAP intoxicated group, **p < 0.01 as compared to APAP intoxicated group, ***p < 0.001 as compared to APAP intoxicated group using one-way ANOVA followed by Bonferroni’s multiple comparison test.
Discussion
The present investigation began with an oral acute toxicity test of fractions of MEJE on experimental rats, which indicated promising safety of this plant extract with no morbidity or mortality during the entire study period, even at higher concentrations, such as 2,000 mg/kg. Many investigations by diverse co-researchers utilising various medicinal plant extracts consistently showed that they were safe, with no evidence of hazardous effects.20–22
Acetaminophen is a commonly used non-narcotic analgesic and antipyretic. 23 When given in therapeutic dose ranges, a significant proportion of APAP is metabolised by phase II conjugating enzymes, predominantly UDP-glucuronosyltransferase (UGT) and sulfotransferase (SULT), resulting in non-toxic compounds excreted in the urine. Only a small proportion is excreted unchanged in the urine. The cytochrome P450 enzymes (CYPs), primarily CYP 2E1, convert the remaining APAP into the highly reactive intermediate metabolite N-acetyl-p-benzoquinone imine (NAPQI). 24 NAPQI is often detoxified quickly by conjugating with GSH. When phase II metabolising enzymes are saturated after an APAP overdose, however, high NAPQI depletes GSH, resulting in covalent sulfhydryl group attachment in cellular proteins, particularly mitochondrial proteins. 25 This causes mitochondrial oxidative stress and malfunction, which eventually leads to hepatocyte necrosis.
Pre-treatment of fractions of MEJE, particularly CF-MEJE (200 mg/kg), HA-MEJE (100 mg/kg) and HA-MEJE (200 mg/kg) preserved nuclear envelope integrity against damage induced by the toxicity of acetaminophen as shown in the histological analysis of hepatocytes (Figure 1). The liver function test (AST, ALT, ALP and TB) is the most important analysis for diagnosing hepatoprotective effects. Our findings reveal that chloroform (200 mg/kg) and hydroalcoholic fractions (100 mg/kg and 200 mg/kg) were significantly (p < 0.001) beneficial in decreasing liver function markers when compared to the toxic group.
Overdose of APAP also lead to decreased GSHt/GSSG ratio and increased MDA content, both of which are indicators of lipid peroxidation.26, 27 Herbal drugs possess phytoconstituents such as flavonoids, phenols, thiols, saponins, which exhibit a protective effect by inhibiting lipid peroxidation, promoting recovery of the damaged cell membrane, preventing mitochondrial damage and eliminating oxygen-free radicals.28, 29 The preliminary phytochemical screening of chloroform and hydroalcoholic fractions of MEJE revealed the presence of triterpenes, flavonoids and saponins, which could be the possible role of hepatoprotective activity by an antioxidant mechanism.
MDA is a significant byproduct of cell membrane oxidation, chloroform and hydroalcoholic fractions were effective in lowering MDA levels. GSH and SOD are antioxidant enzymes that catalyse the dismutation of radical anion superoxide to hydrogen peroxide and oxygen. Chloroform and hydroalcoholic fractions were able to increase GSH and SOD levels in acetaminophen-treated animals.
Also, plant species belonging to the genus Jasminum have been scientifically reported for liver diseases thus indicating the promising potential of crude extracts, solvent fractions and isolated compounds. For example, J. grandiflorum extracts alleviated the hepatotoxicity induced by CCl4 injury by inhibition of oxidative stress.30-31
As a result of this research, it was concluded that fractions of MEJE have hepatoprotective activity against acetaminophen-induced toxicity in rats. Further studies need to be carried out to fully characterise the mechanism responsible for antioxidant activity present in the active fractions and establish their possible mode of action.
Conclusion
In conclusion, this study demonstrated for the first time that the fractions of MEJE were effective in preventing acetaminophen-induced liver damage. Our data indicated that chloroform (200 mg/kg) and hydroalcoholic fractions (100 mg/kg and 200 mg/kg) of MEJE prevented liver injury by inhibiting plasma ALT, AST, ALP and TB activities and lipid peroxidation; also, by reducing the depletion of hepatic antioxidant enzymes such as GSH and SOD in a dose-dependent manner. In addition, the chloroform and hydroalcoholic fraction of MEJE possessed antioxidative activity that likely contributed to its protective effect against APAP-induced liver toxicity. Taken together, this study suggests that chloroform and hydroalcoholic fractions of MEJE have considerable potential for development as a natural hepatoprotective agent.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors acknowledge the grants received from University Grants Commission (UGC) for this project under Maulana Azad National Fellowship (MANF) Scheme. F1-17.1/2010/MANF-MUS-AND-4007/(SA-III/Website).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Summary
In this study, we have explored the hepatoprotective activity of fractions of J. elongatum methanolic extract against acetaminophen-induced hepatotoxicity in albino Wistar rats. Among the three fractions, CF-MEJE and HA-MEJE showed significant hepatoprotective activity, by reducing elevated serum biochemical parameters AST, ALT, ALP and TB, which was confirmed by histopathological changes observed. The antioxidant parameters SOD and GSH were elevated while MDA, a product of lipid peroxidation was reduced by chloroform and hydroalcoholic fractions. However, PE-MEJE failed to exhibit hepatoprotective action against liver injury. The preclinical studies confirm that chloroform and hydroalcoholic fractions of J. elongatum have significant hepatoprotective activity against acetaminophen-induced liver toxicity.
