Abstract
The heterogeneity of lymphoma responses to various treatments remains a significant challenge in clinical practice. Emerging evidence implicates the potential role of the gut microbiome in lymphoma pathogenesis and progression. Advances in high-throughput sequencing and metabolomics have significantly enhanced our understanding of the complex interaction between the gut microbiome and lymphoma. Although causality requires further elucidation, the gut microbiome critically shapes host responses to traditional combined chemotherapy, hematopoietic stem cell transplantation, and targeted therapies, including chimeric antigen receptor T-cell therapy. Notably, the use of antibiotics, particularly broad-spectrum antibiotics, can alter the gut microbiome, thereby impacting treatment efficacy. Prudent antibiotic management should balance infection control with microbiome-dependent immune homeostasis. Strategies to restore gut microbial balance through a high-fiber diet, probiotics, prebiotics, fecal microbiota transplantation, and butyrate supplementation are critically important. Integrating microbiome-based therapies into lymphoma treatment could establish low-toxicity therapeutic paradigms for lymphoma patients.
Plain language summary
Lymphoma treatments often work differently for each patient, and scientists now believe the community of bacteria in our gut may help explain these variations. Research shows these bacteria influence common lymphoma therapies like chemotherapy, stem cell transplants, and CAR-T cell treatment. Problems arise when antibiotics disrupt this delicate bacterial balance, possibly reducing treatment effectiveness. Doctors are now exploring ways to protect helpful gut bacteria while preventing infections. New approaches include special diets, probiotic supplements, and even microbiota transplants that may boost treatment success. These discoveries could lead to more personalized lymphoma therapies with better results and fewer side effects, offering new hope for lymphoma patients.
Background
Lymphoma, originating in lymph nodes or other lymphoid tissues, is a group of immune system malignancies. In recent years, lymphoma has presented a persistently increasing incidence.1,2 Despite advances in treatment, the development of drug resistance in certain patients has led to suboptimal responses, while the limited availability of effective therapies for relapsed/refractory (R/R) lymphoma remains a major clinical challenge.3,4 In addition, the toxicity associated with conventional treatments often results in severe adverse effects, significantly compromising patients’ quality of life. 5 Given these challenges, there is an urgent need to elucidate the key mechanisms driving lymphoma progression and to identify clinically relevant biomarkers and therapeutic targets, which are critical for developing more effective and less toxic treatment strategies.
The gastrointestinal (GI) tract hosts a complex microbial ecosystem, comprising bacteria, archaea (single-celled organisms without nuclei), fungi, and microbial eukaryotes. In healthy individuals, the gut microbiota is dominated by five primary bacterial phyla: Firmicutes (79.4%), Bacteroidetes (16.9%), Actinobacteria (2.5%), Proteobacteria (1%), and Verrucomicrobia (0.1%). 6 Under normal conditions, these microbes aid in digestion and support immune function, 7 but when malfunctioning, the abnormal composition and function of the microbiota are strongly associated with the development of a variety of diseases, such as inflammation, autoimmune diseases, metabolic, and neoplastic diseases.8 –11
Growing evidence 12 suggests a consistent link between gut microbiota and lymphoma. Shi and Zhang proposed the concept of the “microbiota-gut-lymphoma axis” 13 indicating that specific gut microbial taxa may influence the risk of different lymphoma subtypes. Here, we propose that alterations in the gut microbiota composition contribute to the onset and progression of lymphoma, and specific microbial taxa may serve as both biomarkers for disease development and potential targets for personalized, low-toxicity therapeutic interventions. However, the exact cause-and-effect relationship between gut microbiota and lymphoma remains undefined. Systematic characterization of microbiome-host interplay during disease progression could identify novel diagnostic and prognostic biomarkers, thereby establishing a framework for precision management and therapeutic innovation.
Materials and methods
This narrative review is based on the relevant PubMed literature. The main search terms include “lymphoma,” “lymphocyte,” “gut microbiota/microbiome,” “microbial dysbiosis,” “antibiotic,” and “short-chain fatty acids (SCFAs)” combined with Boolean operators AND/OR. The search encompassed literature published until May 2025. The initial search yielded a total of 132 articles. The inclusion criteria were articles that study the gut microbiome in lymphoma. Given the narrative nature of this review, formal evidence grading was not performed. We included all available study designs, predominantly observational studies, along with clinical and preclinical trials. Case reports were restricted to rare clinical scenarios. Sixty-one articles were selected for full-text review. The data, including microbial characteristics, intervention details, main outcomes, and mechanistic roles of gut microbiota, were extracted and analyzed.
Gut microbiome dysbiosis across lymphoma subtypes
Wotherspoon et al.14,15 found that more than 90% of gastric mucosa-associated lymphoid tissue (MALT) lymphoma patients harbored Helicobacter pylori (HP) in their gastric mucosa, and HP eradication led to complete remission (CR) in five out of six cases. Subsequent studies have identified HP as an etiologic factor in gastric MALT lymphoma. 16 Antibiotics have shown potential efficacy in treating certain lymphoma cases. 15 However, their broad-spectrum activity may simultaneously disrupt commensal microbiota. Yuan et al. 17 observed that patients with diffuse large B-cell lymphoma (DLBCL) exhibited elevated levels of Escherichia-Shigella, Enterococcus, Veillonella, and Prevotella-2 compared to healthy controls. Yoon et al. 18 reported decreased alpha-diversity in the gut microbiota of newly diagnosed DLBCL patients, alongside an increased abundance of Enterobacteriaceae. Xu et al. 19 reported that Fusobacteria were increased in primary DLBCL patients compared to healthy controls across all taxonomic levels (phylum to species), while Enterococcus predominated in R/R patients. In patients with cutaneous T-cell lymphoma, Hooper et al. 20 found that the diversity of the gut microbiota was reduced, with a significant decrease observed in advanced disease stages and specific bacterial taxa exhibiting altered abundance, and the gut microbiota may further modulate the response of narrowband ultraviolet B (a common skin-directed therapy). 21 In patients with mycosis fungoides, their latest findings identified a decrease in SCFAs production, suggesting that microbial metabolic changes may underlie variations in therapeutic response. 22 Chiba et al. 23 reported that Klebsiella exhibited significant enrichment in patients with adult T-cell leukemia/lymphoma. Patients with GI follicular lymphoma (FL) also displayed altered duodenal mucosal flora. 24 Xu et al. 25 demonstrated that patients with FL exhibited unique microbiota compositions, characterized by a notable increase in the relative abundance of the Ruminococcaceae family. Kang et al. 26 found that primary central nervous system lymphoma (PCNSL) patients exhibited significantly increased Proteobacteria abundance and an elevated Firmicutes/Bacteroidetes ratio—a recognized signature of gut dysbiosis. Several Mendelian randomization evidence revealed preliminary causal links between gut microbiota and lymphoma. The genus Ruminococcaceae UCG00227,28 may be linked to an increased risk of DLBCL, while the genera Alistipes28,29 and Turicibacter 28 may exert protective effects. Veillonella may be linked to an increased risk of Hodgkin lymphoma (HL), 30 while Faecalibacterium prausnitzii may exert protective effects. 31 Phylum Bacteroidetes may be associated with lymphoma. 32 Notably, Alistipes exhibits a bidirectional interaction with DLBCL: it may protect against lymphomagenesis, yet its abundance is increased in DLBCL patients, 28 possibly reflecting disease-driven dysbiosis or compensatory microbial adaptation. However, given the inherent complexity of gut microbial ecosystems and current limitations in genome-wide association study coverage, these results require further validation.
Influence of gut microbiome on lymphoma treatment
Traditional combined chemotherapy
Traditional combined chemotherapy is a classical therapeutic approach for lymphoma patients. However, there is still a considerable portion of patients who fail to achieve remission due to chemoresistance. Growing evidence highlights the critical role of the gut microbiome in modulating the host’s antilymphoma response, potentially enhancing or diminishing the efficacy of chemotherapy. Nausea and vomiting are frequent side effects that can significantly impair patients’ quality of life during the treatments. In addition, patients often suffer a high risk of febrile neutropenia after chemotherapy, which can cause treatment delay, dose reduction, or even fatal infection. Maintaining gut microbial homeostasis appears critical for modulating febrile neutropenia risk, as specific compositional changes are associated with increased risk, while a balanced microbiota may confer protective effects. Incorporating gut microbiota modulation into therapeutic strategies could improve treatment tolerance and prognostic outcomes.
Influence of gut microbiota and its metabolites on chemotherapy of lymphoma
Diefenbach et al. 33 found that the gut microbiota significantly differed between the chemotherapy responders and nonresponders. Xu et al. 34 found that patients with DLBCL who achieved CR after combined chemotherapy with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) treatment exhibited a higher abundance of family Veillonellaceae both before and after the treatment, while patients without CR exhibited a higher abundance of family Rikenellaceae. The results suggested that the abundance of Veillonellaceae and Rikenellaceae may correlate with outcomes of R-CHOP therapy in DLBCL patients. Another finding was that the presence of Lactobacillus fermentum was associated with more favorable treatment outcomes. While elevated Alistipes levels seem linked to worse treatment response, the overall evidence indicates mixed views. Another study conducted by Yoon et al. 18 suggested that a higher abundance of Enterobacteriaceae at the family level correlated with a shorter progression-free survival (PFS), higher risk of febrile neutropenia, and greater likelihood of relapse and progression. Restoring gut microbiota balance may reduce Enterobacteriaceae overgrowth, potentially lowering febrile neutropenia risk and improving outcomes. Moreover, Hernández-Verdin et al. 35 were the first to report that in PCNSL patients, higher Parabacteroides distasonis abundance and plasma betaine–valine levels were positively associated with prolonged PFS and overall survival (OS). In conclusion, the diversity and composition of gut microbiota correlate with the efficacy of chemotherapy (summarized in Table 1). Although the specific role of gut microbial metabolites in lymphoma chemotherapy remains not fully explored, mechanistic insights may be extrapolated from other malignancies. He et al. 36 found that patients with GI cancer responding to oxaliplatin therapy had significantly higher serum levels of butyrate compared to nonresponders. Butyrate acts as a histone deacetylase (HDAC) inhibitor to modulate CD8 T-cell function and enhance antitumor efficacy. This mechanism may be relevant to lymphoma therapy, as oxaliplatin—used in rituximab, gemcitabine, and oxaliplatin regimen for R/R DLBCL—also relies on T-cell-mediated cytotoxicity.
Clinical efficacy and gut microbiome in combined chemotherapy-treated patients.
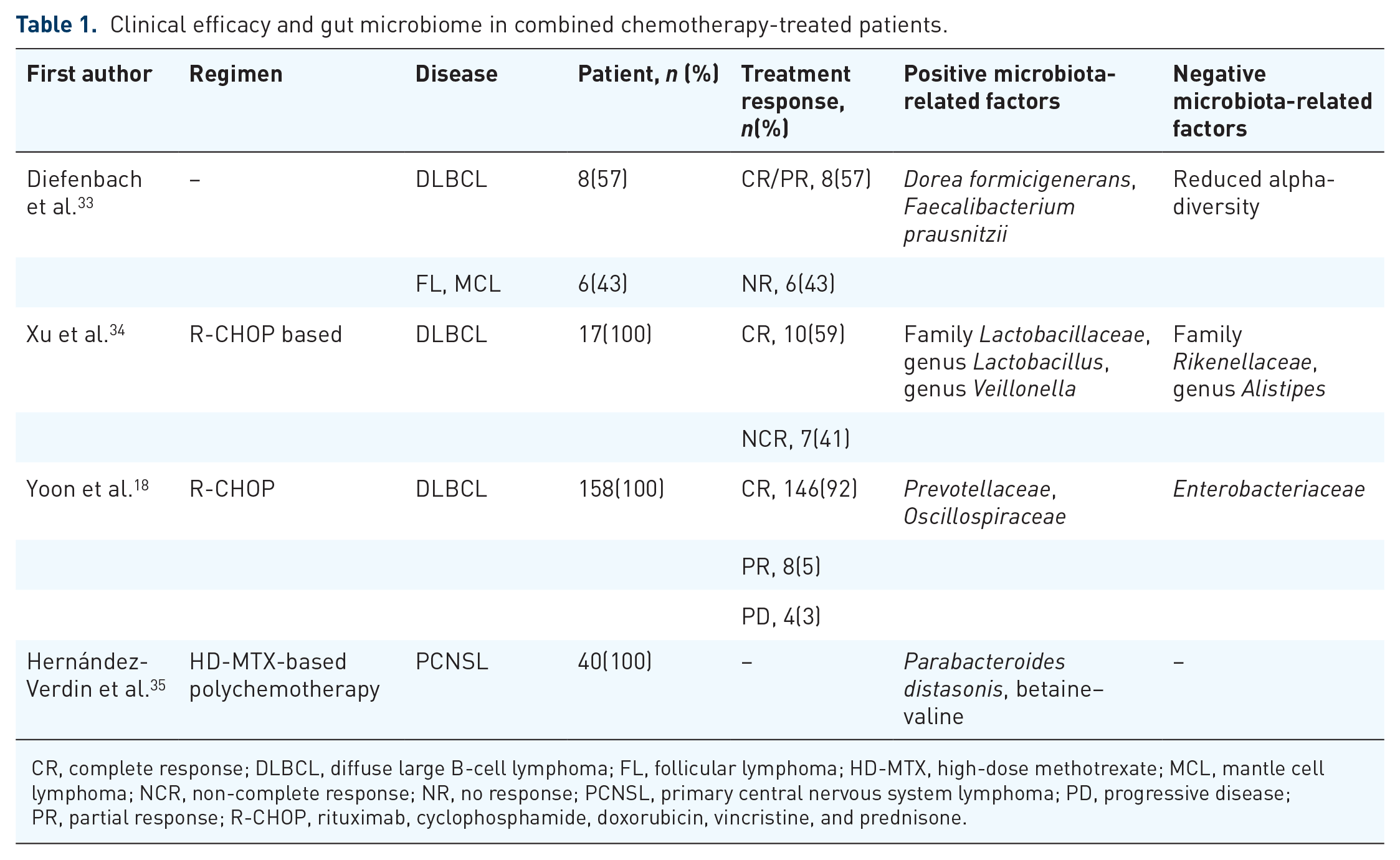
CR, complete response; DLBCL, diffuse large B-cell lymphoma; FL, follicular lymphoma; HD-MTX, high-dose methotrexate; MCL, mantle cell lymphoma; NCR, non-complete response; NR, no response; PCNSL, primary central nervous system lymphoma; PD, progressive disease; PR, partial response; R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone.
Influence of antibiotic management on chemotherapy of lymphoma
Bloodstream infection continues to be a significant contributor to non-relapse mortality among patients experiencing chemotherapy-induced neutropenia. The empirical administration of broad-spectrum antibiotics remains a fundamental aspect of supportive care in lymphoma. It was reported that translocation of gram-positive bacteria into the secondary lymphoid organs caused by cyclophosphamide, a component of the R-CHOP regimen, could promote the generation of pathogenic T helper cell 17 (Th17) cells and memory T helper cell 1 (Th1) immune responses, thereby contributing positively to its antilymphoma effects. However, anti-gram-positive antibiotics will reduce the level of the pathogenic Th17 cells, thus diminishing the antilymphoma effect of chemotherapy. 37 Moreover, Pflug et al. 38 demonstrated that treating relapsed lymphoma patients with anti-gram-positive antibiotics was associated with earlier disease progression, reduced PFS, and worse OS. These findings underscore the importance of understanding the broader implications of antibiotic use in lymphoma management and highlight the need for targeted bacteriotherapy strategies that leverage specific bacterial species or strains to optimize chemotherapy outcomes. 39
Hematopoietic stem cell transplantation
Hematopoietic stem cell transplantation (HSCT) represents a potentially curative therapeutic approach for patients with R/R lymphoma and those presenting with poor prognostic indicators or unfavorable clinical outcomes. It is divided into two types: autologous HSCT (auto-HSCT) and allogeneic HSCT (allo-HSCT). Graft-versus-host disease (GVHD) is a common complication in patients undergoing allo-HSCT. Several studies have shown the changes in gut microbiome in patients undergoing HSCT, characterized by a reduction in gut microbiota diversity and the predominance of specific bacterial taxa.39,40 Moreover, the level of SCFAs in patients receiving HSCT has been observed a reduction. 41 During the pretreatment phase, antibiotics are administered for intestinal cleansing and as a prophylactic measure. In addition, during the whole process, appropriate antibiotics are utilized based on the patient’s clinical manifestations or bacterial culture results. These comprehensive antimicrobial interventions inevitably exert profound impacts on the gut microbiome. Therefore, analyzing the influence of antibiotic management on HSCT cannot be overlooked.
Influence of gut microbiota and its metabolites on HSCT of lymphoma
First, gut microbiota is associated with side effects. High-dose chemotherapy, commonly utilized as a myeloablative conditioning regimen prior to HSCT, often induces several side effects, such as GI mucositis, which manifests in nausea, vomiting, abdominal pain, and diarrhea. Montassier et al. 42 analyzed gut microbiota composition in patients with non-Hodgkin lymphoma (NHL) who received the same myeloablative conditioning regimen without antibiotic exposure. Post-chemotherapy fecal samples exhibited a significant decrease in phylum Firmicutes abundance and a substantial increase in phylum Proteobacteria abundance. They revealed that the compositional disruption induced by chemotherapy can lead to GI mucositis. Moreover, gut microbiota disruption has been implicated in other critical complications of HSCT, including infections and GVHD. Montassier et al. 43 found that diminished gut microbiota diversity was associated with an elevated risk of bloodstream infections. Butyrate is one of the gut microbiota metabolites. Kaundal et al. 44 observed that persistently low fecal butyrate levels before and after auto-HSCT were associated with a higher risk of post-HSCT carbapenem exposure and antibiotic duration. When it comes to GVHD, Shouval et al. 45 reviewed the impact of conditioning regimens with different intensities on gut microbiota disruption. They found that, compared to non-myeloablative regimens, myeloablative regimens correlated with more severe damage to the gut microbiota, which was associated with a higher risk of GVHD occurrence. Qi et al. 40 observed a lower abundance of genus Roseburia before HSCT conditioning regimens in acute GVHD patients. Roseburia has been shown to produce butyrate. Administration of butyrate has been shown to offer protection against GVHD. 46 Gut microorganisms can metabolize nutrients such as choline, phosphatidylcholine, and carnitine into trimethylamine, which subsequently enters the liver and undergoes enzymatic oxidation to produce trimethylamine N-oxide (TMAO). Wu et al. 47 demonstrated that TMAO can aggravate GVHD, acting as a stimulator in GVHD progression. Furthermore, lower diversity of gut microbiota was linked to inferior OS and PFS in patients undergoing auto-HSCT 39 and higher mortality rates in patients undergoing allo-HSCT. 48 Detailed results are summarized in Table 2.
Clinical outcomes and gut microbiome in HSCT patients.

aGI-GVHD, acute gastrointestinal graft-versus-host disease; allo-HSCT, allogeneic hematopoietic stem cell transplantation; auto-HSCT, autologous hematopoietic stem cell transplantation; CLL, chronic lymphocytic leukemia; GI, gastrointestinal; GVHD, graft-versus-host disease; HL, Hodgkin lymphoma; HSCT, hematopoietic stem cell transplantation; NHL, non-Hodgkin lymphoma; OS, overall survival; PFS, progression-free survival.
Influence of antibiotic management on HSCT of lymphoma
Infection and GVHD are two major complications associated with HSCT, with antibiotic management strategies playing a critical role in their development and progression. Weber et al. 49 suggested “restrictive antibiotic strategy” in HSCT. In their findings, this strategy can preserve the diversity of gut microbiota and potentially beneficial taxa without increasing the risk of bacterial sepsis or documented infections. Notably, this approach also demonstrated a reduction in the incidence of severe acute GI GVHD (aGI-GVHD), a life-threatening complication of allo-HSCT that remained a leading cause of transplantation-related mortality due to its challenging treatment. Hayase et al. 50 further demonstrated that microbiome dysbiosis induced by carbapenem antibiotics was correlated with the exacerbation of aGI-GVHD and impaired treatment responsiveness. They also showed that carbapenem use was associated with a reduction in Bacteroides ovatus, which, by metabolizing dietary polysaccharides into monosaccharides, restricts Bacteroides thetaiotaomicron’s mucolytic activity, thereby preserving intestinal mucus integrity. Longer duration of treatment with meropenem and piperacillin-tazobactam was linked to an increase in the detection of genus Staphylococcus following allo-HSCT, including two potentially pathogenic species, Staphylococcus epidermidis and Staphylococcus aureus, which may elevate the risk of bacteremia in these patients. 41 Further, a prospective, multicenter, observational study (ClinicalTrials.gov NCT03727113) focusing on microbiome modulation during HSCT has been carried out. This study was designed to investigate whether a more cautious approach to antibiotic use during HSCT can better preserve the composition and diversity of gut microbiota, thereby reducing severe post-transplant complications. This study may provide stronger evidence supporting the role of gut microbiota modulation in HSCT, though no relevant literature has been published to date, and further research needs to be carried out. Therefore, understanding the impact of gut microbiota on HSCT and the optimal use of antibiotics may decrease the incidence of HSCT complications and benefit patients who undergo HSCT.
Target therapy
Immune checkpoint inhibitors
The evasion of immune surveillance is a primary mechanism that cancer cells utilize for survival and proliferation. The application of immune checkpoint inhibitors (ICIs), especially monoclonal antibodies targeting programmed cell death receptor-1 (PD-1), has shown good efficacy. However, their clinical application is often limited by the fact that more than half of the patients experience disease progression or a loss of therapeutic response over time. The gut microbiome plays an important role in regulating systemic immune responses. Therefore, understanding the interaction between the gut microbiome and ICIs could provide valuable insights to optimize ICI therapy. However, current evidence remains limited, particularly in lymphoma, highlighting the need for further exploration.
Influence of gut microbiota on ICIs of lymphoma
The efficacy of ICIs may be associated with gut microbiota. Casadei et al. 51 showed that patients who responded to anti-PD-1 therapy (nivolumab or pembrolizumab) had a notable increase in the abundance of genus Lachnospira. In contrast, in non-responding patients, the abundance of family Enterobacteriaceae increased. As we already know, Lachnospiraceae can produce SCFAs, mainly butyrate. Butyrate exerts immunomodulatory effects, enhancing antitumor immunity by modulating immune cell populations, including macrophages, dendritic cells, regulatory T cells, T helper cells, and plasma cells.
Influence of antibiotic management on ICIs of lymphoma
Previous research has indicated that the administration of antibiotics may reduce the effectiveness of ICIs in managing solid tumors. A study revealed that antibiotic use within 90 days prior to initiating ICIs therapy in patients with classical HL was associated with worse median OS and PFS. 52 However, larger prospective studies are needed to validate these findings and explore optimal timing windows.
Epigenetic modifications
Epigenetic modifications play a crucial role in modulating hematological tumor cells. 53 Epigenetic modifications enable cells to change gene expression without changing the deoxyribonucleic acid (DNA) sequence, thus causing changes in gene phenotype. Given the reversibility of epigenetic alterations, they emerge as promising therapeutic targets in the field of cancer treatment. 54 Epigenetic regulation includes histone modification, DNA methylation, non-coding Ribonucleic Acid effect, and chromatin recombination. Abnormal hypermethylation and deacetylation leading to the silencing of tumor suppressor genes are crucial regulatory points contributing to chemoresistance in DLBCL.55,56 DNA methyltransferase (DNMT) and HDAC are two key enzymes in epigenetic modification. Inhibiting DNMT or HDAC can promote demethylation and acetylation of tumor suppressor genes, thereby inhibiting tumor growth.57,58 Pera et al. 59 ’s research indicated that combining inhibitors of DNMT and HDAC may offer a delayed but promising chemosensitization effect. This represents a therapeutic strategy to overcome chemoresistance in DLBCL. 55 Our previous study 60 further confirmed the promising effect of a dual epigenetic agent-primed immunochemotherapy regimen, CD-R-GemOx (comprising chidamide, decitabine, rituximab, gemcitabine, and oxaliplatin), in R/R DLBCL patients who had failed salvage treatment. The composition of the human microbiota is very complex, mainly including bacteria, archaea, fungi, viruses, and some tiny protists. The genome of the microbiota is 150 times larger than the human genome, which is named the second human genome. It plays a crucial role in regulating human metabolic processes and is a potentially influential environmental element that may shape our epigenome. Significantly, unlike the immutable genomic composition, the gut microbiome is alterable, making it a promising target for therapeutic interventions. Therefore, epigenetic regulation is increasingly regarded as an effective mechanism for the microbiome to affect host physiology.
Influence of gut microbiota and its metabolites on epigenetic modifications of lymphoma
It has been reported that microbiota-derived metabolites can regulate epigenetic modifications. SCFAs, such as butyrate produced by genera Butyricimonas, Faecalibacterium, Lachnospira, and Ruminococcus, can traverse the boundary of the intestinal epithelium and exert gene expression and epigenetic modulation within the host, including DNA methylation and histone modification. 61 A previous study revealed that butyrate derived from a high-fiber diet enhances histone acetylation and pro-apoptotic gene expression, inhibiting lymphoma cell proliferation and inducing apoptosis. 62 Further, the decreased abundance of butyrate-producing bacteria was related to an increased risk of lymphoma progression and a poorer treatment response.51,63,64 Indole-based metabolites, such as indole-3-propionic acid, which were produced by Lactobacillus johnsonii and Clostridium sporogenes through metabolizing tryptophan, an essential amino acid from protein-rich food, were also reported to be capable of changing immune cell stemness, enhancing the efficacy of anti-PD-1 immunotherapy against lymphoma cells through modulating HDAC processes. 65 However, there is limited understanding regarding the precise function of the interaction between microbial indole metabolites and the epigenetic mechanisms of the host. 66 Together, these findings underscore the importance of gut microbiota and its metabolites in lymphoma biology and suggest potential novel therapeutic strategies targeting the microbiota-epigenome axis.
Preliminary evidence on antibiotic-modulated epigenetics in lymphoma
Limited data are currently available about the function of antibiotic management in epigenetic modifications in lymphoma have been published in the literature. Ragheb et al. 67 found that HDAC inhibitor vorinostat and anticancer antibiotic Mithramycin in combination showed clear synergy or potentiation in patient-derived Sézary syndrome (SS) cells apoptosis as well as epigenetic modifications. Mithramycin broadly affects epigenetic modifications by directly inhibiting Sp1 binding to DNA, binding to histones, and indirectly influencing gene expression through the inhibition of DNMT1 and other histone-modifying enzymes, thereby altering histone acetylation and methylation. The combination of vorinostat and Mithramycin appears to induce changes in histone acetylation, methylation, and other epigenetic marks, leading to a greater impact on gene expression and cell death. This data provided preliminary clues on the individual and combined effects of HDAC inhibitor and antibiotic Mithramycin in SS cells and highlighted a potential therapeutic interest of this novel pair of drugs for the treatment of SS patients. However, the research on the interaction between antibiotic management, epigenetics, and lymphoma is limited currently. As a result, there is a need for further investigation to elucidate these complex interactions.
Chimeric antigen receptor T-cell therapy
Lymphocytes isolated from peripheral blood are genetically modified by incorporating a chimeric antigen receptor (CAR) using a replication-deficient viral vector. 68 Following this modification, these lymphocytes are reintroduced into the patient’s system, enabling them to target and destroy lymphoma cells identified by their T-cell receptors (TCRs). 68 CAR-T cell (CAR-T) therapy has been incorporated into the National Comprehensive Cancer Network guidelines as a recommended second-line treatment for patients with relapsed disease (<12 months) or primary refractory DLBCL, reflecting its substantial therapeutic potential, which has indeed been demonstrated through clinical evidence. However, although the response rate of CAR-T ranges from 40% to 90% in patients with R/R large B-cell lymphoma, only about 30%–40% of patients can achieve durable remission.69,70 Immune-related toxicities, including cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndromes (ICANS), represent pivotal contributors to the mortality rates in CAR-T therapy.71,72 In general, CAR-T therapy indeed demonstrates significant therapeutic potential. However, some patients do not respond to it, and some others experience severe adverse reactions, indicating the need for personalized optimization of the treatment. The role of the gut microbiome in cancer treatment has been established, and increasing evidence suggests that the characteristics of the gut microbiome can be used to predict treatment response or adverse reactions. 73 Targeting the gut microbiome to enhance the antilymphoma efficacy and attenuate CAR-T therapy’s toxicity may be a new approach.
Influence of gut microbiota and its metabolites on CAR-T therapy of lymphoma
Multiple studies have shown that the composition of the gut microbiome relates to the efficacy and toxicity of CAR-T therapy. Smith et al. 64 found that a higher abundance of genera Ruminococcus, Faecalibacterium, and Bacteroides was associated with the response to CD19 CAR-T therapy, and Bacteroides abundance may serve as a biomarker for predicting toxicity, with a higher abundance potentially associated with a higher risk of toxicity. However, the study did not exclude patients who may have previously been exposed to antibiotics, a factor that could potentially interfere with the final conclusion. Focusing on a non-antibiotic-disrupted gut microbiome, Stein-Thoeringer et al. 74 identified Bacteroides, Ruminococcus, Eubacterium, and Akkermansia as the primary taxa that can influence the predictive outcome of CAR-T therapy response. They found that a high abundance of Akkermansia muciniphila was associated with higher levels of peripheral T cells, predicting the possibility of producing better quality CAR-T. It is known that A. muciniphila possesses the ability to produce SCFAs such as acetate and propionate, and F. prausnitzii to produce butyrate. SCFAs can enhance genetically engineered T cells by imparting more stable TCR/CAR transgene expression, optimal functionality, memory potential, and improved therapeutic activity in vivo. Recognizing their pleiotropic effects in vivo, Rangan and Mondino 75 advocated for the use of SCFAs in T-cell manufacturing. Building on this, Prasad et al. 76 proposed an approach for boosting CAR-T effectiveness by increasing the levels of SCFAs during the ex vivo production process of CAR-T. Then they discovered an enhanced CAR-T cytotoxicity and cytokine secretion in prolonged serial killing assays with repeated tumor cell exposures and elevated SCFAs. However, research on gut microbial metabolites, including SCFAs, has been limited to preclinical studies and has not yet been carried out in clinical and real-world studies.
Influence of antibiotic management on CAR-T therapy of lymphoma
Antibiotic management may damage or facilitate CAR-T therapy. A dual-center study 64 found that patients with NHL who received piperacillin/tazobactam, meropenem, and imipenem/cilastatin (broad-spectrum antibiotics) within 4 weeks before CD19 CAR-T therapy exhibited poorer survival outcomes, including OS and PFS. To further investigate these findings, the research team employed A20 tumor-bearing mice to investigate the effect of oral vancomycin, primarily targeting gram-positive bacteria, on CAR-T therapy. Their findings revealed that oral vancomycin administration could enhance the tumor elimination mediated by CAR-T, whereas oral neomycin and metronidazole, which mainly target gram-negative bacteria, failed to exhibit such an effect. 77 Moreover, another multicenter study 74 also revealed that broad-spectrum antibiotics were associated with a worse PFS and OS. The correlation between antibiotic management and the efficacy of CAR-T therapy may be due to a poorer disease state or higher inflammation levels prior to antibiotic exposure, or alternatively, by gut microbiome disruption induced by antibiotic exposure. Recently, data from a single-center cohort 76 further revealed some correlations between antibiotic management, the disruption of metabolome and CAR-T therapy. Patients treated with broad-spectrum antibiotics, particularly those with anaerobic coverage, showed worse survival outcomes after CAR-T therapy and notable changes in their gut and serum metabolic profiles, such as depletion of SCFAs. When it comes to the correlation between antibiotic management and CAR-T-mediated toxicities, Smith et al. 64 found that exposure to broad-spectrum antibiotics in the CAR-T pre-infusion time window raised the rate of ICANS. This finding aligned with the potential link between the gut microbiota and ICANS via the gut-brain axis.78,79 However, data showed no significant correlation between antibiotics and CRS. Based on current studies, prudent use of broad-spectrum antibiotics appears advisable for CAR-T therapy. Detailed results are summarized in Table 3.
Clinical efficacy and gut microbiome in CAR-T-treated patients.

High-risk antibiotic: piperacillin/tazobactam, meropenem, cefepime, ceftazidime.
ANA, target anaerobic commensals; CAR-T, chimeric antigen receptor T-cell therapy; CEF, anti-pseudomonal cephalosporins; CRS, cytokine-releasing syndrome; DLBCL, diffuse large B-cell lymphoma; HR, hazard ratio; ICANS, immune effector cell-associated neurotoxicity syndrome; NHL, non-Hodgkin lymphoma; OS, overall survival; PFS, progression-free survival; P-I-M, piperacillin/tazobactam, imipenem, meropenem; SCFAs, short-chain fatty acids; TMAO, trimethylamine N-oxide.
Specific gut microbial taxa and their metabolites differentially influence treatment responses across lymphoma subtypes through immune modulation, epigenetic regulation, and ecological competition. Summarized in Table 4.
Clinical associations and mechanistic roles of gut microbiota in lymphoma therapeutics.

aGVHD, acute graft-versus-host disease; CAR-T, chimeric antigen receptor T-cell therapy; CSF, cerebrospinal fluid; DLBCL, diffuse large B-cell lymphoma; EPS, exopolysaccharide; GVHD, graft-versus-host disease; HD-MTX, high-dose methotrexate; HSCT, hematopoietic stem cell transplantation; ICIs, immune checkpoint inhibitors; JAK-STAT, Janus kinase-signal transducer and activator of transcription; MR, Mendelian Randomization; NCR, non-complete response; NKTCL, natural killer/T-cell lymphoma; ORR, objective response rate; OS, overall survival; PCNSL, primary central nervous system lymphoma; PFS, progression-free survival; R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone; SCFA, short-chain fatty acids; SOCS1, suppressor of cytokine signaling 1.
Limitations and future
Limitations in gut microbiome research for lymphoma
First, the use of fecal samples to estimate the gut microbiome presents inherent limitations. While fecal samples are widely used due to their accessibility and the ability to obtain sufficient sample sizes, they provide only a partial representation of the gut microbiome. Fecal microbiome analysis does not accurately reflect the composition of the mucosal microbiome in different regions of the GI tract, nor does it fully capture the luminal microbiome, which can differ significantly from the fecal microbiome. 80 Feces are often far from the GI tract that we investigate and are always stored in the rectum, where they undergo active dehydration and ferment bacteria that are not common in other parts of the lumen. In addition, the distribution of fecal microflora in feces is uneven, which is a spatially organized multicellular community. 81 Consequently, there is a need to find a more representative sample to better reflect the composition of gut microbiota. Second, research between gut microbiome and lymphoma remains limited, and the majority of studies have been conducted with a relatively small sample size, which may not adequately represent the broader population, leading to potential biases and increased uncertainty in the findings. Numerous observational studies have already confirmed that lymphoma patients possess distinct gut microbiota compared to healthy individuals, which is associated with disease stage, treatment responsiveness, and adverse reactions. Several clinical trials are now in the recruiting phase to confirm the relationship between gut microbiota and lymphoma. NCT05725720 is a clinical study focusing on the characteristics of gut microbiota during CAR-T therapy to demonstrate its role in treatment prognosis. A clinical trial, NCT06218602, aims to evaluate whether the addition of Fecal Microbiota Transplantation (FMT) can improve the gut microbiota disrupted by antibiotics, and its impact on therapeutic efficacy and prognosis. However, up to now, no prospective study specifically designed for lymphoma has been published to address these gaps. Therefore, larger, prospective clinical trials should be conducted to provide a more comprehensive understanding of the complex interactions between the gut microbiome and lymphoma. Third, the gut microbiome is a complex and dynamic ecosystem, influenced by a multitude of factors such as diet and antibiotics. Given its complexity, it is paramount to rigorously control for confounding variables in research to ensure the accuracy and reliability of findings. Moreover, it is necessary to design personalized treatment plans due to the individual differences in gut microbiome composition. We can potentially enhance therapeutic outcomes and minimize adverse effects by tailoring treatment strategies to each patient’s unique microbial profile and clinical presentation. Fourth, the underlying mechanisms by which the gut microbiome influences lymphoma require further investigation.
Further direction of microbiome therapy in lymphoma
The gut microbiome influences the response to lymphoma therapy via immune and epigenetic mechanisms. A critical condition for the gut microbiome to exert its function is homeostasis. Multiple strategies for microbiome therapy are available and are widely employed to restore gut microbiome homeostasis. Utilizing specific microorganisms or their metabolites as therapeutic agents, such as targeted treatments including special diets, prebiotics and probiotics, FMT, and butyrate supplementation, has demonstrated significant potential in reducing treatment-related complications and enhancing outcomes for patients with hematologic malignancies (Figure 1). We suggest these interventions should be primarily used as adjuncts. Combining microbiome interventions with standard lymphoma regimens can potentially improve treatment efficacy.

Possible strategies and mechanisms for microbiome therapy in lymphoma. Gut microbiota-targeted interventions influence lymphoma progression through immune and epigenetic mechanisms. The upper section presents gut microbiota-targeted interventions, including probiotics, prebiotics, high-fiber diet, butyrate supplementation, protein consumption, and fecal microbiota transplantation. These interventions promote beneficial commensal bacteria and their metabolites, while antibiotics may disrupt this balance. The lower section demonstrates the effects of interventions in the tumor microenvironment. Lactobacillus (one of the probiotics) could stimulate the differentiation of T cells into Th1 and Th17 cells, enhancing antilymphoma immune responses. SCFAs improve intestinal barrier integrity, modulate immune function, and induce epigenetic modifications that suppress lymphoma cell proliferation while promoting apoptosis. IPA expands cytotoxic effector CD8+ T cells through epigenetic regulation, which may enhance apoptosis.
High-fiber diet
A diet high in fiber, which promotes the production of SCFAs, including butyrate, may potentially have a protective effect against lymphoma by enhancing the apoptosis of tumor cells.62,82 So it is suggested to incorporate fiber-rich foods into the diet. However, further research is needed to establish definitive dietary recommendations for lymphoma patients.
Prebiotics and probiotics
Prebiotics are indigestible dietary fibers that selectively promote beneficial gut bacteria growth. These specialized fibers serve as a nourishing food source for the probiotics residing in our intestines, further fostering a healthy gut microbiome. Probiotics are live, nonpathogenic microorganisms that, when consumed in adequate quantities, provide beneficial health effects to the host. Numerous studies have already confirmed the roles of prebiotics and probiotics in various diseases. A study showed that restoring the deficient L. johnsonii in the ataxia-telangiectasia mouse model through short-term oral transfer can reduce cancer risk in susceptible individuals. 83 Xu et al. 34 demonstrated that the abundance of L. fermentum significantly increased after chemotherapy and was associated with better treatment response. In vitro experiments showed that Lactobacillus could stimulate the differentiation of T cells into Th1 and Th17 cells, thereby enhancing antilymphoma immune responses. 37 Shi et al. 84 first demonstrated that F. prausnitzii exhibited tumor-suppressing effects in a natural killer/T-cell lymphoma (NKTCL) mouse model, with butyrate as a key mediating metabolite. This suggested that F. prausnitzii could be a promising probiotic candidate for adjunctive therapy in NKTCL. Savitri et al. 85 and Verma et al. 86 suggested that probiotics may enhance HP eradication protocols, potentially through microbial interference and immune modulation. This enhanced bacterial clearance may theoretically reduce HP-associated lymphomagenesis risk, though direct antilymphoma effects remained unproven.
However, careful evaluation and prudent use of probiotics in immunocompromised individuals should be taken seriously. Chemotherapy and HSCT can induce rendered immunosuppressed for a long time. It was reported that one mantle cell lymphoma patient with compromised immunity after HSCT developed Lactobacillus acidophilus sepsis due to excessive consumption of probiotic-rich yogurt. 87 Another Hodgkin’s lymphoma patient with acquired immune deficiency syndrome developed persistent bacteremia after taking a probiotic containing Lactobacillus. 88 Moreover, it was found that the increase in Lactobacillus was positively correlated with the severity of the disease and the risk of chemotherapy side effects in DLBCL patients. 89 A case report indicated that chemotherapy patients may experience bloodstream infections due to commensal bacteria, but were not necessarily related to probiotics. 90 While these reports highlight potential risks, case reports are inherently constrained by low evidence hierarchy and uncontrolled confounding factors. Therefore, whether to administer probiotics, which type of probiotics to administer, and when to administer them should all be carefully considered before use, especially in HSCT patients.
FMT
The clinical advantage of utilizing a solitary bacterial species is constrained. 91 The use of the whole microbiota is known as FMT, which involves extracting functional microbiota from the feces of healthy individuals and then transferring it into the patient’s intestine. It is the most promising strategy to date for effectively rebalancing the composition and function of the gut microbiome. Montassier et al. 92 observed that the pretreatment chemotherapy before bone marrow transplantation led to significant changes in the gut microbiota, including a sharp decline in alpha-diversity, a reduction in anti-inflammatory bacteria, and an increase in harmful bacteria. Several large observational studies showed that low diversity in the gut microbiota prior to allo-HSCT was closely associated with adverse outcomes post-transplantation, including GVHD and future mortality risk. 93 Studies also suggested that it was possible to restore the pre-disease microbiome diversity and composition by collecting fecal samples from patients and reintroducing the microorganisms back into their bodies. 93 Kakihana et al. 94 successfully applied FMT in four patients suffering from steroid-resistant acute GVHD, with no severe FMT-related adverse effects.
However, the potential risks associated with FMT should not be overlooked. DeFilipp et al. 95 reported that two immunocompromised patients developed bloodstream infection of extended-spectrum β-lactamase-producing Escherichia coli following FMT, and the causative strain was linked to the stool of the FMT donor. This observation may be explained by FMT-induced bacterial translocation across the compromised intestinal barrier, potentially driven by local gut mucosal inflammatory responses. 96 In patients undergoing HSCT, particularly during the early post-transplant period when granulocyte reconstitution is incomplete, the risk of intestinal microbiota-derived infections may be significantly elevated. An exploration of how to balance the risks and benefits of FMT in severely immunocompromised patients is needed. Benech et al. 97 recommended a “case-by-case” approach tailored to each patient’s unique risks, values, and preferences to ensure optimal treatment opportunities are not missed. In addition, stringent donor screening protocols are essential to minimize the transmission of potentially pathogenic microorganisms and reduce adverse infectious outcomes.
Butyrate supplementation
The composition and function of the gut microbiome are influenced by various internal and external factors, with diet, environment, and particularly antibiotics among the most prevalent external disruptors. 98 The butyryl-CoA:acetate-CoA transferase pathway serves as the primary route for butyrate production within the human gut microbiota, predominantly relying on gram-positive bacteria for its process. 98 Certain antibiotics may lead to a decrease in essential bacteria that specifically produce butyrate. Butyrate supplementation has been employed to enhance intestinal mucosa integrity and serve as an anti-inflammatory agent in systemic diseases.99,100 Given its beneficial effects on gut health, administering isolated butyrate during antibiotic use may be advantageous until the recolonization of butyrate-producing bacteria is achieved. 98 Moreover, given its epigenetic modification potential, butyrate may regulate various biological processes, including cell differentiation, proliferation, and apoptosis, as well as lymphoma development and progression. Sodium butyrate administration has been shown to restore the integrity of the intestinal barrier and modulate gut dysbiosis induced by paclitaxel treatment in mice models. 99 As previously noted, lymphoma therapy is also associated with the depletion of butyrate-producing bacteria. By supplementing butyrate levels, sodium butyrate may offer a promising avenue for mitigating chemotherapy-related side effects and improving the treatment response. Further studies are needed to evaluate the safety, optimal dosing, and therapeutic benefits of sodium butyrate supplementation in lymphoma patients. In conclusion, it may be a promising therapeutic target for future treatment and may serve as an adjunctive therapy during lymphoma treatment.
Conclusion
Although more and more studies have focused on the gut microbiome, the correlation between the gut microbiome and lymphoma remains incompletely characterized. Lymphoma patients exhibit a unique gut microbiota profile that differs from healthy individuals, with certain microbial species and metabolites associated with disease progression, responsiveness to treatment, and the occurrence of adverse reactions. To gain a deeper understanding, further research is necessary to elucidate these associations. Future research should focus on conducting larger cohort studies to identify the specific bacterial species and metabolites that are involved in lymphoma progression, as well as the different treatment approaches. In addition, exploring the potential therapeutic benefits of modulating the particular microbial flora may lead to novel treatment strategies for lymphoma. When using antibiotics, a careful evaluation of their benefits and risks is imperative to minimize disruptions to the gut microbiome while maximizing therapeutic efficacy. By addressing these research gaps, we can pave the way for more personalized and effective therapies that leverage the power of the “gut-microbiome-lymphoma axis.”
