Abstract
Tramadol is a weak mu (µ) opioid receptor agonist that acts by inhibiting serotonin and norepinephrine uptake. Tramadol undergoes extensive hepatic metabolism by a number of pathways, including CYP2D6 and CYP3A4, and by conjugation with subsequent renal excretion. The maximum recommended dose is 400 mg/day. One of the most important adverse effects of tramadol is a seizure, which usually occurs at high doses and is often generalized tonic
Introduction
Tramadol is a weak mu (µ)-opioid receptor agonist that acts by inhibiting serotonin and norepinephrine reuptake. 1 Tramadol undergoes extensive hepatic metabolism by a number of pathways, including CYP2D6 and CYP3A4, and by conjugation with subsequent renal excretion. Tramadol is composed of a 1:1 racemic mixture of (+) -enantiomers and (-) +enantiomers.
(+) -Tramadol inhibits serotonin reuptake and (-) -tramadol inhibits norepinephrine reuptake. The major metabolite of (+) -tramadol activates the mu (µ) receptor.
2
The range of blood levels in adults is approximately 100 ng/mL to 300 ng/mL (0.1–0.2 µg/mL). The recommended dose of 400 mg/day
3
is considered as the maximum approved dose. Tramadol hydrochloride is a lipophilic substance that completely penetrates the barrier between the blood and the brain. The level of plasma besieges a peak around 1.5 h after taking, and 5 h to 6 h is the half-life of plasma exclusion. One of the complications of tramadol use is the seizure, which is most often generalized tonic
Case History
A 29-year-old male patient came to the Orthopedic OPD, with a history of low back pain following a fall from height. He was prescribed oral tablets of tramadol hydrochloride 37.5 mg twice daily for five days. After 1 h of taking the first dose of tablet tramadol, he developed morbilliform rashes all over the body. One day later, he developed generalized tonic
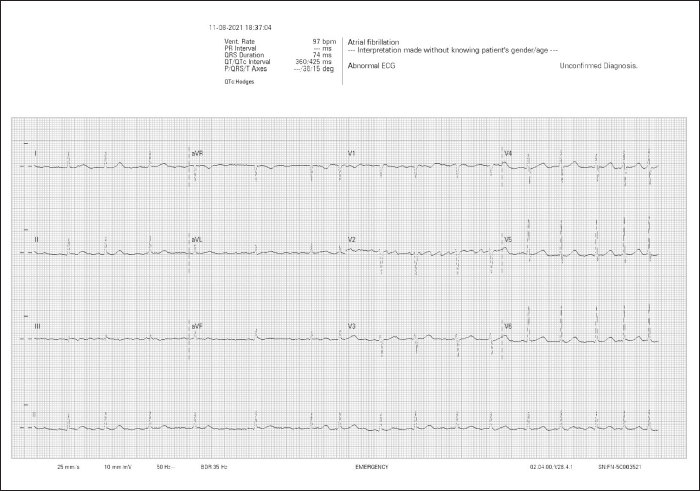
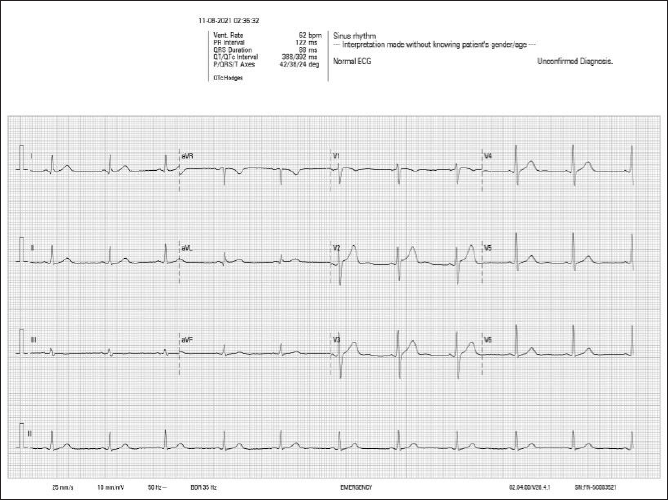
Discussion
Tramadol is a “bimodal” agent that possesses activity in both opioid and monoaminergic (serotonergic and nonadrenergic) pathways in the central nervous system (CNS). Tramadol is commonly used in place of other potent opioid analgesics for the treatment of moderate to moderately severe pain associated with osteoarthritis, rheumatoid arthritis, low back pain, and neuropathic conditions.4, 5 There are controversies about the seizure-inducing effect of tramadol. Some earlier studies suggested that tramadol when given in overdose in patients with extant disorder of seizure or when used along with antidepressants, alcohol, etc., instigates seizure.6, 7 It was also disclosed from other studies that tramadol also instigates seizures when used as monotherapy in an approved dose. It has also been found that the appearance of seizures following tramadol use is not dose dependant.
8
However, in our patient, the seizure occurred at a dose of 37.5 mg oral tramadol hydrochloride. Tramadol-induced seizures have been reported to be generalized tonic
Conclusion
Tramadol has been otherwise a safe drug for many years; however, with evidence of serious reactions, like seizures even at low doses, the scientific community should consider pharmacovigilance with respect to its use, especially in developing countries where such monitoring systems are inadequate.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
