Abstract
Oxidative stress plays a crucial role in the emergence of numerous neurodegenerative diseases, with protein accumulation and mitochondrial damage, which result in neurological disorders. To minimize oxidative stress, several defensive mechanisms protect nerve cells by releasing antioxidants such as nuclear erythroid factor2 (Nrf2)-Kelch-like ECH-associated protein1 (Keap1) signaling pathway activation has been proved to be a prospective treatment to reduce oxidative stress and neuroinflammation for protection of neurons in a variety of neurological disorders. In this review, we focus beneficial role of Nrf2 in Alzheimer’s and Parkinson’s diseases. Nrf2 is proved to be a master regulator of antioxidants by releasing over 250 cytoprotective genes aimed at oxidative stress and neuroinflammation. In animal studies Nrf2 activation is proved to improve autophagy, mitochondrial biogenesis, and Suppression of inflammatory cytokinin which protects neuronal cells and inhibit progressive neurodegeneration.
Introduction
The human brain consists of about 10 11 –10 12 neuronal cells which is maintained by neuroglial cells such as oligodendrocytes, astrocytes, and microglia. Recent research proven a strong linkage between inflammation and neuronal cell death; moreover, this would be a reason this scenario is unclear that it would be a direct cause of neurodegeneration. 1 Despite neurological disorders linked with common risk factors, such as oxidative stress, misfolded proteins, aging, and environmental hazards, this exhibits microglial activation and mitochondrial dysfunction in each disease progression by damage to lipids, proteins, and DNA. 2 Misfolded and aggregation proteins are a similar feature in many neuronal disorders even though misfolded proteins are inactive, but their accumulation of misfolded proteins causes cellular stress and endoplasmic reticulum (ER) stress. Unfolded protein response (UPR) generally maintained by ER gets altered and promotes protein misfolding and reduces ER protein levels by proteasomal degradation and autophagy. 3 In the central nervous system (CNS), there is a very limited rate of regeneration for neurons and they are sensitive to oxidative stress so neurodegeneration must be controlled in the brain. Neuronal loss seen mostly by apoptosis or necrosis. In apoptosis, an intrinsic suicide program initiates by cascade for cellular death. In necrosis accidental cell death with uncontrolled release of inflammatory mediators. 4 The most common neurodegenerative disorders are Alzheimer’s disease (AD), which shows marked neuronal loss by an accumulation of misfolded amyloid plaques, which increases with aging in the hippocampus and the frontal cortex region and secondary to chronic inflammation that result from amyloid accumulation links directly with oxidative stress ultimately leads to severe memory loss. 5 In Parkinson’s disease (PD), inflammation results in the loss of dopaminergic neurons in substantia nigra resulting in an unusual hypokinetic movement disorder. However, activation of inflammatory cytokines is essential for the self-defense mechanism of CNS toward foreign antigens but prolonged expression of inflammatory mediators may lead to cell death. 6 The purpose of this review is to focus on two major neurological problems of AD and PD and the neuroprotective effect of nuclear erythroid factor2 (Nrf2) against oxidative stress has been significantly proven in many neurodegenerative animal models. Nrf2 enhances many transcriptional genes of cytoprotective and Phases I and II drug detoxifying enzymes, and mitochondrial pathways in addition to affecting various antioxidant enzymes can also boost the expression of anti-inflammatory mediators, which shows neuroprotective activity. 7 Cortical culture has shown that Nrf2 knockout mice are vulnerable to oxidative stress, and Nrf2 expression enhances neuroprotection in PD and AD through antioxidative response elements (ARE) activation. 8
Nrf2 Structure
Nrf2 was discovered in 1994 at the laboratory of YuetWaiKan and it belongs to the cap “n” collar (CNC) subfamily of basic leucine zipper (bZIP) transcription factors and is encoded by the NFE2L2 gene.9, 10 It includes members with a CNC homology region of 43 amino acids, which links the N-term DNA-binding domain to the family’s DNA-binding specificity.
11
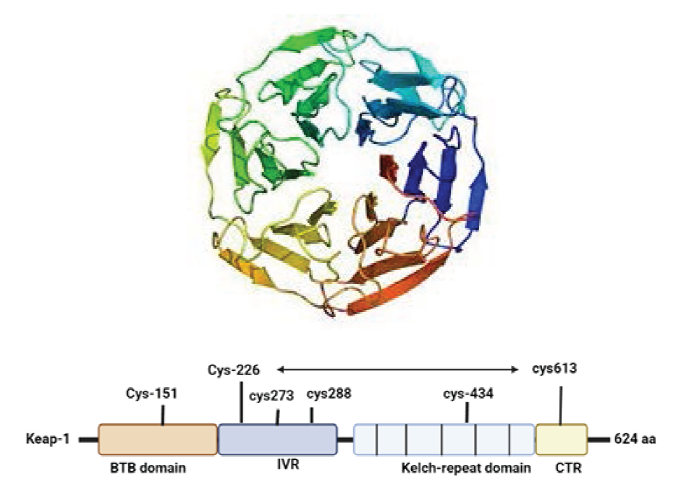
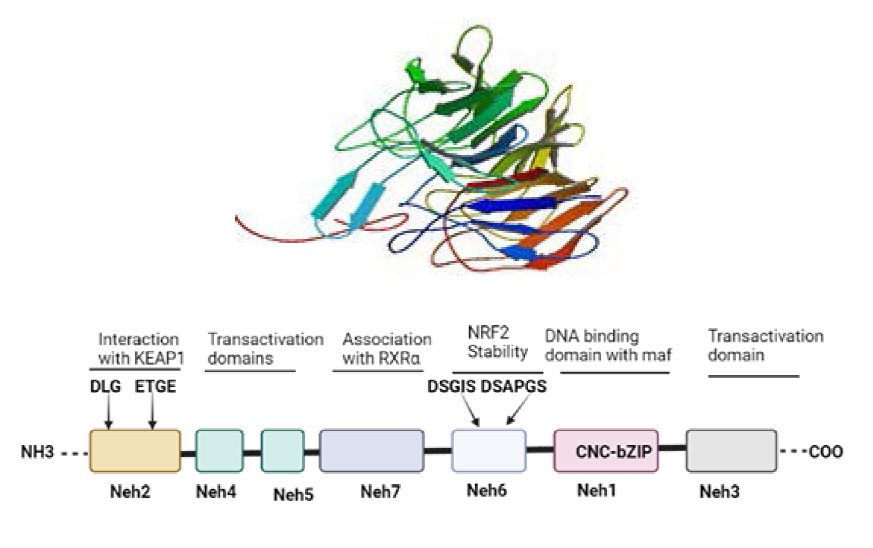
Nrf2 is a complex protein that contains seven functional domains. Neh 1−7 each of which has a specific function, as shown in Figure 1.10, 11 Plafker et al.
16
discovered that Neh1 has a bZIP that forms a dimer from two different monomers with tiny musculoaponeurotic fibrosarcoma protein (
Keap1–Nrf2–ARE Pathway
The Keap1–Nrf2 pathway is a crucial antioxidant signaling pathway against reactive oxygen species, which is one of the major critical players in chronic inflammation associated with PD and AD. The antioxidant enzyme genes glutathione S-transferase (GST) and NAD(P)H quinone oxidoreductase1 (NQO1), as well as heme oxygenase-1 (HO-1), are all enhanced by Nrf2 and induction of an ARE DNA sequence.18, 19 Keap1 suppresses the activity of Nrf2 by binding with Neh2, Keap1 act as an adapter molecule for the Cul3 E3 ligase complex, causing Nrf2 to be degraded via the ubiquitin-proteasome pathway, exposure to electrophiles modifies or reactive oxygen species Keap1 inactivates thereby leading to activation of Nrf2. 20 Causing accumulation of Nrf2 in the nucleus and heterodimerization with small Maf proteins binds to ARE at the regulatory region of the targeted gene21, 22 shown as in Figure 3.
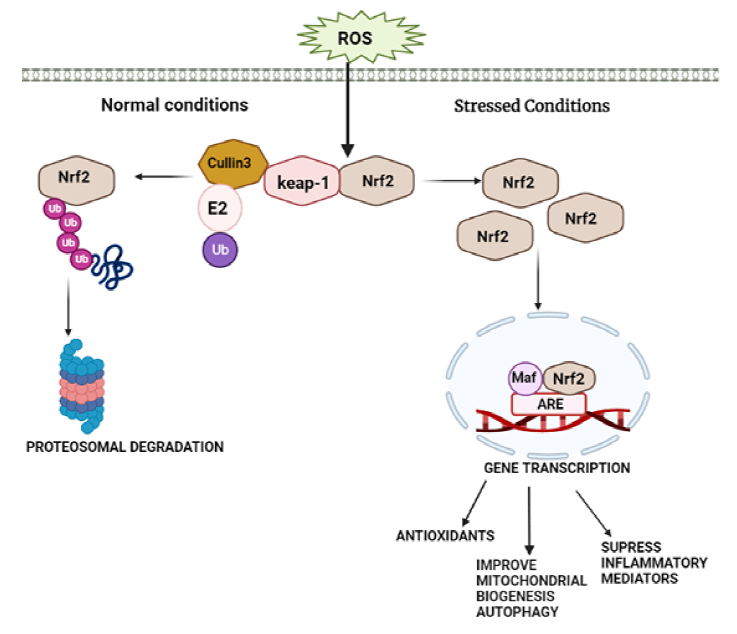
In unstressed conditions, Nrf2 protein level is the low reason being Keap1/Cul3 E3 ubiquitin ligase complex degrades proteasomal proteins.23, 24 Reactive oxygen species modify specific cysteine residue of Keap1 (C257, C273, C288, and C297) causing disassociation of Keap1 and Nrf2 migrate to nucleus causing its accumulation as evidenced in Keap1 knockdown in human cells and KEAP1 deletion in mice, and activation of cytoprotective genes.23–26 Many cytoplasmic proteins that have Keap1 dependent activity that stabilizes Nrf2 by attenuating interaction between Keap1-Nrf2. P62/sequestosome 1 (SQSTM1) also known as Autophagy cargo-adaptor,27–32 dipeptidyl peptidase 3 (DPP3), 31 Wilms tumor gene on the X chromosome (WTX), 33 and Localizer of BRCA2 (PALB2) 34 because they all possess Keap1-interacting region (KIR)-like ETGE motifs, they interact with Nrf2 for Keap1 attachment, allowing Keap1 to be sequestered and Nrf2 to be maintained EDGE suppresses DLG via interacting with the P21 cyclin-dependent kinase p21Cip1/WAF1. 35 BRCA1 promotes stability and activation of Nrf2 36 P300 interacts with Nrf2 and interferes with Keap1-Nrf2 complex formation, 37 which leads to Nrf2 activation and enhance four genes, including Malic enzyme1, Glucose 6-phosphate dehydrogenase, Isocitrate dehydrogenase, and 1,6-phosphogluconate dehydrogenase all of which play a role in the production of NADPH, an antioxidant cofactor and fuel. It moreover regulates cytochrome P450 oxidoreductase activity. Neutrophils and monocytes have the highest levels of NRF2 among blood cells. Nrf2 levels are high in astrocytes. Furthermore, microglia, a kind of monocyte, have larger levels of Nrf2 than neurons.37–40 Nrf2 reduces inflammation in three ways modifying redox metabolism, interacting with (NF-KB), and directly regulating pro-inflammatory mediators responsible for neurodegeneration in PD and AD.40, 41
Nrf2 and Mitochondria
Mitochondria consist of membranes which are separating them from other cellular organelles was thought to be responsible only for energy generation due to their genetic autonomy, however, studies suggest that a transduction network in the cytosol circuit of mitochondria maintains cellular equilibrium.
42
Nrf2 controls mitochondrial biogenesis, and autophagy both directly or indirectly,
43
Role of Nrf2 in Mitochondrial Dysfunctions
The aging mechanism is linked with mitochondrial failure and ROS. The discovery of neurotoxic compound 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) is an accidental human exposure to induced mitochondrial failure was the first proof that mitochondrial dysfunction plays a role in PD, further studies are needed to understand the mechanism of MPTP induced dopaminergic neurotoxicity by converting into 1-methyl-4-phenylpyridinium (MPP+) in brain, damages complex I in mitochondria resulting in mitochondrial ROS and oxidative stress leads to release of cytochrome c reductase in the mitochondrial respiratory chain further lead to mitochondrial failure. 49 Low concentrations of complex I subunit and its activity have been found in postoperative PD individual midbrain biopsies and patient-derived platelets. 50 As a result, the experimental shreds of evidence show that aging reduces cellular protective properties by weakening the Nrf2/ARE pathway’s role. Nrf2-Keap1 protein complex functions as a cellular electrochemical detector, regulating antioxidant gene transcription to maintain redox homeostasis. The Nrf2/ARE activity is influenced by both aging and neurodegenerative disorders. In multiple tissues, a meta-analysis of PD and AD identified 54 impacted genes, 31 of which contain ARE. GSR1 promotes glutathione reduction by employing the reducing power of nicotinamide adenine dinucleotide diphosphate as a subunit of glutamate-cysteine ligase (NADPH).49, 51 Nrf2 regulates the transcription of aldehyde dehydrogenases, which are essential in alcohol elimination and reduce NADP+ to NADPH. DMF and its bioactive metabolite, monomethylfumarate (MMF), have been demonstrated to accelerate mitochondrial oxidative phosphorylation and biogenesis by activating Nrf2. 52 As a result, the Nrf2/ARE system influences both mitochondrial and non-mitochondrial metabolic pathways, indicating therapeutic potential in neurodegenerative diseases.
Nrf2 Expression in Neurodegenerative Diseases
Unlike many tissues, Nrf2 is released at elevated levels in the liver, kidneys, and lungs. Moreover, it is released at a greater level in gastrointestinal tissues (esophagus, stomach, small intestine, and large intestine), studies conducted by Chan et al. 9 reported that the expression of Nrf2 is seen in the lateral ventricle and its mid-wall, as well as in the choroid plexus of the fourth ventricle in the brain, Nrf2 is also observed in the nasal cavity tissue involved in smell, thyroid, submandibular glands, and the brown fat. 5
In AD, Nrf2 is primarily expressed in cytoplasmic hippocampus neurons; it does not translocate to the nucleus; a biochemical extract of the frontal cortex confirmed decreased levels of nuclear Nrf2 in AD. In the nuclei of AD patients, neither neurons nor astrocytes showed substantial Nrf2 staining. The Nrf2 pathway is most likely dysfunctional in AD hippocampal neurons; nevertheless, in PD, Nrf2 is more numerous in dopamine neurons than in SNpC, and dopaminergic Nrf2 exhibits neuronal sensitivity to increased oxidative stress. 6
Role of Nrf2/ARE Pathway in AD
In the last few decades, many studies have discovered different features of Nrf2 expression and relevant antioxidant genes in AD models, a considerable increase in nuclear Nrf2 levels was seen in investigations. The existence of cytoplasmic confinement in the AD patient’s hippocampus was discovered as indicating a reduction in levels of the Nrf2 gene. 53 In AD hippocampal brain areas, NQO1 activity was upregulated. Similarly, the activities of catalase and SOD1 in the frontal lobe and temporal cortex of the AD brain appeared to be diminished. 54 Mishaps in the Nrf2 pathway are associated with peripheral tissues as a reflection of process going on in the brain and spinal cord. Reduced cognitive impairments, oxidative damage, and inflammatory processes in AD models, and counteract against a mediated inflammation-induced, multiple mechanisms involved in the activation of Nrf2 have been identified in AD. The possibility of targeting Nrf2-regulatory proteins in the therapy of AD is demonstrated in this section, 55 p62, a polyubiquitination protein, can promote Nrf2 activity by identifying Keap1 as a target for degradation. Different researchers have discovered that there is a link among Keap1, p62, and Nrf2, which indicates a reduction of protein p62 results decreased Keap1 breakdown and as a consequence, increase in the release of Nrf2, therefore, Aβ insertion into the rat’s hippocampal region enhanced catabolic proteins including LC3-II and beclin1, as well as Keap1, while reduced p62 and Nrf2 in the cortex and hippocampus. 56 In the cortex region of Alzheimer’s brains, however, mRNA and gene expression of p62, and also Nrf2 target genes, were found to be elevated. 57
Several investigations demonstrated that Nrf2 activity is decreased in 3xTg-AD mouse model begins to develop an age-dependent and transformative neurodegenerative genetic makeup, which includes an age-dependent plaque and tangles in the brain, similar to AD patients, due to three protein abnormalities in presenilin-1, amyloid precursor protein, and microtubule-linked tau which never interact in human AD kinship forms. 58 From 3 to 5 months of age, the 3xTg-AD mice reported lower Glutathione and vitamin E levels, but also enhanced GSH-Px activities. Furthermore, 3-month-old PBMC from 3xTg-AD exhibited elevated p(Ser40)Nrf2 which corresponded to Nrf2 genetic translation Sensor same as human MCI PBMCs. 59 In the AD animal model PS1V97LTg, with a change in the PSEN1 gene, the Nrf2 inducer sulforaphane promoted enhanced Nrf2 activation in the brain and the Nrf2-ARE pathway alleviated oxidative imbalance amyloidopathy and improves cognitive performance. When compared to mice of the mutant strain studies shows amyloid proteins build and aggregation of tau protein into neurofibrillary with Nrf2-leakage (AT-Nrf2-KO) that shows an abnormal increase in the number of astrocytes and accumulation of microglial cells as a reaction to injury (AT-Nrf2-WT) see Figure 4. 60
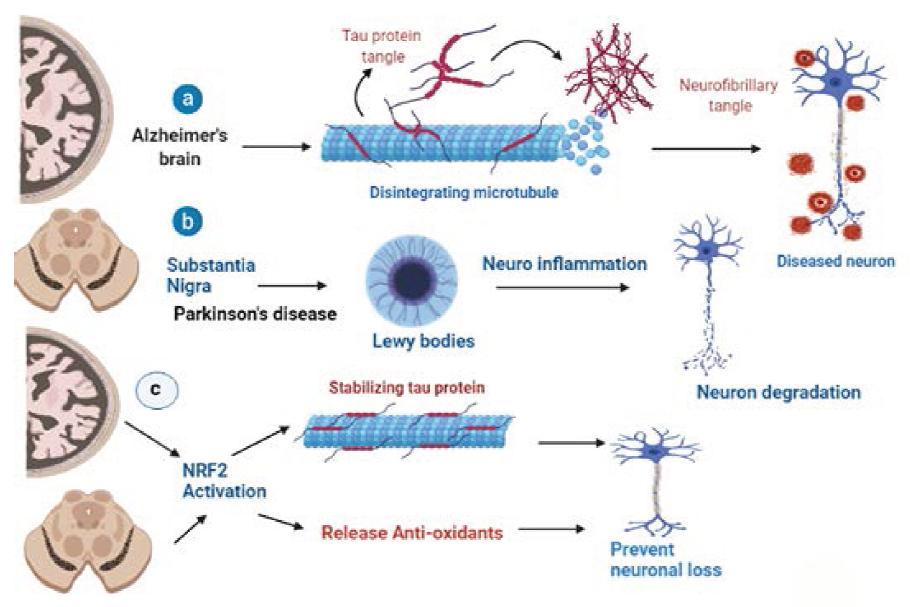
Role of Nrf2/ARE Pathway in PD
Ramsey et al. 10 investigated higher concentrations of Nrf2 in the nucleus of human PD substantial nigra neurons. Multiple investigations have correlated the Nrf2 pathway to various PD models. Interestingly, results from individual samples show that Nrf2 nuclear translocation significantly improved.61–65 Suggesting a higher level of transcriptional regulation than it was in age-matched subjects GSH concentrations were also observed to be decreased in the substantial nigra of individuals with PD brains, whereas HO-1, NQO1 levels were shown to be elevated in astrocytes, endothelial cells, and dopamine neurons from PD autopsy brains. 66 In PD brains, forensic researches reveal that Nrf2 nuclear translocation is significantly greater, along with a higher concentration in certain Nrf2-regulated proteins. α-Syn was stereotaxically delivered in the ventral midbrain of Nrf2/mice using adeno-associated viral vectors to properly understand the purpose of Nrf2 in PD. 67 In Nrf2/mice treated with MPTP or 6-OHDA, which promote oxidative stress and are frequently used to imitate PD in rats, identical results were obtained. Additionally, transplantation astrocytes Nrf2 overexpression into the striatum of wild-type mice reduced sensitivity to 6-OHDA and MPTP during 6-OHDA treatment, and parallel values were demonstrated in cortical neurons from Nrf2+/− animals uncontrolled release of Nrf2 in astrocytes with mice uncontrolled expressing human mutation SYN (hSYNA53T) in neurons resulted in delayed motor dysfunction, lower Synuclin protein buildup, and attenuate ROS along with diminished microglia in the spinal cord see Figure 4. 68 Mild regular physical activity was demonstrated beneficial in mice treated with 6-OHDA through Nrf2 neuron protective action, boosting mitochondrial biogenesis acceleration and limiting Parkinsonism progress. 69 As a result, impaired mitochondrial mitophagy, efficiency, and biogenesis are major pathogenic characteristics of PD, and Nrf2 is a signaling pathway factor that controls mitochondrial biogenesis checks and homeostasis. As a result, there are many shreds of evidence of the role of Nrf2 in the pathogenesis of PD, indicating that increasing the Nrf2/ARE pathway as an effective PD treatment is of interest. 70 As a result, increased p62 availability, which might potentially lead to increased Nrf2 target gene expression, could be a promising therapy for PD. Despite this, there have been no investigations on the usage of p62 oscillators and their influence on Nrf2 transmission in PD. Glycogen synthase kinase 3 (GSK-3) kinase will suppress Nrf2, leading to reduced Nrf2 activity due to increased degradation. Furthermore, there is evidence that GSK-3 activation is modified in PD. Intracellular GSK-3 levels were upregulated with Syn in Lewy bodies from PD human brains in the brainstem and upper parietal lobe, as well as the striatal and inferior frontal lobe. 71 GSK-3 inhibition, PI3K/Akt stimulation, and elevated Nrf2 expression of target genes, PC12 cells, and SH-SY5Y cells reduced oxidative imbalance caused by the cytotoxic agents of MPTP, MPP+, and inhibited apoptosis. Berberine an isoquinoline compound exhibiting PI3K-activating action, also attenuated 6-OHDA-induced dopaminergic neuron loss and motor movement disturbances in zebrafish via enhancing PI3K/Akt activation and Nrf2 activation. 72 Overall, in multiple PD models, manipulation of the PI3K/Akt/GSK-3 pathway and restriction of the p38 MAPK pathway seemed to be the most viable target pathways, leading to Nrf2 activation and subsequent stress response. Minimizing neurotoxicity and promoting the Nrf2/ARE pathway should be an area to explore in PD treatment, based on studies in PD postmortem tissues. 73
Conclusion
ROS or superoxides react with lipids, proteins, and DNA resulting in neuron cell death, which is the primary cause of neurological diseases including PD and AD. The parameters like protein buildup, mitochondrial dysfunction, and proteasomes dysfunction is also reasons that may cause cell death and neurological diseases. Nrf2 pathway activation can help to reduce oxidative stress and maintain mitochondrial biogenesis while also increasing autophagy, which helps to destroy misfolded proteins. In this review, it was focused on the importance of Nrf2 pathway activation that has been proved with scientific studies in neurological diseases. The study suggests that the PI3K/Akt/GSK-3 pathway, which looks to be well-studied for AD and PD therapies, is involved in the positive regulation of Nrf2 activity. Restriction of the NF-kB pathway must also be investigated as a prospective AD and PD therapeutic goal. The evidence clearly states that drugs that activate Nrf2, including those that have been proven to elevate Nrf2-related genes, have a protective effect on AD and PD models.
Footnotes
Abbreviations
Nrf2: Nuclear erythroid factor or Nuclear respiratory factor
Keap-1: Kelch-like ECH associating protein
BTB domain: Broad complex tram track &Bric a brack
IVR: The intervening region
DGR: Double glycine repeats
zip: Basic leucine zipper CNC
Maf: Masculo-aponeuroticfibrosarcoma oncogene
ARE: Antioxidative response elements
UBA: Ubiquitin association
GSK-3: Glycogen synthase Kinase 3
SOD2: Superoxide dismutase enzyme
OPA1: Optic atrophy 1
PINK 1: Phosphatase and tension homolog PTEN, induced kinase
Acknowledgment
The authors would like to thank JSS College of Pharmacy, Ooty, and JSS Academy of Higher Education and Research, Mysuru.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
