Abstract
Tramadol (TR) is a synthetic analgesic drug with central function that can induce seizures even at therapeutic doses. The exact mechanism of TR effect on seizure generation is not clear, but inhibition of the serotonin and nitric oxide pathways and inhibitory effects on GABA receptors are the most common hypotheses about the seizure-inducing mechanism of the TR. This study aimed to evaluate the role of dopaminergic system on the seizure and oxidative damage induced by TR using agonist and antagonist drugs of this system in the Albino mice. Clonic seizure induced by TR was evaluated as seizure threshold. Haloperidol (0.2 mg/kg, IP), a predominantly D2 receptor antagonist, and cabergolin (0.5 mg/kg, IP), a dopamine agonist specific for the D2 receptors, were injected 60 minutes before the seizure induction. The seizure threshold was significantly increased by dopaminergic antagonist, but it was decreased significantly by pretreatment with the selective agonist. Oxidative stress biomarkers (reactive oxygen species, lipid peroxidation, and protein carbonyl content) significantly increased and glutathione content significantly decreased in brain mitochondria by TR compared with the control group, whereas oxidative markers were decreased significantly after pretreatment with haloperidol compared with the TR group. This study revealed that the dopaminergic system is involved in TR-induced seizure, and meanwhile, inhibition of dopamine D2 receptors can increase the TR threshold seizure and decrease the oxidative damage in the brain mitochondria. Conversely, stimulation of dopamine D2 receptors by cabergolin can decrease the TR threshold seizure and glutathione content in the brain mitochondria.
Introduction
Tramadol (TR; 1RS, 2RS)-2-[(dimethylamino)-methyl]-1-(3-methoxyphenyl)-cyclohexanol is a synthetic analgesic drug with central function that is used both parenterally and orally for severe pain treatment. 1 –3 In patients with normal metabolism, oral TR (100 mg) has superior analgesia with peak effect after 1 to 4 hours of drug administration. 4 Maximum plasma level of the TR occurs after 4 hours. The protein binding of TR is about 20% and 60% of the drug excreted as metabolites in the urine. 5 Among different metabolites of TR, the O-desmethyl-TR is the major one that has 200 times μ-affinity compared with the TR. 6 Dosage of the TR is adjusted according to the pain severity. Generally, therapeutic dose for TR is 50 to 100 mg and its total daily doses should not exceed 400 mg. Tramadol therapeutic blood level is 0.1 to 0.3 mg/L for adults. 7 Although TR, as opposed to typical opioids, does not have a remarkable opioid side effects (eg, respiratory depression, sedation, constipation), sometimes it has unfavorable side effects with respect to seizure potential. 8,9 At therapeutic doses, TR slightly suppresses the severity of seizure. However, at high doses, TR paradoxically induces seizure. 10 Two important mechanisms of action for TR are weak agonistic effect at the μ-opioid receptors and inhibition of the monoamines (serotonin, norepinephrine) reabsorption. 8,11 The neurotoxicity of TR mostly occurs within 24 hours of drug administration and 84.6% of seizures occur within 6 hours after TR consumption. The minimum dose of TR that may induce convulsion is not so clear, but in various studies, it has been reported to be about 200 mg. 12,13 The exact mechanism of TR in the generation of seizure remains to be elucidated, but some studies show the involvement of opioid receptor activation-linked γ-aminobutyric acid (GABA) inhibitory pathway in the convulsant effects of TR. It has been reported that TR, at high concentrations, has inhibitory effect on GABA receptors that can potentiate the severity of seizures in animal models. 14–15 Moreover, inhibition of GABA receptors by TR can be due to its agonistic activity of opioid receptors. 10 Other hypotheses about the seizure-inducing mechanism of TR are inhibition of the serotonin and nitric oxide (NO) pathways. 9,16 –18 Also, opioid receptors’ agonist activity of TR has been confirmed to induce seizures due to GABA pathway inhibition. 19,20 Furthermore, seizures in patients receiving TR at recommended doses have been reported. 15
Oxidative stress is an unavoidable result of the seizure. In epilepsy, seizure can increase oxidative stress in the temporal lobe. 21,22 Oxidative stress and mitochondrial dysfunction can induce seizures and neuronal death. 23 Also, oxidative damage is the consequence of seizures induced by neurodegeneration and/or neurological disease. In addition, seizure can induce neuroinflammation, excitotoxicity, and formation of reactive oxygen species (ROS) 24 in brain cells. Reactive oxygen species that produced status epilepticus demolish the mitochondrial natural antioxidant defense mechanism, which leads to mitochondrial dysfunction and mitochondrial DNA damage. 21
It has been demonstrated that a great number of neurotransmitters are involved in the mechanism action of drugs. 25 Hence, it seems that numerous neural pathways may be involved in TR seizure. Dopaminergic system plays a crucial role in seizure susceptibility and in controlling of neuronal excitability. 26 It was reported that the stimulation of dopaminergic receptors by dopaminergic agents can increase seizure threshold, whereas seizures can be enhanced by dopamine-blocking drugs such as antipsychotics and antidepressants. 27 Although it was revealed that TR decreases the binding of 5-HT2A receptors, frontocortical β-adrenoceptors, and α2-adrenoceptors, 28 to the best of our knowledge, there is not report about the involvement of dopaminergic system in the TR-induced seizure.
With regard to the unclear role of the dopaminergic system in the development of seizure, we decided to study the role of dopaminergic system in induction and severity of the seizure created by TR using agonist and antagonist drugs. Also, effects of stimulation and inhibition of dopaminergic receptors on oxidative stress biomarkers in the brain mitochondria were evaluated in the male Albino mice after inducing seizure by TR.
Materials and Methods
Chemicals
All the chemicals used in this study were prepared of analytical grade and purchased from the Merck (Darmstadt, Germany) and Sigma Chemical Co (St Louis, Missouri). Tramadol from Razak Pharmaceutical Co (Tehran, Iran) haloperidol (HAP) and cabergolin from Shahredaru Co (Tehran, Iran) were prepared.
Experimental Design
Male albino mice (25-30 g) were prepared from animal house of the Faculty of Pharmaceutical at Mazandaran University of Medical Sciences (MAZUMS) and used in this study. Animals were housed in a light-controlled room (12-hour day–night cycle) and were allowed free access to food and water. All procedures were done in accordance with the recommendations on animal experiments by the ethics committee of the Mazandaran University of Medical Sciences with the ethic number: IR.MAZUMS.REC.94.1635. Animals were divided into 4 groups (6 mice in each). The first group was the control group. The second group received only TR at a concentration of 25 mg/mL, which was injected via tail vein until the clonus front limb with a full body clonus was observed. 9,29 The third group was mice that received HAP (0.2 mg/kg, IP), then after an hour TR was injected to this group via tail vein until the clonus front limb with a full body clonus was observed. The fourth group was mice that received Cabergolin (CAB) (0.5 mg/kg, IP) and after an hour TR was injected to this group via tail vein until the clonus front limb with a full body clonus was observed.
Determination of the Seizure Threshold
Mice were located in a mouse restrainer with easy access to the tail. After entering the needle (30-gauge butterfly needle) into the tail vein, it was fixed with a piece of tape on the tail and TR (25 mg/mL) was injected with constant speed (0.5 mL/minute) to the moving mouse until observation of the clonus and the first effect of seizure. Afterward, the required dose of TR to induce seizure was recorded. 30,31
Brain Mitochondria Preparation
Mice were sacrificed after 24 hours of injections, and after splitting the skull, brains were removed and washed with mannitol buffer. Then pieces of whole brain were homogenized in mannitol buffer. Samples were prepared for centrifugation. In the first centrifuge (2,000g for 10 minutes), supernatant was collected, and in the second centrifuge (11,000g for 10 minutes), sediments containing mitochondria were scattered in Tris buffer for evaluation of lipid peroxidation (LPO), glutathione (GSH), and protein carbonyl (PC) tests. Some of the sediment was dispersed in breathing buffer for the assessment of ROS test. 32
Determination of ROS
To determine the ROS generated in the brain mitochondria, dichlorofluorescin (DCFH) diacetate was used as an indicator. Briefly, 2 mL of sample (1 mg protein/mL of mitochondrial fractions) was incubated with DCFH for 15 minutes at 37°C. Then, ROS formation was measured at 480 nm (excitation) and 520 nm (emission) wavelength by fluorescence spectrophotometer (Shimadzu RF5000U). 33,34
Measurement of LPO
The content of malondialdehyde (MDA), as LPO marker, was determined by using the method of Zhang et al. 35 Briefly, 0.25 mL phosphoric acid (0.05 M) was added to the 0.2 mL of sample (0.5 mg protein/mL of mitochondrial fractions), then 0.3 mL of 0.2% thiobarbituric acid was added to it. Samples were kept in a boiling water bath for 30 minutes. Finally, the sample tubes were transferred to the ice bath, n-butanol (0.4 mL) was added to each tube, and samples were centrifuged (3,500g) for 10 minutes. The amount of formed MDA in samples was measured in the supernatant at 532 nm with enzyme-linked immunosorbent assay reader (Tecan, Rainbow Thermo, Austria). Content of MDA was expressed as nmol/mg protein, whereas tetramethoxypropane was applied as standard. 35,36
Measurement of GSH Content
Content of the GSH in the samples was determined by spectrophotometer (UV-1601 PC; Shimadzu, Japan) with 5,5′-dithiobis-2-nitrobenzoic acid as an indicator at 412 nm and expressed as μM. 32
Measurement of PC Content
Content of the PC in the samples was assessed by reading the absorbance at the 365 nm wavelength using spectrophotometer. 32
Measurement of Protein Concentration
Protein content was determined using the Coomassie blue protein-binding method. 37 Bovine serum albumin was used as standard for the measurement of protein content.
Statistical Analysis
Results are presented as mean (SD). All statistical analyses were performed using the GraphPad Prism 6 software. Statistical significance was determined using the one-way analysis of variance test, followed by the post hoc Tukey-Kramer test. Statistical significance was set at P < 0.05.
Results
The effect of stimulation and inhibition of dopaminergic receptors on induced clonic seizure threshold showed significant (F 2,15 = 59.21, P < 0.001) changes between the different groups. Seizure threshold significantly (P < 0.01) increased in the TR + HAP group compared to the TR group. Also seizure threshold in pretreated mice with CAB significantly (P < 0.001) decreased when compared to the TR group. Moreover, seizure threshold was significantly (P < 0.001) decreased in the TR + CAB group when compared to the TR + HAP group (Figure 1).
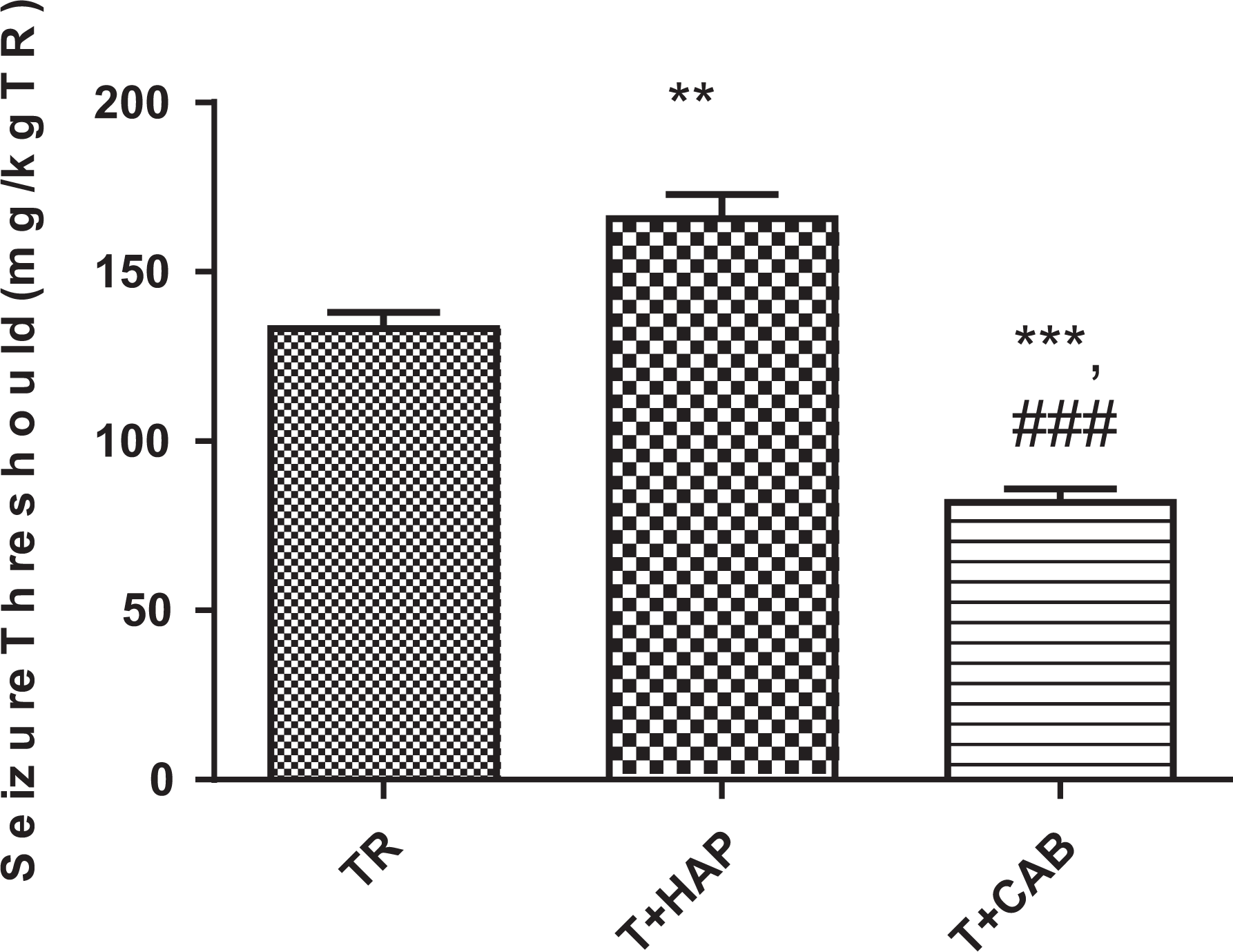
Effect of dopaminergic receptors antagonist and agonist on tramadol (TR)-induced seizure threshold: TR (mice that received TR), TR + HAP (mice that received intraperitoneal injection of haloperidol 60 minutes before determination of the TR-induced seizure threshold); TR + CAB (mice that received intraperitoneal injection of cabergolin 60 minutes before determination of the TR-induced seizure threshold). Values represented as mean (SD; n = 6). **P < 0.01 and ***P < 0.001 compared with tramadol group. ###P < 0.001 compared with the TR + HAP group. HAP denotes haloperidol; TR, tramadol; CAB, Cabergolin; SD, standard deviation.
In this study, ROS formation in mice brain mitochondria showed significant (F 3,20 = 8.565, P = 0.0007) changes between the different groups. Measurement of ROS formation in mice brain mitochondria showed that the levels of ROS were significantly increased (P < 0.01) in the TR group compared with the control group and also ROS levels were significantly decreased (P < 0.01) in the TR + HAP group compared to the TR group. There was no significant change in the amount of produced ROS in the group that received TR + CAB compared with the TR group (Figure 2). HAP denotes haloperidol; TR, tramadol; CAB, Cabergolin; SD, standard deviation.
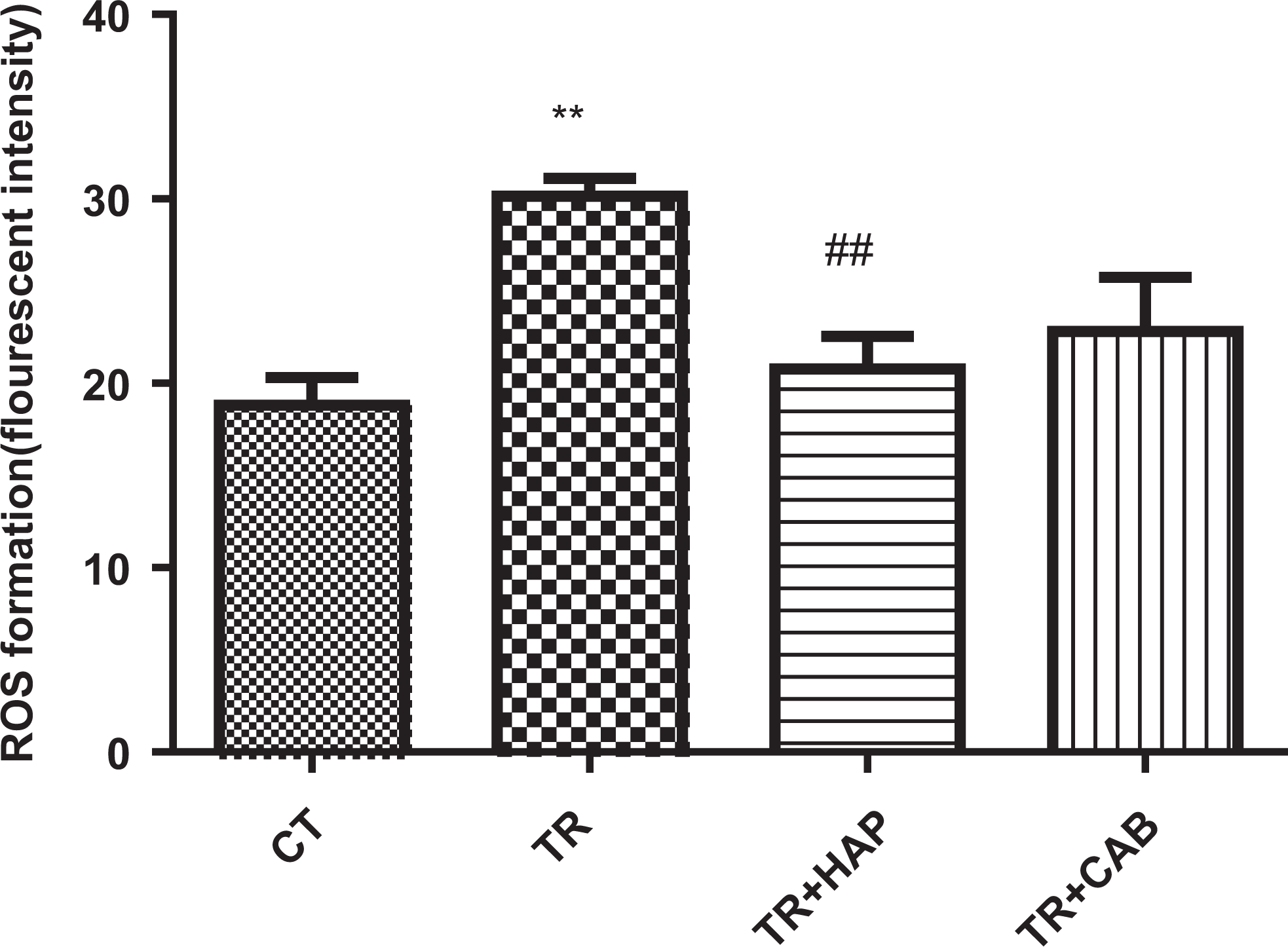
Effect of dopaminergic receptors antagonist and agonist on ROS formation in brain mitochondria: TR (mice that received tramadol), TR + HAP (mice that received intraperitoneal injection of haloperidol 60 minutes before determination of the TR-induced seizure threshold); TR + CAB (mice that received intraperitoneal injection of cabergolin 60 minutes before determination of the TR-induced seizure threshold). Values represented as mean (SD; n = 6). **P < 0.01 compared with the control group. ##P< 0.01 compared with the tramadol group. HAP denotes haloperidol; TR, tramadol; CAB, Cabergolin; ROS, reactive oxygen species; SD, standard deviation.
Significant changes (F 3,20 = 6.487, P = 0.003) were observed in GSH in different groups. Glutathione contents were significantly (P < 0.01) decreased in the TR and TR + CAB groups in comparison with the control group (Figure 3).
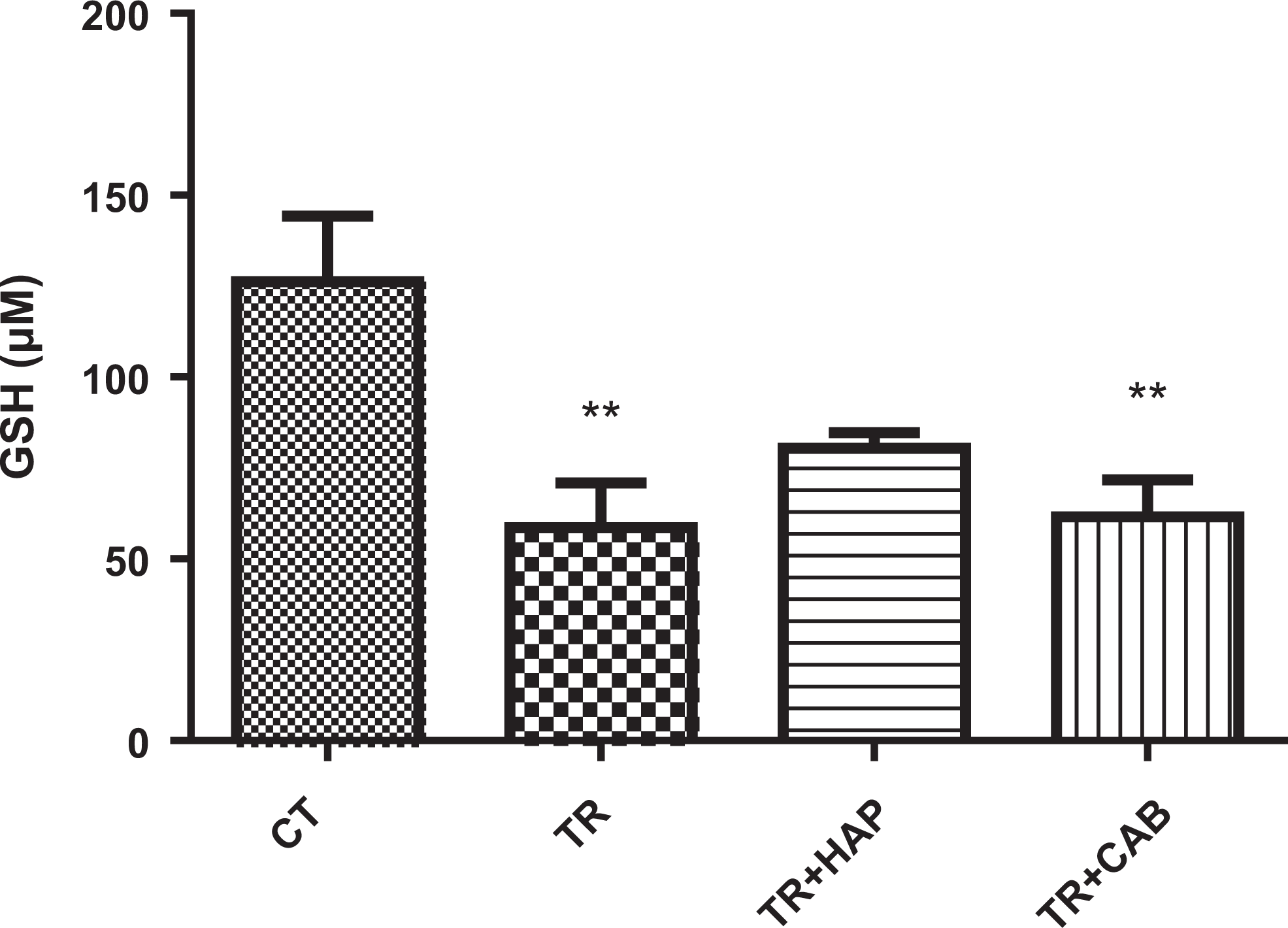
Effect of dopaminergic receptors antagonist and agonist on GSH content in brain mitochondria: TR (mice that received tramadol), TR + HAP (mice that received intraperitoneal injection of haloperidol 60 minutes before determination of the TR-induced seizure threshold); TR + CAB (mice that received intraperitoneal injection of cabergolin 60 minutes before determination of the TR-induced seizure threshold). Values represented as mean (SD; n = 6). **P < 0.01 compared with the control group. GSH denotes glutathione; HAP, haloperidol; TR, tramadol; CAB, Cabergolin; SD, standard deviation.
Protein carbonyl as a marker of protein oxidation was evaluated in this study. The level of mitochondria PC in the brains of male mice was significantly (F 3,20 = 5.441, P = 0.006) changed and it was increased significantly (P < 0.01) in the TR group compared with the control group, whereas it was decreased significantly (P < 0.05) in the TR + HAP group when compared with the TR group (Figure 4).
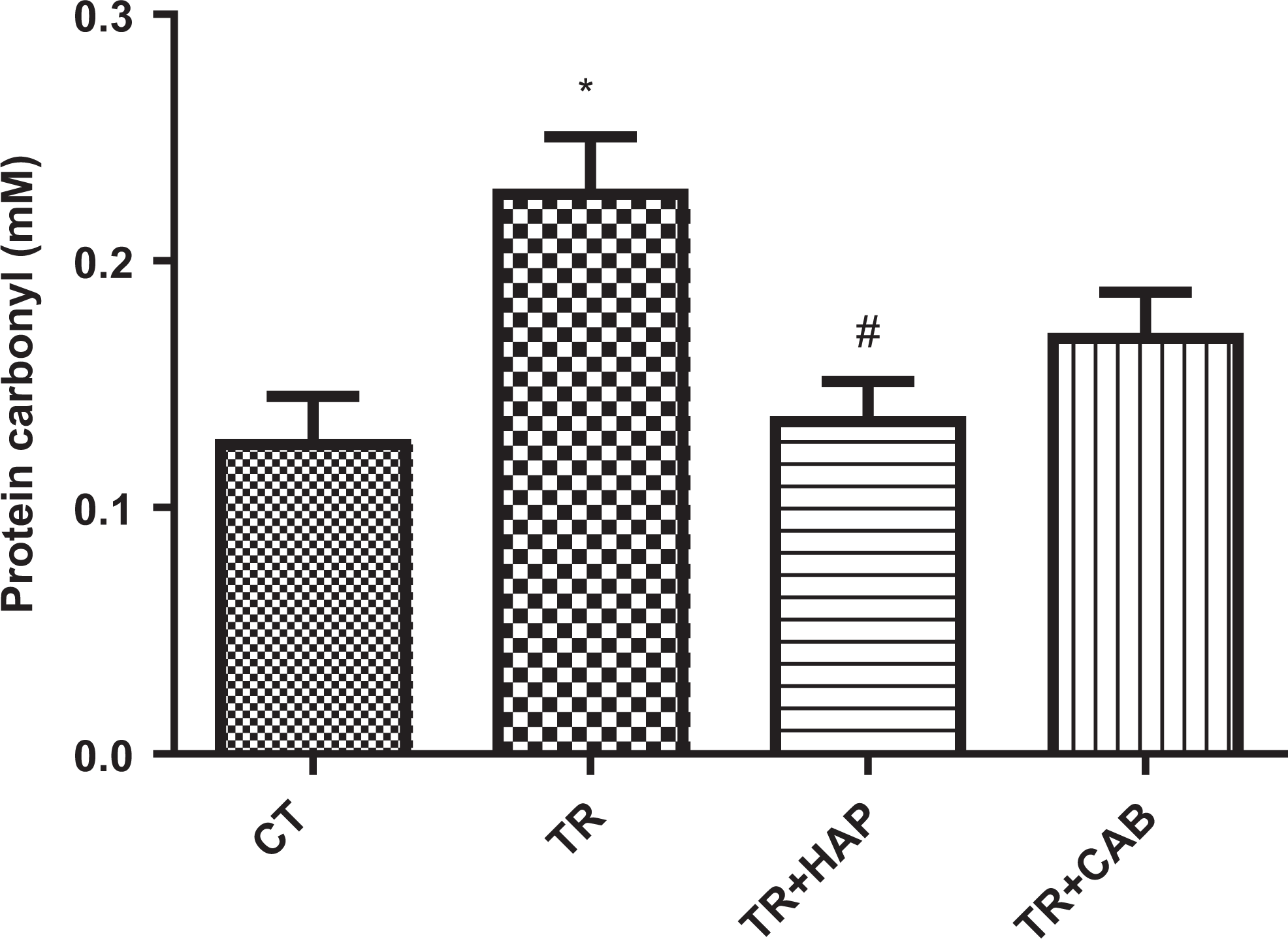
Effect of dopaminergic receptors antagonist and agonist on protein carbonyl in brain mitochondria: TR (mice that received tramadol), TR + HAP (mice that received intraperitoneal injection of haloperidol 60 minutes before determination of the TR-induced seizure threshold); TR + CAB (mice that received intraperitoneal injection of cabergolin 60 minutes before determination of the TR-induced seizure threshold). Values represented as mean (SD; n = 6). *P < 0.05 compared with the control group. #P < 0.05 compared with the tramadol group. HAP denotes haloperidol; TR, tramadol; SD, standard deviation; CAB, Cabergolin.
Level of MDA, as a LPO marker, was determined by measuring the production of MDA in the brain mitochondria and this biomarker showed significant (F 3,20 = 16.17, P < 0.0001) changes in different groups. In this study, the levels of MDA were significantly increased in the TR (P < 0.001), TR + HAP (P < 0.01), and TR + CAB (P < 0.01) groups compared with the control group, but MDA was significantly (P < 0.05) decreased in the TR + HAP and TR + CAB groups when compared with the TR group (Figure 5).
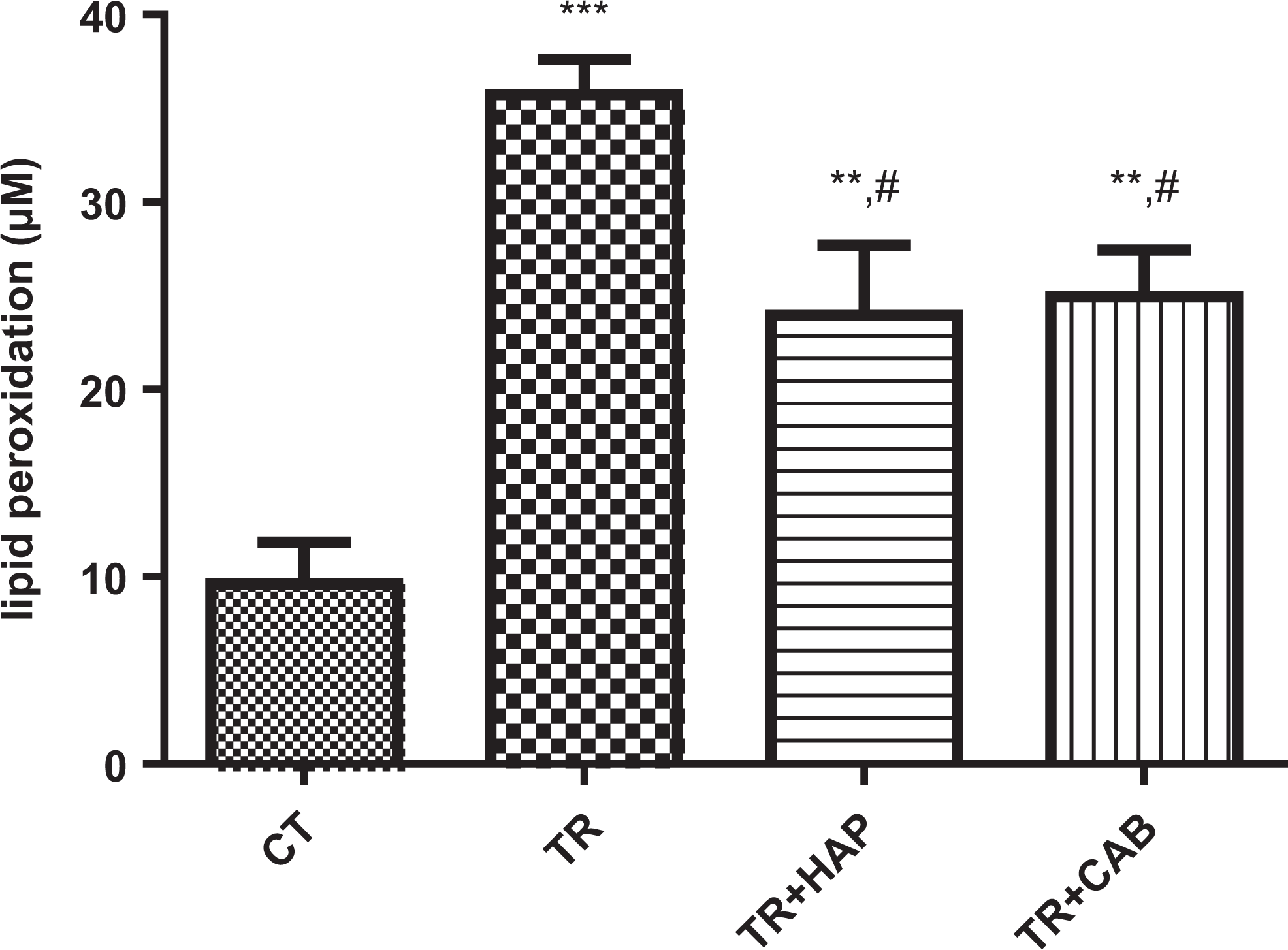
Effect of dopaminergic receptors antagonist and agonist on lipid peroxidation in brain mitochondria: TR (mice that received tramadol), TR + HAP (mice that received intraperitoneal injection of haloperidol 60 minutes before determination of the TR-induced seizure threshold); TR + CAB (mice that received intraperitoneal injection of cabergolin 60 minutes before determination of the TR-induced seizure threshold). Values represented as mean (SD; n = 6). ***P < 0.001 compared with the control group. #P < 0.05 compared with the tramadol group. HAP denotes haloperidol; TR, tramadol; SD, standard deviation; CAB, Cabergolin.
Discussion
The results of this study showed that the dopaminergic system is involved in the seizure and oxidative damage induced by TR. Our study revealed that CAB, a dopaminergic agonist at D2 site, could reduce the seizure threshold, and HAP, a dopaminergic antagonist drug, could increase the seizure threshold consequence of the TR administration. Also, measurement of the oxidative stress biomarkers in the brain mitochondria of mice showed that the inhibition of dopaminergic receptors by HAP could decrease oxidative stress induced by TR associated with seizures.
Administration of TR for severe pain does not have the common side effects of opioids such as respiratory depression, sedation, and constipation, but sometimes TR can induce seizures or increase seizure potential. This drug can induce self-limited seizure even after a single dose of treatment. Unfortunately, the TR abuse is growing in societies due to its euphoric effect. 4,38 Until now, several studies were conducted about TR-induced seizure. Their assumptions about the TR-inducing seizure mechanism include inhibition of the serotonin, GABA, and NO pathways. It was reported that TR can create seizure through inhibition of serotonin reuptake and increased concentration of serotonin to toxic level. 19,39 Also, stimulation of the GABA inhibitory pathways associated with opioid receptors can be effective in TR preseizure. 10 It has been shown that L-NAME inhibits NO synthase and can reduce TR-induced seizures in kindling model. 9 But, to the best of our knowledge, there is no report about the involvement of dopaminergic pathway in TR-induced seizure.
In this study, the role of dopaminergic receptors in seizure created by TR was evaluated in male mice. Haloperidol, a predominantly D2 receptors antagonist and dopaminergic pathway blocker, and CAB, a dopamine agonist specific for the D2 receptor, were used before the seizure induction. Haloperidol increased the threshold seizure and CAB decreased the threshold seizure in this study (Figure 1).
It was reported that a great number of neurotransmitters are involved in the drug’s mechanisms of action. 25 Experimental and clinical studies have shown that different systems such as acetylcholine, serotonin, noradrenaline, and dopamine are involved in the seizure regulation. 26 It has been proven that the dopaminergic system plays a prominent role in the mechanism of seizure propagation. 40 Hence, it seems that numerous neural pathways may be involved in TR seizure. The results of this study showed a significant decrease in the seizure threshold in the group that received CAB before the TR injection compared with the group that received only TR. Also, in comparison with the TR group, a significant increase in seizure threshold was observed in the group that received HAP before TR injection (Figure 1). These results confirm the effect of the dopaminergic system on TR-induced seizures. It was reported that the dopaminergic system plays an important role in mechanisms of seizure propagation and control of the seizures in the limbic system. 40 It has also been reported that signaling from D1-like receptors is usually pro-epileptogenic, while D2-like receptor signaling has anti-epileptogenic effects. Therefore, seizures concerning the limbic system appears to be affected by dopaminergic signaling modulation. 26 Moreover, it was reported that dopamine has an inhibitory effect on hippocampal excitability during activation of dopamine D2 receptors. Conversely, we found that inhibition of dopamine receptors by HAP significantly increased the seizure threshold compared with only TR treatment. In contrast, Starr stated that the use of dopamine antagonist drugs (antipsychotics) in patients with epilepsy lowers the seizure threshold and seizures can occur in patients without a previous history of the disease. Also, inhibition of seizure by stimulation of dopaminergic D2 receptors can occur in patients who use antiparkinsonian drugs such as pergolide and bromocriptine. 41
The role of oxidative stress in the toxicity of TR has been shown in previous studies. 42 Mitochondrial dysfunction is an important side effect of oxidative stress. Some studies reported the relationship between seizure and development of the mitochondrial dysfunction. 43 –46 In this study, oxidative damage induced by seizure was investigated after inhibition and stimulation of dopaminergic D2 receptors and as a consequence of TR injection. The results of our study showed that the mitochondria damage can occur due to the TR seizure induction. We found that formation of ROS, LPO, and PC was significantly increased in the TR group and the level of the GSH significantly decreased in this group compared with the control group (Figures 2 –5). These results suggest that the enhancement of oxidative stress in the mice brain mitochondria can be due to the TR injection. Studies about the relationship between oxidative stress and TR administration indicated that TR at therapeutic dose can reduce oxidative stress in brain tissue. 42 The obtained results in our study can be due to the use of seizure-inducing dose (high dose) of TR, which could increase free radical production in the brain cells and subsequently lead to mitochondria damage. Although in our results the ROS, PC formation, and LPO in the TR + HAP group were significantly increased compared with the control group, which is compatible with previous studies, 47 these markers were decreased significantly in the TR + HAP group when compared with the TR group. Also, the content of GSH in the TR + CAB and TR groups was significantly diminished compared with the control group. It was reported that that HAP reduced the levels of antioxidant enzymes and increased free radicals in the brain that is in contrast to our finding. 47 This difference may be due to the blockage of dopamine receptors by HAP in the dopaminergic pathway that could increase the seizure threshold, and as a result of that, oxidative stress decreased in the brain mitochondria.
In this study, significant decreases in LPO and GSH levels in the TR + CAB group were observed when compared with the TR group. Research about the relationship between CAB and oxidative stress has shown that CAB plays a protective role against oxidative stress-induced damage. In our study, although pretreated mice with CAB have shown lower levels of LPO compared to the TR group, the levels of oxidative damage in the TR + CAB group were significantly increased compared to the control group, which is different to the obtained results from the other studies. 48 This difference might be due to the stimulation of dopamine D2 receptors in the dopaminergic system by the CAB that reduced seizure threshold, and as a consequence of that, oxidative stress can be increased. However, the drugs used are not selective tools for studying agonist/antagonist activity at the D2 but interact with several other targets to a variable degree. It should be mentioned that the selective dopamine D2 receptors antagonist would have been used concurrently in this study, which would allow us to conclude much stronger that it is the shortcoming of our study. This can be a point to be considered in future studies. We used HAP, a predominant D2 receptor antagonist and a dopaminergic pathway blocker, as a common and accessible antagonist drug. For defining the kind of receptors which are more involved in TR-induced seizure and a better understanding of the dopaminergic system role at it, further studies with the use of selective or highly specific dopaminergic receptors are required.
Conclusion
This study revealed the involvement of dopaminergic system in TR-induced seizure. It also shows that the inhibition of dopamine D2 receptors can increase the TR threshold seizure and decrease the oxidative damage in the brain cell mitochondria. Conversely, stimulation of dopamine D2 receptors by CAB can decrease the TR seizure threshold and GSH content in the brain cell mitochondria.
Footnotes
Author Contributions
Behnaz Bameri contributed to conception and design and contributed to analysis and interpretation. Fatemeh Shaki contributed to design and contributed to analysis and interpretation. Nematollah Ahangar contributed to conception and contributed to analysis and interpretation. Ramin Ataee contributed to conception and contributed to analysis and interpretation. Mahedeh Samadi contributed to conception and contributed to analysis and interpretation. Hamidreza Mohammadi contributed to conception and design and contributed to acquisition, analysis, and interpretation. All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed financial support for the research, authorship, and/or publication of this article: Financial support for this work was provided by Mazandaran University of Medical Sciences, Sari, Iran with reference number 1635.
