Abstract
Objective
To find out whether curcumin can be effective in the treatment of traumatic brain injury (TBI).
Methods
A comprehensive and systematic literature search in the PubMed electronic database was performed. Descriptive statistics were used to evaluate the data obtained. The results were presented as frequency and percentage (%) or amount.
Results
Two clinical trials investigated curcumin for the treatment of TBI. One study tested curcumin in living mammalian subjects using an amyloLipid nanovesicle. In three studies, curcumin was investigated together with the drug delivery system for the treatment of TBI.
Conclusion
Drug delivery systems prepared with nanomaterials may have a potential therapeutic effect in treating TBI by increasing neuroprotection because they can penetrate the central nervous system more rapidly.
Introduction
Traumatic brain injury (TBI) is classified as open and closed. Closed head injuries are diffuse axonal injuries that occur after acceleration-deceleration trauma, where the dura is intact, and micro-damages are seen in the deep cerebral region. 1 Open head injuries include gunshot wounds, stab wounds, and falls, where the integrity of the dura is lost, or cerebral tissue damage is observed. 2 No radical and effective treatment for TBI is currently available. 3
TBIs have variable prognoses based on their severity and are classified as mild, moderate, and severe. 4 Open and closed head traumas cause primary and secondary cerebral tissue damage in TBIs via different physiopathological mechanisms. Primary brain injury is caused by cerebral contusion, intracranial hematoma, diffuse axonal injury, and vectorial impact because of acceleration-deceleration. Hypotension, which develops immediately after the trauma, causes neural cell membrane damage, cell membrane destruction after increased intracranial pressure and decreased cerebral perfusion pressure, and this stage is called the secondary injury phase. 5
Primary damage occurs at the time of injury. Secondary injury is the organism’s response to primary mechanisms, and both may be focal or diffuse. Headache, dizziness, incontinence, sensory disturbance, paralysis, spasticity, dysphagia, and/or epileptic seizures are observed in the long term after TBI.6–9
Brain ischemia often occurs after TBI and deaths are common.10, 11 Cerebrovascular autoregulation and CO2 reactivity, which are vital mechanisms that provide cerebral blood flow, are partially or completely impaired following TBI. 12 Chronic depolarization of vascular smooth muscle cells and cerebral vasospasm resulting in TBI are also seen in most cases. 13 Oxygen and glucose consumption are affected in the brain, 14 and the amount of phosphocreatine and ATP in the tissue decreases. 15 An imbalance between cerebral oxygen delivery and cerebral oxygen consumption of the cerebral tissue is an apparent indicator of TBIs, leading to hypoxia in the brain tissue. In addition, excitatory amino acids increase the amount of glutamate, and increased glutamate stimulates receptors in neurons and astrocytes, causing Ca2+, Na+, and K+ flow.16, 17
This event triggers catabolic processes, causing cell damage and disrupting the blood–brain barrier (BBB). Deterioration of antioxidant systems after head trauma causes an increase in free oxygen radicals. In addition, peroxidation, protein oxidation, disruption of electron transport in mitochondria, and DNA damage develop in cells and vascular structures. 18
This results in necrosis or apoptosis. Vasogenic edema because of disruption of the BBB and cytotoxic edema because of intracellular fluid accumulation may occur. Various cytokines, prostaglandins, and other components secreted activate polymorphonuclear leukocytes. Leukocytes stimulate adhesion molecules in the endothelium and secrete intracellular adhesion molecules and vascular adhesion molecules. This inflammatory response creates scar tissue within days and months. 19
Osmotic diuretics such as mannitol or hypertonic saline are administered as first line treatment to reduce the pressure in the head after TBI because of secondary cerebral damage as first-line treatment. If intracranial pressure remains above 20 mm Hg despite osmotic diuretic therapy, surgical interventions such as cerebrospinal fluid drainage, decompressive craniotomy, barbiturate coma, a high-dose phenobarbital administration, or hypothermia can be used.
Along with the treatment for coagulation disorder, antiepileptic drugs such as phenytoin, carbamazepine, and phenobarbital can be used to prevent seizures, especially in the early period of TBI. While agents such as paracetamol are used as antipyretics, antihyperglycemic agents are used for glycemic control. After the patient is discharged, the spectrum of mental and physical functional loss includes a wide range from motor and sensory losses in the extremities to cognitive and behavioral psychology and social defects. Current treatment modalities do not radically treat brain damage. There are no pre- or post-TBI treatment options available. 3
Studies have focused on pharmacomolecular trials for the treatment of TBI which has a high mortality and morbidity and requires long-term treatment and care. In these studies, molecules that can pass through BBB and have the potential to treat TBI, as well as drug delivery systems that can be effective in the central nervous system have been investigated.20, 21 When designing drug delivery systems, many different pharmacological and pharmaceutical agents have been tested alone or using nanomaterials. Studies on nanoparticle-mediated drug delivery systems, especially in cerebral tissue, have provided insight into a highly promising strategy for the treatment of TBIs. 22
Many different drug molecules have been tried in the treatment of such cases. However, BBB penetration poses an important problem for the absorption and distribution of the drugs used. The treatment of TBI with pharmacological agents has been unsuccessful because of the selective permeability of drugs across the BBB. In addition, while designing drug molecules that can pass the BBB or drug delivery systems for TBI, studies have focused on nanomaterials that can pass through BBB or drug molecules absorbed into nanomaterials. 23
Curcumin (C21H20O6) is a pigment in Indian saffron, also known as turmeric. It has been suggested that it may have a potential therapeutic effect on the treatment of TBI. 24 Curcumin is effective against complications observed after TBI by modulating molecular signaling pathways related to oxidative stress, inflammation, apoptosis, and autophagy. However, further studies are needed to determine all the mechanisms involved in the pathogenesis of TBI. 25
This systematic review focuses on the treatment of TBI with curcumin targeted to damaged cerebral tissues using appropriate drug delivery systems.
Materials and Methods
Search Criteria
A comprehensive and systematic literature search of electronic databases, including PubMed, was performed. 26 A combination of keywords was used to retrieve studies broadly associated with the topic of interest. The search criteria were as follows: “traumatic brain injury,” “blood–brain barrier,” “curcumin,” “drug delivery system,” “nano-drug,” “nanomaterials,” and “nano micelles.” All the relevant studies performed until April 1, 2022, were retrieved. Protocol prepared by Lijmer et al. 27 was used to assess and analyze the quality of the selected studies.
Eligibility Criteria
Studies in which curcumin was tested in combination with the drug delivery system were selected for the treatment of TBI.
Data Collection
The studies included were independently selected by the authors. The risk of selection bias because of potential masking was also investigated. All studies were reviewed by the senior authors to ensure accuracy. If there was a disagreement between the authors, the studies were reviewed by all the authors to reach a consensus. This study was conducted per guidelines of the preferred reporting items for systematic reviews and meta‑analysis (PRISMA) guidelines.28, 29
Statistical Analyses
Statistical analyses were performed using Microsoft Office Excel (v. 2010). Descriptive statistics were presented as frequency and percentage (%).
Results
Of the 648,587 studies, 42 were related to the treatment of TBI with curcumin (data are presented in Figure 1).
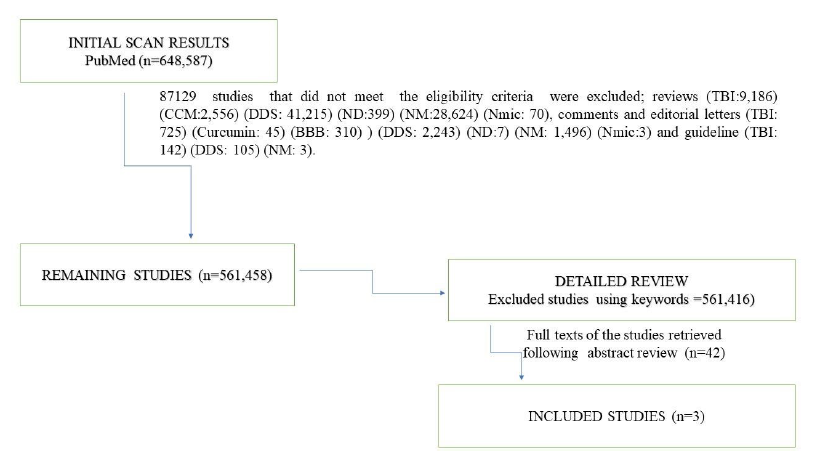
About 59,722 studies performed between 1891 and 2022 were retrieved using the keyword “traumatic brain injury.” Of the studies investigating TBI-related curcumin between 2005 and 2022, 42 were clinical trials.30, 31 The number of studies investigating curcumin in TBI through drug delivery system is three32–34 (Table 1).
Distribution of the Studies Obtained After the Scans by Years (in Amount).
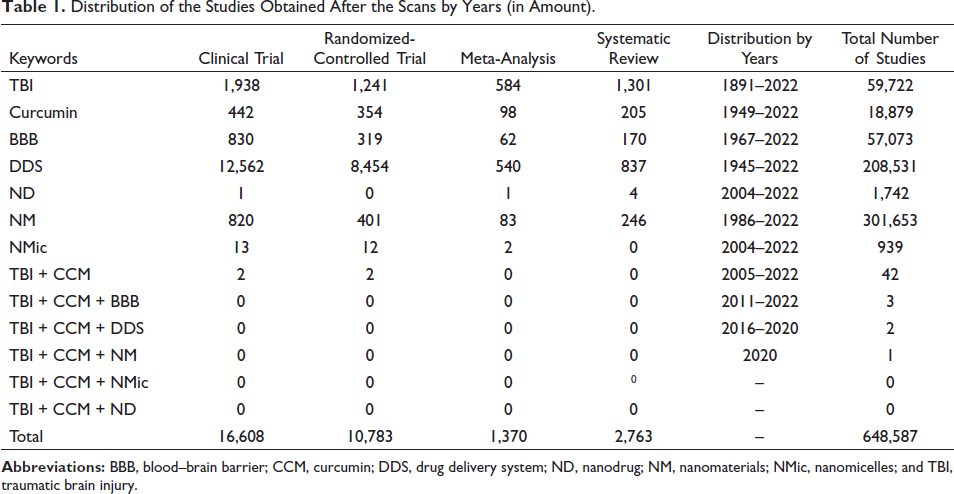
The number of studies in which curcumin was investigated using any drug delivery system for the treatment of TBI was negligible (n = 3).32–34
Discussion
TBI is defined as the transient or permanent neurological dysfunction of the CNS because of external force. The spectrum of motor, mental, and functional loss includes physical losses and cognitive/behavioral, psychological, and social defects. The shape, localization and severity of the damage affect the course and prognosis of the disease. In first stage of TBI, direct tissue damage caused by trauma is characterized by changes in cerebral blood flow and brain metabolism. During this period, lactic acid accumulation because of anaerobic glycolysis, increased membrane permeability, and cerebral tissue edema may be observed. Because anaerobic glycolysis cannot provide enough energy, the ATP-dependent ion pumps are damaged, causing excessive release of excitatory neurotransmitters. Subsequently, activation of N-methyl-D aspartate and voltage-dependent Na+ and Ca2+ channels lead to terminal membrane activation. The continuous intracellular flow of Na+ and Ca2+ triggers apoptosis in cells. Activation of substances such as intracellular translocase and endonuclease may cause necrosis after cell membrane and DNA damage, cell death, or apoptosis. 35
Current conservative/medical and/or surgical treatment modalities have remained unresponsive to TBI-induced cell death. Therefore, pharmacological agents with different pharmaceutical forms are tested in the treatment of TBI. These pharmacological agents include curcumin and drug delivery systems such as carbon nanotubes, dendrimers, hydrogels, liposomes, nanodrugs, nanomaterials, nanomicelles, nanoparticles, and nanovesicles.
Curcumin, a phenolic compound, provides promising outcomes as a natural anti-inflammatory in the treatment of cardiovascular disease, 36 cancer, 37 metabolic syndromes, 38 and various degenerative conditions.39, 40 A study has suggested that curcumin was even more effective than an anti-inflammatory drug in people with rheumatoid arthritis. 41 In addition, curcumin neutralizes free radicals 42 and has an antioxidant capacity. 43 This natural phenolic compound may also have antigenotoxic activity. 44 It has been suggested that curcumin can be used in Alzheimer’s disease, through a retro-inverted peptide that inhibits β-amyloid aggregation by targeting β-amyloid. 45
Parkinson’s disease is a progressive brain disorder. 46 Because of the presence of BBB, conventional pharmaceutical agents are insufficient to treat the diseased nuclei and show their action to prevent or delay the progress of Parkinson’s disease. 46 Curcumin is a promising compound to treat Parkinson’s disease. However, its administration is still challenging in vivo because of its poor druggability and low bioavailability via the BBB. 46
Curcumin-loaded solid lipid nanoparticles and nanostructured lipid carriers have been functionalized with transferrin because of their poor water solubility, low bioavailability, rapid elimination, and difficulties in crossing over the BBB. 47 Accordingly, it facilitates the transport of the mentioned particles to the brain. Curcumin targeting can be used in many central nervous system diseases, especially Parkinson’s diseases. 47
Curcumin also plays a role in memory and learning because of its neuroprotective effect, and it can increase the brain-derived neurotrophic factor protein located in the regions of the brain responsible for nutrition and body weight.48, 49
Curcumin, a natural product with antioxidant, anti-inflammatory, and antibacterial activities, has an anti-inflammatory effect on the brain of BALB/c mice infected with drug-sensitive mycobacteria. Curcumin significantly reduces neuroinflammation and improves behavioral status by slightly increasing the levels of nuclear factor erythroid 2-related to factor 2 brain-derived neurotrophic factor. 50
Shadnoush et al. 30 reported that curcuminoid supplementation has a beneficial effect on serum concentrations of adipokines, but this beneficial effect has not yet been studied in the treatment of critical TBI cases. Placebo-controlled 500 mg/day curcuminoids were administered to 62 critical cases with TBI aged 18 to 65 years. Patients in both the placebo and curcuminoid groups received routine treatments for TBI along with enteral nutrition. The authors reported serum leptin and adiponectin concentrations measured at baseline and at the end of the seventh day. There was a significant decrease in serum leptin levels in the curcuminoids group compared to the placebo group; however, despite supplementation with curcuminoids, serum concentrations of adiponectin in cases with TBI did not change. 30
Zahedi et al. 31 reported that the effects of curcuminoid supplementation on inflammatory and oxidative stress biomarkers, clinical outcomes, and nutritional status in critically ill patients with TBI are unknown, although the beneficial effects of polyphenolic curcuminoids in the treatment of TBI have been proposed in experimental studies. In one such study, serum levels of interleukin-6, tumor necrosis factor-alpha, monocyte chemoattractant protein-1, and C-reactive protein were significantly decreased in patients treated with curcuminoid compared to the placebo group. 31 However, changes in superoxide dismutase and glutathione peroxidase activities in serum were not significant between placebo and curcuminoid groups. In addition, the authors noted that clinical scores were significantly improved following curcuminoid administration in the study group compared to the placebo group and concluded that short-term supplementation with curcuminoids may have beneficial effects on inflammation, clinical outcomes, and nutritional status in critically ill TBI patients. 31
Sintov 32 stated that although curcumin has enormous potential as a therapeutic agent in central nervous system disorders, its effectiveness in reaching cerebral tissue could be limited because of negative conditions such as low bioavailability and rapid body clearance. Curcumin was targeted to the central nervous system via the intranasal route using the amyloLipid nanovesicles (ALNs). The authors designed curcumin-loaded ALNs, which were optimized by producing from microemulsion using starch as a crosslinker agent. Intranasal administration of curcumin at a dose of 160 µg/kg was compared with intravenous administration of the same dose. The authors suggested that ALNs formulation is a promising carrier for brain targeting of curcumin. 32
Although curcumin is an antioxidant compound, in vivo application is greatly limited because of its rapid metabolism combined with its instability under physiological conditions. 33 A thermosensitive hydrogel with a high payload of curcumin was developed and this formulation was tested in Neuro-2a cells to investigate if it reversed oxidative stress. 32 It has been reported that the developed curcumin/thermosensitive hydrogel formulation reduces oxidative stress and may have a great potential for the treatment of TBI. 33
Qian et al. 34 suggested that TBI initiates excess reactive oxygen species (ROS), which can worsen the secondary injury and result in disability and death. ROS pool formation in normal brain tissue adjacent to traumatic cerebral tissue may lead to significant neuronal death. The authors developed an injectable, posttraumatic microenvironment-sensitive, three-parts, curcumin-embedded hydrogel to reduce ROS levels in damaged brain tissue to promote regeneration and healing of neurons and tested their hydrogel in two postoperative TBI models. As a result of the analysis, it was observed that the curcumin loaded hydrogel significantly reduced ROS and cerebral edema. Likewise, reactive astrocytes and activated microglia were decreased, and growth-related protein 43 and doublecortin levels were increased. The authors suggested that this curcumin-loaded drug delivery system has a potent anti-inflammatory effect and effectively promotes nerve regeneration after TBI. 34
Conclusion
The findings suggest that curcumin can modulate pharmacomolecular signaling pathways related to oxidative stress, inflammation, apoptosis, and autophagy by locally targeting traumatic areas in the brain through Drug delivery system developed from suitable nanomaterials such as carbon nanotubes nanotubes, nanofibers, nanomicelles, or nanovesicles. It can therefore be an effective treatment option for TBI in the future.
Footnotes
Author Contributions
IY and NK designed the flow and wrote the subsections of the manuscript, and contributed to the conceptualization, data curation, data investigation, and methodology. IY and HS contributed to project administration, validation, and supervision. HO and OA contributed to manuscript revision and proofreading. All authors approve of the submitted version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
