Abstract
Background:
United States service members injured in combat theatre are often aeromedically evacuated within a few days to regional military hospitals. Animal and epidemiological research indicates that early exposure to flight hypobaria may worsen brain and other injuries. The mechanisms by which secondary exposure to hypobaria worsen trauma outcomes are not well elucidated. This study tested the hypothesis that hypobaria-induced oxidative stress and associated changes in homocysteine levels play a role in traumatic brain injury (TBI) pathological progression caused by hypobaria.
Methods:
Male Sprague Dawley rats were exposed to a 6 h hypobaria 24 h after mild TBI by the controlled cortical impact. Plasma and brain tissues were assessed for homocysteine levels, oxidative stress markers or glutathione metabolism, and behavioral deficits post-injury in the absence and presence of hypobaria exposure.
Results:
We found that hypobaria after TBI increased oxidative stress markers, altered homocysteine metabolism, and promoted glutathione oxidation. Increased glutathione metabolism was driven by differential upregulation of glutathione metabolizing genes. These changes correlated with increased anxiety-like behavior.
Conclusion:
These data provide evidence that hypobaria exposure after TBI increases oxidative stress and alters homocysteine elimination likely through enhanced glutathione metabolism. This pathway may represent a compensatory mechanism to attenuate free radical formation. Thus, hypobaria-induced enhancement of glutathione metabolism represents a potential therapeutic target for TBI management.
Introduction
Traumatic brain injury (TBI) is a public health problem with more than 175 000 victims among United States service members during the Iraq and Afghanistan wars and this number continues to grow as some of them remain in combat zones. 1 Wounded service members, including approximately 10% who suffered from psychiatric disorders such as TBI, were aeromedically evacuated to hospitals in select countries for advanced care critical for survival between 2001 and 2013. 2 During these up to 9 h flights, the cabin pressure is usually maintained at the equivalent to 8000 ft altitude. The mild hypoxia that accompanies such hypobaric conditions is thought to be one of several stressors that could cause secondary injury to TBI patients during these flights.3,4
Over the past decade, there has been significant interest in understanding the impact of hypobaria on TBI pathological progression using various injury models and hypobaria conditions. Mice who underwent 5 h hypobaria (8000 ft) after a closed head injury 3 h earlier presented worse outcomes in cerebral inflammation and neuronal death than those exposed to hypobaria a day later. 5 In a recent report, rats subjected to blast overpressure and exposed 48 h later to hypobaria for 12 h presented with exacerbation of multi-organ injuries, including cerebral lesions. 6 Work from our laboratory showed that 6 h exposure to 8000 ft hypobaria worsened axonal injury in rats subjected to under-vehicle blast. 7 Furthermore, hypobaria-exposure aggravated inflammation, neuronal death, and cognitive deficits in rats subjected to TBI by lateral fluid percussion and exposed to hypobaria up to 7 days post-injury. 8 Despite current advances in understanding the impact of hypobaria-exposure on TBI pathophysiology, hypobaria-associated biochemical changes and their effects on injury pathological outcomes remain unclear.
Elevated levels of the non-proteinogenic amino acid homocysteine (HCY), known as hyperhomocysteinemia (HHCY), is a stress biomarker and stress inducer with substantial neurotoxic properties.9-13 Homocysteine metabolism is tightly regulated to ensure it is continuously remethylated to form methionine, its precursor, or transsulfurated to contribute to glutathione formation, an essential endogenous antioxidant.14,15 There is very limited data on homocysteine metabolism and pathological impact in hypobaria. A previous study by Sharma and colleagues reported that prolonged stay at high altitude (7500 ft) for 3 months, showing an increase in homocysteine levels. 16 We recently reported in our model of methionine-induced moderate HHCY worsens TBI-induced lesion volume, BBB disruption, inflammation, anxiety-like behavior, and working memory in a rat model of mild TBI by controlled cortical impact. 17 In a prior retrospective study involving 150 participants, Rahmani et al 18 found a significant correlation between plasma HCY levels, TBI severity, and associated mortality. In the current study, we tested the hypothesis that hypobaria-induced oxidative stress and associated changes in homocysteine levels play a role in traumatic brain injury (TBI) pathological progression caused by hypobaria.
Mild traumatic brain injury (mTBI) model used in this study is unlike moderate and severe TBI, defined in rodents by no breaking of the dura, subtle pathological changes around the impact region and fast recovery from behavioral deficits.17,19
Materials and Methods
Animals and housing
Male Sprague Dawley rats (300-350 g; Envigo, CA) were used. They were housed in pairs in a temperature-controlled environment, maintained at 23 ± 2°C, with a 12 h light/dark cycle, with continuous access to food and water
Induction of controlled cortical impact injury
The Controlled Cortical Impact (CCI) method was used to induce mTBI.17,19 Animals were maintained under general anesthesia with 2.5% isoflurane throughout surgery. Rat body temperature was maintained at 37°C using a heating pad coupled with a rectal probe. Following craniotomy, CCI was performed over the left parietal cortex. Using a stereotaxically positioned 4 mm diameter piston tip, impact-injury was performed with an electromagnetically controlled impactor (Leica Biosystems, IL) at a piston velocity of 5 m/sec and depth penetration of 1.5 mm. The bone flap was replaced, the skin closed, and the anesthesia terminated. Sham-operated rats were also anesthetized, followed by craniotomy and replacement of the bone flap in the absence of cortical impact.
Rat exposure to hypobaria
At twenty-four hours post-surgery, rats were housed in cages with ad libitum food and water access and placed in a hypobaria chamber. A vacuum pump connected to the chamber was gradually adjusted to depressurize the chamber to 568 mmHg, which is the ambient pressure at 8000 ft., altitude. As previously reported, the chamber was kept normoxic (21% O2) with supplemental O2, were at a near-normal inspired oxygen level of 30%, the SpO2 was 97.0% at a hypobaria level 8000 ft. Rats were maintained under these conditions for 6 h, followed by depressurization every 15 min to sea level ambient pressure (757 mm Hg). 7 As control, other rats were placed in the chamber under normobaric conditions, equivalent to 80 ft., altitude, 21% O2 for the same duration. Some sham or CCI rats (6/group) received intraperitoneal injections of 2-dihydroethidium (2-DHE) in 50% DMSO (6 mg/kg) to track superoxide formation 20 during hypobaria or normobaria-exposure.
Behavioral testing
All behavioral tests were performed at time points described in the experimental time (Figure 1A)

Hypobaria exposure enhances plasma homocysteine elimination: (A) a detailed timeline of behavioral tests and endpoints of experimental procedures starting on day 4 before surgery (study day—4). BW, beam walk test; CCI, controlled cortical impact; HB, hypobaria exposure; NB, normobaria exposure; PM, plus-maze test and (B) histogram of homocysteine quantitation showing that hypobaria enhanced its elimination in sham rats. Homocysteine elimination was potentiated, leading to scanty or undetectable levels among hypobaric CCI-rats (
Beam walk test
This test was performed to assess fine motor movements, as previously described.21,22 Briefly, animals were trained to walk on a 1.5 mm wooden beam for 3 consecutive days, and baseline data determining the number of foot faults over a total of fifty steps were collected on day 4. 22 Test to assess the effect of hypobaria-exposure on fine motor movement in sham and CCI rats overtime was performed on days 3, 9, 16, and 30 post-surgery.
Elevated Plus maze test
The Elevated Plus maze was used to assess rats for anxiety-like behavior on days 2, 8, 15, and 29 post-surgery using a Plus-sign shape device as previously described.23,24 Briefly, rats were individually placed in the central area of the device and allowed to explore the maze for 10 min. Animal movements were analyzed by an overhead camera and data collected and analyzed by Any-Maze software (SD instrument, CA) to provide several behavioral parameters including the time spent in open arms. Anxiety-like behavior was interpreted as inversely proportional to the time an animal spent in the open arms.
Tissue collection and processing
Brain and blood were collected at 2 h, 24 h, or 30-days post-surgery. Rats were anesthetized and blood collected and processed to obtain plasma for biochemical assays as previously described.17,24 Rats were thereafter euthanized and brain collected and processed for slicing in 40 µm thick coronal sections stored at −20°C for histology as described.17,24
Plasma homocysteine measurement
Homocysteine levels were measured in rat’s plasma collected 2 h or 24 h post-hypobaria or normobaria exposure (n = 4/group). The assay was performed using an enzymatic assay kit (BioVision Inc., CA) following the manufacturer’s instructions.
Determination of oxidative stress markers in plasma and brain
Malondialdehyde measurements
Lipid peroxidation levels were determined by measuring the amount of thiobarbituric acid reactive substance (TBARS) malondialdehyde (MDA) in rat plasma samples (n = 6/group). The assay was performed using a kit (Cayman Chemical, MI) following the manufacturer’s instructions.
Superoxide production
The formation of superoxide anion was monitored in rat brains during hypobaria using 2-dihydroethidium (Molecular Probes, OR), a fluorescent probe that incorporates between nucleic acid strands of superoxide producing cells. 20 DHE was dissolved in 50% DMSO and administered intraperitoneally at 6 mg/kg immediately before hypobaria or normobaria exposure (n = 4/group). Rats were euthanized at two-hours post-exposure and brain removed, processed, sectioned, and preserved as described.17,24 DHE incorporated cells were microscopically visualized with a 20× objective at 594 nm wavelength.
Measurements of reduced and oxidized plasma glutathione levels
Plasma samples were collected during euthanasia from sham or brain-injured rats at 2 h, 24 h, or 30 days post-hypobaria or normobaria exposure. Total and oxidized glutathione concentration was measured in rat plasma samples (n = 4/group) using a colorimetric assay kit (Life Technologies Inc, MD). In brief, the assay used a colorimetric substrate that reacted with the free thiol group on glutathione to generate a highly colored product. Reduced glutathione concentration was the difference between total and oxidized plasma glutathione concentration measured separately, following the manufacturer’s instructions.
Transcriptional analysis of glutathione metabolizing genes
Total RNA was extracted from snap-frozen samples of the ipsilateral prefrontal cortex, parietal cortex, and hippocampus from sham and CCI-rats euthanized 2 h post-hypobaria or normobaria- exposure (n = 4/group). RNA extraction was done using the PureLink RNA Mini kit (Ambion/Life Technologies Inc., CA) following the manufacturer’s instructions. Extracted RNA concentration was determined using a Spectrophotometer Nanodrop 2000C (Thermo Scientific, CA), and RNA tubes were stored immediately at −80°C until used.
First, an Access RT-PCR system kit (Promega, WI) was used to amplify 2 µg RNA and determine the optimal annealing temperature of primer sets designed for the quantitative amplification of glutathione metabolizing genes
Presentation of forward (F) and reverse (R) primer sequences used for transcriptional analysis of glutathione metabolizing genes GS, GPX-1, and GPX-4. The table also shows the annealing temperature of these primers, their respective target amplicon size, and the mRNA sequence’s access code from which each primer set was derived.
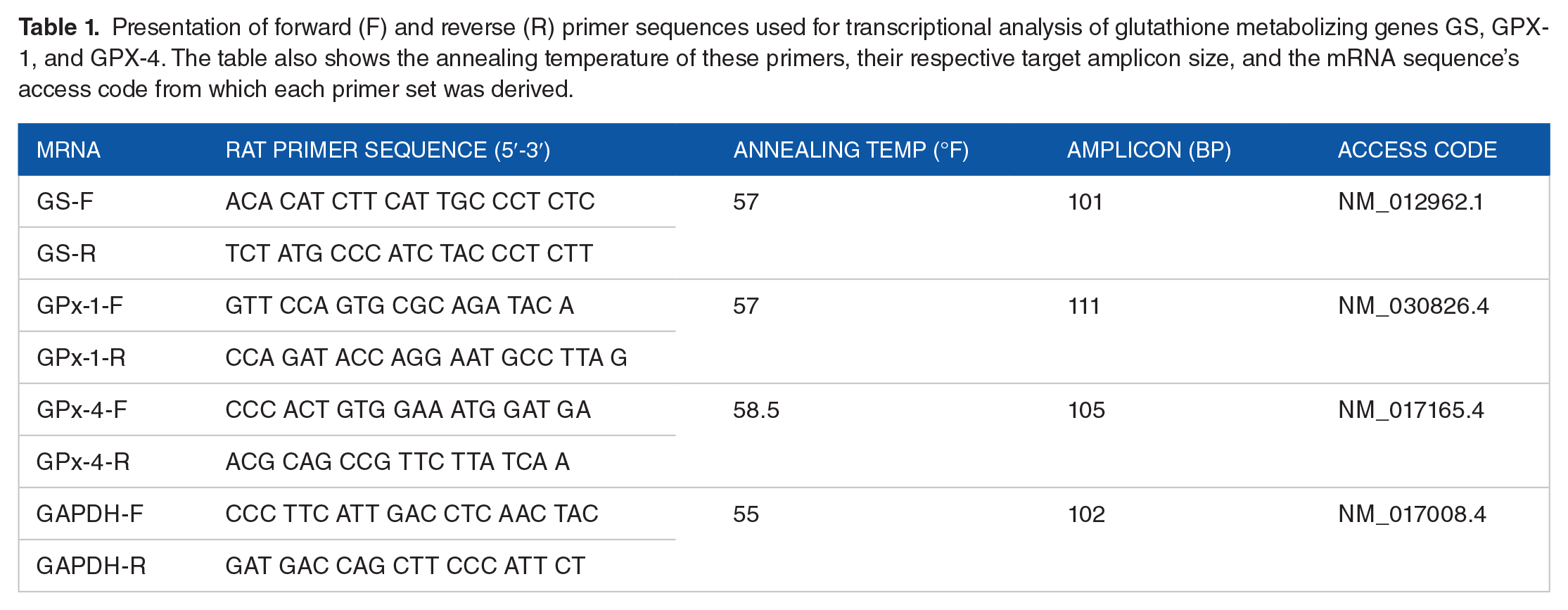
Quantitative reverse transcriptase-polymerase chain reaction (Q-RT-PCR) was performed using the Power SYBR Green RNA-to-CT 1-Step kit (Applied Biosystem, MA). In brief, a complete RT-PCR mix containing 150 nM of primer (IDT, CA) that yielded the best annealing temperature for the respective GS, GPx-1, or GPx-4 gene of interest and GAPDH control was mixed with approximately 15 µg of individual RNA template and placed in a 96 wells plate. Q-RT-PCR test of samples in the 96 wells plate was run in a QuantStudio-3 machine (Applied Biosystems, CA) for a total of 35 cycles after reverse transcriptase reaction at 48°C for 30 min and enzyme activation at 95°C for 10 min (n = 4/group). Each of the 35 Q-PCR cycles consisted of 15-s denaturation at 95°C, and 1 min annealing/extension at the temperature noted in Table 1. Each sample was run in triplicate for the target genes GS, Gpx-1, or GPx-4 and duplicate for the reference gene (GAPDH). At the end of the run, the amplification of a single gene product was verified by the presence of a single peak in the melting curve analysis. Normalized cycle threshold (∆Ct) values were obtained by subtracting the Ct of the reference gene from that of each of the target genes. The double delta Ct (∆∆Ct) for each target gene was determined by deducting the mean ∆Ct of the target gene in the sham normobaric group from the ∆Ct of the respective target gene. The fold-change of each target gene compared to the normobaric sham group was calculated as 2−∆∆Ct.
Lesion volume assessment
Hematoxylin and eosin (Sigma, MO), stained brain sections were dehydrated, cleared, and mounted for lesion volume estimation. Injury lesion volume was quantified from 8 sections corresponding to every 24 serial expanding from bregma 1.05 to −6.30 based to cover the complete lesion area (n = 5/group). Quantification was done using the Cavalieri estimator method of unbiased stereology using the stereo-investigator software (MBF Biosciences, VT). The contusion region (impact-induced damaged region resulting in tissue loss) and penumbra region (the pathologically affected region around impact-induced lesion that could be salvaged with an effective therapeutic intervention) were outlined in the ipsilateral hemisphere of all sections with a grid spacing of 100 µm, and the injury lesion volume was determined and expressed in mm3.25,26
Quantitation and statistical analysis
Image J software (NIH) was used to determine the percent area covered by DHE incorporated cells. To achieve that, 30 microscopic fields of each serial section from DHE or saline-injected rats were captured with SRS fractionator of the stereo-investigator software (MBF Biosciences, VT). Each image was optically segmented with a threshold greyscale value of 0.3 μm, and the percent DHE-positive area was automatically measured. The mean of all DHE positive microscopic fields was determined.
Statistical analyses were performed using GraphPad InStat-3 software (GraphPad Software, Inc., CA). Data from biochemical and histological tests were analyzed using One-Way ANOVA and repeated ANOVA for behavioral data. Both tests were coupled to the Tukey-Kramer post-test to compare differences among the various groups. Results were expressed as mean ± standard error of the mean (SEM). Statistical significance was defined as
Results
Hypobaria exposure reduces circulating homocysteine levels
To determine the effect of acute hypobaria-exposure on HCY metabolism, we measured plasma HCY levels at 2 h and 24 h after 6 h of 8000 ft hypobaria-exposure using normobaria-exposure as control. We found that mTBI alone significantly reduced plasma HCY levels in normobaria-rats after 2 h after exposure, 1.41 ± 0.723 µM, compared to normobaria-shams, 4.32 ± 0.431 µM (Figure 1;
Hypobaria-induced increases oxidized glutathione
We measured reduced and oxidized glutathione levels in that plasma. This showed that hypobaria-exposure caused a modest increase in oxidized glutathione in sham rats at 2 h post-exposure (1.25 ± 0.23 µM) compared to normobaria-shams (0.76 ± 0.19 µM;

Hypobaria exposure enhances glutathione oxidation: (A) quantitative graph of oxidized glutathione levels showing that glutathione oxidation shows a non-significant increase by hypobaria at 2 h post-exposure in sham rats (
The ratio of oxidized to reduced glutathione is an important metric of the metabolic impact of TBI and hypobaria on glutathione metabolism. When we assessed the ratio of oxidized to reduced glutathione, we found that hypobaria-exposure substantially enhanced oxidized glutathione over reduced glutathione in both sham and CCI rats resulting in 1.57 ± 0.202 and 2.38 ± 0.323-ratios respectively, compared to 0.741 ± 0.164 µM and 0.99 ± 0.22 µM for normobaria-sham and CCI rats (Figure 2B,
Hypobaria induces upregulation of glutathione metabolizing genes
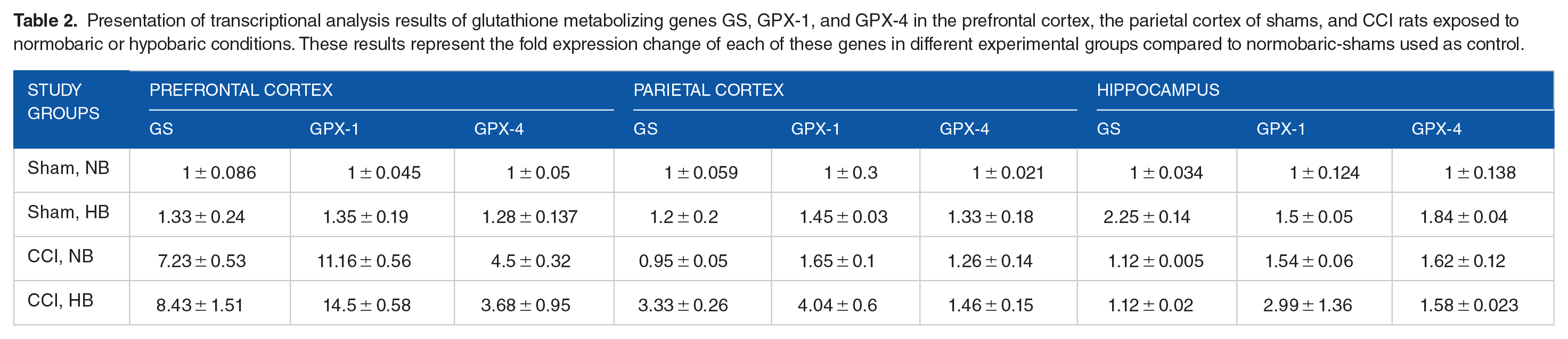
We examined the transcriptional profile GS, GPx-1, and GPx-4 in 3 regions of the ipsilateral hemisphere, the prefrontal cortex, parietal cortex, and hippocampus, at 2 h post-exposure, to determine how their expression responds to hypobaria-exposure. All results are presented in Table 2. These results show a heterogeneous expression pattern in different brain regions. In the prefrontal cortex, the expression of all 3 genes was enhanced by the cortical impact-induced brain injury (Table 2; Figure 3A–C;
Presentation of transcriptional analysis results of glutathione metabolizing genes GS, GPX-1, and GPX-4 in the prefrontal cortex, the parietal cortex of shams, and CCI rats exposed to normobaric or hypobaric conditions. These results represent the fold expression change of each of these genes in different experimental groups compared to normobaric-shams used as control.

Glutathione metabolic genes expression were differentially altered by hypobaria exposure and mTBI. Histograms depicting changes in the transcriptional analysis of
In contrast, the combination of cortical brain injury and hypobaria-exposure upregulated the expression of GS and GPX-1 in the parietal cortex compared to normobaric brain-injured rats as well as hypobaric and normobaric shams (Table 2; Figure 3D, E;
Hypobaric conditions increase oxidative stress
The biochemical markers above indicate that there may be considerable oxidative stress causing a compensatory reduction in HCY and elevation in the processing of glutathione antioxidant systems. Consistent with this conjecture, we found that TBI caused a non-significant 12.3 ± 5.23% increase in plasma malondialdehyde levels in normobaria-CCI rats at 2 h post-exposure compared to normobaria-shams (Figure 4;

Hypobaria exacerbates lipid peroxidation in rats with TBI. Plasma malondialdehyde levels were a non-significant increase in normobaric-CCI rats 2 h post-exposure. Hypobaria-exposure caused a significant increase of this molecule in CCI rats plasma simultaneously (

Superoxide formation intensifies in rat with brain injury after hypobaria exposure: (A) representative microscopic images showing 2-DHE incorporation in superoxide producing cells in the injury penumbra region and the hippocampus dentate gyrus. Quantitation of the %area of 2-DHE positive cells and (B) indicates that brain injury alone caused a significantly increased superoxide formation (
TBI lesion volume was not significantly affected by hypobaria exposure
To assess the effect of hypobaria-exposure on brain injury lesion, we used the Cavalieri method of unbiased stereology to measure injury-induced lesion volume at 2 h and 24 h post-exposure. At 2 h post-exposure, impact-induced injury lesion volume was approximately the same between normobaric, 6.56 ± 2.25 mm3, and hypobaric-CCI rats, 7.09 ± 1.42 mm3 (Figure 6A, B;

Hypobaria exposure does not affect TBI-induced cortical lesion: (A) Representative hematoxylin and eosin-stained coronal brain sections showing cortical impact-induced brain lesions. (B) Lesion volume quantitation indicating that hypobaria did not affect CCI-induced cortical brain lesion 2 h post-exposure. However, 24 h post-exposure, the lesion was greater but non-significant in hypobaric rat brains (
Hypobaria exposure increases anxiety-like behavior in rats with mild TBI
Next, we wanted to determine whether hypobaria exposure following mTBI in animals exacerbates behavioral deficits. We assessed the effect of hypobaria-exposure on anxiety-like behavior usually associated with TBI-related psychiatric impairment such as posttraumatic stress disorder.29,30 Anxiety was assessed with a standard elevated plus-maze in which reduced time spent in the open arms of the maze is interpreted as elevated anxiety-like behavior. As a control, fine motor behaviors classically associated with cerebellar function, a brain region removed from the injury site, were not significantly affected as assessed by the Beam Walk test. On day 2 post-exposure, which is an early time point when changes due to hypobaria are mostly likely detectable, normobaric sham and CCI rats respectively made 1.28 ± 0.98 and 1.87 ± 0.62-foot faults over 50 steps. In comparison, hypobaric sham and CCI rats made 1.39 ± 0.75 and 1.77 ± 1.02 foot faults (
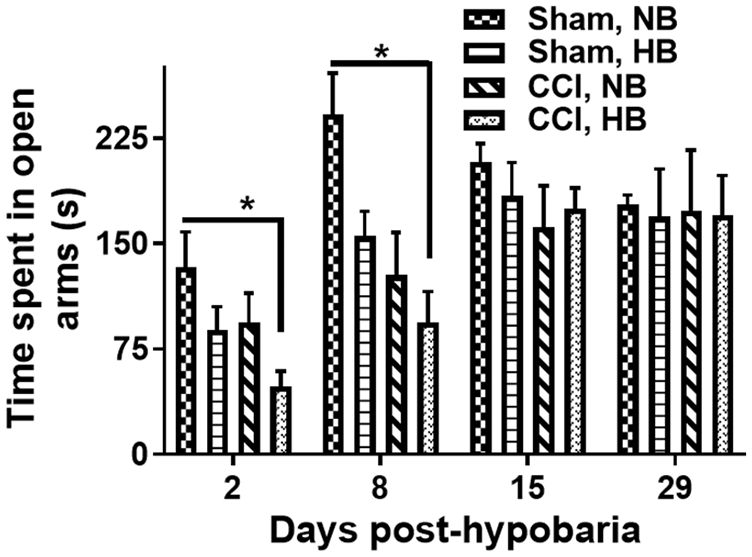
Traumatic brain injury-induced anxiety-like behavior is aggravated by hypobaria exposure. Graph depicting the time spent by rats in the open arms of the elevated plus maze. Both brain injury and hypobaria alone caused a non-significant in anxiety-like behavior. The combination of mTBI and hypobaria significantly increased anxiety-like behavior on days 2 and 8 post-surgery compared to normobaric-shams (
Discussions
Reports from several investigators suggest that flying at high altitude is associated with increased physiological stress,31,32 thus representing a risk for TBI patients who require aeromedical evacuation for advanced care. Results from the present study indicate that exposure of rats with mTBI, which represents up to 80% of all TBI cases,33,34 to hypobaria induces an acute increase of neural oxidative stress, which subsequently triggers an enhanced response of the antioxidant defense system that uses HCY and glutathione in a compensatory attempt to attenuate free radical formation.
Levels of oxidative stress markers malondialdehyde and superoxide increased in rats subjected to mTBI 2 h post-hypobaria (Figures 4 and 5). This observation suggested that oxidative damage during aeromedical evacuation produced changes that could represent targets for therapeutic interventions. This is a key concept, as unmitigated free radical formation mediates the induction of many TBI pathological markers including, inflammation and neuronal degeneration that are exacerbated by hypobaria-exposure.5-8,35,36 Therefore, our findings suggest that hypobaria-induced oxidative stress is a significant mediator or indicator of TBI pathological progression during and after hypobaria exposure.
Hypobaria-induced oxidative stress was coupled with very low to undetectable levels of plasma homocysteine (Figure 1). Homocysteine is a non-proteinogenic amino acid with substantial cytotoxic potential when accumulated.37-39 Therefore, its metabolism is tightly regulated to ensure it is continuously eliminated.15,40 This occurs via its remethylation to methionine, its precursor, or by its transsulfuration to cystathionine, contributing to the downstream formation of glutathione, a very potent antioxidant.14,15 Therefore, we postulated that the drastic reduction of homocysteine levels under oxidative stress at 2-h post-hypobaria was indicative of its flush through the transsulfuration pathway. This hypothesis was potentiated by a significant increase of glutathione oxidation resulting in a two-fold increase of oxidized glutathione versus reduced glutathione at 2 h post-hypobaria exposure (Figure 2A, B). The return of oxidized glutathione to baseline levels at day 30 post-exposure suggests an attenuation of oxidative stress. This observation correlates with the significant improvement in anxiety-like behavior to that of normobaric-sham rats. The therapeutic potential of homocysteine transsulfuration to promote glutathione formation has been underscored in several neurological disorders. For instance, the supplementation of N-acetylcysteine to promote glutathione biosynthesis through the transsulfuration pathway was neuroprotective in models of Huntington’s disease,41,42 Alzheimer’s disease,43,44 and TBI.45,46 Moreover, this therapeutic approach is in a trial for pediatric TBI management. 47
Glutathione has free radical scavenging and anti-inflammatory properties.48,49 Its metabolism is regulated by genes, including
Although the mechanism for the distinctive expression patterns in different brain regions is not fully elucidated, increased GPx expression in the cerebral cortex of aged rats was associated with higher astrocyte density.
58
Furthermore, GPx activity was derived primarily from astrocytes relative to neurons in chicks’ forebrain.
59
Also, increased
In summary, this study provides evidence that hypobaria exacerbates oxidative stress in rats with mTBI. This was associated with an enhanced flush of homocysteine through the transsulfuration pathway to promote glutathione biosynthesis, which is necessary to attenuate free radical formation. Increased glutathione metabolism was putatively driven by the upregulation of glutathione metabolizing genes, suggesting that enhanced glutathione metabolism represents a therapeutic target for TBI management. Further studies would be needed to understand how this antioxidant redox system would respond to interventions during and after hypobaria-exposure following moderate to severe TBI.
Footnotes
Acknowledgements
We acknowledge Ms. Fengying Li and Ms. Lyric Ramsue for their technical assistance
Author Contributions
Experimental design: FT, GF, CM; Laboratory experiments and data collection: FT, MG; Data analysis: FT, AP, GF, MG; Manuscript writing: FT, GF; Manuscript editing: AP, MG, FT.
Funding:
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Defense, U.S Air Force, Grant # FA8650-17-2-6H10
Declaration of conflicting interests:
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
