Abstract
Background
Cladophora goensis and Cladophora glomerata has been widely identified in the Indian Oceans. Marine algae have been identified as potential pharmacological agents useful in the treatment of diabetes, hypertension, infections, and cancers.
Objectives
The present study aimed to evaluate the phytochemicals in C. goensis and C. glomerata using gas chromatography-mass spectroscopy (GC-MS), and to study the molecular interactions with cancer-related proteins using molecular docking techniques.
Materials and methods
GC-MS analysis was done using electron impact ionization at 70eV and the data was evaluated using total ion count for compound identification and quantification. AutoDock 4.0 version was used for the molecular docking analysis.
Results
The methanolic extracts of algae when subjected to GC-MS analysis, 19 compounds from C. goensis and 11 phytocompounds from C. glomerata were identified. The significant molecular interactions of phytochemicals of C. goensis (6-nitro-3H-quinazolin-4-one, Isoquinoline, 1,2,3,4-tetrahydro-7-methoxy-2-methyl-8-(phenyl methoxy) and 9-Decen-1-ol, pentafluropropionate) and C. glomerata (phytol, palmitic acid, and octadec-9-enoic acid) against human epidermal growth factor receptor (4WRG), poly (ADP-ribose) polymerase-1 (4UND), human estrogen receptor alpha ligand-binding domain (3ERT), human peroxisome proliferator-activated receptor alpha ligand-binding domain (3VI8), and human topoisomerase (1EJ9) have been demonstrated.
Conclusion
The phytochemicals of methanolic extracts of C. goensis and C. glomerata showed potential interactions with cancer-related proteins.
Introduction
Cladophora goensis and Cladophora glomerata are green algal seaweeds commonly seen in Polish bays. 1 These species have been identified majorly in the Indian Oceans, Pacific, and Atlantic. 2 Twenty-eight species belonging to Chlorophyceae have been identified in Rameshwaram, Gulf of Mannar. 3 The major portion of the earth is covered by water inhabited by different aquatic plants that have pharmaceutical value. Many research studies have identified novel phytochemicals with different pharmacological activities from this marine vegetation. Algae have been a potential source of pharmacological agents and nutrition for decades. 4
Cladophora species are widely distributed in both marine and freshwater ecosystems. The common name of this species is Mekong weed. These types of weeds placed under the Chlorophyta genus consist of 183 taxonomically identified species. 5 This weed has been used for food as well as medicine in both dry and fresh forms. 6
Many anticancer candidates were investigated, however, over the last few years, it has been thought that choosing targets based on how proteins contribute to the onset as well as the progression of cancer is crucial, making regulatory proteins significant targets for treatment. 7 The well-known anticancer therapeutic targets are human epidermal growth factor receptor (EGFR) (4WRG), poly (ADP-ribose) polymerase-1 (4UND), human estrogen receptor (ER) alpha ligand-binding domain (3ERT), human peroxisome proliferator-activated receptor (PPAR) alpha ligand-binding domain (3VI8), and human topoisomerase (1EJ9).
Approximately 77% of instances of colorectal cancer (CRC) had overexpressed levels of the EGFR. It is well-recognized that EGFR has a role in the carcinogenetic processes of angiogenesis, cell motility, apoptosis, proliferation, and metastasis. Studies both preclinical and clinical have demonstrated that targeting EGFR is a useful approach for anticancer treatment. Cetuximab is a remarkably well-known monoclonal antibody and a unique EGFR inhibitor that is finally approved for use in CRC patients who have metastasized. 8
PARP inhibitors offer treatment potential for the management of nononcologic ailments and a range of malignancies. Enzymes known as PARPs attach ADP-ribose components to a range of amino acid targets. The best way to understand the biological role of ADP-ribosylation is in the broader setting of maintaining genome integrity, where ADP-ribose chains help to attract amino acids to areas of deoxyribonucleic acid (DNA) damage. Olaparib is now used in clinical practice to inhibit poly (ADP-ribose) polymerase-1 and -2 (PARP1, PARP2), and numerous more drugs are in advanced investigation. Poly (ADP-ribose) polymerase-1 (4UND) has been used in various investigations for anticancer screening. 9
The ovarian steroid hormone estrogen acts by attaching directly to the ER. The most often employed target ligands in endocrine therapy for breast cancer (BC), especially for patients with ER-positive metastatic BC, are ER antagonists and inhibitors. The ERs are ligand-inducible receptors, like the antagonist synthetic selective ER modulator tamoxifen and the biosynthesized agonist 17 β-estradiol (EST). The latter has been shown to reduce the risk of BC relapse and is frequently used as an adjuvant treatment for postmenopausal tumors in ER-α positive patients. The ER-tamoxifen combo has mild to severe adverse effects but also prevents the cancer cells from proliferating. Additionally, a large number of individuals who test positive for ERBS acquire intrinsic resistance to hormone therapy, necessitating the use of effective substitute medications. 10
More than 40 years ago, peroxisome PPARs were identified as regulators of peroxisome-increased activity. Part of the nuclear receptor superfamily, PPARs are ligand-induced transcription factors that control energy homeostasis throughout the body during lipid and carbohydrate metabolism, cell proliferation, and the development of cancer. According to earlier research, activation of PPARβ/δ is linked to the advancement of tumors, while the activity of PPARα and PPARγ is linked to the suppression of tumors. Because of the complex regulatory signals of PPARs and the need to fully understand their underlying mechanism, the involvement of PPAR in cancer has progressively emerged as a hot topic of research. 11
DNA topoisomerases are widespread enzymes that are essential to numerous DNA-related biological activities, including DNA unlinking. DNA topoisomerases exist in two varieties: type I and type II. The coumarin and quinolone antibacterial medications’ biological targets have been determined to be DNA gyrase. Many antibacterial and anticancer medications are known to target DNA topoisomerases, particularly type IIA topoisomerases. Several antibacterial substances, such as fluoroquinolones, target DNA gyrase and topoisomerase IV (Top 4), two bacterial type IIA topoisomerases. Clinically significant anticancer medications target the two isoforms of human topoisomerase II (hTop 2) as well as human topoisomerase I (hTop 1). Numerous minor compounds are hTop 1 or hTop 2 inhibitors. 12
The above-discussed therapeutic targets have been selected for the anticancer screening in our study. In this present study, we investigated the phytochemicals present in the two species of Cladophora, namely C. goensis and C. glomerata using gas chromatography-mass spectroscopy (GC-MS), and performed molecular docking studies to reveal the interactions with cancer-related protein and their anticancer activity.
Materials and Methods
Extract Preparation
C. glomerata and C. goensis were collected at Mormugao Bay, Goa, and Hare Island, Tuticorin, Tamil Nadu. They were authenticated at Tamil Nadu Veterinary and Animal Sciences University, Thoothukudi, Tamil Nadu.
The seaweeds (C. goensis and C. glomerata) collected were washed with seawater three times, then successively with tap water and distilled water to remove the epiphytes and other wastes. After thorough washing, they were air-dried under the shade for two weeks and finely powdered using a blender.
The hot solvent extraction process was used for the extraction of the algae. Methanol was used for the extraction of algae. With the use of a Soxhlet device, 25 g of the extract was weighed and extracted with 300 mL of methanol by continuous hot percolation over 10 hours. After being finished, the extract was filtered, condensed with a rotary evaporator at a regulated temperature and lowered pressure of 50°C–60°C (100 mg/mL of concentration), and then stored at room temperature for later use. 13 Methanolic extracts of C. goensis and C. glomerata were subjected to GC-MS analysis.
GC-MS Analysis of Extract
GC-MS analysis was done using electron impact ionization at 70 eV and the data was evaluated using total ion count (TIC) for compound identification and quantification. The spectrums of the components were compared with the database of the spectrum of known components stored in the GC-MS library. Measurement of peak areas and data processing were carried out by Turbo Mass OCPTVS-Demo SPL software.
Molecular Docking
The docking study was carried out using AutoDock tools v1.5.4 and AutoDock v4.2 program (AutoDock, Autogrid, Autotors, Copyright-1991e2000) from the Scripps Research Institute (
The compounds obtained by GC-MS analysis of the methanol marine algae extracts served as a source of ligands. The ligand structures were downloaded as .mol files and using OpenBabel converted into PDB coordinate files (
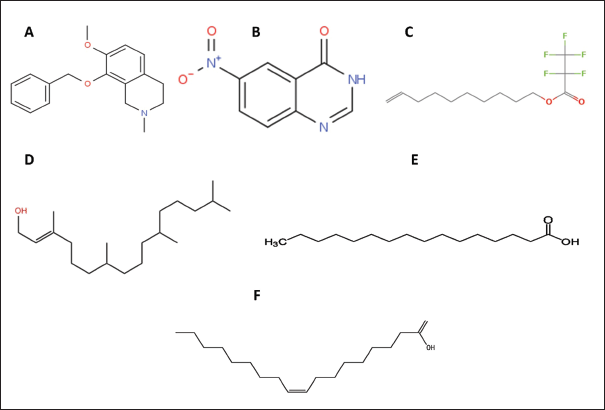
2D Structures of all the Compounds from Both the Extracts. (A) Isoquinoline, 1,2,3,4-tetrahydro-7-methoxy-2-methyl-8-(phenylmethoxy); (B) 6-nitro quinazolin-4-one; (C) 9-decen-1-ol, pentafluropropionate; (D) phytol; (E) palmitic acid; (F) octadec-9-enoic acid.
The 3D (three-dimensional) structures of selected target proteins like human EGFR (4WRG), poly (ADP-ribose) polymerase-1 (4UND), human ER alpha ligand-binding domain (3ERT), human peroxisome PPAR alpha ligand-binding domain (3VI8), human topoisomerase (1EJ9) were recovered from the PDB database (
CASTp was used to look for potential target receptor binding sites. With the aid of Pocket Finder 14 and CASTp analysis 15 , cavities throughout the protein have been investigated. The docking procedure was done as described in the previous study.16, 17
The pharmacokinetic ADMET profile of the study molecules was evaluated using the SwissADME online tool (
Results
Phytochemicals in Cladophora extracts
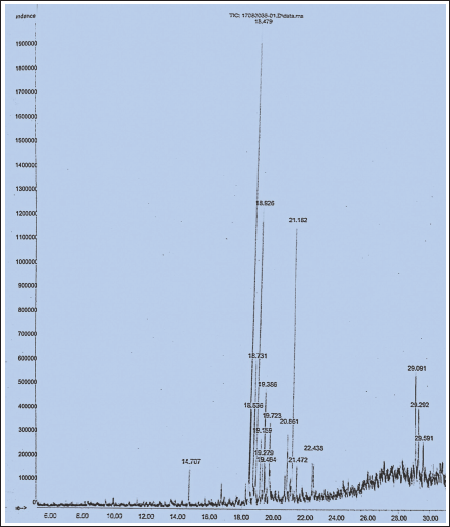
Approximately 19 compounds have been identified from the methanol extract of the marine alga C. goensis, according to the TIC shown in Figure 1. With a peak area of 16.83% and a retention period of 18.479 minutes, the molecule Bicyclo (3,1,1) heptanes-2 carboxaldehyde had the greatest peak area. This substance, which is a derivative of cyclohexane known as cyclohexane-1, 3-dione derivatives, is used to prevent the growth of plants. Other compounds were 6-nitro-3H-quinazolin-4-one, Isoquinoline, 9-decen-1-ol, pentafluropropionate. High concentrations of fatty acids, terpenes, and alkaloids were also detected along with this (Figure 2).
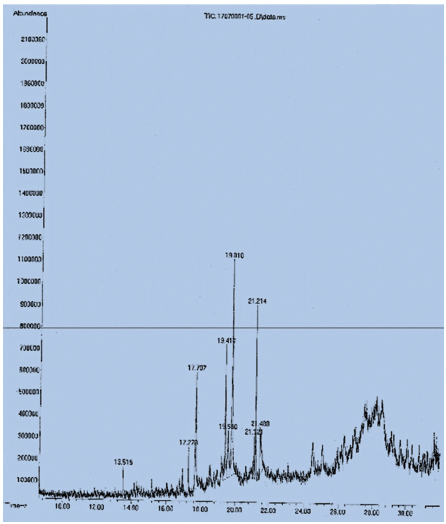
The examination of the methanol extract of the alga C. glomerata revealed approximately 11 phyto-compounds, as can be seen from the TIC in Figure 3. The chemical hexadecanoic acid (palmitic acid) exhibits the highest peak, with a retention period of around 19.810 minutes and a peak concentration of about 30.20%. Tetradecanoic acid overlaps this peak. The methanol extract of C. glomerata contains significant levels of phytol, a chlorophyll derivative, and an excellent source of vitamin K, with a peak area of around 12.39% and a retention period of about 21.213 minutes. There were significant concentrations of long-chain fatty acids present in addition to the substances indicated (Figure 3).
GC-MS Analysis Report of Methanol (Crude) Extract of Cladophora goensis.
GC-MS Analysis Report of Methanol (Crude) Extract of Cladophora glomerata.
Molecular Interactions
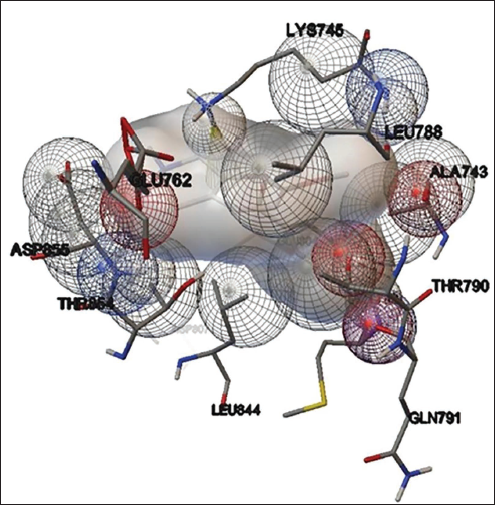
The significant molecular interactions (Binding energy >4.0 kcal/mol) with the selected compounds have been reported in this study. When 14 compounds from C. goensis and 12 compounds from C. glomerata were subjected to in silico docking, only three compounds from each extract showed favorable binding free energies with specified proteins. The molecular interactions of phytochemicals of C. goensis (6-nitro-3H-quinazolin-4-one, Isoquinoline, 1,2,3,4-tetrahydro-7-methoxy-2-methyl-8-(phenyl methoxy) and 9-decen-1-ol, pentafluropropionate) have been tabulated in Table 1, and the molecular interactions of phytochemicals of C. glomerata (phytol, palmitic acid and octadec-9-enoic acid) have been tabulated in Table 2.
Molecular Interactions of Cladophora goensis Phytochemicals with 4UND, 4WRG, and 3ERT Proteins.
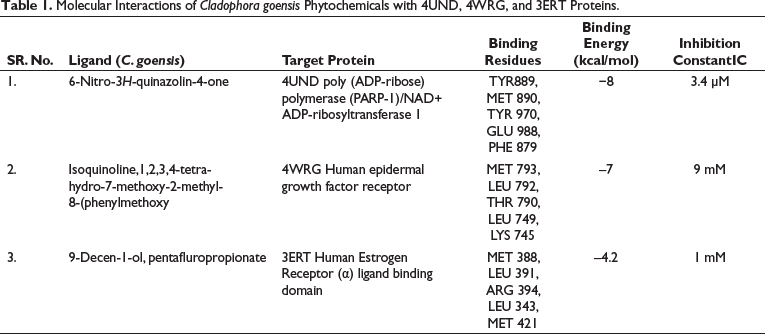
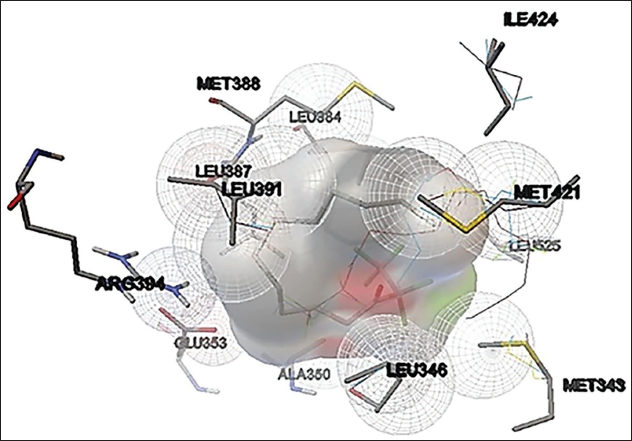
6-Nitro-3H-quinazolin-4-one from the methanol extract of C. goensis showed promising binding activity against poly (ADP-ribose) polymerase (PARP-1)/NAD+ ADP-ribosyltransferase 1. Binding free energy was –8.0 kcal/mol and the Inhibition constant was 3.4µM (Figure 4). Isoquinoline, 1,2,3,4-tetrahydro-7-methoxy-2-methyl-8-(phenylmethoxy) of C. goensis showed promising binding activity against the human EGFR. The binding free energy was –7 kcal/mol and the Inhibition constant of 9µM (Figure 5). 9-Decen-1-ol, pentafluropropionate of C. goensis showed sufficient binding affinity against human estrogen receptor (α) ligand binding domain. The binding free energy was –4.2 kcal/mol and the inhibition constant of 1 mM (Figure 6).
Amino Acid Interaction Poly (ADP-ribose) Polymerase 1 (PARP-1)/NAD+ ADP Ribosyltransferase 1/Poly (ADP-ribose) Synthase 1 in Complex with 6-Nitro-3H-quinazolin-4-one.
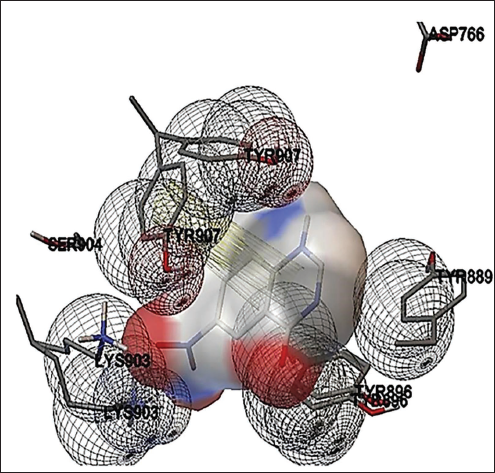
Amino Acid Interactions of Human Epidermal Growth Factor Receptor with Isoquinoline, 1,2,3,4-Tetrahydro-7-methoxy-2-methyl-8- (Phenylmethoxy).
Amino Acid Interaction of Human Estrogen Receptor Alpha Ligand-binding Domain in Complex with 9-Decen-1-ol, Pentafluropropionate.
Molecular Interactions of Cladophora glomerata Phytochemicals with 3VI8, and 1EJ9 Proteins.
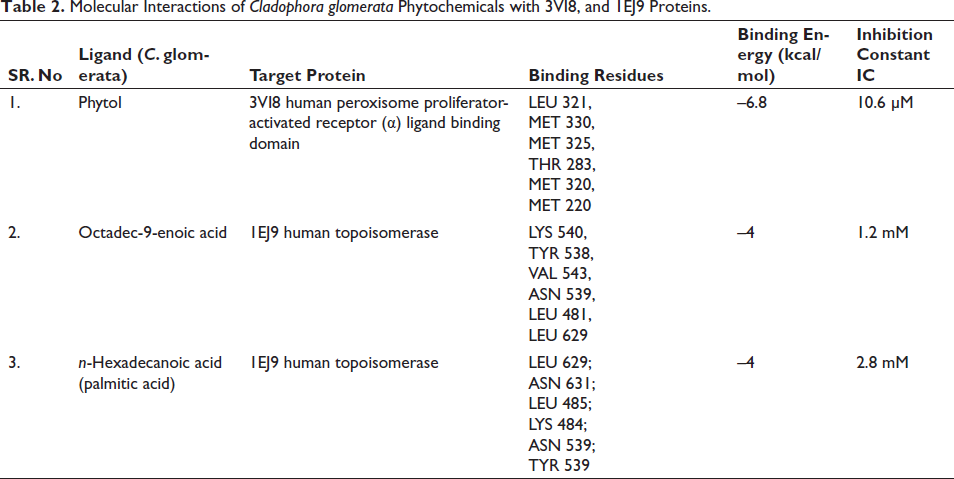
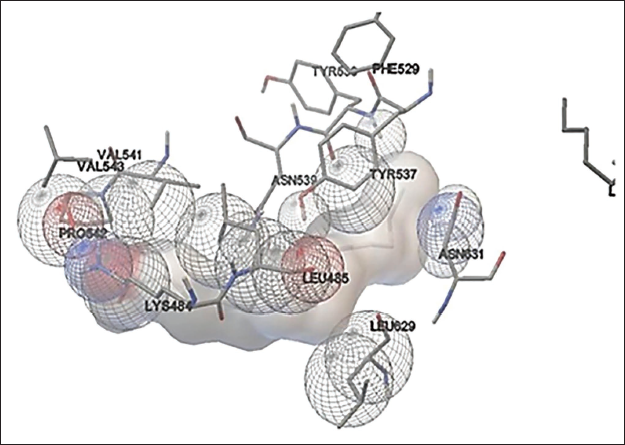
Phytol present in the methanol extract of C. glomerata showed binding activity against the human peroxisome PPARs (α) ligand-binding domain. The binding free energy was –6.8 kcal/mol and the Inhibition constant was 10.6 µM (Figure 7). Octadec-9-enoic acid of C. glomerata showed weak binding affinity against human topoisomerase. The binding free energy was –4 kcal/mol and an Inhibition constant of 1mM (Figure 8). n-Hexadecanoic acid (palmitic acid) of C. glomerata showed weak binding affinity against human topoisomerase. The binding free energy was –4 kcal/mol and the Inhibition constant of 2.8 mM (Figure 9).
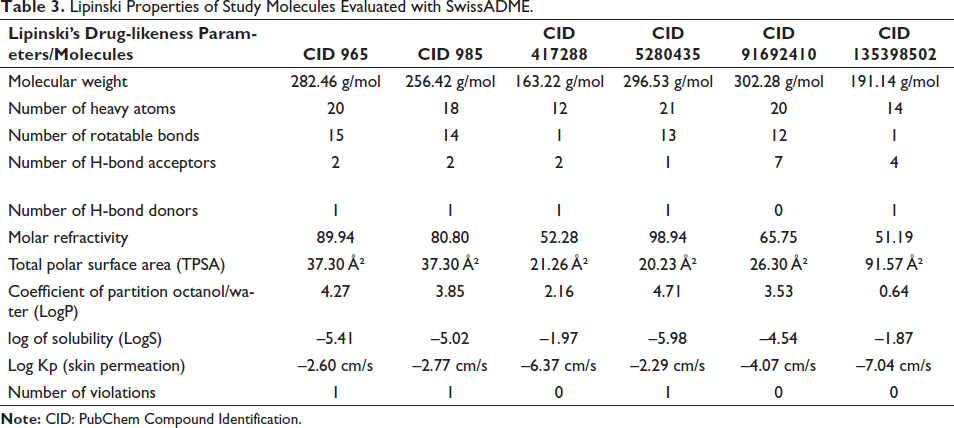
The Lipinski’s drug-likeness properties of the study molecules have been tabulated in Table 3.
Amino Acid Interaction of Human Peroxisome Proliferator-activated Receptor Alpha Ligand-binding Domain in Complex with Phytol.
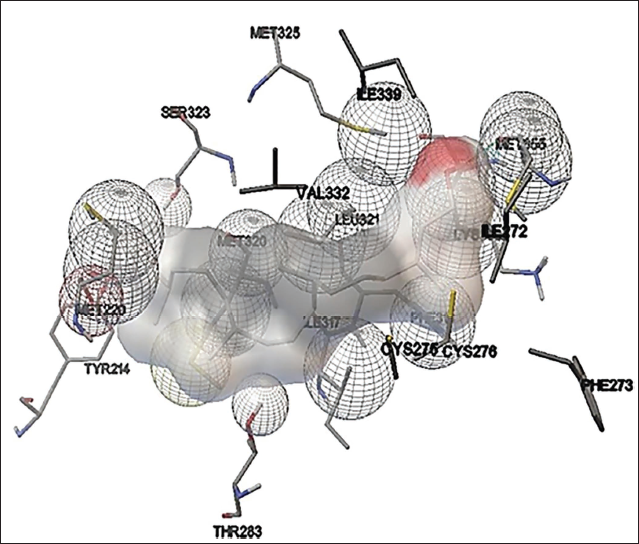
Amino Acid Interaction of Human Topoisomerase in Complex with Octadec-9-enoic Acid-liquorice Model.
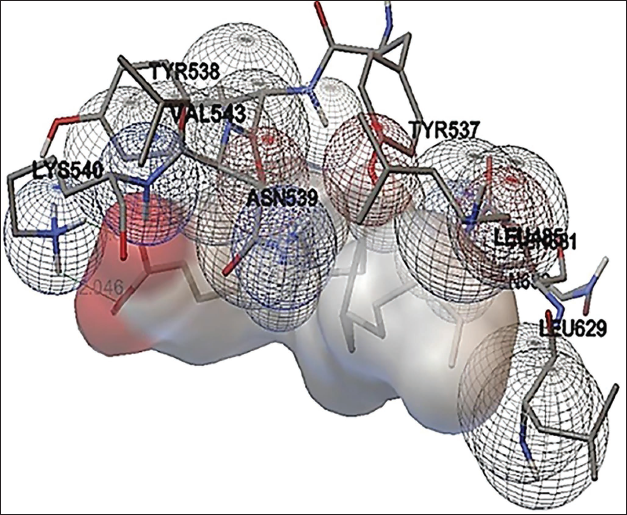
Amino Acid Interactions of Human Topoisomerase Docked with Palmitic Acid.
Lipinski Properties of Study Molecules Evaluated with SwissADME.
Discussion
In structure-based drug design, in silico molecular docking analysis is one of the finest techniques to discover novel ligands for known target receptor structures. 18 Small molecules have been screened by their least score and shape orientations in the binding site of the protein. As a result, novel ligands for receptors of known structure are designed, and their interaction energies are calculated using the scoring functions. 19 Docking score is a measure of interaction and the orientation of the ligand to the active site of the target. More negative values indicate more effective stable conformation of the bound ligand-target. 20
From the data given in the results, we can observe that 6-nitro-3H-quinazolin-4- one from the methanol extract of C. goensis shows promising binding energy of –8 kcal/mol against poly (ADP-ribose) polymerase (PARP-1)/NAD+ ADP-ribosyltransferase. Among the compounds screened from methanol extracts of C. goensis, 6-nitro-3H-quinazolin-4- showed promising results to be developed as a lead anti-cancer drug.
Phytols have also been identified in C. glomerata by previous studies. 21 Phytol from the methanol extract of C. glomerata also shows a promising binding energy of –6.8 kcal/mol against the peroxisome PPARs (α) ligand-binding domain. Among the compounds screened from methanol extracts of C. glomerata, phytol shows promising results to be developed as a lead anti-cancer drug. Phytol, a diterpene alcohol, is a constituent of chlorophyll and is used as a precursor in manufacturing vitamins E and K. 22 Phytol (3,7,11,15-tetramethyl-2-hexadecen-1-ol), which forms the lipophilic side-chain of the chlorophyll. It was reported that metabolites from phytol activate transcription factors and nuclear receptors. Phytol is one of the most important and simplest of the diterpenes. 22 It is highly significant among algal terpenes, as it has a definite role to play in the metabolism of algae. 23 Phytol is a very good lead molecule that can be developed as an anti-cancer drug.
In a study conducted by Silva et al. phytol was found to inhibit inflammatory response by reducing cytokine production and oxidative stress proving that it can also be investigated as a lead molecule for its anti-inflammatory activity. 24 Based on the binding energy values 6-nitro-3H-quinazolin-4-one from the methanol extract of C. goensis shows a better binding energy than compounds from C. glomerata.
The significant interaction of 6-nitro-3H-quinazolin-4-one with 4UND protein was observed at the GLU’988 codon which would be a confirmatory inhibition site as per the previous study 25 . 9-Decen-1-ol, pentafluropropionate which is a bioactive compound was identified in this algae as in other marine algae 26 , has shown similar interactions comparable to active metabolites of tamoxifen at ART’394 and MET’421 amino acid codons of 3ERT 27 . Phytol interaction at LEU’321 was shown to be a significant agonistic activity site for PPAR alpha protein to exhibit anticancer as well as antidiabetic activity. 28 Also, in vitro studies in the HT-29 cancer cell line, with the extracts of C. goensis show significant anti-cancer activity. 29 The presence of hexadecanoic acid in C. glomerata was also reported by other studies. 30 Thus, these phytochemicals from the marine algae Cladophora spp. are good preliminary lead molecules for anti-cancer drug development.
Conclusion
The present study has revealed the phytochemicals of C. glomerata and C. goensis species which consisted of phytols, 6-nitro-3H-quinazolin-4-one and 9-decen-1-ol, pentafluropropionate. These phytochemicals have shown significant interactions at the potent sites of proteins 3VI8, 4UND, and 3ERT respectively. This may provide sufficient preclinical data for continuing further preclinical and clinical research for the development of anticancer molecules from these algae.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
This project was funded by ICMR under the TSS fellowship scheme: PhD (Integrated)/-35-F.T./1/2015).
