Abstract
Objectives
Oxidative stress induces chronic inflammatory diseases in aerobic organisms, and antioxidants from plants represent an efficient strategy to prevent this condition. Morin hydrate (MH), a bioactive flavonoid, has a wide range of pharmacological properties, including anti-inflammatory and anti-oxidant. This study evaluated the protective effects of MH on lipoteichoic acid (LTA)-induced inflammation in RAW 264.7 macrophages by testing the main oxidative and inflammatory biomarkers and also investigating the molecular pathways involved.
Methods
The antioxidant and anti-inflammatory effects of MH were evaluated in a cell-free system and RAW264.7 cells. Quantitative real-time PCR (RT-qPCR) and assay kits were used to measure the expression of nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase 1 (HO-1) mRNA, as well as the activity of antioxidant enzymes. The effects of MH on LTA-induced inducible nitric oxide synthase (iNOS), IL-1β, and TNF-α mRNA and protein expression were also evaluated by RT-qPCR and Western blotting.
Results
MH reduced DPPH and ABTS radicals in a cell-free system and LTA-induced ROS and NO production in RAW264.7 cells. MH upregulated Nrf2 and HO-1 mRNA expression and reversed LTA-mediated reduction of antioxidant enzymes, at a high concentration of 20 µM pretreated cells. MH also effectively attenuated LTA-induced iNOS, IL-1β, and TNF-α mRNA and protein expression, and these effects were reversed by ML385.
Conclusions
The study found that the Nrf2/HO-1 played role in the inhibition of LTA-induced oxidative stress in macrophages by MH. This study may consider to be a promising induced macrophage-targeted strategy via regulating anti-oxidative defense to control inflammatory-related disease.
Introduction
Oxidative stress is a condition caused by an imbalance between oxidants and antioxidants in the body, resulting from exposure to harmful stimuli.
1
This imbalance leads to the overproduction of reactive oxygen species (ROS), which can cause oxidative damage to cells and trigger an inflammatory response.2,3 During the inflammatory response, there is an increase in blood leukocyte influx, oxidative burst, cytokine release, and induction of various enzyme activities. While reactive species play a role in host defense, an overproduction of ROS and reactive nitrogen species (RNS) can also cause damage at inflammatory sites. Additionally, ROS and RNS serve as triggers or signaling molecules that influence protein and lipid kinases and phosphatases, membrane receptors, ion channels, and transcription factors involved in regulating the expression of key cytokines that play a role in inflammation regulation.
4
Lipoteichoic acid (LTA), a crucial component of the cell walls of Gram-positive bacteria such as
Inflammation is a complex physiological response that plays a crucial role in the body’s defense against harmful stimuli, such as pathogens and tissue injury. 14 However, dysregulated or chronic inflammation can contribute to the pathogenesis of various inflammatory diseases, including arthritis, cardiovascular diseases, neurodegenerative disorders, and cancer. 14 These inflammatory diseases are characterized by an abnormal and prolonged inflammatory response, leading to tissue damage and dysfunction. The dysfunctions involve intricate molecular and cellular mechanisms that contribute to the development and progression of these diseases. Imbalances between pro-inflammatory and anti-inflammatory mediators, such as ROS and RNS, further contribute to the detrimental effects of dysregulated inflammation. 15 Understanding the underlying mechanisms of dysregulated inflammation is crucial for the development of targeted therapeutic strategies. This may involve investigating the cross-talk between inflammation and other cellular processes, such as oxidative stress, mitochondrial dysfunction, and epigenetic modifications. 15 By unraveling these complexities, novel approaches for the management and treatment of inflammatory diseases can be explored, leading to improved patient outcomes and quality of life.
Flavonoids are a diverse group of compounds found in many fruits, vegetables, and plants. They are further divided into five sub-categories: flavones, flavanols, flavanones, flavonols, and anthocyanidins. These plant-derived polyphenols possess a wide range of biological properties, including antioxidant and anti-inflammatory effects.
16
Morin hydrate (MH), with a molecular formula of C15H10O7·H2O and a molecular weight of 302.24 MH exhibits radical scavenging activity against DPPH and ABTS radicals. (a) Chemical structure of MH; (b) DPPH and (c) ABTS radical scavenging activity of MH. The values presented are the means ± SEM of four independent experiments (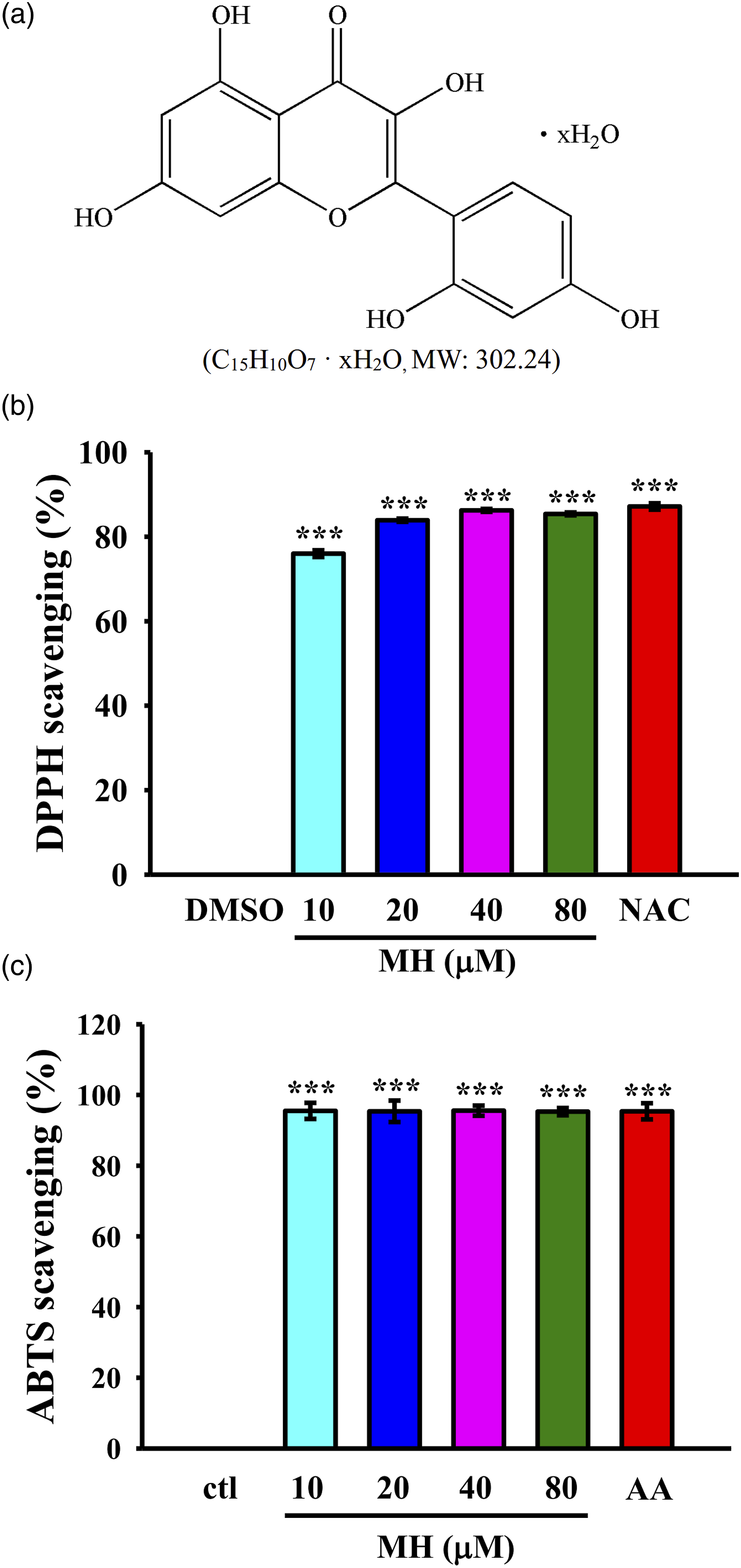
Methods
Chemicals
Morin hydrate (MH), N-acetyl-
2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay
The DPPH scavenging activity of MH was measured following a previous study with slight modifications. 19 Briefly, MH was mixed with DPPH-methanol (400 μM) at concentrations of 10-80 μM and incubated for 30 min. The absorbance at 517 nm was then measured using a Synergy H1 microplate reader (Bio-Tek, VT). N-Acetyl cystein (NAC) was used as a reference control. The DPPH scavenging activity was calculated as ((A2 - A1)/A2) x 100%, where A1 represents the absorbance of MH and A2 represents the absorbance of DPPH.
Total antioxidant activity
The ABTS radical cation was generated by reacting a 7 mM ABTS stock solution and 2.45 mM potassium persulfate. The reaction mixture was allowed to incubate for 12-16 h in the dark at room temperature until the reaction was completed and the absorbance stabilized. 20 The absorbance of the ABTS solution was then adjusted to 0.70 (±0.02) by diluting it with water at room temperature. To test the sample, 1 mL of the ABTS solution was mixed with 10 μL of the test sample which ranged from 0.05 to 10 mg/mL. The absorbance was measured at 734 nm after 6 min. Ascorbic acid (AA) was used as a reference compound for comparison.
Cell culture
The RAW 264.7 cell line (catalog number BCRC 60001) was obtained from the Bioresource Collection and Research Center (BCRC) in Hsinchu, Taiwan. The cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum, 100 units/ml penicillin, and 100 units/ml streptomycin. Cells maintenance was carried out in a humidified atmosphere with a temperature of 37°C and a carbon dioxide-rich environment.
Cell viability (MTT) assay
The colorimetric MTT assay was performed to assess cell viability, following a previously described method. 21
Reactive oxygen species (ROS) assay
The levels of intracellular ROS were determined using DCFH-DA with minor modification. 20 Following pretreatment, RAW264.7 cells were collected by centrifugation at 1200 xg for 10 min at 4°C, washed with PBS three times, and then incubated at 37°C for 20 min with 10 uM DCFH-DA, which is converted to the fluorescent compound 2’,7’-dichlorofluorescin (DCF) within the cells. The fluorescence intensity of the cells was measured using a Synergy H1 microplate reader (BioTek, VT) with an excitation/emission wavelength of 485/528 nm.
Determination of NO production
The supernatant of RAW 264.7 cells was evaluated for nitrite production using a previously described method. 21 In a 24-well plate, cells (1 × 105 cells/well) were treated with MH or 0.1% DMSO for 1 h, followed by stimulation with LTA (5 µg/mL) for 24 h. Nitrite levels were determined by mixing 100 µl of cell culture extract with 100 µl of Griess reagent (1% sulfanilamide and 0.1% naphthyl ethylenediamine in 2.5% phosphoric acid). The absorbance of each sample was measured at 540 nm using a multi-well scanning spectrophotometer. The concentrations of nitrite were determined by comparing the absorbance values with standard solutions of sodium nitrite, prepared in a culture medium and assayed under similar conditions.
Calcium release
The intracellular Ca2+ levels in RAW264.7 cells were measured with slight modifications based on a previous study. 22 Prior to the addition of LTA, the cells were pretreated with 10 and 20 μM MH for 1 h. Subsequently, Fura-2/AM (2 μM) was added, and the cells were incubated at 37°C for 1 h. The fluorescence signals were monitored using an excitation wavelength of 340/380 nm and an emission wavelength of 510 nm.
Analysis of antioxidant enzymes
Antioxidant enzymes, such as superoxide dismutase (SOD) and catalase (CAT), were analyzed in RAW cells. The activities of SOD and CAT were measured using their respective assay kits (#706002 and #707002). The activity of SOD was measured at 450 nm, while the activity of CAT was measured at 340 nm using a microplate reader.
The Glutathione assay kit (#703002) was used to quantify reduced glutathione (GSH). This assay employs an enzymatic recycling method with glutathione reductase to measure the GSH levels. GSH reacts with DTNB (5,5’-dithio-bis-2-(nitrobenzoic acid) to produce a yellow-colored compound called TNB (5-thio-2-nitrobenzoic acid). The rate of TNB production is directly proportional to GSH formation, and it was measured at 412 nm using a plate reader. All assays were performed on the supernatant from the four groups: control, DMSO + LTA, 10 μM MH + LTA, and 20 μM MH + LTA cells. This experiment was carried out according to the experimental protocols provided by the respective assay kit manufacturers.
Quantitative real-time PCR (RT-qPCR)
Primer sequences.

Western blotting
RAW 264.7 cells were harvested and lysed using an ice-cold lysis buffer. 21 The protein samples, containing an estimated total protein of 50 μg each, were separated by 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and subsequently transferred onto PVDF membranes (0.45 μm). The membranes were then blocked with 5% skimmed milk in TBST buffer (10 mM Tris-base, 100 mM NaCl, and 0.01% Tween 20) for 30 min to minimize non-specific binding. Following blocking, the membranes were incubated with specific primary antibodies (anti-iNOS and anti-TNF-α antibodies, Proteintech) targeting the proteins of interest for 2 h at room temperature. After incubation, the membranes were probed with HRP-conjugated secondary antibodies (donkey anti-rabbit IgG or sheep anti-mouse IgG) for 1 h. Protein bands were visualized and quantified using the Biolight Windows Application, V2000.01 (Bio-Profil, Vilber Lourmat, France), allowing for the determination of protein band densities.
Statistical analysis
Each experiment was conducted independently at least four times. The data are presented as the means ± standard error (SEM). Statistical analysis was performed using one-way analysis of variance (one-way ANOVA), and significant differences among the groups were compared using the Newman-Keuls method.
23
A
Results
Spectrophotometric assays for antioxidant capacity of MH
Assays based on DPPH and ABTS radicals are widely used spectrophotometric methods for assessing the antioxidant capacity of pure molecules. As shown in Figure 1(b) and (c), MH demonstrated an increase in DPPH (control (ctl): 0%, NAC: 87.2 ± 0.8%, 10 μM: 76 ± 0.8%, 20 μM: 84 ± 0.4%, 40 μM: 86 ± 0.3%, 80 μM: 85 ± 0.3%) and ABTS (ctl: 0%, ascorbic acid (AA): 95.4 ± 2.3%, 10 μM: 95.5 ± 2.3%, 20 μM: 95.4 ± 3.0%, 40 μM: 95.5 ± 1.4%, 80 μM: 95.3 ± 1.0%) scavenging activity. The result indicated that MH exhibited DPPH radical scavenging activity comparable to that of NAC at all concentrations tested. Similarly, MH displayed robust ABTS scavenging activity, which was similar to that of AA, a well-known antioxidant. These findings suggest that MH possesses the ability to neutralize free radicals, potentially providing cellular protection against oxidative damage. Therefore, MH holds promise as an antioxidant agent.
MH does not exert cytotoxic effects on LTA-induced macrophages at low concentrations
The study aimed to evaluate the effect of MH on cell viability using a concentration of LTA at 5 μg/mL, as reported in a previous study (Figure 2(a) and (b)).
21
The results demonstrated that MH at concentrations ranging from 10 to 40 μM did not significantly affect the viability of RAW264.7 cells induced by LTA after 24 h. However, a decrease in viability was observed at concentrations of 80 and 160 μM (Figure 2(c)). The viability results were further supported by cell morphology images (Figure 2(c) (i)-(vii)), which confirmed that concentrations up to 40 µM of MH did not exert cytotoxic effects on macrophages. Therefore, concentrations of 10 and 20 μM of MH were considered suitable and safe for subsequent experiments. The effect of LTA and MH on nitric oxide production and cell viability in RAW 264.7 macrophages. (a) Cells were treated with various concentrations of LTA to measure nitrite using a Griess reagent. (b) Cell viability was assessed using an MTT assay. (c) Cells were treated with 0.1% DMSO or MH and then with LTA (5 µg/mL) to assess the effects on cell morphology ((i): ctl; (ii): DMSO+LTA; (iii): 10 μM+LTA; (iv): 20 μM+LTA; (v): 40 μM+LTA; (vi): 80 μM+LTA; (vii): 160 μM+LTA) and viability. The values presented are the means ± SEM of four independent experiments (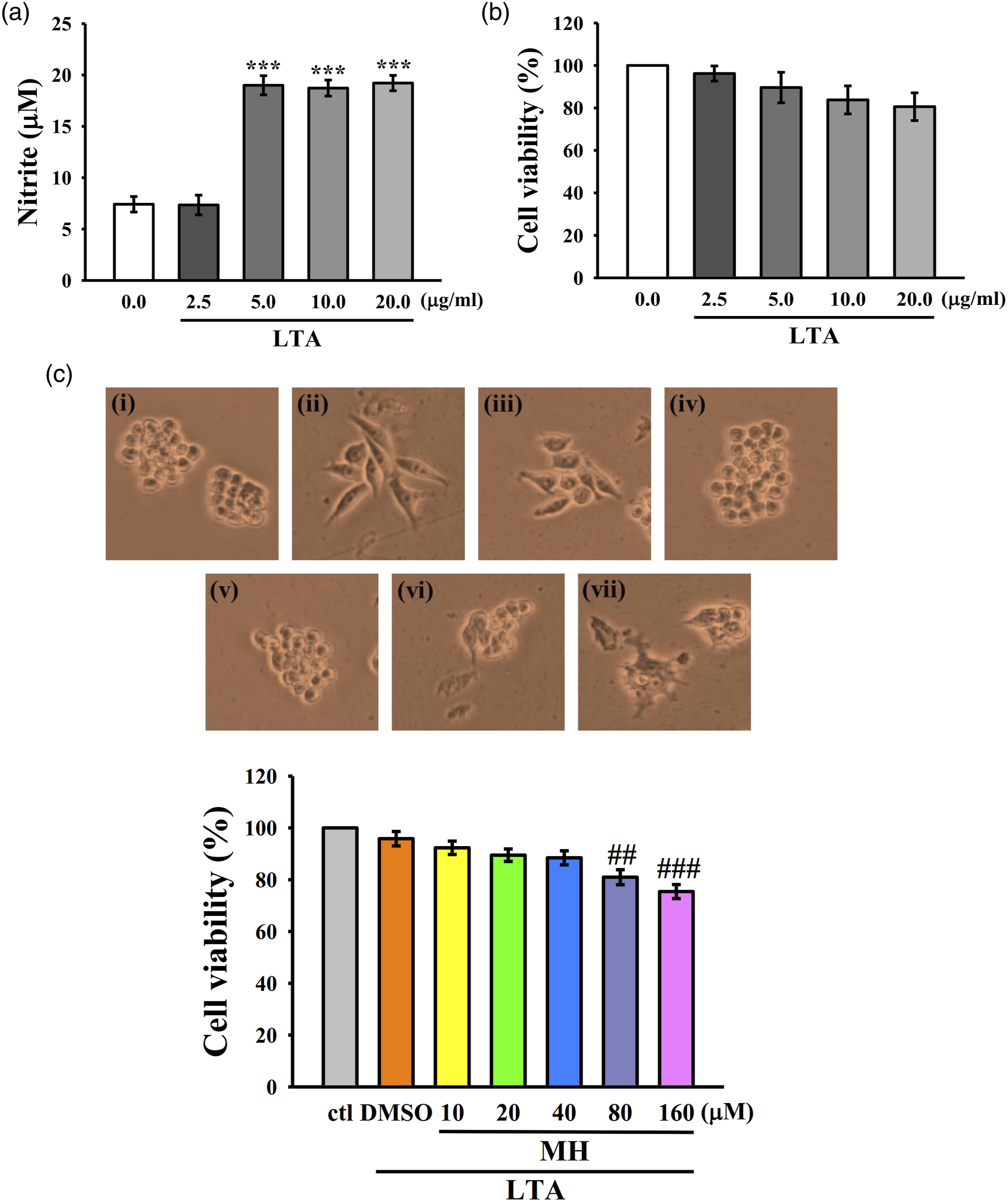
Antioxidative and anti-inflammatory effects of MH on macrophages exposed to LTA
ROS and NO are reactive molecules with a short half-life that can interact with various targets both inside and outside cells, leading to diverse physiological and cellular functions.
24
Following a 24 h exposure to LTA, macrophages exhibited a significant increase in intracellular ROS levels. However, pretreatment with 10 and 20 µM of MH effectively mitigated the LTA-induced rise in intracellular ROS production (Figure 3(a)). Furthermore, MH demonstrated a significant inhibitory effect on NO production in RAW 264.7 cells stimulated by LTA for 24 h (Figure 3(b)). These findings suggest that MH has the potential to modulate inflammatory and oxidative signaling pathways associated with excessive NO production triggered by LTA. Additionally, the results depicted in Figure 3(c) reveal that MH effectively suppressed calcium release in RAW 264.7 cells treated with LTA, indicating a potential regulatory role of MH in calcium-related pathways implicated in inflammatory diseases. Taken together, these results indicate that MH may possess antioxidative and anti-inflammatory properties in macrophages. Effects of MH on (a) ROS formation, (b) NO production, and (c) intracellular calcium release. Cells were treated with MH (10 and 20 μM) and LTA for 24 h. The values presented are the means ± SEM of four independent experiments (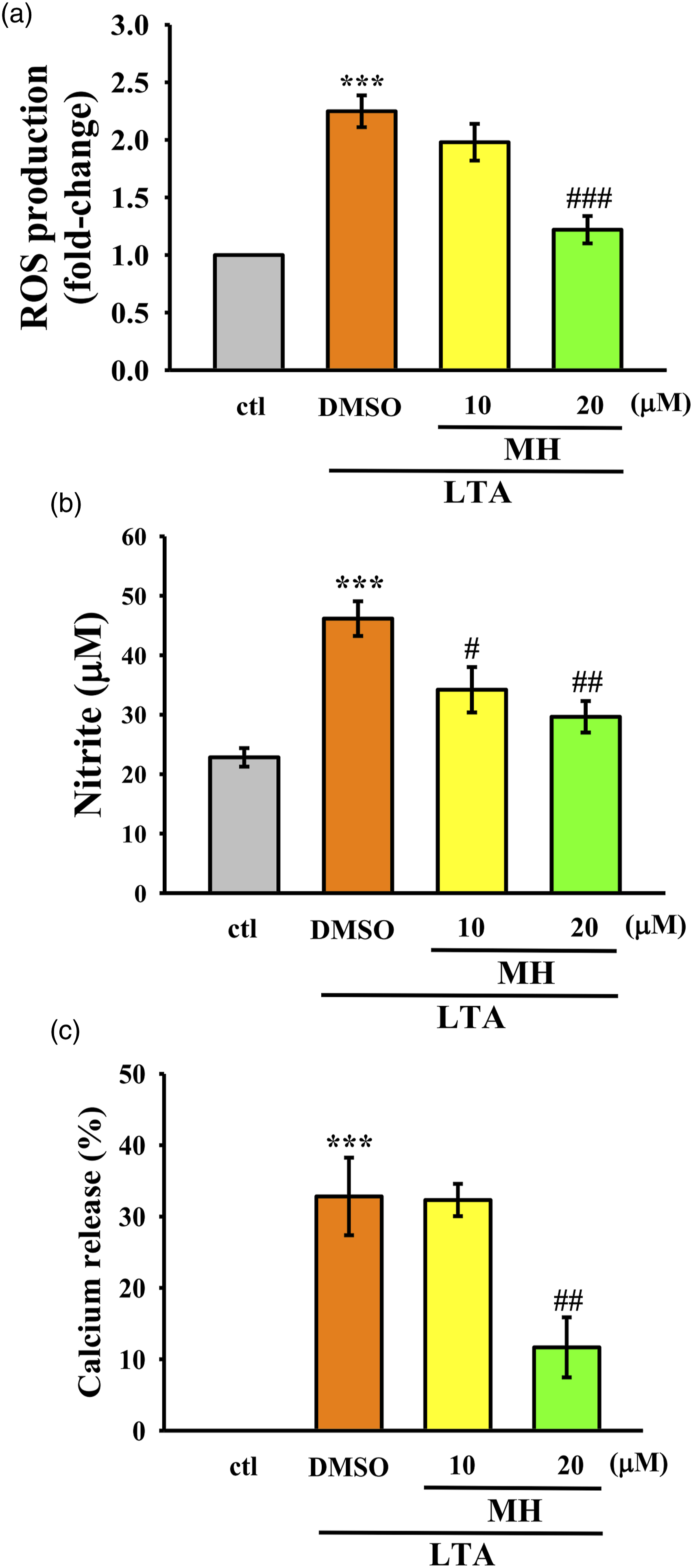
Activation of Nrf2/HO-1 expression by MH in RAW264.7 cells.
To investigate the activation of Nrf2/HO-1 signaling by MH, RAW264.7 cells were treated with 10 and 20 μM MH followed by LTA administration. The mRNA expression levels of Nrf2 and HO-1 were assessed using RT-qPCR. First, the expression levels were evaluated in cells treated with MH alone to determine if MH alone could induce expression (Figure 4(a) and (b)). Subsequently, mRNA expression levels were measured in LTA-stimulated RAW264.7 cells to observe any potential upregulation (Figure 4(c) and (d)). The results revealed no significant increase in Nrf2 mRNA expression in cells treated with MH alone compared to the control group. However, a significant elevation in the mRNA expression of both Nrf2 and HO-1 compared to LTA treatment alone. These findings indicate that MH may have a selective effect on HO-1 expression in RAW264.7 cells. Moreover, the results suggest that MH may enhance the antioxidant activity of macrophages stimulated by LTA by upregulating the expression of Nrf2 and HO-1 expression. The effect of MH on the Nrf2/HO-1 mRNA in LTA-stimulated RAW 264.7 cells. The cells were treated with two different concentrations of MH (10 and 20 μM), either alone (a)-(b) or followed by LTA exposure (c)-(d). The mRNA expression levels of (a),(c) Nrf2 and (b),(d) HO-1 were evaluated using RT-qPCR. The values presented are the means ± SEM of four independent experiments (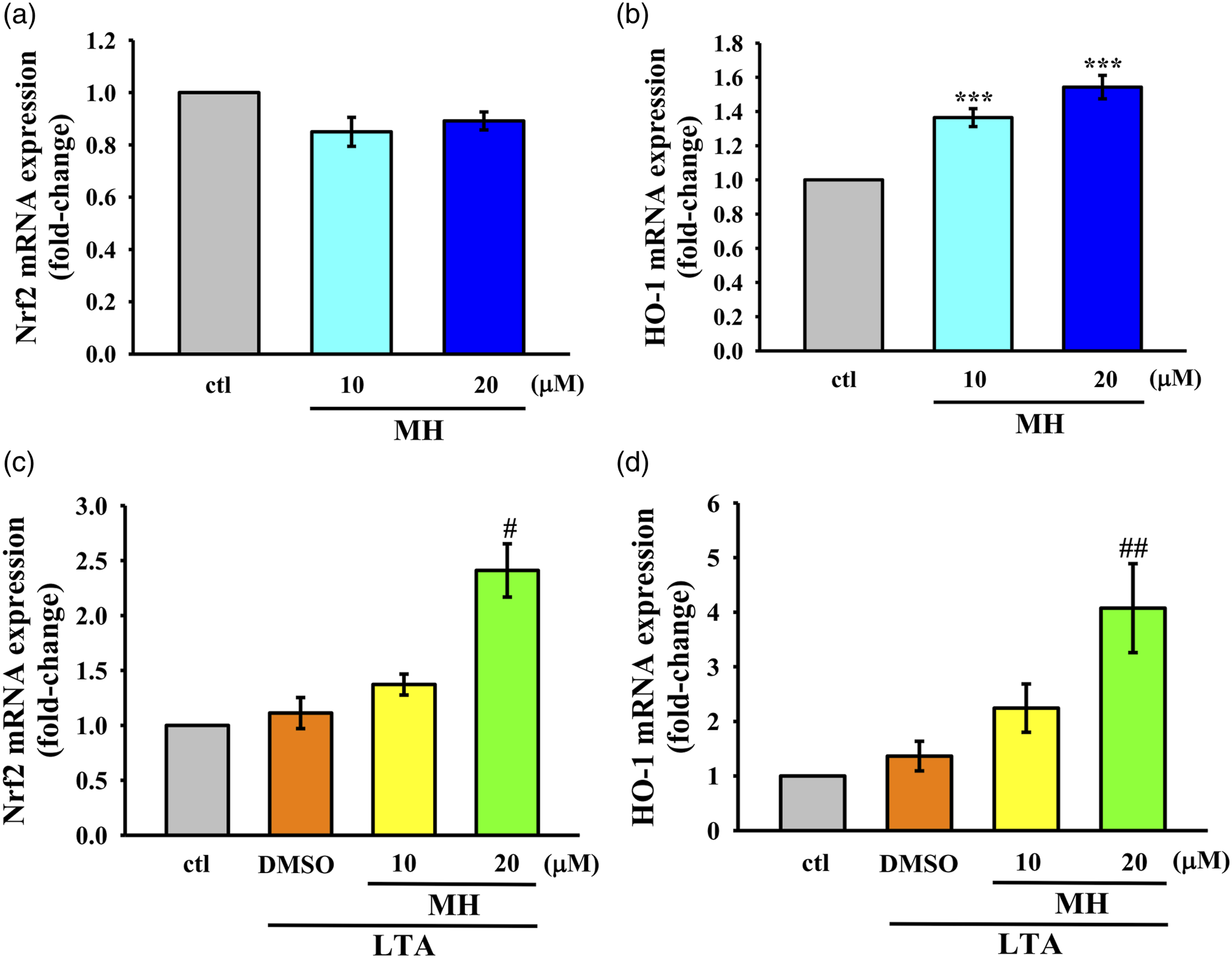
MH activates antioxidant enzyme expression in macrophages
The Nrf2/HO-1 is a crucial antioxidant pathway involved in suppressing oxidative stress and regulating anti-inflammatory responses.
25
It is known that Nrf2 induces the expression of various antioxidative enzymes, including catalase (CAT), superoxide dismutase (SOD), and glutathione (GSH), which are essential for scavenging free radicals and mitigating oxidative damage.
26
Consistent with this, the results presented in Figure 5(a)-(c) demonstrate that treatment with MH leads to increased activities of CAT and SOD, as well as elevated levels of GSH, in LTA induced RAW cells, These findings suggest that MH has the ability to induce the production of antioxidative defense molecules via Modulating the Nrf2/HO-1 pathway. The impact of MH on CAT, SOD, and GSH in LTA-stimulated RAW264.7 cells. Cells were treated with MH for 1 h followed by LTA for 24 h. (a)-(c) The enzyme activity of (a) CAT, (b) SOD, and the level of (c) GSH were detected as mentioned in the Materials and Methods section. The values presented are the means ± SEM of four independent experiments (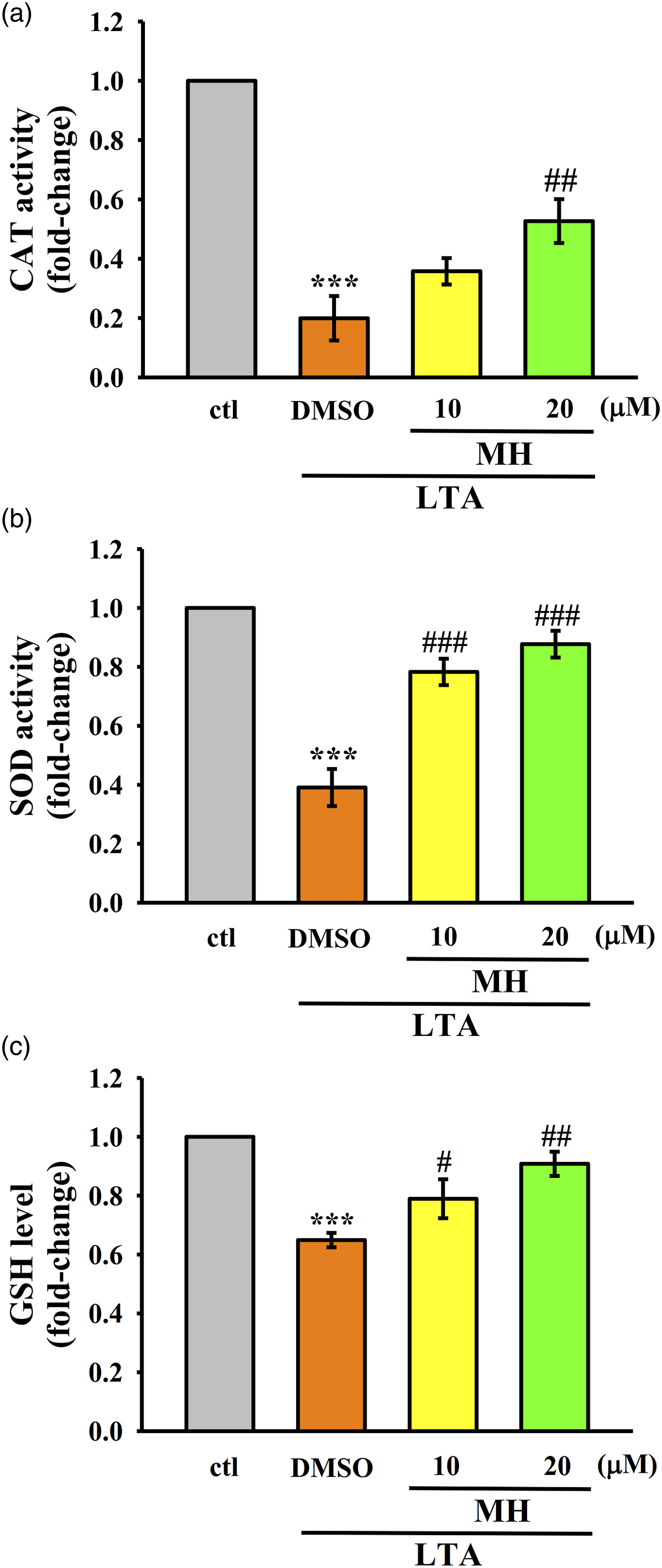
MH reduces the expression of pro-inflammatory genes and proteins in LTA-stimulated RAW264.7 cells via Nrf2/HO-1 pathway
MH was observed to reduce the expression of pro-inflammatory genes iNOS, TNF-α, and IL-1β, as well as the protein expression of iNOS and TNF-α in LTA-stimulated RAW264.7 cells, as depicted in Figure 6(a)-(f). Notably, the anti-inflammatory effects of MH were reversed when co-treated with the Nrf2 inhibitor ML385, suggesting that MH inhibits inflammation by enhancing antioxidative activity through the Nrf2/HO-1 pathway. Effect of MH on the mRNA and protein expression of iNOS, TNF-α, and IL-1β in LTA-stimulated RAW264.7 cells. Cells were treated with MH for 1 h followed by LTA for 24 h. (a)-(c) mRNA expression levels of (a) iNOS, (b) TNF-α, and (c) IL-1β were detected as mentioned in Materials and Methods. (d)-(f) Protein expression levels of (d),(e) iNOS and (d),(f) TNF-α were evaluated. The values presented are the means ± SEM of four independent experiments (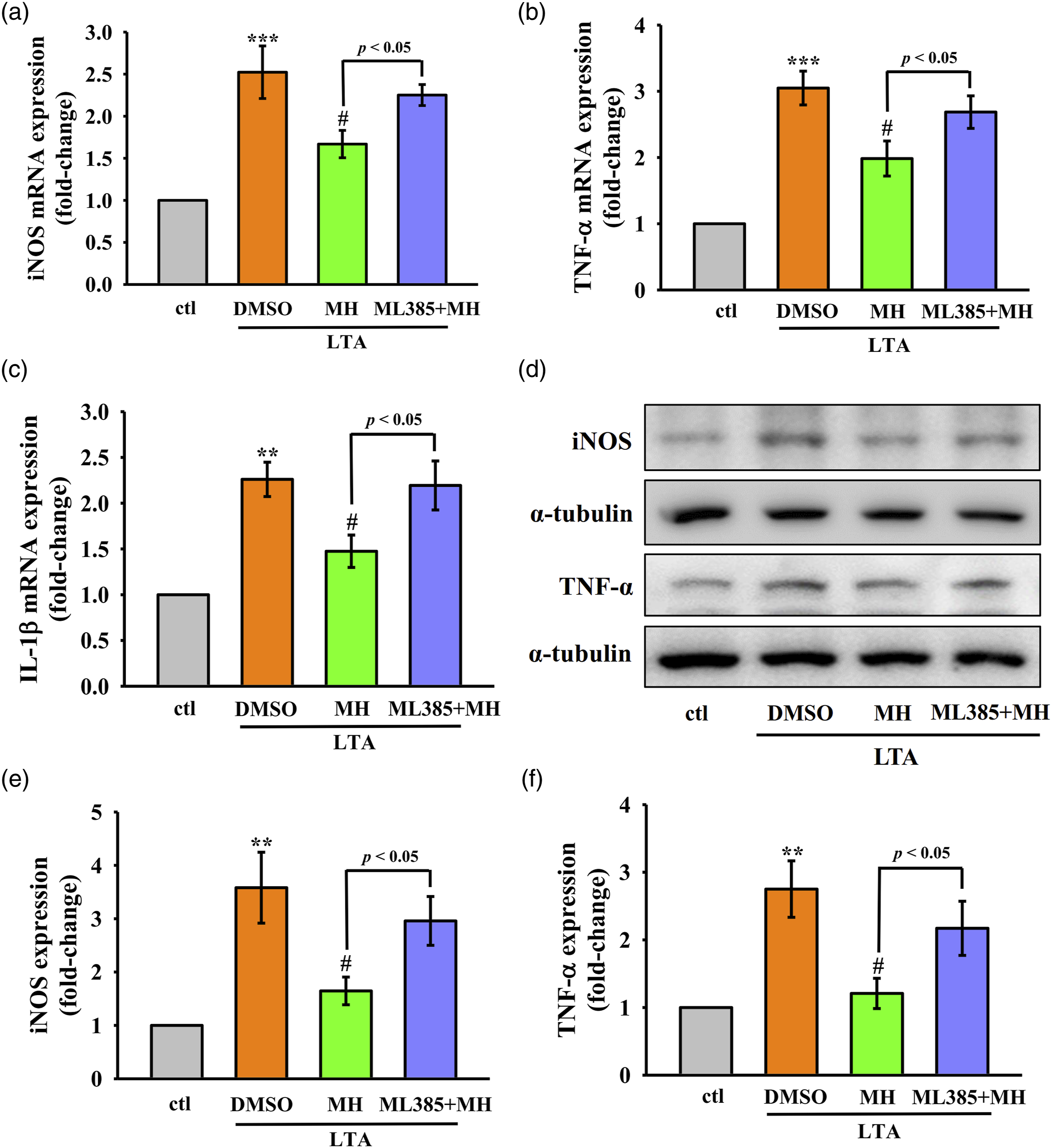
Discussion
This study aimed to investigate the antioxidant and anti-inflammatory effects of MH on LTA-induced macrophages. The results suggest that MH exhibits potential as an antioxidant agent by enhancing scavenging activity against DPPH and ABTS radicals. Additionally, MH effectively reduces the LTA-induced increase in intracellular ROS production and inhibits NO production in RAW 264.7 cells stimulated by LTA. Furthermore, the study demonstrates that MH activates the Nrf2/HO-1 pathway, leading to the induction anti-oxidative enzymes and the downregulation of pro-inflammatory gene expression in LTA-stimulated RAW264.7 cells (Figure 7). A schematic diagram describing the potential anti-oxidant mechanism of MH in LTA-induced RAW 264.7 macrophages. MH increases the anti-oxidant reaction and alleviates the inflammatory response in LTA induced RAW 264.7 macrophages via activation of Nrf2/HO-1 signaling pathways and antioxidant enzyme, and decreasing pro-inflammatory cytokines and iNOS dependent NO production.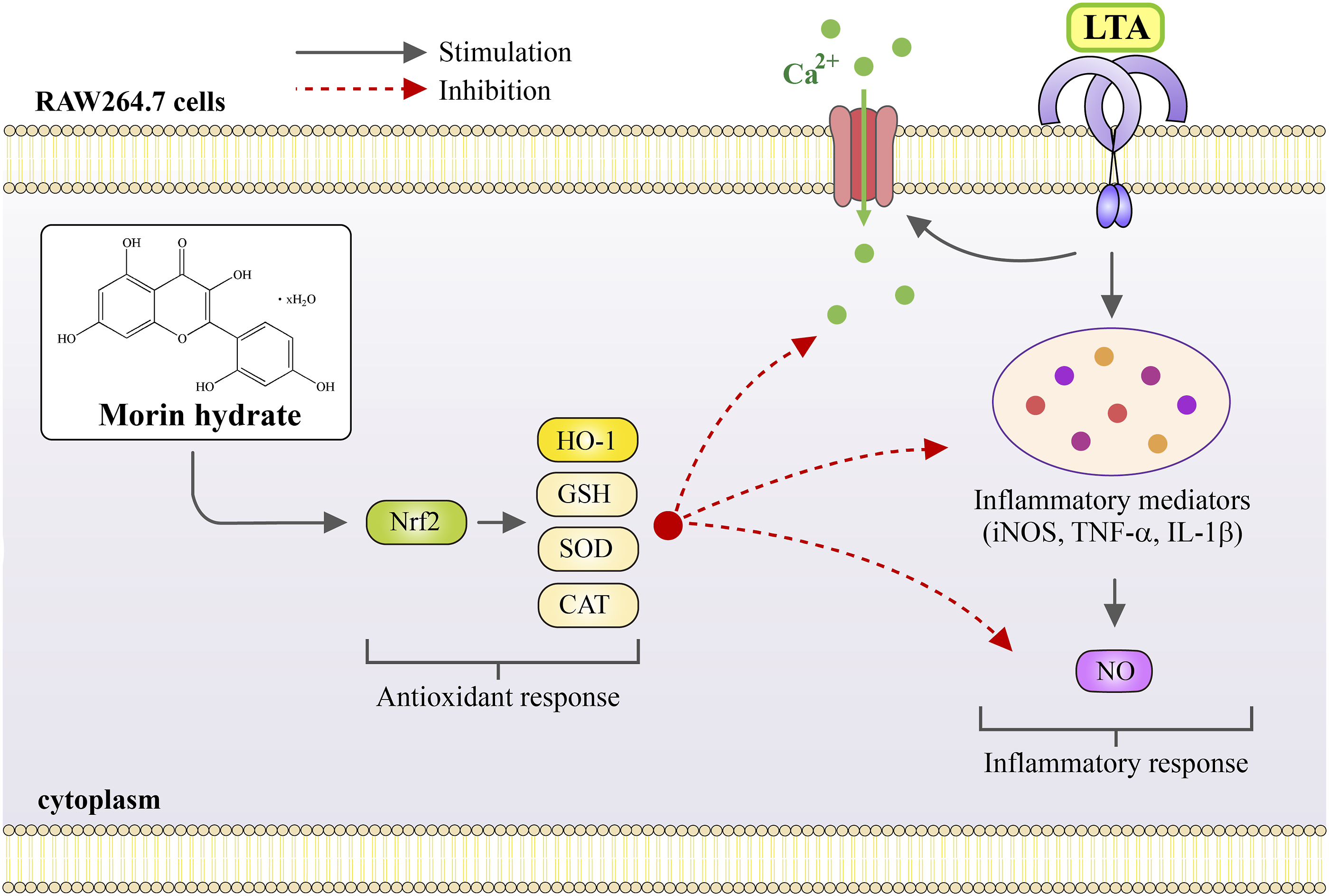
The hydrogen-donating ability of antioxidants is crucial for assessing their physiological functionality. Antioxidant compounds play a significant role in scavenging radicals in various domains, including biological systems, food, and pharmaceuticals.27,28 The DPPH and ABTS assays are commonly employed to evaluate the antioxidant capacity of pure substances due to their simplicity and speed in assessing. 28 In this study, the antioxidant activity of MH was investigated using these two widely employed radical scavenging methods. The results demonstrated that MH exhibited significant scavenging activity against both DPPH and ABTS radicals compared to the control. MH showed notable free radical scavenger ability, similar to NAC and AA, which are recognized as potent free radical scavengers. This finding is consistent with precious studies reporting favorable DPPH and ABTS antioxidant activity of other flavonoid compounds.29,30 NO is a highly reactive free radical and a critical sencondary messenger involved in mediating inflammatory responses.30,31 However, treatment with MH alongside LTA resulted in reduced NO production in RAW264.7 cells. These findings suggest that the antioxidant activity of MH may be attributed to its ability to donate hydrogen atoms to free radicals, thereby neutralizing their reactivity. Furthermore, MH appears to reduce NO generation in inflammatory macrophage cells, indicating its potential anti-inflammatory effects.
The morphological transformation of RAW 264.7 cells provides valuable insights into the relationship between MH concentrations and cell viability. Before LTA treatment, cells show a rounded phenotype, indicating immaturity. LTA exposure induces adhesion, growth, and proliferation, promoting cell maturation and widespread distribution. 29 This morphological response confirms the ability of LTA to modify the cells and elicit an inflammatory reaction which is consistent with the previous study. 21 The viability assessment, combined with the examination of cell morphology in Figure 2(c) (i)-(vii), demonstrates that MH concentrations up to 40 µM do not induce cytotoxic effects on macrophages. The phenomenon observed, where a high dose of MH increases scavenging activity but decreases cell viability in macrophages, can be attributed to a complex interplay of factors. At this concentration, MH exhibits potent antioxidant activity, leading to increased scavenging of ROS in the extracellular environment. However, the same high dose of MH can also exert cytotoxic effects on macrophages, potentially overwhelming cellular antioxidant defense mechanisms and disrupting the cellular redox balance. 31 This imbalance, along with potential interference with intracellular signaling pathways and molecular targets, may induce oxidative damage, mitochondrial dysfunction, DNA damage, and altered gene expression, ultimately compromising cell viability. Careful dose optimization is crucial in therapeutic applications of MH, considering both its beneficial antioxidant properties and the potential cytotoxic effects at high concentrations. Therefore, these findings suggest that MH does not trigger an inflammatory response, and macrophages treated with MH remain in an immature monocyte-like state until stimulated by LTA. Overall, these results highlight the favorable safety profile of MH within the tested concentration range and support its potential as a promising therapeutic candidate for inflammatory diseases.
ROS, NO, and calcium signaling play important roles in inflammation and the immune response. 32 In normal levels, they contribute to defensive mechanisms against pathogens and regulation of cellular activity.33,34 However, excessive production of ROS and NO can damage cells, and dysregulated calcium release can contribute to disease states like autoimmunity or chronic inflammation. 35 As an antioxidant and anti-inflammatory compound, morin reduced ROS and NO levels, thereby decreasing oxidative damage and inflammation in vivo.36,37 Previous studies have demonstrated that LTA moderates the activation of ROS, iNOS, and subsequent NO production.21,38,39 To elucidate the mechanism of the protective effect of MH on LTA-induced oxidative damage and inflammation in macrophages, a fluorescent probe, DCF-DA, was used to detect intracellular ROS levels. Treatment with MH was able to suppress LTA-induced ROS and NO production in macrophages.
Free radicals, such as ROS, have the ability to trigger calcium release from intracellular stores including the endoplasmic reticulum (ER) and mitochondria. 40 These free radicals can modify the redox state of calcium channels and pumps, thereby altering their function and leading to increased calcium release. 40 Consequently, elevated levels of calcium can further stimulate the production of free radicals, creating a cycle of oxidative stress and cellular damage. 40 The interaction between free radicals and calcium release has been implicated in a wide range of physiological and pathological processes, including neurodegenerative diseases, cancer, and cardiovascular disorders. 41 In a cell-based study, it has been observed that flavonoids can restrict calcium release from the ER, thereby reducing its availability for pro-inflammatory signaling. 42 Similarly, the results of this study suggest that MH can decrease ROS, NO, and calcium release induced by LTA. This indicates that the influences of MH on ROS, NO, and calcium may be attributed to its targeting enzymes such as NADPH oxidases, nitric oxide synthases, and ryanodine receptors that are involved in the production or regulation these factors. By reducing the activity of specific enzymes/channels, MH could potentially mitigate inflammation and associated conditions in vivo. 43 In summary, these findings suggest that treatment with MH may alleviate inflammatory diseases by reducing oxidative stress and its related inflammatory signaling.
Nrf2 and HO-1 are important proteins involved in defending cells against oxidative stress and inflammation. Under normal condition, Nrf2 maintains the expression of HO-1 and other protective enzymes, contributing to support cellular homeostasis and antioxidant capacity. The regular induction of Nrf2/HO-1 indicates a healthy basal antioxidant capacity. Research has shown that Nrf2 acts as a transcription factor that can prevent the transcriptional upregulation of pro-inflammatory cytokines induced by LPS. It also helps reduce the production of ROS through its antioxidant pathway.25,43 On the other hand, HO-1 expression plays a critical role in protecting cells during oxidative stress and inflammation. It can be rapidly induced by various oxidative-inducing agents, including LPS.10,44 When cells are exposed to stressors, the induction of Nrf2/HO-1 helps mitigate damage. However, excessive or prolonged induction of these proteins may indicate high cell stress and impaired function. In this study, it was observed that MH inhibited the elevated ROS production induced by LTA and increased the mRNA expression of Nrf2 and HO-1 compared to the LTA group. This increase in Nrf2 and HO-1 expression may contribute to the inhibition of LTA-induced NO production. The results suggest that MH reduces ROS production while simultaneously enhancing the expression of Nrf2 and HO-1, which is consistent with the concept that high levels of Nrf2 and HO-1 can inhibit the production of NO induced by LTA. 45 Therefore, MH may exert anti-inflammatory and antioxidant effects by activating the Nrf2/HO-1 antioxidant pathway.
In order to mitigate cellular damage caused by oxidative stress, the human body relies on a robust antioxidant defense system that regulates various endogenous enzymatic and non-enzymatic antioxidants, including CAT, SOD, and GSH. 46 These antioxidant proteins play a crucial role in maintaining cellular redox balance and protecting against oxidative stress-induced harm. 46 The Nrf2/HO-1 pathway, a well-established antioxidant pathway, holds significant importance in cellular protection against oxidative stress and inflammation. The pathway is governed by the transcription factor Nrf2, which translocates into the nucleus and binds to the antioxidant response element (ARE) present in the promoter region of target genes, including CAT, SOD, and GSH. CAT is an antioxidant enzyme, effectively converts hydrogen peroxide into water and oxygen, thereby neutralizing oxidative species and safeguarding cells against oxidative damage. 46 SOD, another antioxidant enzyme, facilitates the conversion of superoxide radicals to hydrogen peroxide and oxygen, mitigating the detrimental effects of oxidative stress. 46 GSH, a tripeptide antioxidant, exhibits critical antioxidant activity by scavenging ROS and contributing to the reduction of oxidative damage. 46 The present study provides evidence that MH the capability to reverse the imbalance in antioxidant enzymes induced by LTA in macrophages. This is evidenced by the observed increase in the activities and levels of CAT, SOD and GSH following MH treatment which has similar results to other flavonoids. 47 These findings support the notion that MH has the potential in regulating the imbalances in antioxidant enzyme induced by LTA, thereby contributing to cellular protection against oxidative stress.
The Nrf2/HO-1 pathway has been extensively studied and shown to play a critical role in modulating inflammatory responses by blocking the expression and activity of inflammatory mediators. Studies have demonstrated that activation of the Nrf2/HO-1 pathway leads to a reduction in the production of iNOS, TNF-α, and IL-1β, which are key inflammatory mediators involved in various inflammatory diseases.48,49 This inhibitory effect on inflammatory mediators has been observed in both in vitro and in vivo studies. Moreover, research using Nrf2-knockout mice has revealed that the absence of Nrf2 results in an increased expression of inflammatory mediators, 50 highlighting the crucial role of Nrf2 in suppressing inflammation. This suggests that the Nrf2/HO-1 pathway acts as a negative regulator of the inflammatory response, and its activation is associated with the downregulation of pro-inflammatory molecules. In the present study, it was found that MH could inhibit the mRNA and protein expression of inflammatory mediators, including iNOS, TNF-α, and IL-1β. We have additionally observed that MH exhibits a suppressive effect on IL-6 protein expression (Supplemental Figure 1). This indicates that MH possesses anti-inflammatory properties by modulating the expression of these key inflammatory molecules. Furthermore, this study used ML385, an Nrf2 inhibitor, to investigate the involvement of the Nrf2/HO-1 pathway in the anti-inflammatory effect of MH as used in previous study. 21 The results also demonstrated that ML385 reversed the inhibitory effect of MH on the mRNA expression of inflammatory mediators, suggesting that MH’s anti-inflammatory action is mediated through the activation of the Nrf2/HO-1 pathway. These findings provide further support for the notion that the Nrf2/HO-1 pathway plays a crucial role in regulating inflammation. Activation of this pathway can lead to the upregulation of antioxidant defense systems, resulting in the neutralization of excess ROS and a reduction in oxidative damage and inflammation.
By modulating the Nrf2/HO-1 pathway, MH may exert its anti-inflammatory effects by restoring the balance between oxidative stress and antioxidant defense, ultimately suppressing inflammatory responses. However, it is important to recognize that there may exist other molecular mechanisms through which MH exerts its actions, which warrant further exploration. To obtain a more comprehensive understanding of MH’s therapeutic potential, future investigations should encompass the use of in vivo models to assess its effects under physiological conditions.
Conclusion
This innovative study reveals the potential of MH as a therapeutic approach for oxidative stress-induced chronic inflammatory diseases. MH demonstrates impressive antioxidant and anti-inflammatory effects by modulating the Nrf2/HO-1 signaling pathway in LTA-induced RAW 264.7 macrophages. It effectively reduces ROS and NO production, promotes the expression of Nrf2 and HO-1 mRNA, restores antioxidant enzyme activity, and inhibits the expression of key inflammatory mediators. The comprehensive evaluation of MH’s properties and unraveling of underlying molecular mechanisms highlight its potential as a targeted strategy for managing inflammatory-related diseases. These findings contribute to the growing scientific knowledge on plant-derived antioxidants as promising therapeutic interventions against oxidative stress and inflammation. Further research is needed to explore the clinical translatability and broader applications of MH in preventing and treating chronic inflammatory conditions.
Supplemental Material
Supplemental Material - Morin hydrate suppresses lipoteichoic acid-induced oxidative stress-mediated inflammatory events in macrophages via augmenting Nrf2/HO-1 and antioxidant defense molecules
Supplemental Material for Morin hydrate suppresses lipoteichoic acid-induced oxidative stress-mediated inflammatory events in macrophages via augmenting Nrf2/HO-1 and antioxidant defense molecules by Cheng-Ying Hsieh, Thanasekaran Jayakumar, Kao-Chang Lin, Ting-Lin Yen, Chih-Wei Hsia, Wei-Chieh Huang, Joen-Rong Sheu, and Chih-Hsuan Hsia in European Journal of Inflammation.
Footnotes
Acknowledgements
This work was supported by the Ministry of Science and Technology of Taiwan (MOST 110-2320-B-341-001-MY3 and MOST 111-2320-B-038-036-MY3), Chi Mei Medical Center-Taipei Medical University (109CM-TMU-05), and Shin Kong Wu Ho-Su Memorial Hospital (2023SKHAND006).
Author contributions
CYH, JRS and CHH designed the work. TJ and CHH wrote the paper. KCL, TLY, CWH and WCH carried out the experiments and performed data analyses. All authors have read and agreed to the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology of Taiwan (MOST 110-2320-B-341-001-MY3 and MOST 111-2320-B-038-036-MY3), Chi Mei Medical Center-Taipei Medical University (109CM-TMU-05), and Shin Kong Wu Ho-Su Memorial Hospital (2023SKHAND006) were financially supported to this work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
