Abstract
Antimicrobial resistance (AMR) is a serious global health issue, and it is greatly influenced by the gut flora. The rationalization of antimicrobial doses in clinical studies is crucial for preventing AMR. This review analyzes how rationalization tactics affect AMR and gut microbiota in clinical studies. Studies that provided data on the use of antibiotics, AMR, or gastrointestinal microbiota were taken into account for the current review. The AMR rate was found to be low when healthy gut flora was maintained using various antibiotic rationalization techniques, such as limited use of antibiotics or targeted treatments. However, the effectiveness of these strategies differed based on the particular intervention, the research population, and the length of the course of therapy. The rationalization of antibiotic prescriptions in clinical research is one potential method for reducing the prevalence of AMR by maintaining the gut flora. Rationalization techniques may help lower AMR rates and foster the development of good intestinal flora. This review describes various antibiotic rationalization techniques and the importance of maintaining healthy microbial flora to minimize AMR-associated health issues.
Keywords
Introduction
Antimicrobial resistance (AMR) is a global problem that is significantly threatening public health and is growing rapidly. The ability of bacteria, viruses, and fungi to resist the effects of antimicrobial agents is known as AMR. As a result, treating infections has become more difficult, leading to longer hospital stays, higher healthcare costs, and higher mortality rates. 1 AMR is largely attributable to the inappropriate use of antibiotics, which forces micro-organisms to develop resistance. The gut microbiome is home to a diverse array of bacteria, viruses, fungi, and other micro-organisms, which are collectively known as the gut microbiota. 2 This group of bacteria interacts often with the host and is crucial for maintaining human health. The transfer of resistance genes can occur through mechanisms such as conjugation, transduction, and transformation and can result in the rapid spread of resistance among different species of bacteria. 3 The sensitive balance of the gut microbiota is thus disrupted, serving as a reservoir for resistance genes and promoting the emergence of AMR. Antibiotic use can disrupt the gut microbiome by eliminating beneficial microbes, leading to an overgrowth of harmful bacteria as well as Candida albicans. This imbalance can increase the risk of acquiring AMR infections due to the likelihood that most commensal bacteria may acquire resistance genes and pass them on to other bacteria. 4 The misuse and overuse of antibiotics, which puts pressure on bacteria to develop tolerance to these antibiotics, is one of the primary reasons for AMR. When antibiotics are prescribed inappropriately or excessively, bacteria are exposed to the drugs for longer periods, increasing the likelihood of developing resistance. The use of antibiotics can also disrupt the balance in the gut microbiota, which may increase the risk of the emergence of AMR strains. Therefore, it is critical to enhance the justification of antibiotic prescriptions in order to handle the AMR problem. This can be achieved through a number of strategies, including not using antibiotics for primary care or over-the-counter, promoting the use of alternative treatments, and educating patients about the rational use of antibiotics. Health professionals, such as doctors and pharmacists, play an important role in encouraging the prudent use of antibiotics by prescribing them only when it is required and ensuring patients receive the appropriate advice and support. 5
For the purpose of creating strategies for preventing the development of resistance, it is crucial to comprehend how the gut microbiome contributes to AMR. This includes reducing the use of antibiotics, promoting the growth of beneficial bacteria, and developing new methods for treating infections that do not rely on antibiotics. In order to successfully combat this growing public health threat, a comprehensive strategy for AMR that considers the gut microbiome and other aspects is essential. Therefore, rationalizing antibiotic prescriptions is essential to combat AMR and maintain the health of the gut microbiota. This requires a multi-faceted strategy that involves reducing the use of antibiotics, promoting alternative treatments, improving patient education, and developing new diagnostic tools and treatments. Working collaboratively, it is possible to prevent the emergence of AMR and guarantee that treatments for infectious diseases remain efficient for many years to come. 6 The micro-organisms that reside in and on human bodies are collectively referred to as the microbiome. These microbes are essential to our health and well-being. A healthy microbiome helps to keep our body in balance by performing important functions such as (1) boosting the immune system: the microbiome aids in boosting immunity and protecting against harmful bacteria and other pathogens; (2) digestion support: the microbiota aids in food digestion and nutrient extraction; and (3) regulating body metabolism: the microbiota assists in regulating body metabolism, including how we store and utilize food-derived energy.7, 8 However, imbalances in the microbiome can cause a number of health issues, such as (1) inflammatory bowel disease (IBD), a condition that causes inflammation in the digestive system, and its onset has been linked to a microbiome imbalance; (2) obesity and other metabolic problems: usually, an unbalanced microbiome has been linked to type 2 diabetes; and (3) mental health disorders: the microbiome has also been linked to mental health disorders such as depression and anxiety. 8 This current review aims to explore changes in the distribution of normal gut flora when antibiotics are consumed. It also highlights the importance of maintaining a healthy, normal gut microbiome to prevent the development of various clinical conditions. Additionally, this review discusses various strategies recommended by government agencies to prevent the development of AMR in the Indian scenario.6, 9
The Strategy of Data Search
Data was collected from articles in English that were published in journals that were indexed in Scopus, Web of Science, Google Scholar, and PubMed. We included studies published until March 2023, as there was no constraint on the date of publication.
AMR and Gut Microbiome
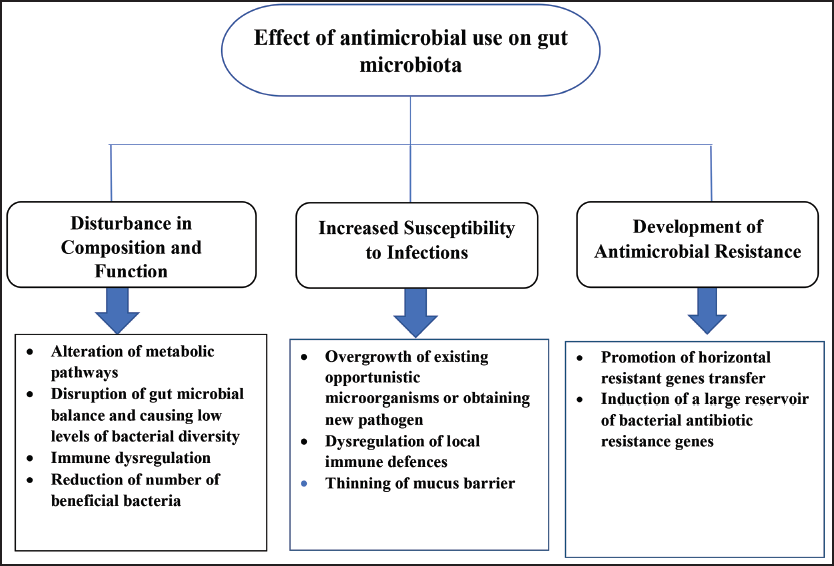
AMR is a condition where the drugs used to regulate the growth of micro-organisms are no longer effective, causing a persistent increase in the number of micro-organisms. AMR is influenced by the gut microbiome that inhabits our digestive system. Antibiotics can kill both pathogenic and commensal bacteria in the gastrointestinal tract, causing an imbalance in the microbiome when a person consumes antibiotics (known as dysbiosis). This dysbiosis can enhance the growth of antibiotic-resistant bacteria, which can easily be disseminated to other people and cause diseases that are difficult to treat. It is evident that there is a connection between AMR and the gut microbiome and that it is caused by the overuse of antibiotics, which can disrupt the equilibrium of bacteria in the gut and increase the growth of bacteria that are resistant to antibiotics.10, 11 Figure 1 shows the effect of antimicrobial use on the gut microbiota.
Effect of Antimicrobial Use on Gut Microbiota.
Global Overview of AMR
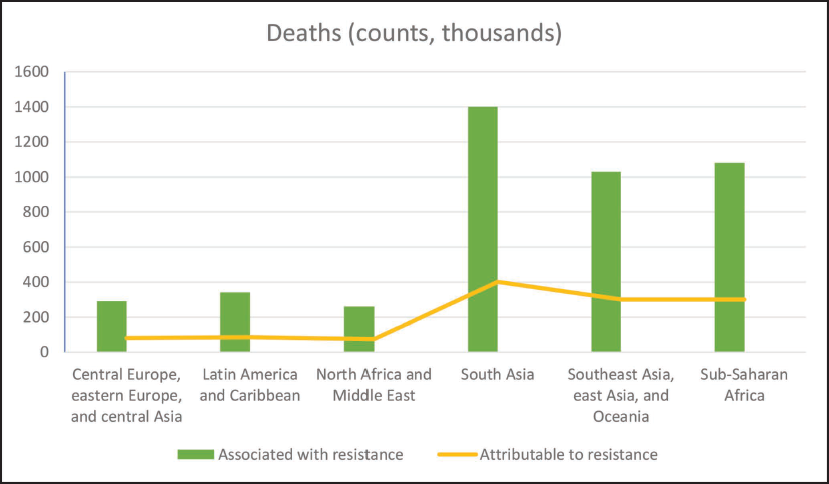
AMR is a global health issue affecting millions of people across the world, and India also confronts major difficulties in managing it. AMR is spreading across the world, and it is estimated that it will result in 10 million deaths a year and may cost the global economy up to 100 trillion USD by 2050. Inappropriate and excessive use of antibiotics has led to the emergence of antibiotic-resistant bacterial strains, both in India and worldwide. In India, the availability of antibiotics without prescription over-the-counter and inadequate infection control measures have contributed to the misuse of antibiotics, resulting in the development of hard-to-treat, antibiotic-resistant bacterial strains.12, 13 This trend is particularly evident in the increasing prevalence of highly infectious drug-resistant multi-drug-resistant tuberculosis and extra-drug-resistant tuberculosis cases in India. Furthermore, AMR poses challenges in treating bacterial illnesses such as pneumonia and urinary tract infections.13, 14 To address the growing threat of AMR, the Indian government launched the National Action Plan on AMR in 2017 as a proactive defensive strategy. This plan emphasizes the importance of appropriate antibiotic use, enhancing infection prevention and control practices, and supporting the development of novel therapeutic agents to combat AMR. However, the current scenario of AMR in India is deeply concerning due to the rising number of infectious diseases and the proliferation of antibiotic-resistant bacterial strains.15, 16 Globally, the misuse of antibiotics and the consequent development of AMR strains pose significant health risks. India’s situation serves as a microcosm of this global challenge, with factors such as over-the-counter antibiotic availability and insufficient infection control measures exacerbating the problem. 17 The National Action Plan on AMR reflects India’s commitment to address the issue by promoting responsible antibiotic usage, strengthening infection prevention and control practices, and facilitating the development of new treatments. The global overview of AMR highlights the urgent need for action to address this growing public health crisis, which poses an important threat to global health.17, 18 Figure 2 shows the deaths associated with or attributable to bacterial AMR. 19
Deaths Associated or Attributable to Bacterial Antimicrobial Resistance. 19
Predisposing Factors for the Development of AMR
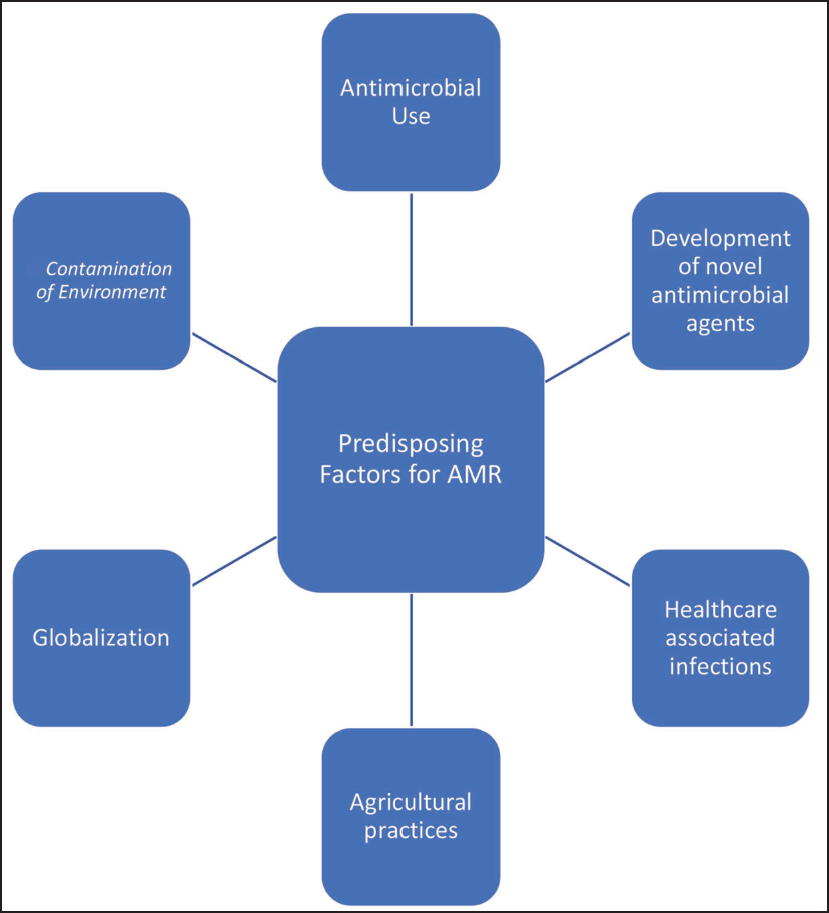
AMR is a phenomenon wherein micro-organisms develop resistance against the effects of antimicrobial drugs, rendering these drugs worthless for the treatment of infections. Figure 3 demonstrates various predisposing factors for the development of AMR. The growth and dissemination of AMR is a complex, multifaceted issue that has an impact on a variety of related factors, as mentioned below:
Antimicrobial Use
Antibiotic overuse and misuse in the context of human health are the primary causes of AMR. Antibiotics are frequently prescribed in human healthcare for viral infections when they are not necessary, which promotes the development of resistant strains. Animal agriculture uses a lot of antibiotics to promote growth, similar to human healthcare, which contributes to creating and spreading resistant bacteria.
20
The survival and spread of resistant bacteria pose significant challenges in the treatment of diseases. To avoid AMR, it is essential to promote responsible antimicrobial use through guidelines, suggestions, and regulations that prioritize proper prescriptions and restrict the use of non-therapeutic antibiotics in humans.
Development of Novel Antimicrobial Agents
The slow pace of the development of new antibiotics and the scarcity of many different types of available drugs make the AMR issue more challenging. Fewer novel antibiotics reach the market as a result of the slow pace of conventional antibiotic research, whereas the targets and structures of current antibiotics are frequently identical, leaving them open to resistance. As bacteria change and gain resistance, the lack of effective antibiotics presents a significant challenge in the management of infectious diseases.
21
To address this issue, efforts are being undertaken to promote the development of antimicrobial drugs through greater financing, research partnerships, and regulatory incentives. The goal is to encourage the development of novel antibiotics with distinct modes of action that will make it more difficult for bacteria to acquire resistance. Alternative methods, such as combination therapy and research into unconventional antibacterial drugs, are available.
22
Healthcare-Associated Infections
AMR is being disseminated in healthcare settings due to insufficient infection prevention and control methods. Patients who have resistant bacteria can readily spread them to others, potentially causing serious infections that are challenging to cure. This issue is exacerbated by elements including poor hand hygiene, inadequate disinfection procedures, congestion, and antibiotic abuse. The problem is made worse by insufficient surveillance, the detection of resistant strains, isolation procedures, and the use of personal protective equipment.
23
Strong infection prevention and control procedures must be prioritized by healthcare facilities in order to stop the spread of AMR. These practices should encourage good hand hygiene, efficient disinfection, practical isolation techniques, and prudent antibiotic usage. Early detection and containment are aided by improved monitoring and quick diagnoses. Healthcare institutions may lessen AMR transmission, safeguard patients, and maintain quality treatment by putting in place comprehensive policies.
24
Agricultural Practices
AMR is a phenomenon that is greatly influenced by agricultural practices. Antibiotics are frequently used in agriculture to prevent illness and promote animal development. Antibiotic exposure caused by preventative usage leads to the selection of resistant types of bacteria. Antibiotics used to promote growth at sub-therapeutic levels also contribute to the emergence of AMR.
25
Agriculture-related resistant bacteria might get into the food chain and endanger the health of people. In order to address this, guidelines and best practices are being used to encourage safe antibiotic usage. Reducing the need for antibiotics and thereby reducing AMR can be achieved through enhanced animal husbandry, biosecurity, and immunization programs. Prioritizing animal welfare and reducing needless antibiotic usage are crucial agricultural practices that preserve both the health of animals and people.
26
Globalization
AMR poses a severe threat to world health due to its rapid spread as a result of increased international travel and trade. Travelers may transport resistant bacteria from one country to another, dispersing them. AMR may spread through the trade of goods, particularly food and agricultural items. Direct touch, contaminated surfaces, or genetic exchange can result in the spread of resistant strains.
23
The effects are severe, lowering the efficacy of antimicrobial therapies, raising morbidity and mortality rates, and raising healthcare expenses. International cooperation, monitoring systems, data sharing, and coordinated policies are needed to combat global AMR. To develop ways to stop AMR transmission and maintain the efficacy of antimicrobial agents internationally, collaboration between healthcare sectors, regulatory agencies, and international organizations is essential. Recognizing the impact of globalization on AMR and taking collective action can prevent further spread, protect public health, and ensure effective treatment options worldwide.23, 26
Contamination of the Environment
A major factor in the spread of AMR is the contamination of the environment with bacteria that are resistant to antibiotics through wastewater from numerous sources, including industrial sites and sewage treatment facilities. Antibiotic-resistant bacteria can reach soil and water sources due to insufficient wastewater treatment and disposal, which serves as a breeding ground for them.
27
The discharge of treated wastewater that still has resistant strains in it can occur if sewage treatment facilities are unable to completely eradicate resistant bacteria and their genetic material because contact with polluted soil or water can spread illnesses that are difficult to cure. This environmental pollution puts human health at risk. Modern technology, safe antibiotic usage, effective wastewater management, and proper wastewater treatment are all necessary to address this problem. Antibiotic-resistant bacteria must be kept to a minimum in the environment in order to control their spread, safeguard ecosystems, and lessen the harm they pose to human health. For successful environmental management methods to address AMR, cooperation between industry, policymakers, and environmental authorities is required.27, 28
Demonstrates Various Predisposing Factors for the Development of Antimicrobial Resistance.
Government Policies on Contamination of Environment
Government policies regarding the contamination of the environment focus on various aspects to mitigate the release of pollutants and minimize their impact on ecosystems and public health. Some common policies include:
National Program on AMR Containment
AMR is a pressing public health concern, with resistance emerging against newer and more potent antimicrobial agents. The fast spread of multidrug-resistant bacteria and the constrained supply of new medicines represent a serious threat to people’s health. As part of its 12th five-year plan, the Indian government introduced the “National Programme on AMR Containment” in 2013 to address this issue.
32
The program, coordinated by the National Centre for Disease Control’s (NCDC) Centre for Bacterial Diseases and Drug Resistance, attempts to put AMR containment tactics into practice. In order to reduce the spread of resistant bacteria and maintain the efficacy of currently available antibiotics, the NCDC emphasizes surveillance, responsible antimicrobial use, and enhanced infection control procedures. India’s “National Programme on AMR Containment” demonstrates a proactive approach to combating the escalating threat of AMR. By prioritizing awareness, surveillance, and control measures, the program strives to safeguard public health and ensure viable treatment options for infections both now and in the future. Through these efforts, India is taking significant steps to address the challenges posed by AMR and protect the well-being of its population.32, 33 The primary objectives of the National Program on AMR Containment include:
AMR Surveillance
A critical component of India’s National Program on AMR Containment is AMR Surveillance. To assess the prevalence and trends of AMR in various parts of the nation, the National AMR Surveillance Network (NARS-Net) was created. Government medical colleges that participate in NARS-Net have their laboratories supported for capacity enhancement. These labs are essential to gathering and reporting data on AMR for seven important priority bacterial diseases for public health. These pathogens include Salmonella enterica serotypes Typhi and Paratyphi, Acinetobacter baumannii/Acinetobacter calcoaceticus complex, Enterococcus spp., Klebsiella spp., Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus. 32
NARS-Net
BJ Medical College, Ahmedabad, Gujarat
BJ Medical College, Pune, Maharashtra
Medical College and Hospital, Chandigarh
GSVM Medical College Kanpur, UP
Lady Hardinge Medical College, Delhi
Mysore Medical College and RI Mysore, Karnataka
SMS Medical College, Jaipur, Rajasthan
VMMC and associated Safdarjung Hospital, Delhi
Government Medical College, Thiruvananthapuram, Kerala
KAPV Government Medical College, Tiruchirappalli, Tamil Nadu. 32
Effect of Antimicrobial Use on the Gut Microbiota and Resistome
Antibiotic consumption may have a major impact on the gut flora, changing the makeup, diversity, and function of the microbial community. The complex and dynamic population of bacteria known as the gut microbiome plays crucial roles in maintaining gut barrier function, immune system regulation, and food metabolism. Antibiotics can disrupt the gut microbiota by selectively targeting specific bacterial populations, altering the overall microbial composition, and reducing microbial diversity. This can result in increased antibiotic resistance in bacteria and the development of an AMR reservoir, known as the resistome, within the gut.6, 34 Studies show that antibiotic consumption can increase the number of bacteria, like P. aeruginosa, Klebsiella pneumoniae, and E. coli, in the gut that are resistant to them. This may lead to the emergence and dissemination of AMR because pathogenic and commensal bacteria may exchange antibiotic-resistant genes. The use of antibiotics can also disrupt the metabolic functions of the gut flora, altering the production of short-chain fatty acids, vitamins, and other metabolites that are important nutrients for good human health. To reduce the influence on the gut microbiota and resistance, antibiotic usage should be carefully monitored and administered judiciously. This can limit the spread of AMR and keep the beneficial effects of the gut flora intact.6, 35 Figure 4 shows the effect of antimicrobial use on the gut microbiota.
Effect of Antimicrobial Use on the Gut Microbiota.
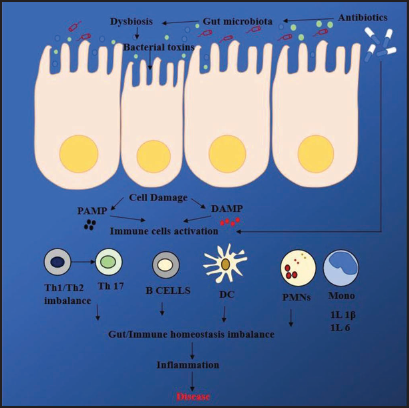
Figure 4 illustrates that the use of antibiotics may destroy the normal gut flora, causing dysbiosis. This dysbiosis may result in the overgrowth of pathogenic bacterial strains that produce various virulence factors, thereby damaging the normal intestinal epithelial cells. This damage further induces the pathogen-associated molecular patterns and damage-associated molecular patterns-mediated signaling pathways of the immune response, causing an acute inflammatory process. Various immune cells that take part in the acute inflammatory process include Th (T helper cells), B cells, dendritic cells, peripheral nuclear lymphocytes, and monocytes (Figure 4).
Methods of Preventing AMR
AMR must be avoided to reduce the prevalence of diseases caused by antimicrobial-resistant bacterial strains. Multiple AMR prevention strategies are listed below.
Hygiene and Barrier Precaution
A substantial source of contamination from drug-resistant bacteria is healthcare personnel. Gloves and gowns are advised by guidelines when caring for infected patients. Studies have looked at how cleanliness standards and good hand hygiene affect the transmission of bacteria that are resistant to antibiotics. Glove and gown contamination rates among healthcare professionals range from 4% to 67%.
36
Attending patients with particular medical devices and making contact with specific patient body parts are factors linked to higher contamination rates. There was no link between the length of time spent in a patient’s room and a greater chance of developing drug-resistant microbes. The study emphasizes the importance of protective barriers like gloves and gowns, as 17.5% of healthcare workers’ protective clothing was found to be contaminated. Meta-analysis indicates that both home and industrial laundering can reduce microbial contamination on uniforms. Overall, a comprehensive approach, including gloves, gowns, proper hand hygiene, and garment removal, is necessary to protect patients and healthcare workers.
37
Environmental Cleaning
Hospital environmental cleaning procedures can be improved to lessen the spread of MRSA and VRE, two bacteria that are resistant to several common antibiotics. In a medical intensive care unit (MICU), researchers performed a multistage intervention that included improved daily cleaning practices and a hand hygiene campaign. Infection rates for VRE dropped from 33.47 cases per 1000 patient days to 10.40 cases per 1000 patient days, and clinical culture detection rates for VRE similarly dropped. In another investigation, an ICU patient’s MRSA infection was shown to be positively correlated with environmental pollution. Patients were more likely to get MRSA in rooms that had previously been inhabited by MRSA carriers.
38
Prophylaxis and Topical Decolonization
S. aureus colonization offers a serious risk for infection, especially in the nose. To stop S. aureus infections, preventative methods and decontamination techniques have been investigated. Studies have demonstrated that preoperative administration of the antibiotic drug mupirocin helps to lower S. aureus-related infections in healthcare settings. A cost-effective technique that saved money and avoided infections was to screen patients for S. aureus and provide mupirocin to carriers. Pre-admission fast testing for S. aureus nasal carriage and subsequent decontamination treatment in patients undergoing elective surgery also produced significant cost savings and avoided in-hospital fatalities. Intranasal mupirocin, chlorhexidine gluconate wash, oral rifampin, and doxycycline demonstrated effectiveness in curing MRSA infections. These findings emphasize the need for preventative measures, including topical decontamination, to reduce the burden of S. aureus infections and improve patient safety in healthcare settings.
39
Antibiotic Overuse
The misuse of antibiotics, which frequently results in incorrect prescriptions and the establishment of antibiotic resistance, is a serious problem in medical practice. An effective way to reduce the use of antibiotics and combat resistance is the Swedish Strategic Programme for the Regional Use of Antimicrobial Agents and Surveillance of Resistance. Sweden observed a decrease in both the overall usage of antibiotics and the number of prescriptions thanks to concerted efforts, showing that resistance may be reduced. Additionally, it is critical to avoid using antibiotics in situations where they are not essential, such as the treatment of asymptomatic bacteriuria, since eliminating micro-organisms might hasten the emergence of bacteria that are resistant to certain antibiotics. Implementing strategies like a monthly rotation policy for broad-spectrum antibiotics and post-prescription reviews with physician input in hospitals has shown promise in lowering antibiotic use and enhancing treatment appropriateness. With these steps, the overuse of antibiotics will be addressed, and ethical antibiotic prescription procedures will be encouraged.
40
Prevention of Infection and Control
Infection control and prevention practices can help to a significant extent in the prevention of AMR. By implementing practices like hand hygiene, environmental cleaning, and isolating ill people, infections can be prevented from spreading. These prophylactic measures minimize the selection pressure that promotes the establishment of resistance by reducing the overall demand for antibiotics.
40
Vaccination
Vaccination is another important strategy for avoiding AMR; by providing protection against specific diseases, immunizations help to lower the incidence of infections. The use of antibiotics can be cut back by avoiding illnesses in the first place and limiting the spread of antibiotic resistance. Therefore, in addition to protecting individuals from sickness, vaccination also helps curb the use of antibiotics generally and the growth of antibiotic-resistant germs.
41
Development of New Antibiotics and Alternative Treatments
New antibiotic research and alternative treatment modalities are crucial for combating AMR. Funding research and innovation in the discovery and development of antimicrobial medications are beneficial in addressing the issue of drug-resistant infections. Healthcare systems can improve their options for treating infections and reduce their reliance on current antibiotics by fostering the development of new antibiotics and investigating alternative therapies such as phage therapy or combination medications.
41
Education and Awareness
Programs for education and awareness are essential for preventing AMR. The prevalence of AMR can be reduced by promoting judicious antibiotic usage, educating medical professionals and the public on proper administration and treatment compliance, and raising awareness of the risks of AMR. Additionally, educational efforts emphasize how important it is to complete all recommended antibiotic regimens, refrain from abusing them, and distinguish between bacterial and viral illnesses.40, 41
Antibiotic Stewardship and Rationalization of Prescription
Healthcare workers, the pharmaceutical industry, and the government must collaborate to improve the proper use of antibiotics and reduce the onset of AMR. In clinical practice, rational prescription involves choosing the right medication at the right dose for the right amount of time and only prescribing antibiotics when they are necessary. This serves to avoid the emergence of AMR by limiting the amount of time that bacteria are exposed to antibiotics and reducing the pressure of selection that promotes the evolution of resistant strains. The rationalization of prescriptions also helps to reduce the spread of AMR by ensuring that antibiotics are used most appropriately and effectively. A multidisciplinary approach is necessary to prevent AMR, including the prudent use of antibiotics, infection prevention and control methods, vaccination, surveillance, and monitoring, as well as the research and development of new antibiotics and alternative treatments.42, 43
National AMR Policy of India
The National AMR policy was developed in India in response to the growing public health threat presented by AMR. India has a high incidence of infectious diseases, and inappropriate use of antibiotics and poor storage of medications have increased the spread of bacterial strains that are challenging to treat and resistant to common antibiotics. India needed a national AMR plan due to a variety of factors, including the high prevalence of contagious diseases, the overuse and abuse of antibiotics, and the lack of effective preventative measures. To lower the prevalence of AMR, the National AMR Policy of India was introduced in 2017. Its objectives include encouraging proper antibiotic usage, enhancing infection prevention and control procedures, and assisting in the discovery of novel antimicrobial agents. The strategy also intends to encourage research and the development of novel antibiotic therapies for infections, enhance the monitoring and surveillance of AMR, and raise awareness of the dangers of AMR and the appropriate use of medicines.44, 45
National Action Plan on AMR
The National Action Plan on AMR (NAP-AMR) in India is a comprehensive strategy created to combat the threat that AMR poses to public health. The approach adheres to the Global Action Plan on AMR that was authorized by the World Health Organization. The Indian government has made numerous attempts and engaged in high-level discussions to demonstrate its commitment to battling AMR. There are six strategic priorities that direct the NAP-AMR. Raising public knowledge and comprehension of AMR through effective communication, instruction, and training is the top goal. The second goal focuses on advancing information and data through monitoring AMR, which includes laboratory amplification and surveillance in the human, animal, food, and environmental sectors. Reducing the occurrence of infection through efficient infection prevention and control practices in the community, animal health, food, and healthcare is the third goal. The fourth priority focuses on regulating, controlling access to, and implementing stewardship programs to optimize the use of antimicrobial agents in food, medicine, and animal care. 46 The fifth objective encourages funding for AMR initiatives, research, and breakthroughs, such as the creation of novel treatments and diagnostics. The sixth objective focuses on enhancing India’s leadership in AMR through partnerships at the international, federal, and state levels. Specific actions, activities, and outputs have been outlined under each strategic priority, along with their associated responsibilities and timetables. The plan comprises short-, medium-, and long-term goals to effectively tackle AMR. The creation of governance structures like the Intersectoral Coordination Committee, Technical Advisory Group, and Core Working Group on AMR is necessary for the implementation of the NAP-AMR. These organizations ensure that the strategy is developed and carried out with technical knowledge, oversight, and coordination. In general, the NAP-AMR in India shows a multifaceted strategy to combat AMR, including awareness campaigns, monitoring systems, infection control measures, responsible use of antibiotics, research and innovation, and national and international partnerships. The six strategic priorities of the NAP-AMR aim to address key areas for combating AMR effectively. These goals aim to increase awareness, improve understanding and evidence, lower the risk of infection, utilize antibiotics more effectively, encourage investment, and boost India’s position as a leader in AMR control.46, 47
The focus areas of the six strategic priorities of NAP-AMR are:
Improve awareness and understanding of AMR through effective communication, education, and training.
In order to increase knowledge of AMR and its effects among policymakers, the general public, and farmers, it is essential to use communication and IEC resources. Providing comprehensive education and training programs to professionals in relevant fields to enhance their knowledge and skills.
46
Strengthen knowledge and evidence through surveillance.
Increasing the capability of labs in many industries to increase performance. Using surveillance techniques to detect and monitor the occurrence of antibiotic resistance.
46
Reduce infection incidence through effective prevention and control.
Optimize antimicrobial agent use in health, animals, and food.
Implementing effective regulations, ensuring access to antimicrobials, and strengthening surveillance of their use. Encouraging the use of antimicrobial stewardship techniques in the healthcare industry. Improving agricultural practices and animal health optimization measures.
46
Promote investments in AMR activities, research, and innovations.
Promoting the creation of fresh treatments and diagnostics for better illness management. Supporting creative methods for controlling infectious illnesses. Developing long-term financial strategies to assist AMR control initiatives.
46
Strengthen India’s leadership on AMR.
Increasing international cooperation and alliances to combat AMR globally. Encouraging partnerships and collaborations at the federal and state levels to fight AMR in particular areas and jurisdictions.
46
The NAP-AMR is a comprehensive strategy for addressing the threat posed by AMR in India, with a focus on enhancing surveillance and methods for preventing and controlling infections, encouraging the prudent use of antibiotics, helping in the development of novel therapies, and informing the public about the risks of AMR. The NAP-AMR is a crucial step in lowering the prevalence of AMR while preserving the efficiency of the current drugs.46, 47
Gut Microbiome Modulation as a Tool Against AMR
The community of bacteria that inhabits the gut, known as the gut microbiome, is essential for preserving human health and controlling the immune system. Dysbiosis, commonly referred to as an unbalanced gut microbiome, has been linked to infections caused by resistant bacteria. Several methods can be employed to alter the gut microbiota as a defence against antibiotic resistance.
Prebiotics
Prebiotics are dietary fibers that serve as food for beneficial gut bacteria, promoting their growth and helping to maintain a healthy gut microbiome. Prebiotics can be used to modulate the gut microbiome as a tool against AMR by encouraging the development of helpful micro-organisms that can outcompete antibiotic-resistant bacteria. 48
Probiotics
Live bacteria known as probiotics are available as supplements or in fermented foods like yogurt, kefir, and sauerkraut. Probiotics can be used to modulate the gut microbiome as a tool against AMR by colonizing the gut with beneficial bacteria that can outcompete antibiotic-resistant bacteria and enhance the body’s natural defence against infections.48, 49
Fecal Microbiota Transplantation
Fecal microbiota transplantation (FMT) is a method that restores healthy gut microbiota by transplanting the gut microbiome of a healthy individual into the recipient’s gut. FMT has been demonstrated to be successful in treating Clostridium difficile infections, which are frequently brought on by micro-organisms that are resistant to antibiotics. 50
Stewardship of Antibiotics
Antibiotic stewardship is the sensible use of antibiotics to prevent the emergence of bacteria that are resistant to them. This can be accomplished by using antibiotics just as needed, selecting the proper drug for the ailment, and avoiding antibiotic misuse. By reducing the selection pressure that promotes the evolution of resistant strains, it is feasible to manipulate the gut microbiome as a defence mechanism against AMR. 51 The gut microbiome can be modulated as a tool against AMR by using prebiotics, probiotics, FMT, and promoting antibiotic stewardship. These methods can assist in restoring a balanced gut microbiome and preventing the growth of bacteria that are resistant to antibiotics.
Antibiotic Resistance Genes Found in the Microbiota
Antimicrobial resistance genes (ARG) that are primarily prevalent in different microbiomes have been identified by regular metagenomics research. These genes are linked to common clinically used medications and are carried by species that are not the gut’s dominant inhabitants. As a result, the conventional technique cannot recognize these genes because these ARGs amplify genes that are insignificantly represented. Other techniques, like tailored metagenomics, have been created to amplify thousands of resistance genes with clinical significance. These techniques have the benefit of improving gut gene detection but have the disadvantage of being focused (as they amplify only known genes). There are many different resistance genes in the gut microbiota, but those that have been documented most often are those that are specifically aimed against tetracycline, beta-lactams, aminoglycosides, glycopeptides, chloramphenicol, and macrolides, respectively. 52 The lung microbiota mainly consists of beta-lactamase genes, while the epidermis microbiome is a significant source of genes for resistance to a wide range of antibiotics, including lactamases, glycopeptides, aminoglycosides, fluoroquinolones, and macrolides. Lincosamide-synergists efflux pump mechanisms are a common resistance mechanism across different microbiomes. Fecal samples often contain resistance alleles for antibiotics such as tetracycline or chloramphenicol, though it is unclear why these resistance genes are present at such high levels. Extensive agricultural use or unknown factors in the gut microbiota may have contributed to their prevalence. However, standard metagenomic techniques can make it challenging to identify which organisms carry ARGs. Recently, a novel method called epic-PCR (emulsion, paired isolation, and concatenation PCR) has been developed to analyze the functional diversity and roles of individual members of an ecosystem. The emulsion technology used in this approach enables the separation of cells into individual reactions through partitioned reaction drops. Additionally, the concatenated PCR method is employed to identify the sequence of the gene of interest and the species to which it belongs, targeting both the ARG and bacterial 16S rRNA. Although this technique has been primarily used to investigate sewage water, it could be used to explore the gut microbiota as well.43, 52
Various researchers pointed out the scenario of drug resistance across the globe and discussed several strategies to tackle it. Chandra et al. emphasize the issue of AMR in India and the requirement for judicious antibiotic usage in the nation. 34 They underline the growing issue of AMR in India and the requirement for an efficient strategy that can slow down the emergence and spread of bacterial drug resistance. This further emphasizes the necessity for education and awareness campaigns aimed at educating the general public and healthcare professionals about the risks of antibiotic overuse and the significance of taking antibiotics cautiously. 34 However, the focus of Brinkac et al. 53 is on how AMR affects the human microbiome. The human microbiome has been defined by the authors as a complex population of bacteria that is significant for human health in several ways. This draws attention to the growing problem of AMR, which is being fuelled by the improper use of antibiotics. They describe how the overuse of antibiotics may disturb the delicate balance of the microbiome and cause the emergence of antibiotic-resistant bacteria. However, this issue may be effectively overcome by fractional antibiotic use, the creation of novel antibiotics and complementary medicines, and the implementation of infection control measures. This also explains the necessity for an international response to AMR as well as the need for the creation of global health policies and plans to deal with the issue. The other method to minimize the risk of AMR development is using I-AMRSS, which is a comprehensive and integrated surveillance system that aims to monitor and control the spread of AMR in India, including the collection of data on antimicrobial use, resistance patterns, and the appearance of new resistant bacteria. They also discuss the potential capacity of the I-AMRSS as a tool for global AMR surveillance, including its ability to track the proliferation of resistant bacteria and monitor the impact of AMR on public health. The other initiatives to prevent AMR in India include the Chennai Declaration, which was created in response to the growing threat posed by AMR and fits this description. The declaration lays out a plan for combating AMR, which calls for more funding for research, the development of novel antimicrobial substances, and the implementation of effective strategies to promote the wise use of antibiotics. To address the problem of AMR, this article also covers the need for international collaboration in the research of novel antimicrobial agents, the sharing of information on resistance trends, and the implementation of coordinated strategies to promote moral antibiotic consumption.54, 55 The Chennai Declaration concludes by highlighting the importance of international cooperation in resolving this issue and providing a comprehensive and integrated approach to addressing the growing AMR crisis. In addition to these, additional approaches to bacterial infection control in animal feed are being developed. These include improved hygiene and animal health management practices, as well as the promotion of appropriate antibiotic treatment through education and awareness initiatives. 56 The changes in the gut microbiota can create an environment that allows multidrug-resistant organisms (MDROs) to colonize and persist, and gut microbiota modulation can be used to reduce the number of MDROs in the gut. Modulation of the gut microbiota using probiotics and prebiotics can minimize the risk of the development of AMR to some extent. A multidisciplinary approach that integrates microbiology, medicine, and biotechnology to address the complex interplay between gut microbiota and MDROs.56, 57
Summary
In summary, one of the biggest threats to world health in the 21st century is the growth of AMR. Micro-organisms can become resistant to the effects of antimicrobial medications, which results in a condition known as AMR. AMR is developed and disseminated as a result of the diverse population of microbes that live in the human gut, or gut microbiome. The selection and spread of bacteria that are resistant to antibiotics can result from the frequent use of antibiotics to treat infections spurred on by the gut microbiome. Additionally, the spread of AMR can be facilitated by the gut microbiota. The difficulty of eliminating AMR and preserving antibiotic efficacy for future generations has been greatly exacerbated by the misuse and overuse of antibiotics. Antibiotics can acquire resistance if they are used improperly, making them useless against bacterial infections. In India, where the availability of antibiotics over-the-counter without a prescription is common, the misuse and abuse of antibiotics are particularly prominent. The rationalization of antibiotic prescribing in clinical practice is required to address the problem of AMR. Before prescribing antibiotics, healthcare professionals ought to take into consideration an array of parameters, including the severity of the infection, the type of pathogen, and the pathogen’s susceptibility profile to the antibiotic. Improved hygiene habits, vaccinations, and the application of infection control measures might fall under this category. For the purpose of informing public health policy and ensuring successful AMR management in the future, it is crucial to monitor and track the evolution of AMR and the gut microbiota. The early detection of outbreaks and the identification of emerging resistant microorganism strains can be made easier by a systematic monitoring system. Healthcare professionals can choose the best antibiotics for treating bacterial infections by maintaining records of antibiotic effectiveness and resistance trends. The development and spread of AMR are greatly influenced by the gut microbiota. Further, to combat the problem of AMR, it is crucial to rationalize the prescription of antibiotics in clinical practice, establish infection control measures, and monitor and track AMR and the gut microbiome. By undertaking such measures, we may safeguard public health and ensure that antibiotics are still effective for future generations.
Footnotes
Abbreviations
AMR: Antimicrobial resistance; ARG: Antimicrobial resistance genes; FMT: Fecal microbiota transplantation; IBD: Inflammatory bowel disease; MDRO: Multidrug-resistant organism; NAP: National action plan.
Acknowledgment
The authors acknowledge the School of Pharmacy, Graphic Era Hill University, Dehradun, India for support.
Authors Contribution
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent Statements
Not Applicable.
Funding
The authors received partial funding to cover article processing charge from the School of Pharmacy, Graphic Era Hill University, Dehradun, India.
