Abstract
Colorectal cancer (CRC) remains a leading cause of cancer-related deaths worldwide, with its progression intricately linked to gut microbiota dysbiosis. Disruptions in microbial homeostasis contribute to tumor initiation, immune suppression, and inflammation, establishing the microbiota as a key therapeutic target. Fecal microbiota transplantation (FMT) has emerged as a transformative approach to restore microbial balance, enhance immune responses, and reshape the tumor microenvironment. This review explores the mechanisms underlying FMT’s therapeutic potential, evaluates its advantages over other microbiota-based interventions, and addresses challenges such as donor selection, safety concerns, and treatment standardization. Looking forward, the integration of FMT into personalized CRC therapies requires robust clinical trials and the identification of predictive biomarkers to optimize its efficacy and safety.
Plain language summary
Colorectal cancer (CRC) progression is tied to gut microbiota dysbiosis. Fecal microbiota transplantation (FMT) restores balance and boosts immunity, offering promise for personalized CRC therapies with further clinical validation.
Introduction
Fecal microbiota transplantation (FMT) has recently emerged as a promising strategy for restoring gut microbial balance, with the potential to bolster immune function and improve colorectal cancer (CRC) treatment outcomes. 1 CRC remains one of the leading causes of cancer mortality worldwide, with incidence rates expected to rise significantly by 2040. 2 Standard CRC treatments, including surgery, radiotherapy, and chemotherapy, are widely used for tumor management but often induce acute or chronic intestinal injuries. Chemotherapy, while initially effective in slowing tumor growth, presents serious side effects and resistance risks. Over time, tumor cells can develop resistance to chemotherapy, reducing efficacy, increasing DNA repair activity, and leading to treatment failure, which may require long-term immunosuppressive therapy and frequent hospitalization. 3 Radiotherapy, although beneficial, disrupts gut microbiota and impairs the gut’s ability to repair radiation-induced damage, causing adverse effects and limiting efficacy. 4 Despite these treatments, many CRC patients exhibit limited responsiveness. FMT offers a potential alternative to address these issues by restoring gut microbiota balance; however, its efficacy, safety, and optimal application in CRC treatment remain inadequately understood.
Alongside lifestyle factors like smoking and diet, gut microbiota imbalance is now recognized as a significant contributor to CRC risk, influencing immune response and inflammation. 5 The gut microbiota, comprising bacteria, fungi, and viruses, plays a crucial role in intestinal health. Disruptions in this microbiota balance can promote CRC by allowing pathogenic organisms to breach the mucosal barrier and trigger chronic inflammation. 6 Once the intestinal barrier is compromised, the relationship between the microbiota and the host is broken, leading to an increase or decrease, or even the disappearance, of specific portions of the flora. The imbalance of the microbiota allows pathogenic bacteria to become dominant in the intestinal tract, penetrate the barrier, and enter the lamina propria, inducing an abnormal immune response and leading to the development of CRC. 7 FMT has currently been applied to reduce radio- and immunotherapy-related toxicity and enhance their efficacies by restoring the gut microbiome after irradiation for long-term persistence. 8 Even if this approach was less acceptable to patients in earlier years, the number of publications on FMT treatment studies in patients with inflammatory bowel disease (IBD), immunocompromised patients, or cancer has nearly doubled in the past decade, 9 while its safety and efficacy have been examined.
This review explores the therapeutic potential of FMT in CRC treatment, examining its mechanisms, advantages over other interventions, and the challenges and future directions. A particular focus is placed on understanding how FMT restores gut microbiota balance in CRC patients, the factors that shape its efficacy, and the strategies needed to address safety concerns in clinical practice.
Influence of microbiota on CRC pathogenesis
Intestinal barrier dysfunction and microbiota dysbiosis
The outermost mucus layer functions as a selective barrier against potential pathogens in the intestinal lumen while fostering a nutrient-rich microenvironment that supports the resident microbiota. Beneath the mucus layer lies a tightly sealed epithelial cell barrier, essential for nutrient absorption, metabolite exchange, water transport, and defense against antigens and pathogens. 10 Immune cells reside within and beneath the epithelial layer, engaging in intricate interactions with epithelial cells to strengthen barrier integrity and immune defense. Interactions among these three layers—the microbiome, epithelial cells, and immune system—are pivotal in maintaining intestinal permeability. 11 These three barriers are intricately linked. The mucus layer protects epithelial cells from pathogens and sustains a microbiota that, in turn, supports its integrity. The epithelial barrier relies on the mucus for pathogen filtration and on immune cells for secondary defense. Immune cells, in turn, depend on epithelial cells to detect pathogens and are activated when either barrier is compromised. 12 This coordinated interplay is essential for intestinal homeostasis, with disruptions potentially initiating CRC-related changes. Disruption of this complex barrier system can lead to disease pathogenesis by facilitating microbial translocation and promoting chronic inflammation.
Mucosal barrier integrity and microbial defense
The mammalian gut hosts a diverse microbial ecosystem—collectively termed the gut microbiota—that is integral to colonic health. Commensal bacteria within the gut contribute essential nutrients and signaling molecules, which are crucial for host tissue development and function (Figure 1). The microbiota’s beneficial effects also extend to the other two barriers. Nutrients and signaling molecules from commensal bacteria enhance epithelial cell function, fortifying the epithelial barrier, while also modulating immune cell activity to optimize the immune barrier’s response. 13 Alghetaa et al. reported an increase in Tenericutes and Actinobacteria, with Tenericutes degrading dietary carbon sources and defending against viruses, thereby preserving mucosal integrity. Actinobacteria produce antibiotics, strengthening microbial defense and protecting the mucosal barrier. 14 Weisburger et al. 15 observed that germ-free rats exposed to N-methyl-N'-nitro-N-nitrosoguanidine developed more colon adenomas than conventional rats, suggesting a potential role of the gut microbiota in early tumorigenesis. This finding indicates that the absence of a normal microbiota disrupts the balance between the mucosal, epithelial, and immune barriers. Without protection, the mucosal barrier becomes more vulnerable, compromising epithelial integrity. 16 This, in turn, can trigger abnormal immune responses, creating a microenvironment that promotes tumorigenesis. However, no significant difference in adenocarcinoma incidence was observed, implying that factors beyond immune status, such as carcinogen metabolism, may be pivotal in CRC progression. These findings highlighted a potential association between gut microbiota and colon cancer, sparking decades of research into microbial contributions to CRC. Within the healthy human gut, beneficial bacteria colonize the intestinal surface, forming a physical barrier that impedes pathogen adherence and proliferation. 17 Although the underlying mechanisms remain incompletely understood, these effects may involve competition for adhesion sites, stabilization of the mucosal barrier, nutrient competition, and antimicrobial production. 18 The abundant presence of Helicobacter pylori, Haemophilus hepaticus, Escherichia coli, Streptococcus gallolyticus, Streptococcus fragilis, Streptococcus anaerobius, Enterococcus faecalis, and Pseudomonas nucleicola in the intestinal tract is often predictive of a high incidence of CRC. 19 In addition, specific microbial metabolites, including free fatty acids, tryptophan derivatives, vitamins, and choline, have been shown to influence intestinal barrier permeability. 20 Recent studies indicate that tryptamine, via 5-HT4 receptor activation, modulates gastrointestinal (GI) motility and acts as a key signaling molecule in host–microbe interactions. 21 Tryptophan metabolism to kynurenine can induce neuroinflammation and impair brain health, whereas microbial conversion of tryptophan to indole supports gut and brain protection. 22 Extensive studies indicate that dysregulated gut microbiota can compromise intestinal mucosa and cellular integrity through metabolite secretion, immune modulation, and toxin production, thereby influencing tumor initiation, progression, and prognosis.23–25

A schematic representation of the intestinal barrier. The intestinal barrier includes a microbial barrier, a mechanical barrier, and an immune barrier, and plays a significant role in regulating the gut microbiota. The microbial barrier exists in a mucous layer acting as a scaffold, including Escherichia coli, Helicobacter pylori, and Fusobacterium nucleatum, to name a few. The mechanical barrier is made up of a single layer of intestinal epithelial cells that is sealed by tight junctions, the cells of which include stem cells (ISCs), progenitor cells (TA cells), and specialized cells (enterocytes, M cells, goblet cells, Paneth cells, enteroendocrine cells, and tuft cells). Immune cells in the lamina propria constitute the immune barrier and support the gut barrier by helping to maintain the host–microbiota interface.
Epithelial barrier and junctional complex integrity
The intestinal epithelium functions as a critical physical barrier against pathogens, stressors, and microbial invasion; its disruption may promote CRC initiation and accelerate tumor invasion and metastasis.26,27 The intestinal epithelium comprises six distinct mature cell types, broadly categorized as absorptive and secretory cells (Figure 1). The integrity of the epithelial barrier is closely interconnected with the other two barriers. The mucus layer protects epithelial cells from harmful substances in the intestinal lumen, while immune cells help monitor and defend the barrier. 28 Dysbiosis can lead to the secretion of toxins or metabolites that damage epithelial cells, compromising the mucus layer and triggering abnormal immune responses. Absorptive cells, including enterocytes and microfold cells, and secretory cells such as goblet cells, Paneth cells, enteroendocrine cells, and tuft cells, are crucial for barrier function. 29 In CRC patients, alterations in the proportion and function of these cells occur. For example, reduced mucus production by goblet cells weakens the mucus layer, increasing the epithelial barrier’s vulnerability to pathogenic bacteria, which can interact with underlying tissues and promote CRC development. 30 Collectively, these cells establish a dynamic barrier that isolates luminal contents from underlying host tissue. The epithelial layer’s filtering capacity relies on both the surface mucus layer and the intermicrovillar bridging structures, which enhance mechanical integrity and resistance to luminal stress. 31
Intercellular junctions include desmosomes, adherens junctions, and tight junctions. 32 Positioned at the apical region of the junctional complex, tight junctions primarily function as both a barrier and a fence. 33 Tight junctions are essential for regulating the paracellular transport pathway, with a size-selective leak pathway that restricts large solutes (e.g., proteins, bacterial lipopolysaccharides (LPS)) and a charge-selective pathway mediated by claudin-based pores. 34 Tight junction function can be compromised by the microbiota and other barriers. In CRC, toxins like colibactin from E. coli disrupt tight junctions, while microbiota-induced alterations in the mucus layer allow pathogens to damage epithelial cells. 35 This increases intestinal permeability, enabling harmful substances to reach underlying tissues, triggering inflammation, activating tumorigenic pathways such as NF-κB, and promoting CRC progression.
Immune barrier and intestinal immunity
The immune response is critical at every stage of CRC progression, influencing both tumor control and immune escape. 36 Dysbiosis disrupts immune cell activation, impairing normal immune surveillance and defense against tumor cells. Antigen-presenting cells (APCs), such as macrophages, dendritic cells (DCs), and B cells, play key roles in antigen presentation and immune activation (Figure 1). In CRC, APCs operate within an immunosuppressive tumor microenvironment (TME), facilitating immune evasion, a situation exacerbated by the dysfunction of the mucosal and epithelial barriers, and in CRC patients, the dysbiotic microbiota further impairs APC function. 37 Pathogenic bacteria can skew macrophage polarization to a pro-inflammatory M1-like phenotype, which, while initially protective, promotes chronic inflammation—a known CRC risk factor that drives tumor proliferation and immune escape. Disruption of the mucosal and epithelial barriers further hampers immune activation, weakening defenses and promoting CRC progression. (1) Intestinal macrophages normally protect against inflammation, but disrupted polarization can lead to chronic inflammation, creating a tumor-promoting microenvironment. Cytokines like TNF-α and IL-6, released by activated macrophages, activate pathways in tumor cells that enhance survival, proliferation, and invasion, while further driving DNA damage and genomic instability. 38 (2) Plasmacytoid dendritic cells (pDCs) produce high levels of IFN-I during viral infections, but this function is impaired in the TME, allowing tumor cells to evade immune detection and promoting unchecked growth. Dysbiosis can exacerbate this effect by releasing metabolites that inhibit pDC function, enhancing the immunosuppressive microenvironment. 39 (3) B cells contribute to antitumor immunity by secreting IgA and IgG1 antibodies and presenting tumor antigens. In CRC, dysbiosis impairs B-cell functions, hindering antibody production and antigen presentation, weakening the immune response, and allowing tumor cells to evade destruction. In addition, disrupted B-cell interactions with other immune cells further impair immune defense. 40
Collectively, microbiota dysbiosis disrupts the intestinal barrier system, triggering a cascade of events that foster CRC development. As outlined in the following section, specific bacterial species and signaling pathways further drive CRC progression, exploiting the compromised barrier and the ensuing pro-inflammatory and immunosuppressive microenvironment.
Mechanisms of microbiota-driven CRC progression
A 2014 study using 16S rRNA sequencing of fecal and digestive tissue samples identified consistent decreases in Lactobacillus, Faecalibacterium spp., and Bifidobacterium in CRC, while Staphylococcaceae, Alistipes, Porphyromonadaceae, and Fusobacteria increased. 41 This alteration in microbial composition deepens the dysbiotic state, amplifying its impact on CRC progression via specific bacterial species and signaling pathways. Eleven microorganisms are classified as human carcinogens primarily based on epidemiological links to cancer, with only H. pylori showing directly in vivo carcinogenic potential. 42 The gut commensal microbiota resists pathogen colonization mainly through inter-microbial competition for nutrients and space. 43 In CRC, dysbiosis alters the microbial composition, enabling pathogen colonization and worsening microbial imbalance. Immune and inflammatory responses in the CRC microenvironment may favor the growth of potentially oncogenic bacteria. 43 Thus, whether dysbiosis is a cause or consequence of CRC remains unclear; however, microbiota promote tumor growth through three main mechanisms: pathogenic bacteria/toxins, a pro-inflammatory environment, and immune modulation. 44
Pathogen-induced DNA damage and genomic instability
Campylobacter jejuni
The gram-negative bacterium Campylobacter jejuni promotes tumor growth in a mouse CRC model. Cytolethal distending toxin (CDT), a genotoxin from C. jejuni, comprises subunits CdtA, CdtB, and CdtC. 45 CDT and colibactin are unique genotoxins that directly damage DNA and induce genomic instability.46,47 CDT, a major virulence factor, has DNase activity that induces DNA double-strand breaks. CDT activates DNA repair mechanisms by inducing cell cycle arrest at the G2/M phase.48,49 He et al. 50 observed that E. coli-induced DNA damage in vitro is likely linked to the CDT subunit CdtB. In addition to upregulating oncogenes, CDT suppresses humoral and cellular immunity by inducing immune cell apoptosis and intestinal epithelial cell necrosis. 51 Immune cell apoptosis disrupts gut immune surveillance, while the resulting cellular debris and inflammation drive metabolic alterations. This can upregulate glycolysis in neighboring cells, meeting the energy demands of damaged tissue and creating a more favorable environment for tumor growth (Figure 2). 52

Several groups of intestinal bacteria with carcinogenic effects. (a) Escherichia coli. Escherichia coli is a genotoxic substance produced by strains of E. coli carrying the pathogenicity island polyketide synthase (pks+). Its pathogenic mechanism is to cross-link DNA and induce DNA ds breaks, leading to DNA damage in colonic epithelial cells and tumor induction. (b) Campylobacter jejuni. Campylobacter jejuni produces CDT, genotoxins that also cause DNA ds breaks. (c) Bacteroides fragilis. ETBF treatment of colorectal cancer cells promoted Th17 cell differentiation by down-regulating miR-149-3p in exosomes in vitro and in vivo, which was dependent on METTL14-mediated m6A methylation. ETBF down-regulation of miR-149-3p and further promotion of PHF5A-mediated RNA variable shearing of KAT2A ultimately induced colorectal carcinogenesis.
Escherichia coli
Escherichia coli, a Gram-negative symbiotic bacterium, is clearly linked to CRC when attached to mucosal surfaces. Studies show elevated mucosal E. coli colonization in CRC patients compared to healthy controls. 53 Swidsinski et al. detected E. coli in only 3% of healthy biopsies, but in 62% of adenoma and 77% of CRC patients using 16S PCR. Colibactin-producing E. coli (pEPEC), which synthesizes the genotoxin colibactin, exhibits carcinogenic effects. 54 The carcinogenic mechanisms involve DNA double-strand breaks and nucleotide modification, causing genomic instability and mutation, which drive carcinogenesis.53,55 pEPEC attracts immune cells, but its toxins impair their function, hindering immune response. 56 DNA damage activates the pentose phosphate pathway, supplying nucleotides for repair and promoting tumor cell proliferation. Nouri et al. 57 reported a higher prevalence of virulence factor genes in E. coli strains from CRC patients (Figure 2).
Bacteroides fragilis
Bacteroides fragilis is an anaerobic, gram-negative, opportunistic pathogen. Bacteroides fragilis in the gut microbiome is suggested as an early biomarker for CRC. 58 Bacteroides fragilis has two subtypes: non-toxigenic and enterotoxigenic (ETBF). ETBF produces BFT toxins that disrupt mucosa, potentially leading to acute diarrhea, IBD, and CRC. ETBF promotes CRC cell proliferation by downregulating miR-149-3p in vitro and in vivo, 59 with miR-149-3p dysregulation linked to cancer and other diseases, including cancer, 60 autoimmune disorders, 61 and inflammatory diseases. 62 This downregulation impairs CRC growth and alters the immune microenvironment, reducing antitumor immune cell recruitment and promoting immunosuppressive cell infiltration. Meanwhile, metabolic changes in surrounding tissue increase metabolites like lactate, supporting tumor survival and invasion. 63 The bft gene in ETBF encodes BFT, a metalloproteinase toxin that disrupts the gut barrier by binding to intestinal epithelial receptors.64,65 Zakharzhevskaya et al. 66 observed higher levels of enterotoxin-producing B. fragilis in CRC patients, especially those with recurrence. A study found that Anaplasma fragilis and Pseudomonas aeruginosa were more prevalent in CRC cases compared to other strains studied. 67 This was also confirmed by Hwang et al. 68 who showed that in the AOM/DSS mouse model, Mimicronium fragilis induced the formation of polyps and colitis and exacerbated the development of colorectal tumors. These findings implicate B. fragilis as an enterotoxigenic, tumor-promoting bacterium in CRC (Figure 2).
Inflammatory signaling pathways and tumorigenic inflammatory microenvironment
Activation of the innate and adaptive immune systems, coupled with increased mucosal permeability and bacterial translocation, triggers chronic inflammation, while microbiota imbalances and opportunistic infections further exacerbate this process. 69 Dysbiosis activates signaling pathways such as MAPK, NF-κB, and PI3K/AKT (Figure 3), increasing reactive oxygen species (ROS) and pro-inflammatory cytokines, including nitric oxide and IL-8, which promote cancer development.1,70 Short-chain fatty acids (SCFAs), key microbiota-derived metabolites, interact with G-protein-coupled receptors on immune and epithelial cells. 71 At low concentrations, SCFAs exert anti-inflammatory effects, but in dysbiosis, elevated levels can excessively activate the NF-κB pathway, driving pro-inflammatory cytokine production and attracting immune cells to the tumor site. 72 Chronic inflammation disrupts immune cell metabolism, shifting them from antitumor to tumor-promoting phenotypes. In addition, microbial toxins like LPS from Gram-negative bacteria activate the MAPK pathway via toll-like receptors, increasing ROS production. Elevated ROS not only induces DNA damage but also disrupts normal cellular metabolism, fostering tumorigenesis. 73 Elevated levels of specific bacterial populations in advanced CRC patients induce inflammatory mediators (e.g., ROS, IL-6, IL-8, IL-1ß, TNF-α) and recruit CD11b+TLR4+ cells.19,74,75 Chronic inflammation drives carcinogenesis by inducing oxidative DNA damage, activating oncogenes, and inactivating tumor suppressor genes. 76 However, in addition to these pro-inflammatory responses, the role of anti-inflammatory pathways, such as IL-10 and TGF-β, is crucial in maintaining immune homeostasis. 77 Dysbiosis can impair the function of these regulatory pathways, leading to a breakdown of immune tolerance and promoting tumor progression. For instance, the reduced production of IL-10 in the context of microbiota dysbiosis impairs immune suppression, allowing persistent inflammation to fuel cancer development. 78 In CRC mouse models, pks+ E. coli promotes DNA damage and tumorigenesis in the context of active mucosal inflammation, but not post-inflammation. 79
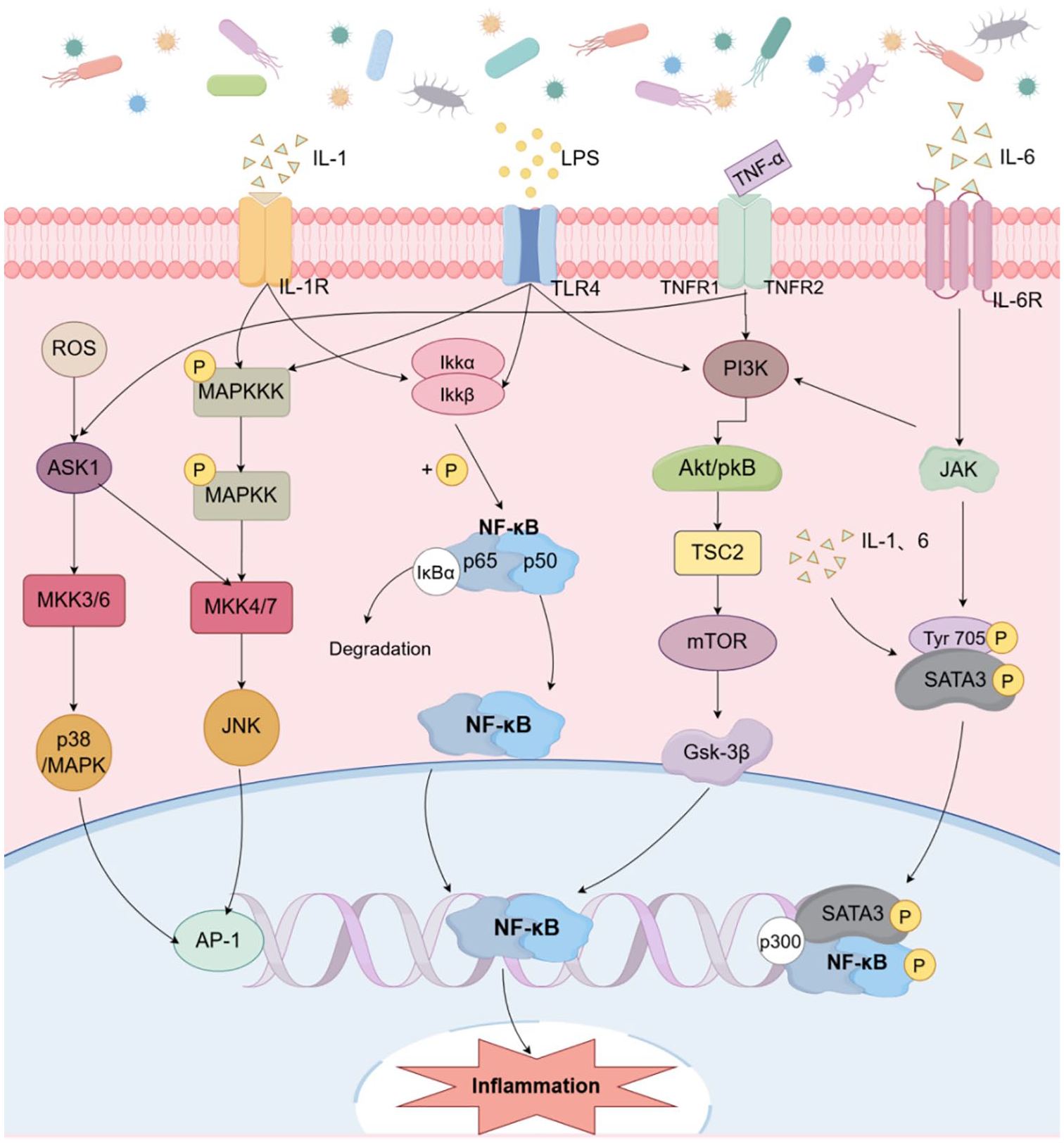
Mechanisms of microbiota disruption leading to an inflammatory environment. (1) The NF-κB heterodimer is usually composed of the p50 and p65 subunits. It is present in the cytoplasm and is inactivated by the inhibitory molecule IkBα. The IkB protein is activated when a specific IKK acts on the IkB protein. This causes NF-κB to translocate to the nucleus and initiate transcription of the target gene. (2) JAK/STAT signaling pathway. Cytokine binding induces receptor dimerization. Upon ligand binding, JAKs are activated and phosphorylate each other, then recruit, bind, and phosphorylate STATs in the cytoplasm, which are then dimerized and transported to the nucleus. The dimerized STATs bind directly to DNA, altering chromatin structure and inducing gene expression of inflammatory mediators. (3) MAPK signaling pathway. The pathway of MAPK consists of MAPK kinase (MKKK), MAPK kinase (MKK), and MAPK, that is, the MAP3K-MAP2K-MAPK chain. These three kinases can be activated sequentially to transmit signals from upstream to downstream. Of the four MAPK signaling pathways, those associated with inflammation, apoptosis, and growth are JNK and p38. (4) PIK3CA/AKT/mTOR signaling pathway. AKT is activated by phosphorylation of PDK1 and also by the RAS/MAPK pathway. On the cell surface, members of the tyrosine kinase receptor activate intracellular cofactors that regulate the PIK3 pathway. MAPK interacts with transcription factors in the nucleus, thereby affecting gene expression.
Microbiota-mediated immune modulation and tumor immune evasion
Mouse models with altered immune and inflammatory responses provide robust evidence that dysbiosis alone can induce cancer.80,81 These findings indicate that the immune system plays a central role in mediating gut microbiota–CRC interactions. 19 Barrier disruption and increased permeability activate immune cells in the lamina propria.82,83 Activated immune cells interact with microbiota-derived metabolites, such as tryptophan metabolites like indole-3-acetic acid, which influence immune cell function. In CRC, these metabolites can shift the balance between regulatory T cells and effector T cells, with an elevated regulatory T-cell ratio promoting immune suppression and aiding tumor immune evasion. Concurrently, tumor cells may enhance the expression of transporters to uptake these metabolites, utilizing them to support growth and invasion. 84 In mice, adenomas induce colonic barrier defects that allow microbial products to drive IL-23/IL-17-mediated tumor growth. 82 Brennan et al. 83 found that in the colonic lamina propria, F. nucleatum strain Fn7-1 modulates Th17 cell frequency and IL-17A/F expression, impacting intestinal immunity. Elevated ROS and microbial metabolites in the TME induce DNA damage and suppress CD8+ T-cell function. 85 Microbial metabolites, like secondary bile acids, accumulate in the TME due to microbiota dysbiosis. These acids interact directly with CD8+ T cells, disrupting their signaling and reducing cytotoxic activity. 84 In addition, they modulate the metabolic reprogramming of tumor cells, enhancing the Warburg effect, where tumor cells preferentially rely on glycolysis for energy production, even in the presence of oxygen. 86
The role of FMT in CRC treatment
FMT has emerged as an innovative therapeutic strategy to mitigate gut dysbiosis, a condition frequently worsened by standard CRC treatments such as chemotherapy, radiotherapy, and surgical resection.87,88 In non-cancer GI diseases, FMT is an effective treatment for recurrent Clostridium difficile infection, which led to its approval by the FDA for this disease. 89 Currently, this is the only indication to use FMT in patients. Although FMT has not yet been directly used to treat CRC, radiation-induced dysbiosis and GI side effects during CRC chemoradiotherapy might be reverted by FMT. 8 However, its application in other GI disorders remains limited, and the long-term stability of the restored microbiota post-treatment requires further investigation (Table 1).
Summary of FMT in clinical trials.

FMT, fecal microbiota transplantation; SBM, spontaneous bowel movements; UC, ulcerative colitis.
In CRC treatment, evidence supporting the use of FMT during radiotherapy, chemotherapy, or immunotherapy is sparse. Although FMT has shown promise in enhancing immune checkpoint inhibitor (ICI) efficacy in melanoma patients, its therapeutic potential in CRC remains unproven. 90 Furthermore, research exploring FMT’s role during radiotherapy and chemotherapy is even more limited. 91 FMT can be utilized both as a standalone therapy and as an adjunct in CRC treatment. As a primary approach, it restores gut microbiota balance and modulates the immune response to directly impact tumor progression. When combined with conventional treatments like chemotherapy, radiotherapy, and immunotherapy, FMT enhances efficacy by strengthening the gut barrier, mitigating side effects, and amplifying immune responses. 8
Mechanisms of FMT in CRC treatment
The microbiota–gut–brain axis in CRC therapy
The microbiota–gut–brain (MGB) axis represents a dynamic communication network between the gut microbiota and the central nervous system (CNS), mediating systemic and local effects critical for CRC treatment. 92 A pivotal component of this axis is the vagus nerve (VN), which exerts anti-inflammatory effects by activating the hypothalamic–pituitary–adrenal axis and suppressing pro-inflammatory cytokines, such as TNF, via the cholinergic anti-inflammatory pathway. Acetylcholine released from VN terminals interacts with α7 nicotinic receptors on intestinal macrophages to mediate this process. 93 In addition, vagal afferents within the GI tract detect immune and endocrine signals, enabling bidirectional interaction between the gut microbiota and the CNS. 94
FMT restores microbial diversity, which enhances VN-mediated signaling and contributes to a reduction in intestinal permeability, facilitating gut barrier repair. 95 Specific microbial metabolites, such as SCFAs like butyrate, play a critical role in strengthening tight junctions and promoting immune cell function. Butyrate also activates vagal afferents, further stimulating the cholinergic anti-inflammatory pathway via α7 nicotinic receptors on intestinal macrophages. This modulation of immune cells helps to counteract inflammation in the TME. 96 Moreover, by re-establishing microbial homeostasis, FMT can regulate the inflammasome, a key player in linking gut microbial changes to systemic immune responses and alleviating CRC-associated inflammation. 97 Experimental studies underscore FMT’s potential in reversing dysbiosis-induced inflammation, highlighting its therapeutic value through the MGB axis (Figure 4). 98
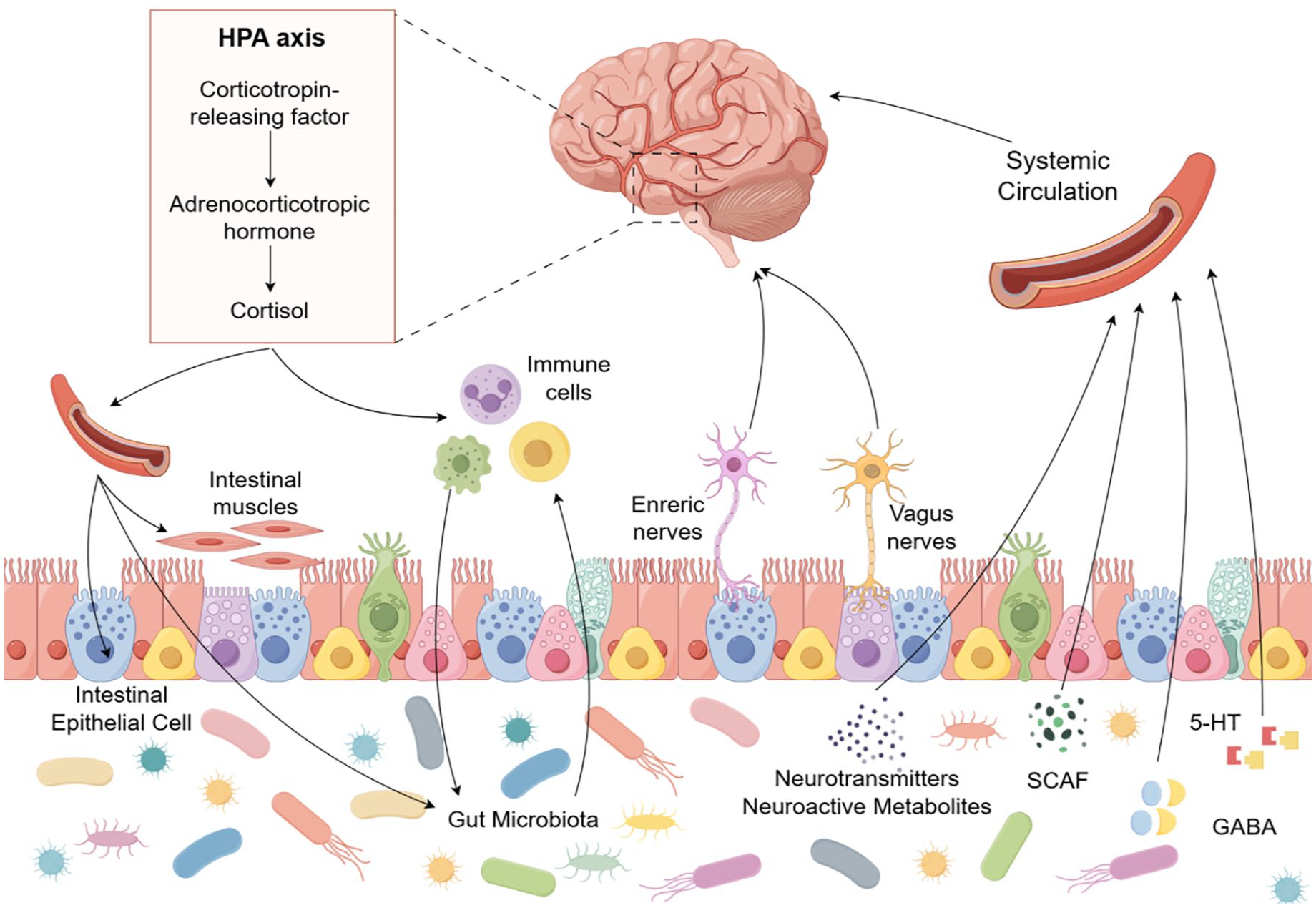
Bidirectional lines of communication between the microbiota, gut, and brain primarily involve neural, endocrine, and immune pathways. When gut microbes are out of balance, a wide range of microbial products including metabolites, neurotransmitters, and cytokines, including 5-HT, SCAF, and GABA, enter the brain through the bloodstream. The unidirectional and bidirectional movement of metabolites in the gut and brain or distal organs in direct press constitutes a co-metabolic interaction.
Microbial restoration and immune modulation by FMT
FMT emerges as a critical intervention to counteract microbiota dysbiosis caused by standard CRC therapies, such as chemotherapy and radiotherapy, which often disrupt the gut microbiome and cause significant intestinal injury. 87 By introducing fecal matter from healthy donors, FMT replenishes beneficial microbial populations, enhancing gut barrier integrity and mitigating treatment-associated toxicities. 1
FMT modulates the TME by reshaping microbial composition and promoting immune cell infiltration. Donor-derived bacteria could colonize the TME, increasing the abundance of antitumor immune cells, such as CD8+ T cells and DCs. The presence of Akkermansia muciniphila and Bifidobacterium longum has been linked to improved antigen presentation and heightened effector T-cell activity. 99 Clinically, a trial investigated the combination of FMT and anti-PD-1 (NCT04130763), revealing that FMT enhances anti-PD-1 efficacy by increasing microbial diversity and promoting CD8+ T-cell infiltration, thereby boosting antitumor immunity. 8 Such changes promote immune cell infiltration, strengthening antitumor immunity. In addition, the restoration of microbial metabolites, such as SCFAs, contributes to improved gut barrier function, reduced inflammation, and enhanced immune responses, further highlighting FMT’s systemic benefits. 100
In summary, by integrating MGB axis mechanisms with immune modulation, FMT provides a multifaceted therapeutic approach for CRC, addressing both local dysbiosis and systemic immune regulation. Future strategies optimizing FMT protocols could further enhance its efficacy as a cornerstone therapy in CRC management.
FMT-enhanced microbiota modulation for improved ICI efficacy in CRC
Enhancing the efficacy of ICIs with FMT
ICIs, targeting pathways such as the PD-1/PD-L1 axis, have revolutionized cancer treatment by restoring T-cell cytotoxicity and boosting immune responses.101,102 However, their efficacy remains limited in CRC, benefiting only approximately 15% of patients. 103 This underscores the need for adjunctive strategies, where the gut microbiota plays a critical role in modulating ICI effectiveness.
FMT enhances the efficacy of ICIs by modulating the gut microbiota and amplifying effector T-cell responses. Specific bacteria, such as B. longum, A. muciniphila, and B. fragilis, improve CD8+ T-cell function and antigen presentation in the TME, enhancing the response to PD-1 blockade therapy. Moreover, microbial metabolites like butyrate boost CD8+ T-cell cytotoxicity while reducing Treg-mediated suppression, further reinforcing the antitumor immune response.104,105 In addition, B. fragilis and Bifidobacterium spp. activate DCs, promoting IL-12 production and stimulating type I interferon signaling, which collectively enhances antitumor immunity. 104 Microbial metabolites, such as butyrate, also contribute to this process by directly enhancing CD8+ T-cell cytotoxicity in an ID2-dependent manner through the IL-12 pathway. 106
FMT has been shown to restore microbial diversity and enhance ICI efficacy. 107 For instance, transferring microbiota from ICI responders into germ-free mice significantly improved PD-1 blockade effectiveness, underscoring the gut microbiota’s pivotal role in modulating immune responses. In addition, differences in glycerophospholipid metabolism between ICI responders and non-responders suggest that FMT may promote immune responses through IFN-γ and IL-2 signaling pathways. 108 For example, a study demonstrated that FMT from ICI responders enhanced IFN-γ production and T-cell activation in the TME, improving the efficacy of PD-1 blockade in CRC. 8
Comparison with probiotics, prebiotics, and postbiotics
FMT demonstrates significant advantages over other microbiota-modulating strategies, such as probiotics, prebiotics, and postbiotics. Although probiotics like B. lactis Bl-04 and L. acidophilus NCFM have shown a potential to enrich butyrate-producing species in some CRC patient studies, their capacity to consistently enhance anti-tumor immunity remains uncertain. 109 Key challenges include the difficulty in identifying strains with reliable immunomodulatory properties and the risk of adverse events, such as bacteremia, particularly in immunocompromised individuals. 110 Prebiotics, while capable of increasing the abundance of immunogenic gut bacteria, lack robust evidence of efficacy in CRC-specific contexts. Similarly, neither prebiotics nor postbiotics can fully restore microbial diversity or address the complex dysbiosis associated with CRC.1,110
By contrast, FMT transplants a complete and diverse microbial community, re-establishing intricate microbiota–host interactions essential for immune modulation. A recent clinical trial (ChiCTR2100046768) in patients with microsatellite stable metastatic colorectal cancer (MSS-mCRC) implemented a novel triple therapy combining FMT with PD-1 inhibitors and VEGFR inhibitors. 111 Preliminary findings indicated that this approach effectively slowed tumor progression, reduced serum tumor marker levels, enhanced immune function, and facilitated gut microbiota homeostasis. Microbiome analysis further revealed that treatment responders exhibited an increased abundance of Proteobacteria and Lachnospiraceae, alongside a comparatively lower presence of Actinobacteria and Bifidobacterium. These findings underscore the therapeutic potential of FMT in reshaping the gut microbiome to improve immunotherapy outcomes.
Moreover, FMT has been shown to mitigate the adverse effects of antibiotics, which can deplete beneficial bacteria necessary for ICI efficacy and may foster resistance in pathogenic microbes.104,112,113 By restoring microbial metabolites such as SCFAs, FMT enhances gut barrier integrity, reduces inflammation, and promotes systemic immune responses, addressing both local and systemic challenges in CRC treatment. 1
Overcoming challenges and addressing limitations of FMT in advancing CRC therapy
Risks and safety concerns in FMT
Pathogen transmission and donor screening
The therapeutic potential of FMT in CRC is accompanied by critical risks and safety considerations, particularly related to pathogen transmission, adverse effects, and long-term consequences. FMT involves transplanting live microbiota from donors, which raises the risk of transmitting multidrug-resistant organisms (MDROs) or unknown pathogens. DeFilipp et al. reported two cases of bacteremia caused by extended-spectrum beta-lactamase-producing E. coli, one of which was fatal. Genome sequencing confirmed that both infections were linked to the same fecal donor, highlighting the importance of stringent donor screening protocols.114,115 Donors must be free from infections, chronic illnesses, or metabolic conditions, as studies suggest that traits such as obesity or diabetes may also be transferred to recipients. 116
Proper processing and handling of donor feces are essential to minimize risks. Homogenizing and diluting stool samples under sterile conditions ensures consistency and safety.117,118 Techniques that maximize the transfer of viable aerobic and anaerobic strains are crucial for comprehensive microbiota remodeling. 118 Delivery methods such as nasogastric or nasojejunal routes, as well as transendoscopic approaches, are commonly used to improve safety and efficacy.113,119
Adverse effects and long-term safety
Most adverse effects of FMT are mild and self-limiting, including abdominal pain, diarrhea, constipation, and fever. 120 Adverse reactions occur in approximately 10%–20% of patients, with severe complications, like bacteremia, being rare but serious. In the National FMT Registry of the United States, 1% of patients developed potentially drug-resistant bacterial infections 1 month after FMT, with the incidence of infection events within 6 months at about 4%. 121 In addition, some patients may not benefit from FMT and may experience worsened symptoms, particularly in cases of refractory GI diseases like C. difficile infection. The exacerbation rate of IBD in adults is 26.8%, and 10.8% in children. 122 Effective management strategies include pre-screening for infections, close monitoring during the procedure, and the use of post-FMT antibiotics in cases of confirmed infections. Prompt recognition and intervention can help mitigate severe outcomes. However, pre-treatment regimens, such as administering oral antibiotics to recipients for enhanced decolonization, may inadvertently increase susceptibility to MDROs. 123 Optimizing pre-treatment strategies is essential to minimize these risks while maintaining microbiota balance.
The long-term safety of FMT remains uncertain, particularly regarding the potential for transferring unknown pathogenic genes or traits linked to donor microbiota. These risks emphasize the need for robust monitoring and follow-up in clinical settings.
Factors influencing the efficacy of FMT
The efficacy of FMT can differ based on patient characteristics. In early-stage CRC, FMT may help address microbiota imbalances that contribute to tumorigenesis. However, in late-stage CRC, it primarily serves to support other treatments, such as immunotherapy, by enhancing immune response.1,124 Moreover, CRC patients with immune dysregulation or comorbidities may experience varying outcomes, as the altered immune environment may influence how microbiota impacts treatment.
FMT relies heavily on factors such as donor selection, preparation of donor material, and the delivery techniques employed. The characteristics of both donors and recipients, including gender, age, geographic background, immune function, and nutritional health, play a pivotal role in determining treatment outcomes. 125 Healthy stool donors, free from infections or chronic diseases, are essential for safe and standardized procedures. Ideal donors should be willing to provide stool regularly as needed, ensuring a consistent supply of high-quality microbiota for transplantation.126–128 Equally important is the preparation and handling of donor material. Donor stool must be thoroughly diluted and homogenized to ensure consistency and safety during administration.117,129 Advanced preparation techniques focus on maximizing the transfer of viable aerobic and anaerobic strains, enabling comprehensive remodeling of both small and large intestinal microbiomes. 118
FMT delivery methods influence its effectiveness. These methods include oral-fecal capsules and transdigestive injections of fecal suspensions, with the latter being more commonly used for its direct targeting of the GI tract. For targeting the upper and middle GI tract, delivery is achieved through gastroscopy, nasogastric, or nasojejunal tubes, whereas the lower GI tract is treated via retention enemas or transendoscopic techniques. 119 Stool suspensions are typically prepared using sterile saline solutions, with naso-duodenal or nasogastric routes often preferred for their reliability and safety in clinical settings (Figure 5). 113 These interconnected factors collectively determine the safety, standardization, and therapeutic success of FMT in clinical applications. Oral capsules are noninvasive and convenient but may result in reduced microbiota viability due to gastric acid exposure. By contrast, enema and transendoscopic methods offer higher local efficacy for targeting the lower GI tract but are more invasive. Each method’s choice depends on the treatment goals and the patient’s clinical condition. 130
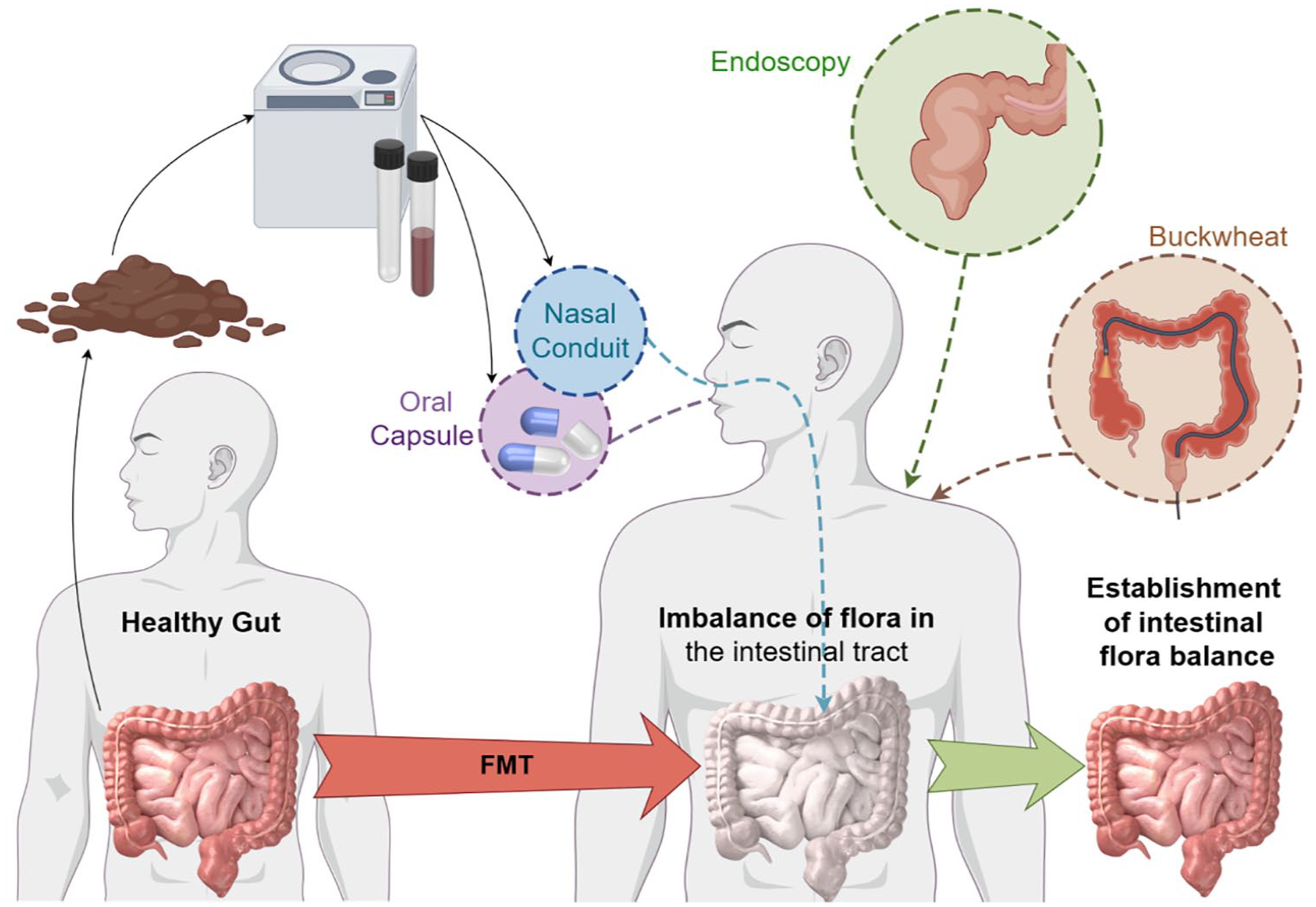
The main path of the operation of fecal bacteria transplantation is (1) through the introduction of enteroscopy to the end of the small intestine where the immune tissues and cells are particularly high, put the bacteria liquid; (2) through the upper digestive tract, to reach the jejunum nutrient tube or nasojejunostomy tubes; and (3) an enema, but the enema can only be enrolled in a small section. Currently, many companies are also trying to do fecal bacteria capsules.
Conclusion
FMT holds transformative potential in CRC therapy by reshaping the gut microbiota to restore microbial balance, mitigate dysbiosis-driven inflammation, and reprogram the TME. These effects not only suppress tumor-promoting signals but also enhance antitumor immune responses, positioning FMT as a powerful tool for improving CRC treatment outcomes. However, the real clinical significance of FMT lies in its potential to personalize CRC therapy by addressing dysbiosis as a modifiable risk factor and improving the efficacy of standard treatments such as chemotherapy, radiotherapy, and immunotherapy. Its success hinges on overcoming key challenges, including identifying robust biomarkers to predict treatment response, standardizing donor selection criteria, and leveraging advanced tools like artificial intelligence to optimize microbiota composition and donor matching. Establishing standardized protocols for FMT preparation, administration, quality control, and follow-up monitoring is essential to ensure safety, efficacy, and reproducibility across diverse clinical settings.
Future efforts should focus on large-scale, well-designed clinical trials to validate FMT’s therapeutic efficacy, elucidate its long-term impact, and assess its suitability for distinct CRC stages, including early-stage patients where microbiota modulation may reduce progression risk and late-stage patients requiring adjunctive support for immune-based therapies. Integrating FMT into personalized CRC treatment strategies, supported by innovations such as precision microbiome editing, computational modeling, and advanced bioinformatics, offers a transformative approach to improving patient outcomes. By addressing core gaps in clinical translation—such as standardizing protocols, defining patient selection criteria, and conducting longitudinal safety studies—FMT could evolve from an experimental therapy to a cornerstone of microbiota-based CRC treatments.
