Abstract
Objective:
To investigate potential interactions between rivaroxaban, an oral direct Factor Xa inhibitor approved for the management of thromboembolic disorders, and digoxin or atorvastatin.
Methods:
Two randomized, phase 1 clinical trials were undertaken in healthy men to assess pharmacokinetic and pharmacodynamic interactions between rivaroxaban and digoxin or atorvastatin, and the safety of these drug combinations.
Results:
Steady-state rivaroxaban did not affect the pharmacokinetic profile of steady-state digoxin (n = 17). Digoxin did not significantly influence the pharmacokinetic profile of single-dose rivaroxaban and had minimal effects on rivaroxaban-induced inhibition of Factor Xa activity and prolongation of clotting time. Similarly, steady-state atorvastatin did not affect the pharmacokinetic profile or the pharmacodynamics of rivaroxaban and vice versa (n = 19). All drugs (alone or in combination) were well tolerated.
Conclusions:
There were no clinically relevant pharmacokinetic or pharmaco - dynamic interactions between rivaroxaban and digoxin, or between rivaroxaban and atorvastatin, suggesting that rivaroxaban can be coadministered with either drug. This study also confirmed that rivaroxaban does not interact with substrates for permeability (P)-glycoprotein alone (digoxin) or P-glycoprotein and cytochrome P450(CYP)3A4 (atorvastatin).
Introduction
Thromboembolic disorders are associated with significant mortality and morbidity, and anticoagulant therapy plays a key role in the management of these conditions. New oral anticoagulants that selectively and specifically target key coagulation factors (i.e. Factor Xa and thrombin) have been developed in recent years. These new agents have favourable pharmacological profiles compared with vitamin K antagonists, which have traditionally been used to provide oral anticoagulant therapy. 1 Rivaroxaban (Xarelto® Bayer Pharma AG, Berlin, Germany) is an oral, direct Factor Xa inhibitor that has been evaluated in a number of large-scale, randomized phase 3 studies in the prevention and treatment of thromboembolic disorders.2–9 Based on the outcomes of these studies, rivaroxaban has received regulatory approval in many countries for the prevention of venous thromboembolism in adult patients undergoing elective hip or knee replacement surgery. Approval has also been granted for the treatment of deep vein thrombosis (DVT) and the secondary prevention of DVT and pulmonary embolism following an acute DVT in adults (EU and Canada), and for the prevention of stroke and systemic embolism in adult patients with atrial fibrillation (AF; EU, Canada, Japan and the USA). In clinical practice, it is likely that many patients treated with rivaroxaban will also receive other commonly used drugs for cardiovascular disorders, including cardiac glycosides – such as digoxin – and statins.
Digoxin is used for the control of ventricular rate in patients with heart failure, as well as in patients with AF, 10 which is a common cardiac arrhythmia associated with an increased risk of stroke. 11 The transport protein permeability (P)-glycoprotein (P-gp) is thought to play a role in the elimination of some drugs, including digoxin. 12 Drug interactions involving transport proteins are increasingly being reported and such interactions may alter the pharmacokinetics of digoxin. For example, competitive inhibition of P-gp can lead to increased, toxic, digoxin plasma concentrations.10,13,14 Because digoxin toxicity can result in potentially life-threatening cardiac arrhythmias and other adverse events, 14 it is important to determine whether coadministration with other drugs can alter digoxin plasma concentrations.
Atorvastatin (Lipitor® Pfizer, Berlin, Germany) is a synthetic lipid-lowering drug of the 3-hydroxy-3-methyl-glutaryl-CoA reductase class, more commonly referred to as statins. It is commonly used to treat hypercholesterolaemia and hyper -lipidaemia, and to prevent cardiovascular disease.15–17 Atorvastatin is metabolized by cytochrome P450 (CYP)3A4 to its 2-hydroxy (ortho-) and 4-hydroxy (para-) forms. 18 In addition, atorvastatin has been reported to be a substrate of P-gp. 18 Many drugs are metabolized by CYP3A4, meaning that the potential for drug–drug interactions with atorvastatin is substantial. 19 Investigation of potential interactions is warranted, particularly because increased atorvastatin plasma concentrations resulting from drug interactions have been associated with adverse events that can be severe, such as rhabdomyolysis.20,21
The novel oral anticoagulant rivaroxaban exerts its anticoagulant effect by directly inhibiting Factor Xa activity, thereby blocking thrombin generation.22,23 After oral administration, rivaroxaban is rapidly absorbed, with maximum plasma concentrations being reached within 2 – 4 h.24,25 Of the administered rivaroxaban dose, approximately two-thirds undergoes metabolic degradation, with half then being eliminated renally and the other half eliminated by the hepatobiliary route. The final one-third of the administered dose is excreted renally as unchanged active drug in the urine. Rivaroxaban is metabolized via CYP3A4, CYP2J2 and CYP-independent mechanisms, and is a substrate for P-gp.26,27 Thus, it is important to identify any potential interactions between rivaroxaban and commonly used cardiovascular drugs that are also substrates of P-gp and/or CYP3A4, such as atorvastatin and digoxin.
Two drug interaction studies in healthy male subjects were undertaken to determine potential interactions between digoxin and rivaroxaban, and between atorvastatin and rivaroxaban.
Subjects and methods
Study Population and Design
Healthy Caucasian men, aged 18 – 55 years and with a body mass index (BMI) of 18 – 30 kg/m2, were recruited in two studies: a digoxin–rivaroxaban interaction study and an atorvastatin–rivaroxaban interaction study. Both took place between November 2003 and April 2007. The exclusion criteria included known coagulation disorders, an increased risk of bleeding, and a history of drug allergy or active liver disease (atorvastatin– rivaroxaban interaction study only). Both studies were single-centre, randomized, nonblinded, crossover studies conducted at ClinPharmCologne, MEDA Manufacturing GmbH, Cologne, Germany. The studies were approved by the Ethics Committee of the North-Rhine Medical Council, Dusseldorf, Germany, and were conducted in accordance with the Declaration of Helsinki, the International Conference on Harmonisation Good Clinical Practice guidelines and German drug law. All subjects provided written informed consent to participate in the studies.
Digoxin has a narrow therapeutic window, which necessitates monitoring of its plasma concentration. 28 The digoxin– rivaroxaban interaction study determined the effects of a concomitant intake of multiple doses of rivaroxaban on the steady-state pharmacokinetics of digoxin. The safety and tolerability of rivaroxaban and digoxin coadministration, and the effects of multiple doses of digoxin on the pharmacokinetics and pharmacodynamics of rivaroxaban, were also investigated.
The objective of the atorvastatin– rivaroxaban interaction study was to determine the effects of steady-state plasma concentrations of atorvastatin on the safety, tolerability, pharmacokinetics and pharmaco -dynamics of rivaroxaban, and vice versa.
All subjects were admitted to the study unit the day before receiving the first dose of study drug, and had physical and electrocardiogram (ECG) examinations before both studies began. Blood samples for laboratory analyses were also taken. In both studies, subjects were randomized before the start of the trial (after the prestudy examination) by use of a computer-generated randomization schedule.
Both studies used 20 mg rivaroxaban because this dose has demonstrated substantial pharmacodynamic effects in healthy subjects in previous studies;24,25 should either of the concomitantly administered drugs evaluated (digoxin or atorvastatin) increase rivaroxaban plasma concentrations, the 20-mg dose is below the maximum, well-tolerated dose tested in healthy subjects (a single 80-mg dose).24,25
Digoxin–Rivaroxaban Interaction Study
All subjects received a single dose of 20 mg rivaroxaban orally on day 0. From day 2, all subjects were given 0.375 mg digoxin (Lanoxin Mite® GlaxoSmithKline, Research Triangle Park, NC, USA) orally once daily for a period of 28 days to achieve steady state. Subjects were randomized to receive a single dose of 20 mg rivaroxaban on either day 6 (group A) or day 20 (group B), followed by 20 mg rivaroxaban twice daily for 9 consecutive days to achieve steady state (group A, days 7 – 15; group B, days 21 – 29; Fig. 1). The data were analysed as a two-way crossover trial. The treatment sequences A and B were applied in parallel. Study days were mapped to a crossover design.
Design of the current single-centre, randomized, nonblinded crossover study to investigate potential pharmacokinetic (PK) or pharmacodynamic (PD) interactions between rivaroxaban and steady-state digoxin in healthy male subjects
Subjects self-administered digoxin alone on an outpatient basis on days 18 – 27 in group A and on days 4 – 13 in group B (which was documented for compliance), and were inpatients for the remainder of the study. On profile days (see below), study drugs were administered with water in the morning after an overnight fast. On the other days, study drugs were administered within 2 h of breakfast.
Digoxin–rivaroxaban interaction study pharmacokinetics
The pharmacokinetics of steady-state digoxin alone on day 29 (group A) and day 15 (group B), and in the presence of steady-state rivaroxaban on day 15 (group A) and day 29 (group B), were determined (Fig. 1). Blood samples were taken immediately before and 0.5, 1, 1.5, 2, 2.5, 3, 4, 6, 8, 12, 15 and 24 h after digoxin administration. Blood samples were collected on days 30 (24h), 31 (48h), 32 (72h) and 33 (96h). Urine was collected and pooled over 24 h on days 15 and 29 in each group. In addition, trough serum concentrations of digoxin were determined using blood samples obtained immediately before the next digoxin dose on the last 3 consecutive days of rivaroxaban dosing. Trough (rather than peak) concentrations were measured owing to the substantial interindividual variability in serum digoxin concentrations after administration, 29 which makes peak serum concentrations difficult to define.
The pharmacokinetic profile of single-dose rivaroxaban alone was determined on day 0, and in the presence of steady-state digoxin on day 6 (group A) and day 20 (group B; Fig. 1). Blood samples were collected on day 0 immediately before and 0.3, 0.7, 1, 1.5, 2, 3, 4, 6, 8, 12, 15, 24, 36 and 48 h after administration of rivaroxaban. Blood samples were also collected immediately before and 0.3, 0.7, 1, 1.5, 2, 3, 4, 6, 8, 12, 15 and 24 h after rivaroxaban administration on day 6 or day 20 for groups A and B, respectively. Urine was collected and pooled over a 24 h period after rivaroxaban administration on day 0, and on day 6 or day 20 for groups A and B, respectively.
Digoxin–rivaroxaban interaction study pharmacodynamics
The pharmacodynamic effects of single-dose rivaroxaban alone were measured on day 0. The pharmacodynamic profiles of single-dose rivaroxaban in the presence of steady-state digoxin were measured on day 6 in group A and day 20 in group B, and for steady-state rivaroxaban in the presence of steady-state digoxin on day 15 in group A and day 29 in group B (Fig. 1). Pharmacodynamic effects were assessed in plasma samples obtained before and 1, 1.5, 2, 3, 4, 6, 8 and 12 h after rivaroxaban administration.
Atorvastatin–Rivaroxaban Interaction Study
In a three-way crossover design, subjects were randomized to receive 20 mg rivaroxaban alone on day 0 (group A), atorvastatin alone to steady state (10 mg once daily on days 0 – 2 and 20 mg once daily on days 3 – 6; group B), and the combination of both 10 mg (days 0 – 2) and 20 mg (days 3 – 6) atorvastatin with 20 mg rivaroxaban on day 6 (group C; Fig. 2). Study drugs were administered in the morning with water after a standardized breakfast following an overnight fast. Subjects self-administered atorvastatin alone for 6 days on an outpatient basis and were inpatients for the remainder of the study.
Design of the current single-centre, randomized, nonblinded crossover study to investigate potential pharmacokinetic (PK) or pharmacodynamic (PD) interactions between rivaroxaban and steady-state atorvastatin in healthy male subjects (R, randomization)
Atorvastatin–rivaroxaban interaction study pharmacokinetics
The pharmacokinetic profiles of single-dose rivaroxaban alone and in the presence of steady-state atorvastatin were determined on day 0 in group A and on day 6 in group C, respectively (Fig. 2). Blood samples for determination of the pharmacokinetics of rivaroxaban alone were collected 0, 0.5, 1, 2, 3, 4, 6, 8, 12 and 15 h after administration on day 0, at 0 and 12 h on day 1, and at 48 and 72 h in subjects in group A. The pharmacokinetic profiles of steady-state atorvastatin alone and in the presence of single-dose rivaroxaban were determined on day 6 in groups B and C, respectively (Fig. 2). To determine the effect of steady-state atorvastatin on the pharmacokinetics of rivaroxaban, blood samples were collected 0, 0.5, 1, 2, 3, 4, 6, 8, 12 and 15 h after administration on day 6; at 0 and 12 h on day 7, and at 0 h on days 8 and 9, in group C. In addition, urine samples were taken on day 6 at 0, 4, 8 and 12 h, and at 0 h on days 7, 8 and 9.
Atorvastatin–rivaroxaban interaction study pharmacodynamics
The pharmacodynamic effects of single-dose rivaroxaban alone and in the presence of steady-state atorvastatin were determined on day 0 in group A and on day 6 in group C, respectively (Fig. 2). Blood samples for the determination of the pharmacodynamics of rivaroxaban alone were collected 0, 0.5, 1, 2, 3, 4, 6, 8, 12 and 15 h after administration on day 0, at 0 and 12 h on day 1, and at 0 h on days 2 and 3 in group A. To determine the effect of steady-state atorvastatin on the pharmacodynamics of rivaroxaban, blood samples were collected 0, 0.5, 1, 2, 3, 4, 6, 8, 12 and 15 h after administration on day 6, at 0 and 12 h on day 7, and at 0 h on days 8 and 9 in group C.
Plasma, Serum and Urine Sample Analyses
In the digoxin interaction study, blood samples for determination of rivaroxaban in plasma were collected in ammonium heparin tubes. In the atorvastatin interaction study, blood samples for determination of rivaroxaban and atorvastatin in plasma were collected in lithium heparin tubes. If the sampling tubes were completely filled, the resulting heparin concentration was approximately 15 IU/ml blood. Blood samples for pharmacokinetic analysis of atorvastatin or rivaroxaban were centrifuged (digoxin–rivaroxaban interaction study, 3900 rpm for 10 min; atorvastatin– rivaroxaban interaction study, 3000 rpm for 5 min) and the resulting plasma samples were frozen and stored below –15 °C until analysis. All pharmacokinetic analyses were performed using fully validated methods and analyses were carried out in a nonblinded fashion.
Concentrations of digoxin in serum and urine were determined after appropriate dilution using the Elecsys® Digoxin electro -chemi luminescence immunoassay (Roche Diagnostics, Mannheim, Germany). Serum samples were prepared by allowing whole blood samples to clot, followed by centrifugation at 1500
Plasma rivaroxaban concentrations were measured using a close chemical analogue of rivaroxaban as an internal standard, as described previously. 30 Briefly, they were determined using a validated liquid chromatography–mass spectrometry/ tandem mass spectrometry method using the SCIEX API 3000™ LC/MS/MS system (Applied Biosystems, Foster City, CA, USA) after solid-phase extraction. Plasma concentrations of rivaroxaban above the LLOQ (0.50 μg/l) were determined with an accuracy of 92.1 – 98.9% and a precision of 4.3 – 6.6% across both studies. Rivaroxaban concentrations in urine were determined by high-performance liquid chromatography with ultraviolet detection after solid-phase extraction. Concentrations of rivaroxaban above the LLOQ (0.01 mg/l) were determined with an accuracy of 95.5 – 101.0% and a precision of 0.9 – 7.1% across both studies.
Atorvastatin is metabolized by CYP3A4 to 2-hydroxy- and 4-hydroxy atorvastatin (found as both acid and lactone in human plasma), 18 so plasma concentrations of the parent compound and its CYP3A4-mediated metabolites were determined. Plasma concentrations of atorvastatin and its metabolites above the LLOQ (0.05 μg/l) were determined with an accuracy of 96.7 – 103.4% and a precision of 2.6 – 10.3%.
Pharmacokinetic and Pharmacodynamic Assessments
The primary pharmacokinetic parameters determined for digoxin included the area under the serum concentration–time curve (AUC) during any dosing interval at steady state (AUCτ, ss) and the serum digoxin concentration at the expected minimum (trough) concentration at steady state (Ctrough, ss). Other pharmacokinetic parameters determined for digoxin were: AUCτ, ss normalized to dose and body weight; maximum serum concentration (Cmax); Cmax normalized to dose and body weight (Cmax, norm); time to peak serum concentration (tmax); renal clearance (CLR); amount of drug excreted via urine (Aeur); and half-life (t½).
The pharmacokinetic parameters of rivaroxaban determined in both studies included AUC and Cmax (primary parameters in the atorvastatin study), AUC normalized to body weight (AUCnorm), Cmax, norm, tmax, CLR, Aeur and t½.
The primary pharmacokinetic parameters determined for atorvastatin were AUC from time 0 to 24 h (AUC0 – 24 h), Cmax, tmax and t½.
In both studies, the following pharmacodynamic parameters for rivaroxaban were determined as described previously: 30 inhibition of Factor Xa activity, prolongation of prothrombin time (PT), activated partial thromboplastin time (aPTT) and HepTest (Haemachem, St Louis, MO, USA). Pharmacodynamic effect–time curves were plotted and parameters calculated included area under the effect–time curve (AUEC), maximum effect (Emax) and tmax.
Safety and Tolerability of Rivaroxaban, Digoxin and Atorvastatin
Subjective tolerability was assessed by interviewing subjects on the occurrence of any adverse event and by spontaneous reporting of events. Objective tolerability was assessed by measuring haematological parameters, clinical chemistry and resting heart rate, blood pressure and ECG examination. The narrow therapeutic window of digoxin meant that subjects in the study unit were required to undergo haematological and clinical chemistry assessments at least once every 2 days, and cardiovascular and ECG readings were taken at least once daily.
Statistical Analyses
Statistical analyses were carried out using the SAS software package, version 9.1 (SAS Institute Inc., Cary, NC, USA). The primary pharmacokinetic parameters of digoxin (AUCτ, ss and Ctrough, ss) were analysed assuming log-normally distributed data. The logarithms of these parameters were determined by analysis of variance (ANOVA), including sequence, period and treatment effects. Point estimates (least squares [LS]-mean) and confirmatory 90% confidence intervals (CIs) for the ratio of digoxin plus rivaroxaban versus digoxin alone were calculated by retransformation of the logarithmic data using the intraindividual SD of the ANOVA.
The AUC and Cmax of single-dose rivaroxaban were analysed using ANOVA, including a period effect in the digoxin– rivaroxaban interaction study, and sequence, period and treatment effects in the atorvastatin–rivaroxaban interaction study, assuming log-normally distributed data. Point estimates (LS-mean) and 90% CIs (exploratory in the digoxin–rivaroxaban interaction study; confirmatory in the atorvastatin–rivaroxaban interaction study) were calculated for the ratios of rivaroxaban alone versus digoxin plus rivaroxaban or atorvastatin plus rivaroxaban. In addition, similar exploratory analyses were undertaken for the AUC and Cmax of atorvastatin and its CYP-mediated metabolites atorvastatin lactone and 2-hydroxy atorvastatin, alone and in the presence of rivaroxaban. Furthermore, the pharmacodynamic effects of rivaroxaban (i.e. the AUEC and Emax of inhibition of Factor Xa activity and prolongation of PT) were analysed in an exploratory manner using the same method, including a sequence effect. On Day 6, point estimates (LS-mean) and 90% CIs were calculated for ratios of the pharmacodynamic effects of: single-dose rivaroxaban plus steady-state digoxin versus single-dose rivaroxaban; steady-state rivaroxaban plus steady-state digoxin versus single-dose rivaroxaban; steady-state rivaroxaban plus steady-state digoxin versus single-dose rivaroxaban plus steady-state digoxin.
In all analyses (pharmacokinetics and pharmacodynamics), if the calculated 90% CIs for the ratios were within the accepted limits of bioequivalence (0.80 – 1.25), a clinically relevant interaction was excluded.14,31
Results
Study Population
In the digoxin–rivaroxaban interaction study, 20 subjects were enrolled: mean age 34 years (range 22 – 45 years), mean ± SD weight 78.8 ± 10.6 kg and BMI 24.5 ± 2.8 kg/m2. One subject was withdrawn before receiving study drugs; accordingly, 19 subjects were included in the safety analysis (10 in group A and nine in group B). One subject withdrew consent after receiving digoxin for 18 days, and one subject was noncompliant with the study drug protocol. Thus, a total of 17 subjects (10 in group A, seven in group B) were available for the pharmacokinetic and pharmacodynamic analyses. Unless stated otherwise, data sets from group A and group B were pooled for statistical analyses.
In the atorvastatin–rivaroxaban interaction study, 26 subjects were enrolled: mean age 41.9 years (range 24 – 53 years), mean ± SD weight 87.1 ± 11.7 kg and BMI 26.6 ± 2.7 kg/m2. Three subjects did not receive study drugs because of inclusion/exclusion criteria violations or withdrawal of consent. Thus, 23 subjects were available for the safety analysis. A further four subjects terminated the study prematurely as a result of protocol violation, withdrawal of consent or adverse events (microscopic haematuria and creatine phosphokinase elevation); thus, 19 subjects were available for the pharmacokinetic and pharmacodynamic analyses. In total, 21 subjects received a single dose of 20 mg rivaroxaban, 23 received atorvastatin alone and 19 received the combination in accordance with the study protocol.
Rivaroxaban, Digoxin and Atorvastatin Pharmacokinetics
Rivaroxaban had no effect on the steady-state pharmacokinetic characteristics of digoxin (Table 1, Fig. 3A). The LS-mean ratios for AUCτ, ss and Ctrough, ss in the absence and presence of steady-state rivaroxaban were near unity, with 90% CIs within the limits of bioequivalence, indicating no pharmacokinetic interaction between rivaroxaban and digoxin. Urinary excretion of digoxin was not affected by concomitant rivaroxaban administration (Table 1).
Serum concentration–time profile of: (A) steady-state digoxin (0.375 mg once daily) alone and in the presence of steady-state rivaroxaban (20 mg twice daily for 9 days); and (B) single-dose 20 mg rivaroxaban alone and in the presence of steady-state digoxin (geometric mean values from healthy male subjects [n = 17])
Effect of concomitant administration of steady-state 20 mg rivaroxaban twice daily on the pharmacokinetic parameters of steady-state digoxin in healthy male subjects

Data presented as geometric mean (geometric coefficient of variation), unless otherwise stated.
If 90% confidence intervals (CI) were within accepted limits of bioequivalence (0.80 – 1.25), a clinically relevant interaction was excluded.
LS-mean ratios were calculated by retransformation of the logarithmic results given by analysis of variance (ANOVA); ANOVA was only performed for the parameters AUCτ, ss and Ctrough, ss.
Terminal half-life (t½) after the last digoxin dose (n = 10 for steady-state digoxin alone; n = 7 for digoxin plus rivaroxaban, both at steady state).
LS-mean, least-squares mean; AUCτ, ss, area under the serum concentration–time curve at steady state during any dosing interval; Cmax, maximum drug concentration in serum; Ctrough, ss, serum concentration at expected minimum (trough) concentration at steady state; tmax, time to peak serum concentration; CLR, renal drug clearance; Aeur, amount of drug excreted via urine.
Steady-state serum concentrations of digoxin had no significant effect on the pharmacokinetics of single-dose rivaroxaban (Table 2, Fig. 3B). Although the AUC and Cmax of single-dose rivaroxaban decreased slightly when given with steady-state digoxin, the LS-mean ratios for these parameters showed that rivaroxaban given alone and with steady-state digoxin met the prespecified bioequivalence criteria (Table 2). In the presence of digoxin, the tmax of rivaroxaban was prolonged from 1.5 to 2 h. Urinary excretion of rivaroxaban was not affected by concomitant steady-state digoxin (Table 2); approximately 20% of the administered dose of rivaroxaban was excreted in the urine, irrespective of whether digoxin was present or not.
The effect of digoxin at steady-state serum concentrations on the pharmacokinetic parameters of a single dose of 20 mg rivaroxaban in healthy male subjects
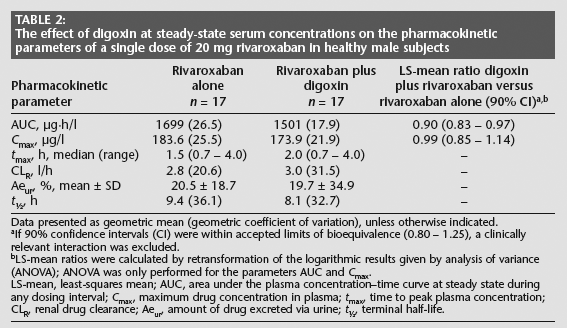
Data presented as geometric mean (geometric coefficient of variation), unless otherwise indicated.
If 90% confidence intervals (CI) were within accepted limits of bioequivalence (0.80 – 1.25), a clinically relevant interaction was excluded.
LS-mean ratios were calculated by retransformation of the logarithmic results given by analysis of variance (ANOVA); ANOVA was only performed for the parameters AUC and Cmax.
LS-mean, least-squares mean; AUC, area under the plasma concentration–time curve at steady state during any dosing interval; Cmax, maximum drug concentration in plasma; tmax, time to peak plasma concentration; CLR, renal drug clearance; Aeur, amount of drug excreted via urine; t½, terminal half-life.
Steady-state atorvastatin plasma concentrations did not affect the pharmacokinetics of rivaroxaban (Table 3, Fig. 4A). The LS-mean ratios for the AUC and Cmax of single-dose rivaroxaban alone and in the presence of a steady-state plasma concentration of atorvastatin confirmed the lack of effect of atorvastatin on these rivaroxaban pharmacokinetic parameters (Table 3). The excretion of rivaroxaban in urine was similar when rivaroxaban was administered alone and with steady-state atorvastatin.
Plasma concentration–time profile of: (A) single-dose 20 mg rivaroxaban with and without steady-state atorvastatin; and (B) steady-state atorvastatin after administration of steady-state atorvastatin with and without single-dose 20 mg rivaroxaban (geometric mean values from healthy male subjects [n = 19])
The effect of atorvastatin at steady-state plasma concentrations on the pharmacokinetic parameters 20 mg rivaroxaban in healthy male subjects

Data presented as geometric mean (geometric coefficient of variation), unless otherwise indicated.
If 90% confidence intervals (CI) were within accepted limits of bioequivalence (0.80 – 1.25), a clinically relevant interaction was excluded.
LS-mean ratios were calculated by retransformation of the logarithmic results given by analysis of variance (ANOVA); ANOVA was only performed for the parameters AUC and Cmax.
LS-mean, least-squares mean; AUC, area under the plasma concentration–time curve; Cmax, maximum drug concentration in plasma; tmax, time to peak plasma concentration; CLR, renal drug clearance Aeur, amount of drug excreted via urine; t½, terminal half-life.
Rivaroxaban had no effect on the pharmacokinetic characteristics of atorvastatin (Fig 4B) or its CYP3A4-mediated metabolites 2-hydroxy atorvastatin and atorvastatin lactone (Table 4).
Effect of single-dose 20 mg rivaroxaban on the pharmacokinetic parameters of atorvastatin and its cytochrome P450 3A4-mediated metabolites 2-hydroxy atorvastatin and atorvastatin lactone in healthy male subjects
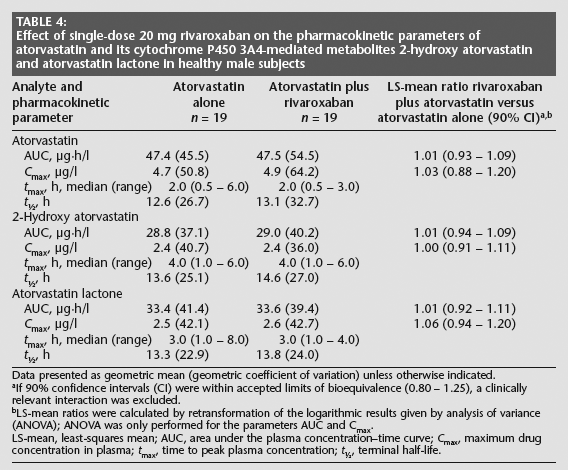
Data presented as geometric mean (geometric coefficient of variation) unless otherwise indicated.
If 90% confidence intervals (CI) were within accepted limits of bioequivalence (0.80 – 1.25), a clinically relevant interaction was excluded.
LS-mean ratios were calculated by retransformation of the logarithmic results given by analysis of variance (ANOVA); ANOVA was only performed for the parameters AUC and Cmax.
LS-mean, least-squares mean; AUC, area under the plasma concentration–time curve; Cmax, maximum drug concentration in plasma; tmax, time to peak plasma concentration; t½, terminal half-life.
Effects of Steady-State Digoxin or Atorvastatin on Rivaroxaban Pharmacodynamics
The Emax of inhibition of Factor Xa activity was not affected by the presence of steady-state serum concentrations of digoxin in combination with single-dose rivaroxaban compared with single-dose rivaroxaban alone (Table 5, Fig. 5A). There was a slight increase in Emax when steady-state rivaroxaban plus steady-state digoxin was compared with single-dose rivaroxaban alone or single-dose rivaroxaban plus steady-state digoxin (Table 5). The AUEC of inhibition of Factor Xa activity induced by single-dose rivaroxaban in the presence of steady-state digoxin was slightly reduced compared with single-dose rivaroxaban alone (Table 5, Fig. 5A). When steady-state rivaroxaban was combined with steady-state digoxin, there was a slight increase in the AUEC of inhibition of Factor Xa activity associated with rivaroxaban compared with single-dose rivaroxaban alone, which was enhanced further when compared with single-dose rivaroxaban combined with steady-state digoxin (Table 5).
Effect of single-dose 20 mg rivaroxaban alone and in the presence of steady-state digoxin (0.375 mg once daily), and steady-state rivaroxaban (20 mg twice daily for 9 days) in the presence of steady-state digoxin on: (A) inhibition of Factor Xa activity over time; and (B) prolongation of prothrombin time (× fold change from baseline) (median values for each individual time point in healthy male subjects [n = 17])
Effect of single-dose 20 mg rivaroxaban alone, with steady-state digoxin and with both drugs at steady state on inhibition of Factor Xa activity and relative prolongation of prothrombin time in healthy male subjects (n = 17)

LS-mean, least-squares mean; CI, confidence interval; AUEC, Area under effect–time curve, Emax, maximum effect.
The Emax (mean of maximal values for each individual) of PT prolongation was 1.61-fold of baseline at run-in and 1.56-fold of baseline on the first day of rivaroxaban treatment with digoxin. The Emax of PT prolongation at rivaroxaban and digoxin steady state was slightly increased to 1.82fold of baseline (Fig 5B). Prolongation of PT occurred within 2 h of rivaroxaban administration in all groups. The LS-mean ratios and their 90% CIs for AUEC and Emax indicated that coadministration with steady-state digoxin had no significant effect on the prolongation of PT induced by rivaroxaban (Table 5).
Statistically significant differences between groups A and B (rivaroxaban after short-term or after longer-term digoxin, respectively) were demonstrated for the AUEC and Emax of PT prolongation relative to baseline (P < 0.01; data not shown). Both groups (alone or pooled together) showed no significant differences compared with rivaroxaban alone (as demonstrated by LS-means ratios and 90% CIs; data not shown). Digoxin did not have a marked effect on rivaroxaban-induced prolongation of aPTT or HepTest (data not shown).
Atorvastatin alone did not affect Factor Xa activity. Steady-state atorvastatin had no relevant impact on rivaroxaban-mediated inhibition of Factor Xa activity. Maximum inhibition of Factor Xa activity after rivaroxaban alone was 56% at 4 h postdose, and was similar when coadministered with steady-state atorvastatin (53% at 3 h postdose) (Fig. 6A).
Effect of single-dose 20 mg rivaroxaban alone, steady-state atorvastatin alone and single-dose rivaroxaban in combination with steady-state atorvastatin on: (A) inhibition of Factor Xa activity over time; and (B) prolongation of prothrombin time (× fold change from baseline) (median values for each individual time point in healthy male subjects [n = 19])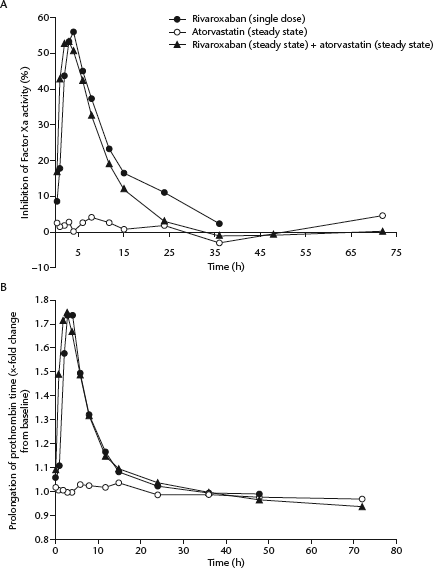
Steady-state atorvastatin alone had no impact on PT (Fig. 6B). After the administration of single-dose rivaroxaban alone, PT was prolonged up to a maximum of 1.74 times baseline (median relative change) at 3 – 4 h postdose. An identical prolongation of PT was observed after 3 h when rivaroxaban was administered with steady-state atorvastatin. Atorvastatin did not have a marked effect on rivaroxaban-induced prolongation of aPTT or HepTest (data not shown).
Safety and Tolerability
All subjects reported at least one adverse event. Of the 93 treatment-emergent adverse events reported, 45 were considered to be drug related. Single-dose rivaroxaban alone was not associated with any drug-related adverse events. With one exception, all drug-related events were mild to moderate in severity and had resolved by study completion; one subject experienced a serious nondrug-related psychosis event starting 13 days after receiving a single dose of rivaroxaban followed by 9 days of digoxin (the subject stopped self-administering digoxin 2 days before the event). The psychotic episode was triggered by alcohol and amphetamine abuse, and was not considered to be drug related. The subject was hospitalized and the event resolved after treatment. Treatment-emergent adverse events were reported by 11 of 19 (58%) subjects receiving steady-state digoxin alone and by 15 of 19 (79%) subjects receiving steady-state digoxin plus rivaroxaban. Headache was the most frequent adverse event, occurring in eight subjects receiving digoxin plus rivaroxaban (both at steady state) and two subjects receiving steady-state digoxin alone.
Psychiatric disorders (such as insomnia, abnormal dreams, nightmares, or sleep or psychotic disorders), all considered to be study drug-related, occurred in 21% of subjects (four of 19) receiving combination therapy and in 5% of subjects (one of 19) receiving digoxin alone.
Eighteen of the 23 subjects reported a total of 47 treatment-emergent adverse events; two occurrences of mild and one occurrence of moderate headache were possibly related to rivaroxaban, and seven adverse events were thought to be related to atorvastatin. Three events were considered moderate in intensity (conjunctivitis after receiving rivaroxaban plus atorvastatin; vomiting and headache after rivaroxaban alone); all other adverse events were considered to be mild in intensity. All but two events (intermittent glutamate dehydrogenase elevation and microscopic haematuria) had resolved by the end of the study. Adverse events were reported by 29% (six of 21) of subjects receiving rivaroxaban alone, 39% (nine of 23) of subjects receiving atorvastatin alone who had achieved steady-state plasma concentrations, and 79% (15/19) of subjects receiving rivaroxaban who had achieved steady-state atorvastatin concentrations. The most frequently reported adverse events were changes in laboratory parameters – one subject terminated the study because of creatine kinase elevation (which was believed to be atorvastatin related) before receiving rivaroxaban in addition to atorvastatin.
No additional relevant changes of laboratory parameters after rivaroxaban administration were observed in either study, and rivaroxaban had no impact on vital signs or ECG parameters.
Discussion
Anticoagulant drugs are a cornerstone therapy for thromboembolic disorders. Ongoing oral anticoagulant therapy is recommended for stroke prevention in patients with AF who are elderly or at a high risk of stroke. Unless contraindicated, oral anticoagulant therapy is the preferred choice for stroke prophylaxis if one of the following risk factors for stroke is present: prior ischaemic stroke; transient ischaemic attack or systemic embolism; moderate/severely impaired left ventricular systolic function and/or congestive heart failure; or history of hypertension or diabetes mellitus.32,33 Patients with AF – particularly those with risk factors for stroke that would indicate anticoagulant therapy – frequently have comorbidities, some of which require cardiac glycoside or statin therapy with drugs such as digoxin and atorvastatin. Until recently, vitamin K antagonists have been the only oral agents for long-term anticoagulation, but their use is complicated by a narrow therapeutic window, variable dose response and numerous food and drug interactions; these issues necessitate routine coagulation monitoring and dose adjustment. 34 For stroke prophylaxis in patients with nonvalvular AF, rivaroxaban is given as fixed, once-daily doses, and can be used without any requirement for routine coagulation monitoring. 27
The present studies demonstrated that rivaroxaban did not interact with digoxin (a substrate for P-gp) or atorvastatin (a P-gp substrate metabolized via CYP3A4). Steady-state plasma concentrations of rivaroxaban did not affect the pharmacokinetics of digoxin at steady state, which is important because digoxin has a narrow therapeutic window. 28 Steady-state serum concentrations of digoxin had a small effect on the pharmacokinetic characteristics of single-dose rivaroxaban, reducing the AUC and Cmax of rivaroxaban when both drugs were coadministered, compared with single-dose rivaroxaban alone; however, exploratory analyses demonstrated no significant effect of digoxin on the pharmacokinetic parameters of rivaroxaban.
Concomitant daily administration of rivaroxaban, when serum digoxin concentrations were at steady state, slightly increased the AUEC and Emax of Factor Xa activity inhibition associated with rivaroxaban compared with single-dose rivaroxaban administered alone. This effect was predominantly caused by the cumulative effect of repeated daily dosing of rivaroxaban, as demonstrated by the increased inhibition of Factor Xa activity observed at trough plasma concentrations before the final day of administration. A previous study investigating multiple doses of rivaroxaban showed similar results. 25 These observations confirm the predictable pharmacodynamic profile of rivaroxaban, and the exploratory analysis indicated that the interactions between rivaroxaban and digoxin were not significant. Furthermore, coadministration of rivaroxaban with steady-state digoxin had no significant effect on the prolongation of PT induced by rivaroxaban alone. Thus, any pharmacodynamic interaction between rivaroxaban and digoxin in this study setting was not considered to be clinically relevant. The data from the present studies also confirmed the findings of previous in vitro studies, which showed no interaction between rivaroxaban and digoxin. 35
The present results also demonstrated that there were no significant pharmacokinetic or pharmacodynamic interactions between rivaroxaban and atorvastatin. Rivaroxaban showed no effect on the pharmacokinetics of steady-state atorvastatin or its CYP3A4-mediated metabolites. Steady-state plasma concentrations of atorvastatin had no clinically relevant effects on rivaroxaban-induced inhibition of Factor Xa activity or clotting-time prolongation.
Rivaroxaban was well tolerated, alone and in combination with digoxin or atorvastatin, further confirming its favourable tolerability profile.24,25 No evidence of compromised liver function attributable to rivaroxaban or the combination treatments was observed in either study. Psychiatric disorders occurred more frequently in the digoxin–rivaroxaban interaction study than has been shown with rivaroxaban alone (data on file); this is consistent with previously published case reports of the neuropsychiatric effects of digoxin at therapeutic serum concentrations 36 and the summary of the product characteristics of digoxin. 37
A strength of the present studies was that steady-state concentrations of digoxin and atorvastatin were reached before rivaroxaban administration, permitting a superior simulation of the real-world clinical situation. This is in contrast with previous studies of novel oral anticoagulants in which only single doses of digoxin or atorvastatin were administered.38,39 One limitation of these studies is that they were performed in healthy volunteers with no comorbidities. In addition, study drugs were administered over a much shorter period compared with potentially lifelong treatment in patients receiving these drugs in everyday clinical practice.
The results of these studies are consistent with the findings of previous investigations of the influence of atorvastatin and digoxin on the pharmacokinetic and pharmacodynamic characteristics of other anticoagulants in healthy subjects.38–40 Concomitant administration of digoxin or atorvastatin and the direct thrombin inhibitor ximelagatran showed no pharmacokinetic or pharmacodynamic interactions in healthy subjects.38,39 Similarly, coadministration of digoxin and the indirect Factor Xa inhibitor fondaparinux showed no pharmacokinetic interaction. 40
In conclusion, there were no clinically relevant interactions between digoxin and rivaroxaban, or between atorvastatin and rivaroxaban, in this population of healthy male subjects, suggesting that rivaroxaban can be coadministered with either drug. These data confirm previous in vitro studies demonstrating that rivaroxaban does not interact with substrates for the P-gp transport protein and/or CYP3A4.27,35
Footnotes
Acknowledgements
We acknowledge Mark Hillen and Kelly Farrell, who provided editorial support with funding from Bayer HealthCare Pharmaceuticals and Janssen Research & Development LLC.
All authors listed on this manuscript are employees of Bayer Pharma AG.
