Abstract
Objective
To study the incidence of potential drug−drug interactions (DDIs) and evaluate their pattern and severity in cancer inpatients.
Materials and Methods
A detailed clinical data and prescriptions of 150 inpatients with different malignancies were subjected to DDI screening using Micromedex software. The frequency of potential DDIs and their types, patterns, and severity were investigated.
Results
A total of 360 potential DDIs were present in 111 (74%) of 150 inpatients, dominated by female (67.33%) and breast cancer (30%) patients. The incidence of severe interactions was 63.88%, moderate interactions 35.83%, and mild interactions 0.27%. The potential mechanisms of DDIs were 38.33% pharmacodynamic, 48.33% pharmacokinetic, and 13.33% unspecified. The drug interactions were found to be positively correlated (p < 0.01) with the 6–10 number of prescribed medicines.
Conclusion
According to this study, the number of medicines prescribed to cancer inpatients increased the chance of DDIs. As a result, the drug surveillance program could save a sizable number of patients from the potentially hazardous clinical effects of DDIs.
Keywords
Introduction
In India, cancer is an increasing burden that has serious social and economic repercussions, frequently resulting in family poverty and societal inequality. More than 7 lakh new cancer patients were reported each year. An estimated 6–7 lakh fatalities each year are attributed to cancer-related illnesses, while approximately 2.5 million people are living with cancer. 1 Surgery, chemotherapy, radiation therapy, immunotherapy, and hormone therapy are some of the modalities in the management of cancers. Of these, chemotherapy is one of the main modalities, in which anti-cancer drugs are used to prevent the progression and recurrence of various cancers like carcinoma breast, carcinoma lung, and carcinoma cervix. The anti-cancer drugs include alkylating agents, platinum compounds, antimetabolites, antibiotics, natural products, monoclonal antibodies, and hormones.2, 3
Drug−drug interactions (DDIs) are the major clinical or pharmacological reaction to the concomitant exposure of two or more medications interacting in a way that changes the drug’s toxicity or efficacy. 4 Since the majority of cancer patients frequently use many medications as part of their treatment, DDIs are particularly important in oncology. 5 As the majority of cancer patients are aged, they need additional medicines for treating metabolic comorbidities such as neurological, cardiovascular, gastro-intestinal, and age-related disorders. Interactions with other drugs might create changes in a chemotherapeutic agent’s pharmacokinetics or pharmacodynamics, which can result in decreased efficacy or increased toxicity. 6 Drug dose can be affected by DDIs, which is crucial for maximizing the anti-tumor efficacy of therapy and reducing their toxicity to healthy tissues. Human enzymes and transporters play a significant role in the metabolism and elimination of anti-cancer medications. 5 Factors like gender, intrinsic drug clearance, diet, genetics, and concurrent diseases of patients influence the DDIs, and the drug-specific factors include dosage, route of administration, drug formulation, and the sequence of drug administration. 7 Although precise information on the frequency of DDIs in oncology is not yet available, drug interactions may increase the risk of hospitalization and raise healthcare expenses. A reasonable, informed approach to DDIs that is founded on scientific understanding can thereby lower the probability of unfavorable results. With this context in mind, the current study sought to determine the incidence of potential DDIs as well as their pattern and severity in cancer patients undergoing chemotherapy.
Materials and Methods
Study Design and Data Collection
Cancer inpatients at MS Ramaiah Hospitals (medical oncology department), a tertiary care facility in South India, were recruited for this cross-sectional study from January to December 2017 after obtaining the institutional ethical committee approval (No. SS-1/EC/08/2016 dt. 18/10/2016). The patients who were less than 18 years old, pregnant and lactating, on concomitant Ayurvedic and herbal drugs, and undergoing radiotherapy were excluded from this study. A minimum sample of 100 newly diagnosed cancer patients was determined by considering the expected proportion of 33.9% with a relative precision of 15% and the desired confidence level of 95% with the assumption of the number of drugs in each prescription to be four. 6 But we were able to collect 150 cases. A detailed patient’s demographics, type of cancer, cancer stage, pre-existing diseases, and drugs prescribed (including anti-cancer drugs, supportive care medications, and drugs to treat the comorbidities) were recorded by interviewing the patient, consulting the oncologists, and reviewing the patient’s case records. Patients were advised to declare any medication self-taken at the time of enrolment, including over-the-counter medications. Each drug was counted separately in the analysis if a medication comprised two or more pharmacologically active ingredients. When a patient took the same prescription on different regimens (such as long-acting and short-acting morphine), twice daily, thrice daily, or both, the medication was only counted once. All cancer patients were included, irrespective of tumor staging.
Screening for DDIs
The potential DDIs among all the prescriptions were assessed using Micromedex software v2.0. Based on recently released data, this software has demonstrated higher accuracy, thoroughness, sensitivity, and specificity of medication interactions. According to the description of the software, all the detected interactions were divided into various groups based on the severity levels: ‘contraindicated’ refers to the drugs being contraindicated for concurrent use, ‘severe’ refers to the interaction being life-threatening and/or requiring medical intervention to minimize or prevent adverse effects, ‘moderate’ refers to the interaction resulting in an exacerbation of the patient’s condition and/or requiring an alteration in therapy, and ‘mild’ refers to the interaction having limited clinical effects. Manifestations may include an increase in the frequency or severity of the side effects, but generally, these would not require a major alteration in therapy. Additionally, the frequency of DDIs, their many subtypes, the relationship between age, the kind of cancer, and the quantity of medications prescribed were examined, and the pattern and severity of DDIs were analyzed.
Statistical Analysis
Quantitative variables like age and the number of drugs prescribed per patient were expressed as means and standard deviations, while the type of drug and the comorbidities were presented as frequencies and percentages. A Chi square test was used to find the association between DDIs and age, number of drugs prescribed, comorbidity, type of cancer, and so on. All analyses were performed using IBM SPSS Statistics version 20, and a p-value less than 0.05 was considered statistically significant.
Results
Clinical and Demographic Characteristics of Subjects
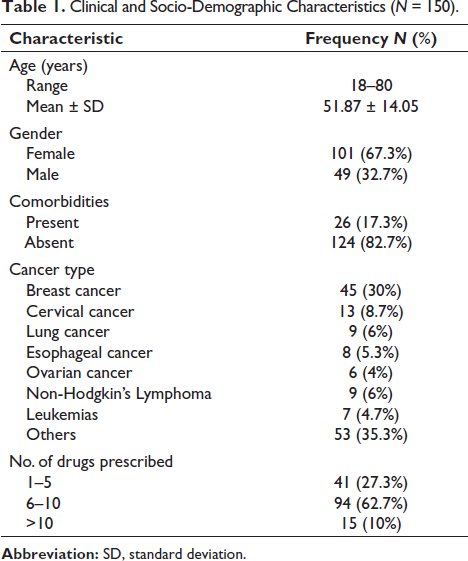
Among the 150 cancer inpatients included in the study, the majority of them were females. The clinical and socio-demographic characteristics are given in Table 1. The major cancer types present were breast cancer (30%), cervical cancer (8.7%), lung cancer (6%), non-Hodgkin’s lymphoma (6%), esophageal cancer (5.3%), leukemias (4.7%), ovarian cancer (4%), and others (35.3%). Comorbidities were present in 26 (17.3%) subjects, and the majority of them had diabetes (7), hypertension (3), followed by hypothyroidism (3), asthma (2), ischemic heart disease (1), seizure disorder (1), and the remaining nine subjects had >1 comorbid condition. The cancer inpatients were prescribed 1–20 drugs among the 1081 drugs used in the study, in which the majority of patients took 6–10 drugs (62.7%), followed by 1–5 (27.3%), and >10 drugs (10%) (Table 1).
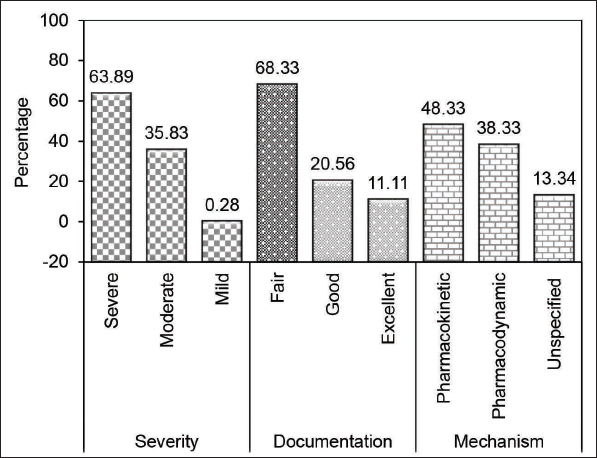
Clinical and Socio-Demographic Characteristics (N = 150).
Incidence of Potential DDIs
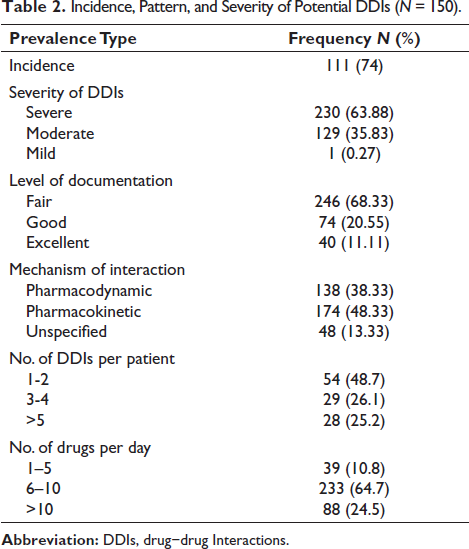
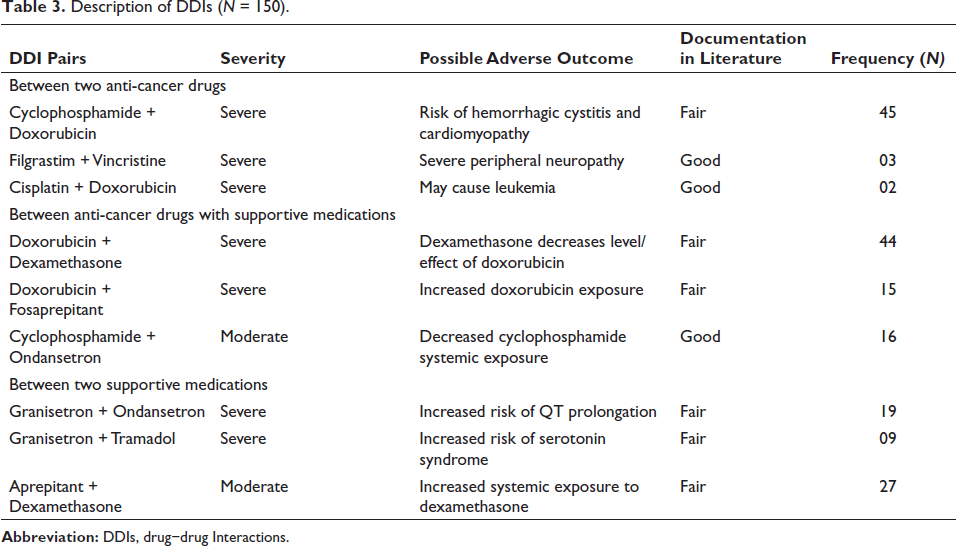
Among the 150 subjects, 111 experienced 360 potential DDIs, with an overall incidence of 74% in this tertiary care center (Table 2). Of the 360 potential DDIs, 230 (63.89%) interactions belonged to the severe category, 129 (35.83%) were of moderate severity, 1 (0.28%) was of mild severity, and none were found in the contraindicated category (Figure 1). In addition, depending on the level of documentation, the potential DDIs were classified as having fair (246; 68.33%), good (74; 20.55%), and excellent (40; 11.11%) documentation in the current literature according to the Micromedex tool (Figure 1). In terms of mechanism, 174 (48.33%) DDIs had a pharmacokinetic mechanism, 138 (38.33%) DDIs had a pharmacodynamic mechanism, and 48 (13.33%) DDIs had an unspecified mechanism of interaction (Figure 1). Among the 111 subjects who have potential DDIs, the incidence of 1–2 DDIs was 48.7% (n = 54), 3–4 DDIs was 26.1% (n = 29), and >5 DDIs was 25.2% (n = 28). In addition, the maximum number of DDIs was observed in patients who were taking 6–10 drugs per day (Table 2). Distribution of DDIs among different classes of chemotherapeutic drugs includes majorly platinum compounds to 71 patients, followed by alkylating agents (54), anthracycline antibiotics (49), taxanes (41), pyrimidine analogues (26), vinca alkaloids (9), and monoclonal antibodies (8). The most frequent pairs of DDIs were observed for the drug combination of cyclophosphamide and doxorubicin (Table 3).
Incidence, Pattern, and Severity of Potential DDIs (N = 150).
Description of DDIs (N = 150).
Discussion
According to the World Health Organization, DDI is the ‘modification of the effect of a drug can occur when administered with another drug; the effect may be an increase or a decrease in the action of either substance or it may be an adverse effect that is not normally associated with either drug’ 8 . This study determines the incidence of potential DDIs in cancer inpatients and classifies based on the severity, documentation level, and mechanism of interaction using Micromedex software. This study involved 150 inpatients who were on chemotherapy drugs, of which 101 were females (Table 1). Similar observations have been made in the study conducted by Ussai et al. in Italy, in which 69% of patients were female, and carcinoma breast (43%) was the most common tumor. 9 The mean age of the studied subjects was 51.87± 14.05 years, with a range of 18–80 years (Table 1). In a study conducted in a south Indian tertiary care teaching hospital, the median age of cancer patients was observed to be 56 years, with a range of 23–74 years. 6
The most common cancer diagnosed in this study was carcinoma breast (30%), followed by carcinoma cervix (8.6%), carcinoma lung (approximately 6%), non-Hodgkin’s lymphoma (6%), carcinoma esophagus (5.3%), leukemia (4.6%), and carcinoma ovary (4%). The rest of the cases were admitted for other malignancies like carcinoma tonsil, carcinoma epiglottis, carcinoma pancreas, and papillary carcinoma bladder (Table 1). A pilot study was conducted on cancer patients by Ussai et al., who recruited 64 cancer patients and the most common tumors included were carcinoma breast (43%), colorectal (27%), head and neck cancer (18%), and other malignancies contributed to 12% of the study population. The maximum number of cases included were carcinoma breast, similar to the present study. 9 The comorbidities observed in this study were associated with malignancy in 26 (17.33%) cancer patients. In a prospective study conducted by van Leeuwen et al., 302 patients were included, and 57% of them suffered from at least one comorbid disease. 10
The occurrence of DDIs and the disease condition were found to have no significance in this study. According to Tavakoli-Ardakani et al., in a study done in a developing country, the occurrence of potential DDIs in patients with hematological malignancy was significantly higher (54.09%) compared to the patients with solid tumor (31.28%). 11 A total of 1081 drugs were prescribed among 150 newly diagnosed cancer patients who are on their first cycle of chemotherapy. The incidence of DDI was found to be 74% of 150 patients, with at least one DDI observed in 111 patients (Table 2). Similar observations were made by Popa et al., who reviewed the medical records of 244 cancer patients and identified 769 potential drug interactions among 184 patients (75.4%) by screening potential interactions among all drugs, supplements, and herbal drugs using Drug Interactions Facts software. 12 However, in a study by Nightingale et al., who enrolled 142 cancer patients, they identified 310 major DDIs in 69% of patients using Lexi-Interact and 315 major DDIs in 61% of patients using Micromedex software. 13 Among 111 patients, 54 cancer inpatients developed 1–2 DDIs, 29 patients developed 3–4 DDIs, and 28 patients developed more than five DDIs (Table 3). The number of interactions ranged from 1 to 10, with the mean ± SD as 2.40 ± 2.416. There were no studies available to compare this result with. The cancer inpatients in this study were prescribed with minimum of 4 drugs and a maximum of 21 drugs (mean ± SD was 7.21 ± 2.6), which includes the chemotherapeutic regimen, antiemetics, analgesics, supportive care medications, drugs used for comorbidities, and over the counter medications.
Among DDIs between two anticancer drugs, about 45 DDIs were due to the cyclophosphamide and doxorubicin combination, with a possible outcome of risk of hemorrhagic cystitis and cardiomyopathy and were considered severe with respect to severity (Table 3). In a study conducted by Ussai et al., who enrolled 64 cancer patients, they also classified DDIs as neoplastic DDIs, involving at least one anti-tumor drug and non-neoplastic DDIs, involving other classes of drugs. 9 A similar study by Espinosa and others found that most of the DDIs occurred between non-antineoplastic agents (98.6%) of the total prescribed 7939 drugs. 14 A total of 26 anticancer drugs of various classes were administered to 150 cancer patients, and 360 potential DDIs were identified. The most common anticancer drugs were platinum compounds, which were administered to 71 patients, followed by alkylating agents (54), anthracycline antibiotics (49), taxanes (41), and other anticancer drugs (43). Similar studies were not available to compare the results.
Based on the severity of the DDIs, around 63.90% were categorized as severe, 35.83% as moderate, and 0.27% as mild interactions (Figure 1). Comparable results were found by Riechelmann et al. in Brazil, where 63 of 100 cancer patients experienced the potential interactions, of which 18.3% were severe, 56.7% were moderate, and 25% were mild interactions. 15 Furthermore, the DDIs observed in this study were also categorized based on the level of documentation in the current literature 246 (68.33%) of interactions were found to have fair documentation, 74 (20.55%) were found to have good, and 40 (11.11%) were found to have excellent documentation according to Micromedex software (Figure 1). Nightingale et al. identified 315 major DDIs in 61% of 142 outpatients using Micromedex tool, of which 195 interactions (62%) had fair reliability reporting, 95 interactions (30%) had good reliability, and 25 interactions (8%) had excellent reliability. 13 From the analyzed material, out of 360 drug interactions, 174 (48.33%) were pharmacokinetic interactions, 138 (38.33%) were pharmacodynamic interactions, and 48 (13.33%) were of an unspecified mechanism (Figure 1). In a similar study by Kannan et al., 88% DDIs were classified as pharmacokinetic, 5% as pharmacodynamic and 7% of DDIs with the unknown mechanism of the 213 interactions among 75 patients. 6 However, a Spanish hospital-based study conducted by Espinosa and others found 62.6% of pharmacodynamic interactions and 22.6% of pharmacokinetic interactions based on the mechanism of interaction in 246 patients. 14 The usability and appropriateness of commercially available electronic databases such as Lexi-Interact, Micromedex, Drug-Reax, Drug Interaction Facts, and Pharmavista software were systematically assessed by comparing each other to assess the incidence of potential DDIs. It was discovered that there was a significant discrepancy in the number and relevance of observed DDIs, but that there was an overlap of 11% in some cases. 16
Conclusion
The number of prescribed medicines increased the risk of DDIs, as was observed in cancer inpatients in this study. Therefore, adopting the medication surveillance program could prevent the patients from experiencing potentially harmful clinical consequences of DDIs and thereby improve their quality of life. Overall, the identification of potential DDIs may enable the prescribing clinician to avoid their occurrence through intense monitoring, careful dose titration, and frequency of drug administration. It also helps to restrict the coadministration of one or more interacting drugs.
Footnotes
Abbreviations
DDI: Drug−Drug Interaction.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical approval was obtained form the Institutional Ethical Committee (No. SS-1/EC/08/2016 dt. 18/10/2016), Department of Pharmacology, MS Ramaiah Medical College, Bangalore, Karnataka, India.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
