Abstract
Background
Mild traumatic brain injury (mTBI) generally resolves within weeks. However, 15-30% of patients present persistent pathological and neurobehavioral sequelae that negatively affect their quality of life. Hyperhomocysteinemia (HHCY) is a neurotoxic condition derived from homocysteine accumulation above 15 μM. HHCY can occur in diverse stressful situations, including those sustained by U.S. active-duty service members on the battlefield or during routine combat practice. Mild-TBI accounts for more than 80% of all TBI cases, and HHCY exists in 5-7% of the general population. We recently reported that moderate HHCY exacerbates mTBI-induced cortical injury pathophysiology, including increased oxidative stress. Several studies have demonstrated hippocampus vulnerability to oxidative stress and its downstream effects on inflammation and cell death.
Objective
This study aimed to assess the deleterious impact of HHCY on mTBI-associated hippocampal pathological changes. We tested the hypothesis that moderate HHCY aggravates mTBI-induced hippocampal pathological changes.
Methods
HHCY was induced in adult male Sprague-Dawley rats with a high methionine dose. Rats were then subjected to mTBI by controlled cortical impact under sustained HHCY. Blood plasma was assessed for homocysteine levels and brain tissue for markers of oxidative stress, blood-brain barrier integrity, and cell death. Endothelial cell ultrastructure was assessed by Electron Microscopy and working memory performance using the Y maze test.
Results
HHCY increased the hippocampal expression of nitrotyrosine in astroglial cells and decreased tight junction protein occludin levels associated with the enlargement of the endothelial cell nucleus. Furthermore, HHCY altered the expression of apoptosis-regulating proteins α-ii spectrin hydrolysis, ERK1/2, and AKT phosphorylation, mirrored by exacerbated mTBI-related hippocampal neuronal loss and working memory deficits.
Conclusion
Our findings indicate that HHCY is an epigenetic factor that modulates mTBI pathological progression in the hippocampus and represents a putative therapeutic target for mitigating such physiological stressors that increase severity.
Introduction
More than 5.3 million people live with traumatic brain injury (TBI) or related disabilities in the US.1,2 Mild TBI (mTBI) accounts for more than 80% of all TBI cases.3–5 It is defined in animal models by no breaking of the dura, transient behavioral deficits associated with pathological changes mostly confined around the lesion side, or the perivascular and periventricular regions.6,7 In humans, mTBI symptoms generally resolve within weeks.8,9 However, 15-30% of mTBI patients experience lasting pathological and behavioral sequela10,11 associated with a poor quality of life.12,13 However, the causes of mTBI interpatient variability are still unclear. A limited number of studies have been conducted in animal models and humans to understand genetic and epigenetic factors that influence mTBI interpatient pathological variability. These studies demonstrated the involvement of aging,14,15 apolipoprotein E deficiency, 16 and the single nucleotide mutation in the ABCC8 gene. 17 Hyperhomocysteinemia (HHCY) is a neurotoxic condition derived from the accumulation of non-proteinogenic amino acid homocysteine above 15 μM18,19 and can be classified as mild (15-30 μM), moderate (31-100 μM), or severe (>100 μM,18,19). HHCY can occur in diverse stressful situations, including those sustained by US active-duty members on the battlefield or during combat routine practice, among whom HHCY prevalence is 9-12% 20 and 5-7% in the general population.21,22 We recently reported that moderate HHCY exacerbates mTBI-induced cortical pathologies, including lesion volume, blood-brain barrier (BBB) disruption, inflammation, and anxiety-like behavior in rats subjected to mTBI by the controlled cortical impact method (CCI). 7
Although CCI-induced mTBI pathologies primarily affect the cortical regions, the subcortical location of the hippocampus renders it sensitive to the injury.23,24 The hippocampus’s dentate gyrus (DG) granular cell layer harbors a high density of NMDA receptors,25,26 which hyperactivation by excitatory amino acids leads to excitotoxicity. Homocysteine is a potent NMDA receptor agonist,27–29 suggesting mTBI-induced hippocampal injury may be exacerbated by HHCY. In the current study, we tested the hypothesis that HHCY worsens hippocampal pathology in rats with mTBI, which overwhelmingly represents more than 80% of all TBI cases.3–5 Some aspects of the study associated with neuronal death and oxidative stress focused on the DG, which is vulnerable to HHCY-induced excitotoxicity and plays a key role in hippocampal memory formation, including working memory.30,31 Following mTBI, the hippocampus of HHCY rats showed increased oxidative stress, BBB dysfunction, neuronal loss, and altered expression of pro-apoptotic and cell survival-promoting proteins consistent with neuronal loss. We found impairments of working memory performance classically ascribed to hippocampal function. These findings indicate that moderate HHCY is an epigenetic factor exacerbating mTBI pathophysiological progression in brain regions close to (the ipsilateral hippocampus) and distant to (the contralateral hippocampus), the direct injury impact site.
Materials and methods
Animals and housing
Male Sprague Dawley rats (300-350g, 9-10 weeks old; Envigo, CA) were used. They were housed two per cage in a temperature-controlled room, maintained at 23 ± 2°C, with a 12 h light/dark cycle. They had continuous access to food and water ad libitum. Animals were randomly assigned to four experimental groups. First, all animals were weighed at the beginning of each experiment and stratified into subgroups, including animals within five grams of body weight difference. Animals within each subgroup were then evenly randomly assigned to experimental groups. The four experimental groups included, a group of animals subjected to increased HCY levels named hyperhomocysteinemia (HHCY), and those injected with saline non-hyperhomocysteinemia (NHCY). HHCY and NHCY rats were either subjected to traumatic brain injury by controlled cortical impact (CCI) or were sham-operated. Therefore, there were four animal groups in each experiment, and the total weight of animal in all groups was similar. All animals involved in each experiment were born on the same date. After surgery, animals were subjected to behavioral testing (10-12/group). Other animals, 4-6 rats/group, were used for immunohistochemistry or biochemical analyses. The sample size was decided using a power analysis based on previous studies as explained in the animal protocol. A total of 84 animals were used in this study.
Induction of hyperhomocysteinemia
Moderate hyperhomocysteinemia was induced in rats by intraperitoneal injections of methionine (Sigma-Ulrich, MO) at 0.3 g/kg once daily for 7 days.7,32 Methionine administration was continued after surgery until euthanasia on day 1, day 7, or 30 post-surgery. Baseline body weight was measured and continued once weekly during methionine injections and was reported in our recent publication on the effects of HHCY on TBI-associated cortical injury. 7 Control animals received intraperitoneal injections of normal saline.
Induction of controlled cortical impact injury
Mild traumatic brain injury (mTBI) was induced by the CCI method.6,7 Anesthesia was induced with 4% isoflurane and maintained with 2.5-3% isoflurane throughout the surgery. Rat body temperature was maintained at 37°C using a heating pad coupled with a rectal probe. Following craniotomy, CCI was performed over the left parietal cortex. Using a stereotaxically positioned 4 mm diameter piston tip, impact injury was induced with an electromagnetically controlled impactor (Leica Biosystems, IL) at a 5 m/sec piston velocity and depth penetration of 1.5 mm. The bone flap was replaced, the skin closed, and the anesthesia terminated. Sham-operated rats were also anesthetized and sustained craniotomy and replacement of the bone flap without cortical impact.
Behavioral testing
Animals were assessed for hippocampus-dependent working memory performance using the Continuous Spontaneous Alternation method 33 on days 1, 7, 14, and 28 post-surgery (10-12 rats/group) to evaluate the impact of HHCY on working memory performance over time. The test used a Y-shaped device with arm dimensions of 35 cm long, 5 cm wide, and 15 cm tall and was performed as we previously described.34–36 The test was conducted between 9:30-10:30 am. Animals were allowed to acclimate for 45-60 min before the test. In brief, animals were individually placed at the center of the Y maze and allowed to explore the three arms for 5 min. The sequence and number of each arm entry were tracked with a video camera and recorded for 5 min. The recorded videos were individually analyzed manually. An arm visit referred to the animal moving all four paws into the arm, and each alternation was defined as a consecutive entry in three different arms. The percent spontaneous alternation is calculated based on the formula: total number of alternations/(total number of arm entries-2). 37
Tissue collection and processing
Brain tissues were collected on days 1, 7, or 30 post-surgery, and the effects of HHCY on mTBI pathological outcomes were assessed at the acute and sub-chronic time points of day 1 and 7 post-injury, respectively, using biochemical and histological analysis (4-6 rats/group).
For histological analysis, rats were first anesthetized with an intraperitoneal injection of an anesthetic cocktail composed of ketamine (160 mg/kg) and xylazine (120 mg/kg). They were then euthanized by exsanguination via transcardial perfusion with artificial cerebrospinal fluid (148 mM NaCl, 5 mM glucose, 3.0 mM KCl, 1.85 mM CaCl2, 1.7 mM MgCl2, 1.5 mM Na2HPO4, and .14 mM NaH2PO4 (pH 7.4) at a rate of 2 mL/min for 5 minutes. The flush was followed by perfusion with a fixative containing 4% paraformaldehyde in 50 mM K2HPO4 and 50 mM KH2PO4 (pH 7.4). Brains were removed, maintained in fixative for 24 h, transferred to 30% sucrose, and individually sliced in 40 μm thick coronal sections. Sections were preserved in a cryoprotectant (66 mM NaH2PO4, 190 mM Na2HPO4, .87 M sucrose, 30% ethylene glycol, and 1.25 mM povidone), and stored at −20°C for histology and immunohistochemistry.
For Western blot analysis, brains were dissected to collect fresh (not paraformaldehyde-perfused) hippocampal and cortical tissues immediately after decapitation with a guillotine. The tissues collected were flash-frozen in liquid nitrogen and stored at −80°C until analyzed.
Blood samples were collected via cardiac puncture during euthanasia and transferred to EDTA tubes. They were immediately processed into plasma and stored at −80°C until used for HCY measurements.
Histological analysis
Immunofluorescence staining
Brain sections were co-stained for the expression of the oxidative stress marker nitrotyrosine and the neuron-specific neuronal nuclear antigen (NeuN), nitrotyrosine, and the astrocyte marker the glial fibrillary acidic protein (GFAP) or the apoptotic marker cleaved caspase-3 and NeuN as previously described.34,35,38 Briefly, free-floating sections were rinsed in phosphate-buffered saline (PBS) and blocked in 1% horse serum in PBS containing .3% Triton X for 1 h. Sections were then transferred in an antibody mixture containing rabbit anti-nitrotyrosine polyclonal antibody (1:2500; Sigma, MO) and chicken anti-GFAP polyclonal antibody (1:3000; Novus Biologicals, CO), rabbit anti-nitrotyrosine polyclonal antibody (1:2500; Sigma, MO) and mouse anti-NeuN monoclonal antibody clone, A60 (1:1500; Millipore/Sigma, MO) or rabbit anti-cleaved caspase 3 polyclonal antibody (1:7500; Millipore, MA) and mouse anti-NeuN monoclonal antibody clone, A60 (1:1500 Millipore/Sigma, MO) and incubated at 4°C overnight. Sections were washed in PBS and incubated in a mixture of corresponding secondary antibodies containing Alexa Fluor 594 and Alexa Fluor 488 or Alexa Fluor 547 (1:2000; Invitrogen, NY) for 1 h at room temperature. Sections were washed with PBS, counterstained with 4,6-diamidino-2-phenylindole (DAPI), and mounted with an anti-fade mounting medium (Vector Labs, CA).
Nickel DAB immunostaining
Brain sections were antibody-stained for the expression of the neuronal marker NeuN using nickel DAB staining. In brief, free-floating sections were rinsed in PBS, blocked with 1% horse serum, and incubated in mouse anti-NeuN monoclonal antibody (1:40 k; Millipore/Sigma, MO) overnight at 4°C. Sections were washed and incubated for 1-hr at room temperature in biotinylated horse anti-mouse secondary antibody (1/1000 respectively; Vector Laboratories Inc, CA). Then, sections were incubated in Vestastain solution and nickel DAB, followed by PBS washes. Sections were then mounted with DPX (Sigma, MO)
Hematoxylin and eosin staining
Four brain sections (40 μm) from NHCY-sham animals were stained with hematoxylin and eosin (Sigma, MO) to locate the hippocampal fissure, highly vascularized to present endothelial cells. Stained brain sections were dehydrated, cleared, and mounted with DPX (Sigma, MO).
Transmission electron microscopy
For the transmission Electron microscopy, the hippocampal dentate gyrus upper blade (40 μm) containing the hippocampal fissure was micro-dissected, fixed, dehydrated, and embedded in Epoxy resin following standard procedures. 70 μm ultrathin sections were collected and imaged with a Transmission Electron Microscope (Technai T12, ThermoFisher) at 80 KeV using an AMT digital camera to capture the blood vessel ultrastructure.
Western blot analysis
Western blotting was performed to determine the expression of occludin, alpha-2 spectrin, ERK1/2, phosphorylated ERK1/2 (pERK1/2), AKT, pAKT, and β-actin (loading control) in the ipsilateral hippocampus of animals in different experimental groups. The ipsilateral hippocampi were manually homogenized in ice-cold lysis buffer, and respective supernatants were processed for electrophoresis as we previously described.34–36,39 Total proteins (25 μg) from each supernatant were separated by electrophoresis on 4-12% SDS-polyacrylamide gels (Invitrogen, CA), then transferred to a nitrocellulose membrane (Sigma, MO). Membranes were blocked with 5% non-fat milk in Tris-buffered saline with .1% Tween-20 (TBST) and incubated overnight at 4°C in rabbit polyclonal antibodies against occludin (1:2000; Abcam, MA), α-ii spectrin (1:3000; Invitrogen/ThermoFisher Scientific, NY) ERK, 1/2, pERK1/2, AKT, pAKT (1:1000; Cell Signaling Technology, MA) or a mouse anti-β-actin (1:4000; Sigma, MO). Blots were washed in Tris-buffered saline with .1% Tween-20 and incubated for 1 h at room temperature with corresponding HRP-conjugated secondary antibodies (1:5000; Millipore, MA). Horseradish peroxidase-labeled proteins were detected by enhanced chemiluminescence (ECL, Thermo Scientific, IL), and protein bands were visualized using a digital blot scanner (LI-COR, NE).
Quantitative methods
The optical fractionator method of stereology of the stereo-investigator software (MBF Biosciences) was used to quantify nitrotyrosine, cleaved caspase 3, and NeuN immuno-reactive cells in the hippocampal dentate gyrus region. For all measurements, six sections were obtained to cover the entire impact region, −1.60 mm to −6.3 mm from Bregma, corresponding to every 12 serial sections for each brain. For the stereological quantitation, we used a grid spacing of 75 μm × 75 μm in the x and y-axis and guard zones of 2 μm at the top and bottom of each section where immuno-positive cell bodies were counted. The total number of nitrotyrosine, cleaved caspase 3, and NeuN-positive cells in the volume of interest were automatically determined and expressed as cells/mm3.
Image J software (NIH) was used to measure immunoblot protein band signal intensity, the internal length, and the central width of each endothelial nucleus.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 9 software (GraphPad Software, Inc, CA). Data from biochemical and histological assessments were analyzed using Two-Way ANOVA and behavioral data with repeated Two-Way ANOVA. All analyses were coupled with the Tukey-Kramer multiple comparisons post-test to compare differences among all the groups. Results were expressed as mean ± standard deviation of the mean (SD) at 95% CI. All data points were included. Statistical significance was defined as P < .05.
The individuals who performed the histologic, biochemical, and behavioral assays were blinded to animal group identifications using codes revealed after data collection.
Results
Hyperhomocysteinemia increased oxidative stress-induced nitrotyrosine in astroglial cells in the dentate gyrus
Nitrotyrosine was expressed mainly by stellate-shaped cells across all layers of the DG, consistent with the morphology and distribution of astrocytes (Figure 1). Nitrotyrosine-positive cells increased significantly in the DG of HHCY-sham rats, 12 000 ± 5237 cells/mm3 compared to 1100 ± 52 cells/mm3 in non-hyperhomocysteinemic (NHCY) shams (Figure 1(A), (B)), indicating that HHCY in normal sham animals is a pro-oxidative stressor as predicted. HHCY also potentiated nitrotyrosine expression in the DG of rats with mTBI, 18,000 ± 5863 cells/mm3, compared to 7200 ± 2190 cells/mm3 in NHCY-CCI rats (Figure 1A and B). This indicates that TBI and HHCY had an additive effect on inducing oxidative stress. In addition, the co-staining of brain sections from HHCY-CCI rats against nitrotyrosine and the astrocyte marker GFAP demonstrated that astrocytes expressed nitrotyrosine in the DG (Figure 2A). By contrast, around the impact side, nitrotyrosine was expressed by different cell types, including astrocytes (Figure 2B) and neurons (Figure 2C), which are immuno-negative against nitrotyrosine in the DG (Figure 1A). Results of plasma HCY levels were reported in our recent publication.
7
Increased nitrotyrosine expression in the dentate gyrus of brain-injured rats and exacerbation by hyperhomocysteinemia (A) Representative immuno-fluorescent images demonstrating the presence of neuronal protein NeuN (red), oxidative stress marker nitrotyrosine (green), and DAPI used for nuclear counterstaining (blue) in the DG of rat hippocampus. The scale bar is 100 μm. (B) Quantitation of nitrotyrosine positive cells showed that nitrotyrosine expressing cells were significantly higher in the DG of HHCY sham and CCI rats on day 1 post-injury compared to NHCY-sham and -CCI rats, respectively (n = 5; #P < .05; **P < .01 and ***P < .01). These cells were counted in the entire DG region using the FITC channel to detect nitrotyrosine immunopositive cells revealed by Alexa fluor 488 secondary binding. Nitrotyrosine in the dentate gyrus was expressed in astroglial cells (A, B) Representative fluorescent images showing the presence of astrocyte-like cells (red), nitrotyrosine (purple), and DAPI (blue) in rats’ dentate gyrus or motor cortex on day 1 post-surgery. (C) Representative fluorescent images showing the presence of neurons (red), nitrotyrosine (green), and DAPI (blue) in rats' motor cortex around the impact side. The scale bar is 100 μm.

Homocysteine accumulation suppressed tight junction protein expression and altered the ultrastructure of the vascular endothelial nucleus after mTBI
To investigate the impact of HHCY on BBB function, we assessed the expression of the tight junction protein occludin. Occludin expression decreased by 40.27 ± 4.2% in NHCY-CCI rat hippocampi on day 7 post-injury (pi) compared to NHCY-sham rats (Figure 3A and B). HHCY aggravated the effect of mTBI on occludin expression, causing a 52.2 ± 2.9% and 48.1 ± 3.6% reduction, respectively, on days 1 and 7 pi compared to NHCY-shams (Figure 3(A), (B)). Moreover, occludin levels decreased by 32.5 ± 4.3% in HHCY-CCI rats vs NHCY-CCI rats on day 1 pi (Figure 3A and B). Altered occludin expression and vascular endothelial cell ultrastructure in CCI-injured rat brain hippocampus were worsened by hyperhomocysteinemia. (A) Representative immunoblots illustrating the expression of occludin in rat hippocampus. (B) Quantitation of protein band intensities indicates that occludin expression was decreased in the NHCY-CCI rat hippocampus on day 7 post-injury. This reduction was more profound in the hippocampus of HHCY-CCI rats on days 1 and 7 post-injured (n = 5; *P < .05, **,##P < .01, ***P < .001). Beta-actin was used as a loading control. (C) Representative hematoxylin and Eosin staining of the hippocampal fissure region. (D) Representative TEM images showing endothelial cells and their respective nucleus with a pointed arrow on day 1 post-surgery. The scale bar is 500 μm. (E-F) Quantitation of randomly captured images of endothelial nucleus internal length and central width indicated a significant increase in endothelial cell nucleus internal length and central width in HHCY-CCI compared to HHCY-sham and NHCY-CCI rats (n = 11-12 cells/group; **P < .01, ***P < .001). There was also a significant increase in endothelial cell nucleus internal length in NHCY-CCI compared to HHCY-sham rats (n = 11-12 cells/group; #P < .05).
Next, we determined if HHCY affected the ultrastructure of vascular endothelial cells following mTBI, which could further disorganize the vascular endothelium and increase BBB breakdown. Focusing on the impact of HHCY on endothelial cell ultrastructure in mTBI rats compared to NHCY-CCI rats and NHCY-sham controls, TEM imaging of the hippocampal fissure region on day 1 post-surgery (Figure 3D) showed that mTBI alone significantly increased the length of the endothelial cell nucleus (ECN) in NHCY-CCI rats, 3.42 ± 1.64 μm, compared to NHCY-sham rats, 1.88 ± .55 μm (Figure 3D and E). The combination of mTBI and HHCY further increased the length of the ECN, 6.47 ± 1.82 μm, compared to NHCY-sham and NHCY-CCI rats). Moreover, the ECN width was also significantly increased in HHCY-CCI rats, 1.18 ± .55 μm compared to NHCY-CCI rats, .66 ± .23 μm, and NHCY-shams .51 ± .15 μm (Figure 3(D), (F)).
Elevated homocysteine increased mild TBI-induced expression of cleaved caspase 3 and reduced neuronal cell density in the dentate gyrus
We examined the impact of HHCY on hippocampal apoptotic cell death post-mTBI and found a significant increase of C-Casp-3+ cells in HHCY-sham rats, 2660 ± 492 cells/mm3 compared to NHCY-shams, 171 ± 75 cells/mm3 (Figure 4A). Mild TBI alone resulted in more C-Casp-3+ cells in NHCY-CCI rats, 3977 ± 1237 cells/mm3, than HHCY alone. Moreover, the number of C-Casp-3+ immuno-positive cells was significantly increased by the combination of HHCY and mTBI, 6352 ± 1563 cells/mm3, compared to HHCY or mTBI alone (Figure 4A and B). Brain injury-induced cleaved caspase 3 expression in the hippocampal dentate gyrus exacerbated by hyperhomocysteinemia (A) Representative immunofluorescent microscopic images showing cleaved caspase 3 (CCASP-3; green) and NeuN (red) immuno-positive cells and DAPI stained nuclei in the DG of HHCY and NHCY-CCI and sham rats on day 1 post-surgery. The scale bar is 100 μm. (B) Quantitation found a significant increase in C-casp-3+ cells in the DG of HHCY-sham and NHCY-CCI compared to NHCY-shams (n = 5; **P < .01 and ***P < .001). This condition was aggravated in HHCY-CCI rat DG, resulting in a significant increase of CCASP-3+ cells in their DG than those of HHCY-sham and NHCY-CCI rats (n = 5; #P < .05, ##P < .01).
To determine how apoptotic cell death impacted neuronal cell density in the dentate gyrus, we immuno-stained against the neuronal marker NeuN in the ipsilateral and contralateral dentate gyrus. NeuN Immunostaining in the ipsilateral and contralateral DG found that mTBI alone significantly reduced neuronal density in the ipsilateral DG of NHCY-CCI rats, 295,877 ± 70,341 cells/mm3, compared to NHCY-shams (592,889 ± 47,296 cells/mm3, Figure 5A and B). Remarkably, a significant reduction of neuronal density was found in both the ipsilateral and contralateral DG of HHCY-sham rats, 416,395 ± 55,464 cells/mm3 and 433,932 ± 21,801 cells/mm3, respectively, compared to 592,889 ± 47,296 cells/mm3 and 613,226 ± 30,645 cells/mm3 (Figure 5A and B). The combination of HHCY and mTBI exacerbated the decrease of neuronal density, resulting in neuronal cell counts of 194,495 ± 22,242 cells/mm3 and 290,445 ± 46,670 cells/mm3 in the ipsilateral and contralateral hemispheres, respectively (Figure 5A and B). Hyperhomocysteinemia aggravated the reduction of neuronal density in HHCY brain-injured rat’s hippocampal dentate gyrus (A) Representative Nickel-DAB immuno-stained microscopic images showing NeuN immuno-reactive cells in the ipsilateral and contralateral DG of HHCY and NHCY-CCI and sham rats on day 1 post-surgery. The scale bar is 50 μm. (B) Quantitation indicated a significant decrease in NeuN immuno-reactive cells in the ipsilateral DG of HHCY-sham, NHCY-CCI, and HHCY-CCI rats compared to NHCY-shams (n = 4-5; **P < .01 and ***P < .001). In addition, brain injury caused a significant loss of neurons in the ipsilateral DG of NHCY-CCI rats than HHCY-shams (n = 5, ##P < .01), and that condition was exacerbated in HHCY-CCI rat brains (n = 5, ###P < .001). Moreover, there was a significant decrease in neuronal cell density in the DG HHCY-CCI rats that NHCY-CCY (n = 5, #P < .05). In the contralateral hemispheres, there was a significant loss in neurons in the DG of HHCY-sham rats (n = 4-5; ###P < .001). Neuronal loss was potentiated by brain injury in HHCY-CCI rats compared to HHCY-shams (***P < .001).
Hyperhomocysteinemia upregulated α-ii spectrin proteolytic breakdown and suppressed ERK and AKT phosphorylation following mild TBI
To determine molecular changes associated with the loss of neuronal cells observed in the hippocampal DG, we investigated if HHCY affected the proteolytic breakdown of the pro-apoptotic protein α-ii spectrin. We found a significant 1.57-folds increased α-ii spectrin proteolytic breakdown products in HHCY-CCI rats' hippocampus on day 7 post-injury compared to NHCY-shams and HHCY-sham rats (Figure 6A and B). In addition, more significant effects of HHCY on alpha spectrin breakdown product 70-45 kdal were observed among HHCY-CCI rats at day 7 post-injury compared to NHCY and HHCY-shams NHCY-CCI rats. There was a 2.2-folds difference between HHCY-CCI rats and shams rats, both NHCY and HHCY. Compared to NHCY-CCI rats, there was also a significant 1.6-folds difference (Figure 6C). Increased proteolytic breakdown of α-ii spectrin in the hippocampus of hyperhomocysteinemic brain-injured rats. Representative immunoblots illustrating the expression of alpha ii spectrin and breakdown products (A) in hippocampal tissues from NHCY or HHCY sham and CCI rats on days 1 and 7 post-surgery. Beta-actin was used as a loading control. Quantitation of target protein band intensities over actin showed a significant increase in (B) α-ii spectrin breakdown products significantly increased in HHCY-CCI rats hippocampi on day 7 post-surgery compared to NHCY and HHCY-sham rats (n = 5, *P < .05). (C) α-Ii spectrin, 70-45 kdal breakdown product, showed a more significant increase in HHCY-CCI rats on day 7 post-injury compared to NHCY and HHCY shams as well as NHCY-CCI rats (n = 5, 
We also examined the effect of HHCY on the phosphorylation of the cell survival-promoting proteins ERK1/2 and AKT. Results showed a 1.8-fold reduction in ERK phosphorylation in HHCY-shams and a 1.6-fold reduction in NHCY-CCI rats on day 7 post-surgery compared to NHCY-shams (Figure 7A and C). Moreover, the combination of HHCY and mTBI further suppressed ERK phosphorylation by 2.15-folds on day 1 post-injury and by 2.5-folds on day 7 post-injury compared to NHCY-shams (Figure 7A and C). These results suggest that HHCY-CCI rat hippocampal cells were more prone to apoptotic cell death. On the other hand, mTBI alone had a biphasic impact on AKT phosphorylation at different experimental time points. On day 1 post-injury, pAKT levels increased by 1.98-folds and 2.35-folds in NHCY-CCI rats respectively, compared to HHCY-shams and HHCY-CCI rats (Figure 7B and D). We also found a 2.1-folds significant decrease in pAKT levels in HHCY-CCI rats on day 7 post-injury compared to NHCY-shams (Figure 7B and D). Furthermore, AKT phosphorylation showed a significant 1.6-folds decrease among HHCY-sham rats on day 1-pi compared to NHCY-shams (Figure 7B and D). Brain injury-induced decreased phosphorylation of cell survival-promoting proteins ERK1/2 and AKT potentiated by hyperhomocysteinemia. Representative immunoblots illustrating the expression of phosphorylated and total ERK (A) and AKT (B) in the hippocampal of NHCY or HHCY sham and CCI rats on days 1 and 7 post-surgery. Beta-actin is used as a loading control. (C, D) Quantitation of phosphorylated protein band intensities over the total protein showed a significant decrease in pERK in the hippocampus of HHCY-sham rats on day 7 post-surgery and NHCY-CCI on days 1 and 7 post-surgery compared to NHCY-shams (n = 5, *P < .05, **P < .01). HHCY aggravated pERK reduction on days 1 and 7 post-injury (n = 5; ##P < .01 and ***P < .001). On the other hand, AKT phosphorylation was significantly reduced in HHCY-sham on day 1 post-injury compared to NHCY-shams, and the combination of HHCY brain injury exacerbated the condition compared to HHCY-sham and CCI rats (n = 5; *P < .05, **P < .01). Phosphorylated AKT levels showed a significant increase in the hippocampus of NHCY-CCI rats at day 1 post-injury compared to HHCY-shams and HHCY-CCI rats (n = 5; ##P < .01).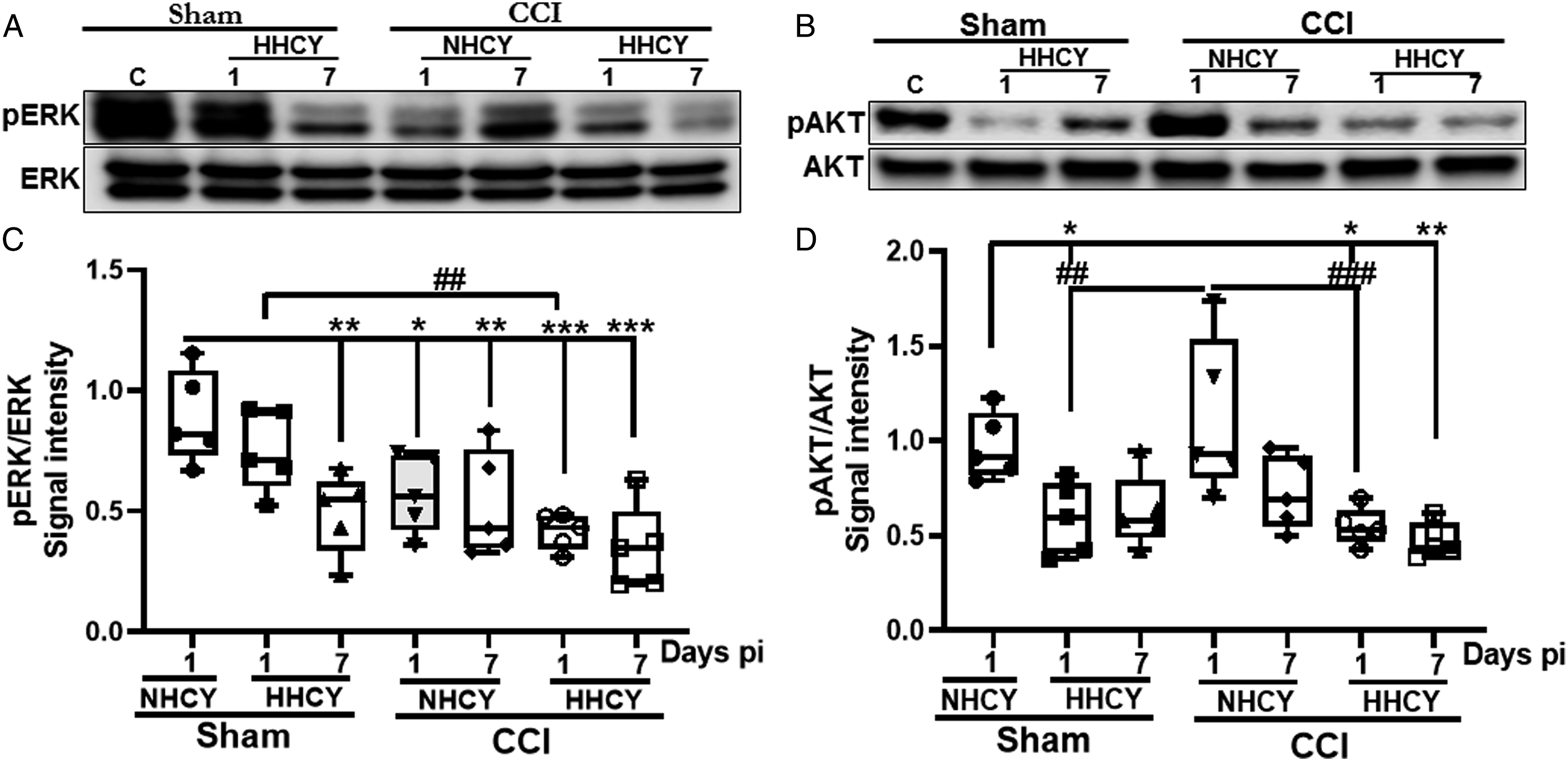
Homocysteine accumulation impaired Hippocampus-dependent working memory performance in rats with mTBI
The hippocampal formation is a key determinant in the working memory process. We assessed if HHCY-induced exacerbation of several TBI pathological markers affected the hippocampus-dependent working memory performance in rats with mTBI. We found that HHCY-CCI rats achieved significantly reduced spontaneous alternations on days 1 pi, 50.8 ± 13.5%, and on 7 pi, 46.76 ± 19.36% compared respectively to 72.9 ± 9.58% and 73.89 ± 11.44% for NHCY-shams (Figure 8D). In addition, HHCY-CCI rats presented significantly reduced percent alternation on day 7 post-injury, 46.76 ± 19.36%, compared to NHCY-CCI rats, 65.3 ± 14.79% (Figure 8D). Moreover, HHCY prolonged the time some mTBI rats took to complete their first spontaneous alternation in the Y maze device. Consequently, some of these animals completed a spontaneous alternation after 77 secs in the Y maze. NHCY-sham and NHCY-CCI had completed 3 or more spontaneous alternations after that time in the Y maze (Figure 8A-C). Mild-TBI alone had no significant effect on working memory performance compared to NHCY-shams (Figure 8D). Hyperhomocysteinemia caused a significant reduction of the percent spontaneous alternation in brain-injured rats in the Y maze (A-C) Images of the Y maze exploration tracks showing the exploration frequency of NHCY-sham, NHCY-CCI, and HHCY-CCI rats in the Y maze device during the working memory performance test on day 1 post-surgery. These representative tracks show that HHCY-CCI mice completed their first alternation after up to 77 secs in the maze, while rats in other groups including HHCY-rats, had achieved 3 or more spontaneous alternations at that time. (D) Graph of the Y maze test results demonstrating that HHCY-CCI rats had significantly reduced % spontaneous alternation in the Y maze on days 1 and 7 post-injury compared to NHCY-shams (n = 10-12; *P < .05, **P < .01). In addition, HHCY-CCI rats also exhibited significantly reduced % spontaneous alternation than NHCY-CCI rats on day 7 post-injury (n = 10-12; #P < .05).
Discussion
TBI interpatient pathological variability represents a challenge for developing an effective therapeutic agent. Mild TBI accounts for more than 80% of all TBI cases. Although the condition usually resolves within weeks, 15-30% of victims suffer long-term pathological and behavioral sequela,10,11 and susceptibility factors are not elucidated. One candidate we have been investigating is HHCY, the neurotoxic accumulation of the non-proteinogenic amino acid homocysteine. We recently reported on the detrimental effects of HHCY on mTBI pathological outcomes in rat cortex in a CCI model. 7
HHCY can occur in diverse stressful situations, including those sustained by US active-duty members on the battlefield or during ‘routine’ deployments to regions of potential conflict, 20 and is present in 5-7% of the general population.21,22 HHCY’s neurotoxic properties include acting as a potent agonist to NMDA receptors,27–29 which are highly expressed in the DG of the hippocampus.25,26 Thus, a large population of soldiers at risk of mTBI from conflict are also subject to chronic stressors leading to HHCY. Therefore, we investigated the impact of HHCY on mTBI-associated neuropathology on the hippocampal DG, responsible for several brain-associated memory functions, including hippocampus-dependent working memory.40,41 Our results present novel findings showing additive harmful effects of HHCY on mTBI-associated oxidative stress, BBB dysfunction, neuronal loss, and related molecular mechanisms in the rat hippocampus. We also found that the overall impact of these neuropathological changes decreased hippocampus-dependent working memory working performance.
Oxidative stress following TBI is an important marker of the injury’s pathological progression. It promotes reactive chemical species production and mediates several TBI-associated pathophysiological events, including BBB damage, inflammation, and cell death.35,42–44 HHCY has been shown to trigger nitrotyrosine expression mediated by dimethylarginine-dimethylaminohydrolase, which causes increased nitrotyrosine formation from peroxynitrite.45,46 Our results showed that the combination of HHCY and mTBI-induced oxidative stress aggravated the nitrotyrosine expression in the hippocampal DG of HHCY-CCI rats. Nitrotyrosine immunopositive cells in the DG co-expressed GFAP. Although these cells predominantly presented stellate-shaped consistent with astrocyte morphology, GFAP can also be expressed neural stem cells in the hippocampal DG and could be differentiated by their ability to co-express neural stem cell biomarkers such ad nestin or Sox2.47,48 Due to its stress-inducing effects, elevated homocysteine has been shown to promote the death of neuronal precursor cells via multiple cell death mechanisms, including apoptosis and autophagy.49,50 The hippocampal DG sub-granular zone is one of the two areas of the brain where adult neurogenesis occurs, 51 followed by new cell integration into the hippocampal circuit.52,53 This may make the DG particularly sensitive to HHCY neurotoxicity.
The BBB is a neurovascular unit that regulates the movement of substances and blood-borne immune cells into the brain parenchyma. 54 Therefore, changes to the molecular and cellular components of the BBB may increase the presence of neurotoxins in the brain parenchyma, which could alter disease progression. The combination of mTBI and HHCY had a more substantial impact in decreasing occludin expression after injury. Moreover, it also caused a significant alteration to the endothelial cell nucleus suggesting structural and molecular perturbations of the BBB. Although the pathological implication is not elucidated, recent findings of the presence of enlarged endothelial cell nucleus variants in the microvasculature of allograft kidney biopsies were believed to result from endothelial cell injuries. 55 Moreover, HCY has injurious effects that cause vascular microbleeding associated with vascular injuries. 56 The hippocampus vascular architecture composed of several transversals, longitudinal arteries, and veins in the hippocampal fissure suggests hippocampal vulnerability to BBB disruption, which correlates with previous reports of HHCY-induced of BBB leakage,57,58 exacerbated by mTBI. 7
Homocysteine neurotoxic cell death may result from its excitotoxic activation of NMDA receptors27–29 or DNA fragmentation associated with the poly-ADP-ribose polymerase (PARP) or caspase activation. 29 Novel findings from our study determined that the combination of HHCY and mTBI altered α-ii spectrin proteolysis in the rat hippocampus. The timeline of these impairments revealed that α-ii spectrin proteolysis is a delayed process that precedes cleaved caspase-3 upregulation. Notably, changes in 145-150 kdal fragments of α-ii spectrin breakdown products are primarily reported in the cortex following TBI.59–61 Here, we observed breakdown products at 100 and 70-45 kdal, suggesting a brain region-specific breakdown pattern, and HHCY had more impact on 70-45 kdal breakdown products. Contrary to pro-apoptotic proteins, cell survival-promoting proteins such as ERK1/2 and AKT are required under normal homeostasis conditions to promote cell survival through their protective effects on the brain-derived neurotrophic factor. 62 However, in pathological conditions, including TBI, their phosphorization is suppressed34,36 to promote brain cell loss. In our experiment, HHCY alone reduced AKT phosphorylation in sham rats, exacerbated by HHCY and TBI combined. AKT phosphorylation was also upregulated in the hippocampus of NHCY-CCI rats on day 1 post-injury before returning to normal on day 7. HHCY had similar effects on ERK phosphorylation. These results suggest that the sustained decrease of AKT and ERK phosphorylation in HHCY-CCI rats on days 1 and 7 post-injury indicates a still active TBI pathology, while their phosphorylation shows signs of TBI recovery in NHCY-CCI rats on day 7 post-injury.
Brain cell death following CCI-induced mTBI is typically confined to the impact side. However, we found a significant increase in neuronal loss in the ipsilateral and contralateral DG of the HHCY-sham rat, which HHCY and mTBI aggravated. These results suggest that while mTBI mainly impacts the ipsilateral brain hemisphere, HHCY has a systemic effect in both hemispheres. One possibility is that HHCY primes cells in the contralateral DG, making them more vulnerable and prone to death after mTBI. Second, mTBI-induced stress renders the cells vulnerable to HHCY-induced loss of neurons.
The hippocampus is the epicenter of memory and cognition.63,64 Mild TBI-induced cell loss exacerbated by HHCY in the DG may have affected both neural progenitor cells and adult neurons. Interestingly, neural progenitor cells and stem cells are known to drive neurogenesis, and neuronal cells located in the DG, which is the center of hippocampal memory consolidation, play a pivotal role in memory and learning.47,65,66 Consistently, the Y maze test found significant deficits in the hippocampus-dependent working memory performance in HHCY-CCI rats on days 1 and 7 post-injury that did not exist in NHCY-CCI rats. Although HHCY-CCI rats showed improvement thereafter, they maintained at least a 15% deficit up to day 28 post-injury. In agreement with our observation, prior studies reported that HHCY exacerbated memory deficits in other neurological disorders, including stroke and Alzheimer’s disease.67,68 We also observed a non-significant decline in working memory performance in HHCY-sham rats occurring later on day 14-pi, underscoring the worsening of TBI pathological outcomes by HHCY.
Although this study focused on the effects of HHCY on the hippocampus, a limitation is that other brain regions, such as the prefrontal cortex and amygdala, mainly associated with changes in anxiety-like behaviors, are also vulnerable to stress. In addition, we only used male rats in this study. Although males represent at least 60% of all TBI cases, females with the same TBI severity experience worse symptoms than males.69–71
Conclusions
In summary, we used in this study a CCI model of mTBI defined by no breaking of the dura, transient behavioral deficits associated with pathological changes mostly confined around the lesion side or the perivascular and periventricular regions.6,7 Findings demonstrate that moderate HHCY exacerbated mTBI-associated pathological markers. They included the markers of oxidative stress, BBB integrity, and cell death in the hippocampus of rats with mTBI. All these HHCY-associated pathological changes after TBI caused significant working memory deficits. HHCY also reduced neuronal density in DG, resulting in a delayed decline in working memory performance. Together, these findings suggest that HHCY is an epigenetic factor that modulates mTBI pathological heterogeneity in the hippocampus without causing a modification of the DNA sequence. Therefore, HHCY represents a putative therapeutic target.
Footnotes
Acknowledgements
We acknowledge Ms Fengying Li and Mr. William Jackson for their technical assistance.
Ethics statement
This was an in vivo study, and all animal housing and experiments were conducted following the institutional Guidelines for Care and Use of Laboratory Animals as well as the ARRIVE guidelines. All procedures were approved by the University of Maryland, Baltimore, Animal Use and Care Committee (IACUC # 1118014), and the US. Air Force Surgeon General’s Office of Research Oversight and Compliance. These experiment were conducted in a facility accredited by AAALAC, in accordance with the Guide for the Care and Use of Laboratory Animals (NRC, 2011), and were performed in compliance with DODI 3216.1.
Authorship contribution
Flaubert Tchantchou: Conceptualization, Formal analysis, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Ru-ching Hsia: Conceptualization, Methodology, Visualization, Writing – review and editing, Adam Puche: Conceptualization, Methodology, Writing –” review and editing, Gary Fiskum: Conceptualization, Formal analysis, Methodology, Writing – review and editing
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the US Air Force, FA8650-17-2-6H10
Availability of data and materials:
Data are available in ftchantchou OpenDrive, University of Maryland School of Medicine
