Abstract
Introduction:
Systemic autoimmune diseases are known to have a significant impact on renal function, with varying degrees of severity that can influence overall patient outcomes. This study aims to assess the impact of Coronavirus Disease-2019 (COVID-19) on the renal outcomes associated with the spectrum of systemic autoimmune diseases.
Material and Methods:
This observational study was conducted in Jawaharlal Nehru Medical College, Aligarh, from 1 November 2020 to 31 October 2021, to evaluate outcomes in patients with systemic autoimmune diseases and COVID-19 who developed acute kidney injury (AKI).
Results:
A total of 129 patients with systemic autoimmune diseases were admitted, out of which 51 patients with AKI were included in the study. Of those, 18 patients were COVID-19 RT-PCR positive. The most common systemic autoimmune disorder observed was systemic lupus nephritis (35.3%) followed by rheumatoid arthritis (RA) (19.6%) and seronegative spondyloarthropathy (SpA) (17.6%). The most common renal manifestation observed was drug-induced kidney injury (37.2%) followed by lupus nephritis (29.4%), thrombotic microangiopathy (7.8%) and renal artery stenosis (7.8%). Patients who were COVID-19 RT-PCR positive (P <.01), those who required inotropic support (P <.01), ventilatory support (P <.01), Intensive Care Unit admission (P <.01), haemodialysis (P <.01) and with peripheral oxygen saturation <94% at presentation (P <.01), were significantly associated with mortality. The need for ventilatory support was observed as an independent predictor of mortality in patients with renal involvement and COVID-19 (P <.01).
Conclusion:
The present study sheds light on the outcome of AKI in systemic autoimmune diseases and COVID-19, with the use of ventilatory support significantly contributing to mortality.
Keywords
Abbreviations
AKI: Acute Kidney Injury
ANCA: Anti-Neutrophil Cytoplasmic Antibodies
AAV: ANCA Associated Vasculitis
CKD: Chronic Kidney Disease
COVID-19: Coronavirus Disease 2019
GFR: Glomerular Filtration Rate
HSP: Henoch-Schönlein purpura
ICU: Intensive Care Unit
KDIGO: Kidney Disease: Improving Global Outcomes
LDH: Lactate Dehydrogenase
RA: Rheumatoid Arthritis
RT-PCR: Real-Time Reverse Transcriptase Polymerase Chain Reaction
SLE: Systemic Lupus Erythematosus
SpA: Seronegative Spondyloarthropathy
SPO2: Peripheral Oxygen Saturation
SS: Systemic Sclerosis
SPSS: Statistical Package for the Social Sciences
Introduction
Systemic autoimmune diseases represent a heterogeneous group of chronic diseases characterised by aberrant responses by the immune system, targeting the body’s own tissues and organs.[1] Renal involvement in systemic autoimmune diseases manifests in a spectrum ranging from mild proteinuria to severe glomerulonephritis and chronic kidney disease (CKD).[2] The presence of renal disease significantly influences the prognosis of the diseases, often contributing to increased morbidity and mortality.[3]
The global emergence of the Coronavirus Disease 2019 (COVID-19) pandemic, which was caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2 virus), has been associated with a wide range of renal complications, including acute kidney injury (AKI), proteinuria, and hematuria.[4] It has introduced new challenges in patient management and has further complicated the complex interrelationship between systemic autoimmune diseases and their renal outcomes.[5–7] The literature on the impact of COVID-19 on various systemic autoimmune diseases has been described; however, the majority of these studies focus on specific diseases, and there is a lack of large-scale research. Data regarding the interaction between COVID-19, systemic autoimmune diseases and renal characteristics that impact patient prognosis are limited.
To our knowledge, the current research is the only study conducted in the Indian subcontinent to investigate the intersection of AKI and systemic autoimmune diseases, during the COVID-19 crisis. The outcomes of the present study provide insights into the management of patients with systemic autoimmune diseases who have renal involvement in the context of multisystem organ involvement by the SARS-CoV-2 virus, providing crucial data for future research for better understanding of these complex diseases and better management of patients.
Material and Methods
This observational study was conducted at Jawaharlal Nehru Medical College and Hospital (JNMCH), Aligarh Muslim University, Aligarh, Uttar Pradesh, India. This study was conducted from 1 November 2020 to 31 October 2021, for a duration of one year. The study included all patients above 12 years of age, those who were admitted with Systemic Autoimmune Diseases, diagnosed as per standard disease criteria, and with renal involvement defined as the development of AKI, as defined by the Kidney Disease: Improving Global Outcomes (KDIGO)-2012 guidelines.[8] Patients who were already diagnosed with systemic autoimmune diseases with documented renal involvement, who were immunocompromised, those who had a history of renal transplant, CKD patients on renal replacement therapy and those who refused to provide consent were excluded from the study.
Data Collection
Patients who presented to the out-patient department and emergency trauma centre with history, clinical signs and symptoms suggesting systemic autoimmune disorder or those who were already diagnosed with systemic autoimmune disorder without renal involvement were enrolled in the study. Prior to admission, each patient underwent screening for COVID-19 using a nasal or oropharyngeal swab, which was analysed using real-time reverse transcriptase polymerase chain reaction (RT-PCR). The age and gender of patients were documented. Mean Arterial Blood Pressure and peripheral oxygen saturation, using a pulse oximeter (DS100A-1 Nellcor™ SpO2 sensor and Schiller TRUSCOPE Elite A6 monitor) were also noted. On the basis of history and a thorough clinical assessment a screening autoimmune profile was done using standard serological methods with additional serological tests that were conducted based upon results of screening tests [Supplementary Table 1]. The patient’s diagnoses were determined based on the latest criteria set by widely accepted standard guidelines for various Systemic Autoimmune Diseases [Supplementary Table 2].
Renal function test, electrolytes, arterial blood gas, urine routine and microscopic examination were conducted as an initial assessment to determine renal involvement. Hemogram, general blood picture, Serum Lactate Dehydrogenase (LDH) levels and iron studies were also performed for determination of type of anaemia, if present. Further work-up was done based on the patient’s clinical and biochemical findings including renal ultrasound, total serum protein and albumin, Total urinary protein, urine culture and renal biopsy, if indicated and noted. Patients who tested positive for COVID-19 RT-PCR their biopsy was performed at an interval after they had recovered from the disease. Classification of AKI and protein excretion was done according to KDIGO - 2012 guidelines.[8,9]
Patient Management and Outcome
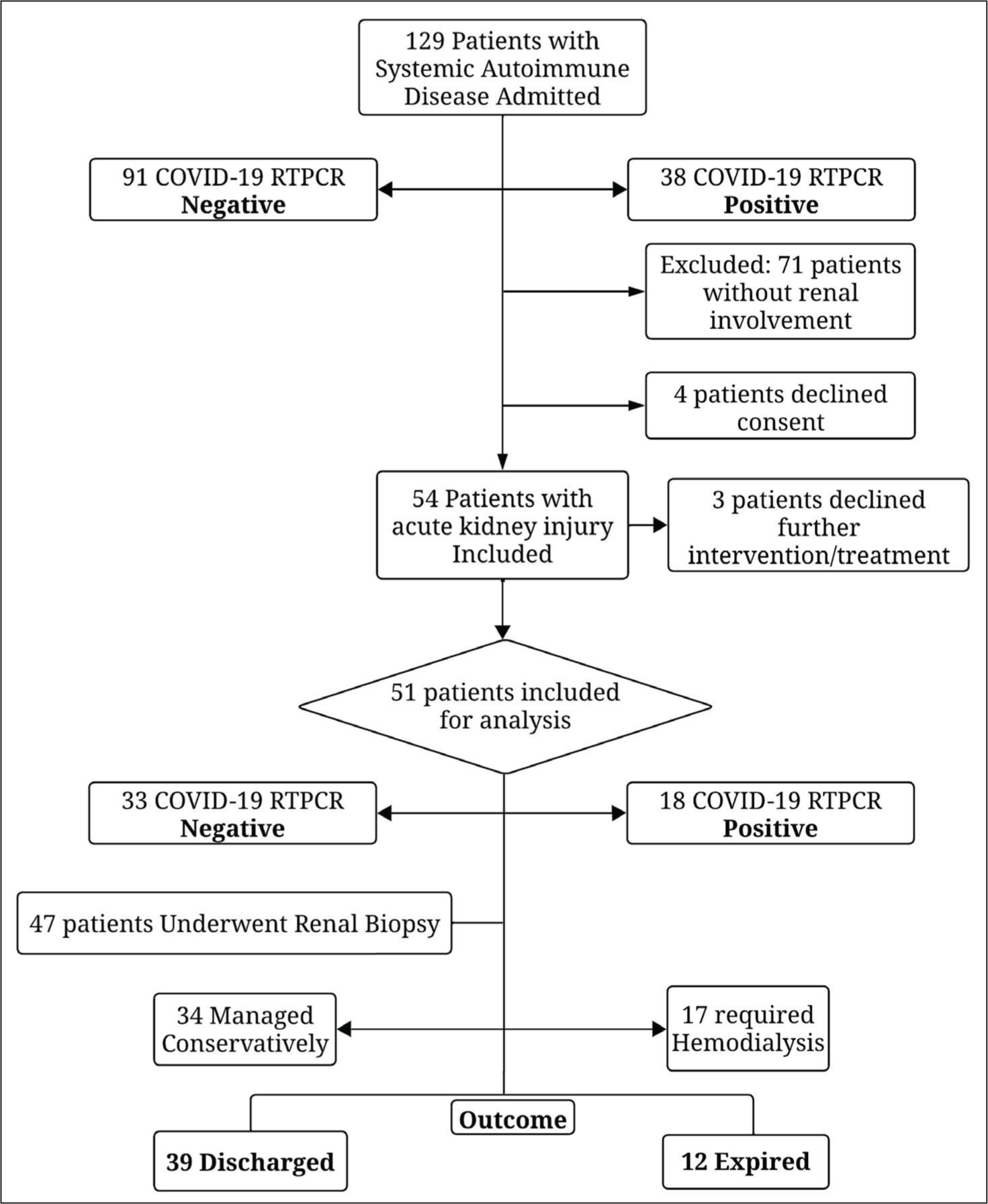
Patients who tested positive for COVID-19 via RT-PCR were managed in dedicated isolation units. Systemic autoimmune diseases were managed as per their latest standard clinical guidelines [Supplementary Table 2]. All patients who developed AKI received initial medical management in accordance with the institutional protocol based on standard accepted guidelines [Supplementary Table 2]. Renal replacement therapy was performed using the Fresenius 4008S NG haemodialysis machine, when indicated. Patients who needed inotropic assistance or ventilatory support were managed in Intensive Care Units. The total length of hospital stay was recorded as the number of days the patient remained under hospital care until being discharged or expired. The outcome was determined as patients who were discharged under satisfactory conditions or those who expired [Figure 1].
Flowchart showing patients included for final analysis. COVID-19: Coronavirus Disease 2019
Data Management and Statistical Analysis
The presentation of the categorical variables was done in the form of numbers and percentages (%). On the other hand, the quantitative data with normal distribution were presented as the means ± standard deviation and the data with non-normal distribution as median with 25th and 75th percentiles (interquartile range). The data normality was checked by using the Kolmogorov–Smirnov test. In the cases, in which, the data were not normal, non-parametric tests were used. The comparison of the variables which were quantitative and not normally distributed in nature was analysed using the Mann–Whitney test and variables that were quantitative and normally distributed in nature were analysed using independent t-test. Categorical variables between the two groups were compared by chi-square test (or Fisher’s exact test when at least one cell had an expected value of <5). Patients were categorised based on their COVID-19 status as either positive or negative, and their outcome, as either discharged or expired. The parameters were correlated with the COVID-19 positive status and outcome. A logistic regression analysis was done to assess the independent predictors of mortality. The data entry was done in the Microsoft Excel spreadsheet and the final analysis was done with the use of the Statistical Package for the Social Sciences (SPSS) software, IBM manufacturer, Chicago, USA, version 25.0. For statistical significance, a P < .05 was considered statistically significant.
Results
A total of 129 patients diagnosed with Systemic autoimmune diseases were admitted. Out of 129 patients, 51 patients who developed AKI were included for final analysis. 23.5% (n = 12) of the 51 patients included in the study had AKI stage 1, 33.3% (n = 17) patients had AKI stage 2, and 43.1% (n = 22) patients had AKI Stage 3. Out of the 22 patients who developed AKI Stage 3, only 5 were managed conservatively, and 17 patients eventually required haemodialysis. Of the 51 patients, 35.3% (n = 18) tested positive for COVID-19 RT-PCR [Figure 1].
The mean age of patients under study was 38.7 ± 14.7 years and female patients (82.3%; n = 42) were more prevalent than male patients (17.6%; n = 9) (Female-to-male ratio: 4.6:1). The study patients’ median hospital stay was 8 days, with a range of 7–11 days.
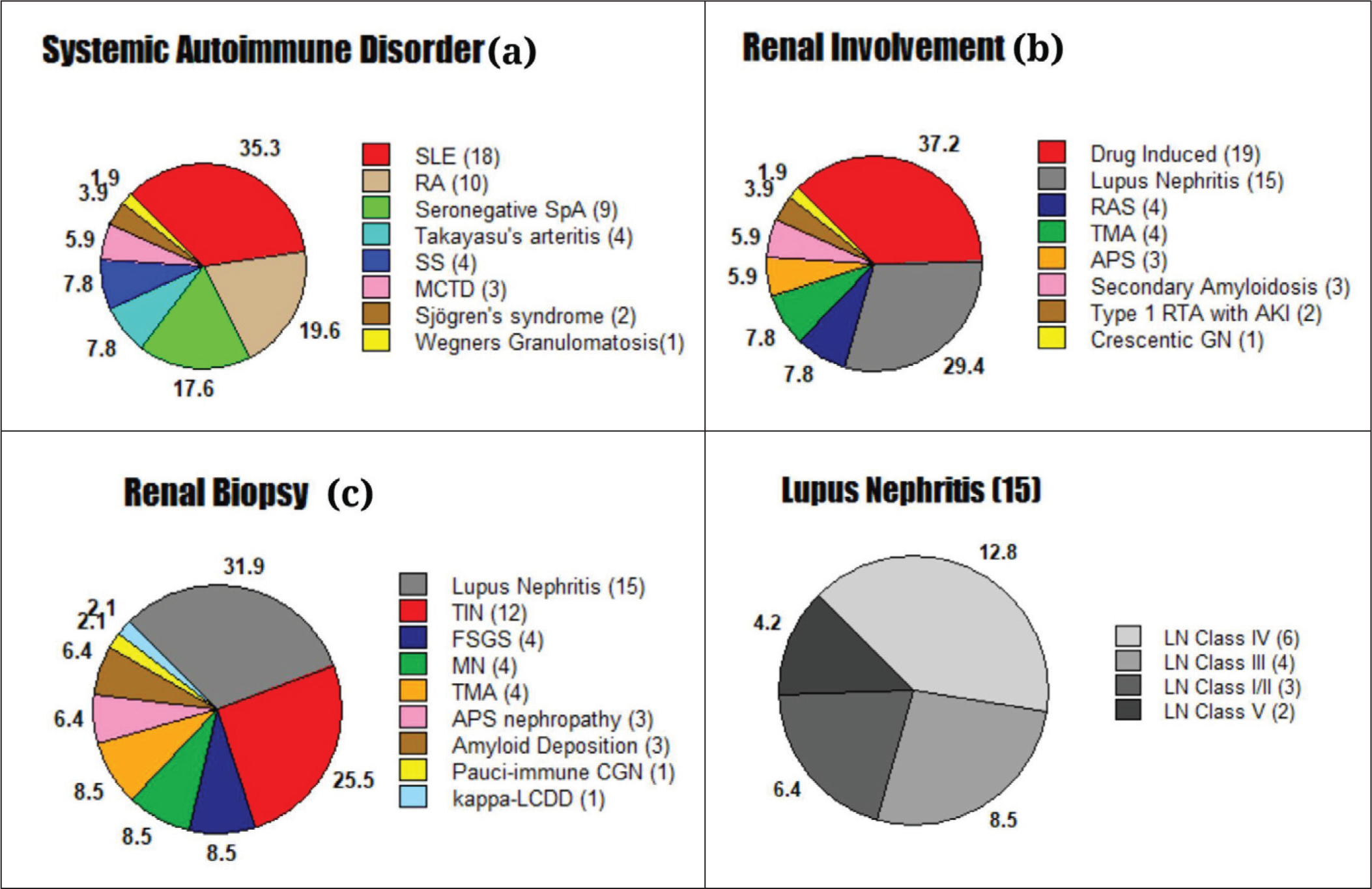
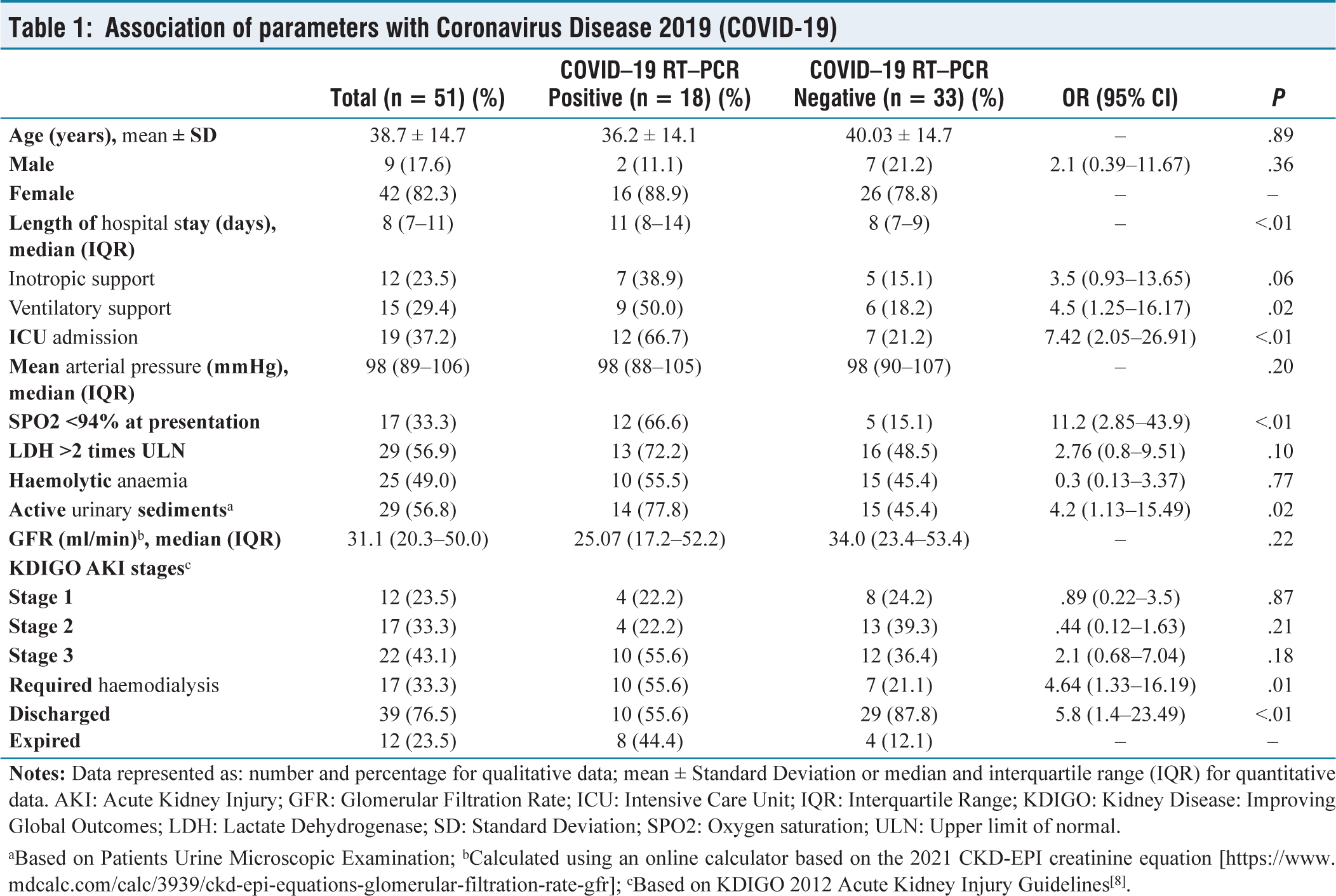
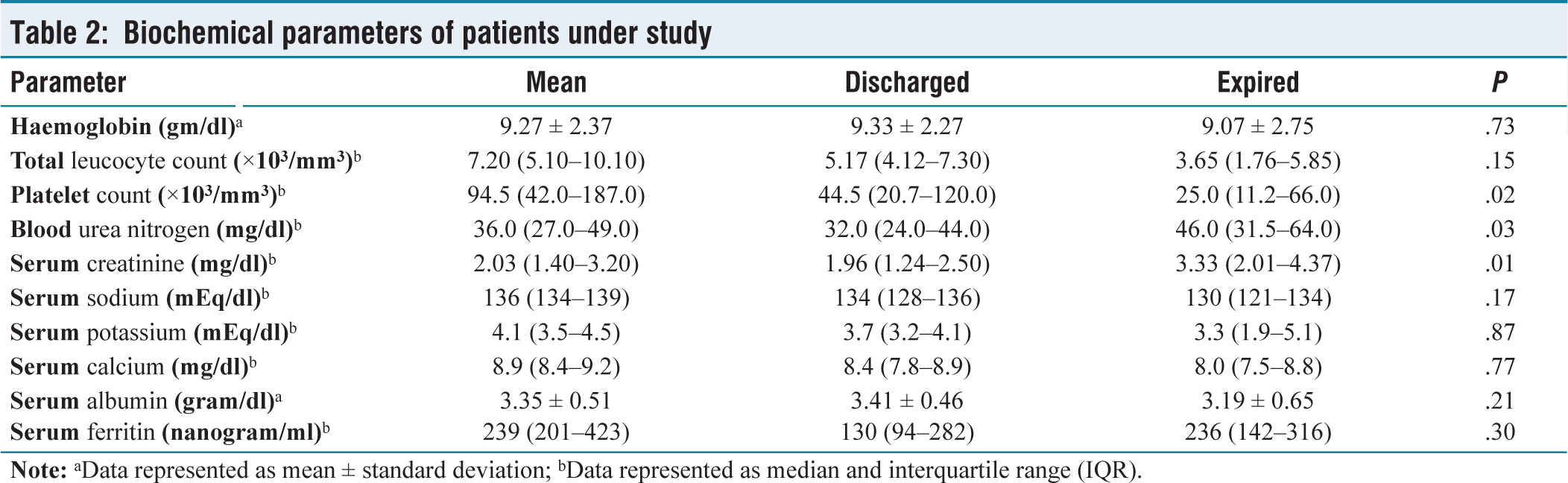
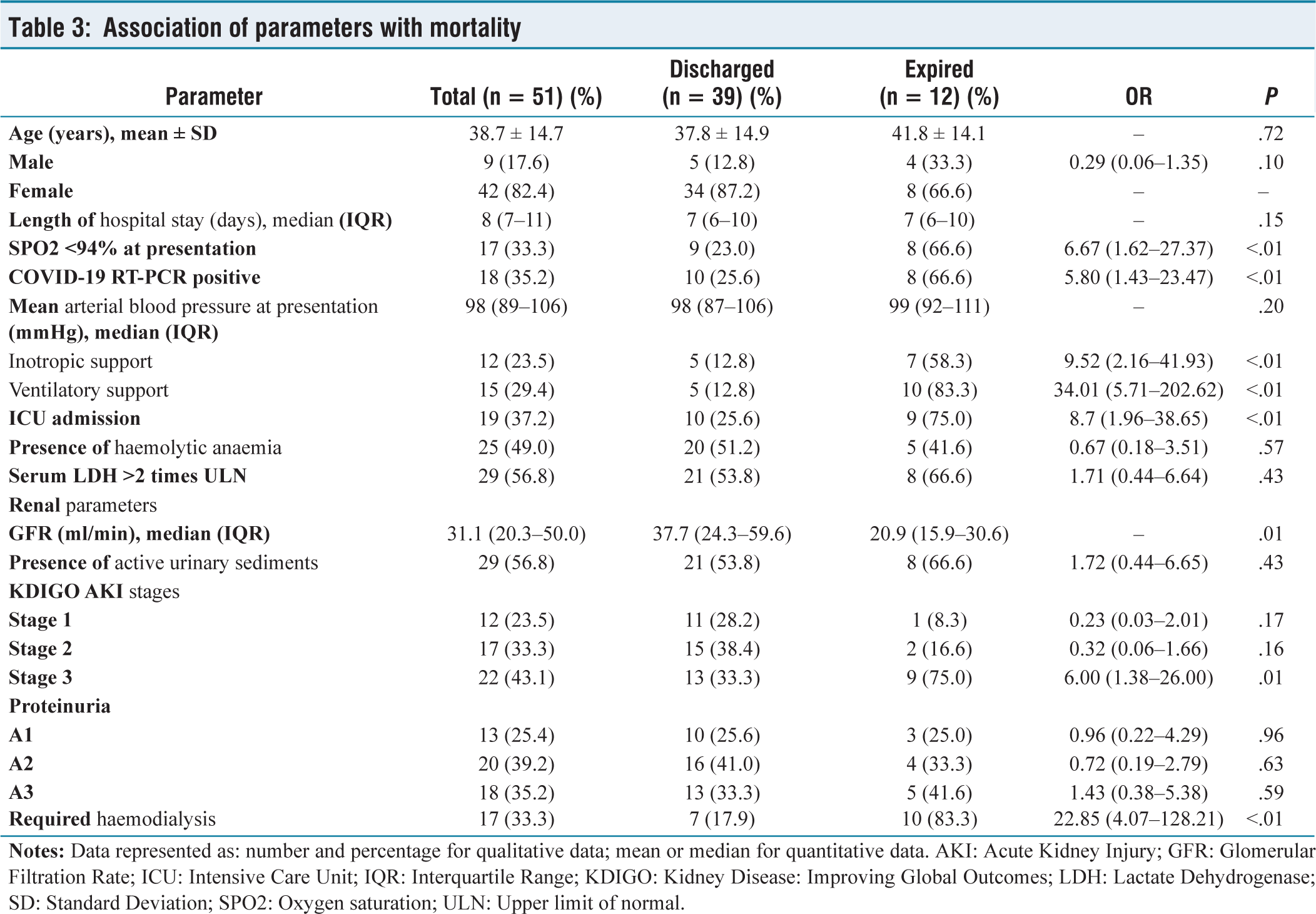
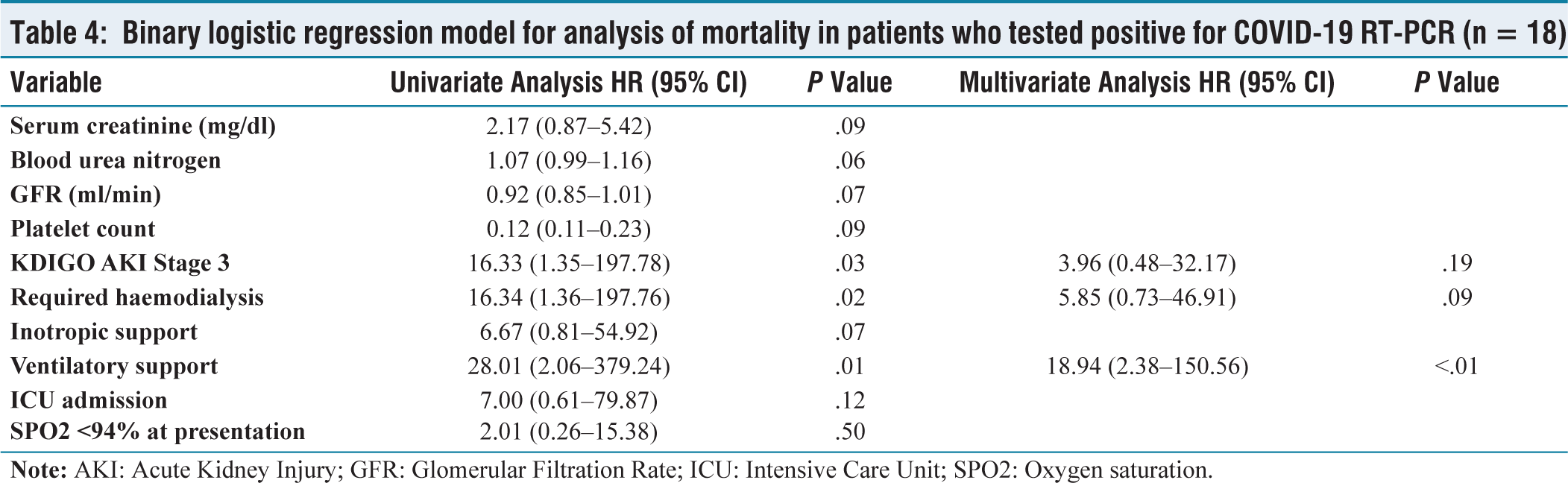
The most common systemic autoimmune disease observed among patients with renal involvement, during the study period was Systemic Lupus Erythematosus (SLE) (35.3%) followed by Rheumatoid Arthritis (RA) (19.6%), Seronegative spondyloarthropathy (SpA) (17.6%), Systemic Sclerosis (SS) (7.8%) and Takayasu Arteritis (7.8%). Three patients were diagnosed with Mixed Connective Tissue Disorder; two of them had an overlap between SS and SLE, while one patient had an overlap between SLE and RA [Figure 2a]. The most common renal involvement observed was drug-induced kidney injury (37.2%) followed by Lupus Nephritis (29.4%) and Renal Artery Stenosis (7.8%). Furthermore, Secondary Amyloidosis, Myeloma Nephropathy, and Crescentic Glomerulonephritis were also observed [Figure 2b]. Renal Biopsy was performed in 47 patients and their observations are shown in Figure 2c. Compared to patients in COVID-19 negative group, more patients in the COVID-19 positive group had SPO2 less than 94% at presentation (12 vs. 5, P <.01) and had longer duration of hospital stay [11(8–14) vs. 8(7–9) days; P <.01). No significant association between age (P = .89), gender (P = .36) and mean arterial blood pressure at presentation (P = .20) was observed. More patients required inotropic support (7 vs. 5; P = .06), ventilatory support (9 vs. 6; P = .02) and ICU admission(12 vs. 7; P <.01). On urine microscopy, a significant number of patients had active urinary sediments (P = .02). More number of patients in the COVID-19 positive group required haemodialysis as compared to COVID-19 negative group (10 vs. 7; P = .01) [Table 1]. Out of the 51 patients included 39 patients were discharged under satisfactory condition and 12 patients expired. The development of acute respiratory distress syndrome and subsequent ventilator-associated pneumonia (n = 9) was the most common cause of mortality. One patient expired due to a diffuse pulmonary haemorrhage resistant to treatment, one patient expired due to septic shock and another suffered sudden cardiac arrest from refractory hyperkalaemia. Biochemical parameters of the patients under study are shown in Table 2. Patients who expired had lower platelet counts (P = .02) higher serum creatinine (P = .01) and blood urea nitrogen levels (P = .03). The association between mortality in patients with systemic autoimmune disease and renal involvement is shown in Table 3. As compared to patients who were discharged the mean age of patients who expired was more (41.8 ± 14.1 years vs. 37.8 ± 14.9 years). There was no significant association of age (P = .72), gender (P = .10), length of hospital stay (P = .15) and mean arterial blood pressure at presentation (P = .20) with mortality. A significant association with mortality was observed in patients who presented with SPO2 less than 94% at presentation (P <.01); who required inotropic support (P <.01), ventilatory support (P <.01) and ICU admission (P <.01). More patients expired in COVID-19 positive group as compared to COVID-19 negative group (10 vs. 8; P <.01). Association of renal parameters and mortality is also shown in Table 3. Glomerular filtration rate (GFR) of patients who expired was lesser compared to patients who were discharged [20.9 (15.9–30.6) ml/min vs. 37.7 (24.3–59.6) ml/min; P = .01) . In comparison to patients who were discharged, a greater proportion of patients who developed AKI Stage 3 (75.0% vs. 33.3%; P = .01) and required haemodialysis (83.3% vs. 17.9%; P <.01) had expired. The univariate analysis of patients in the COVID-19 positive group showed patients who developed AKI Stage 3 (OR 16.3; P = .03), required haemodialysis (OR 16.34; P = .02) and ventilatory support (OR 28.01; P = .01) was significantly associated with mortality. The multivariate analysis revealed that the need for ventilatory support was an independent predictor of mortality (OR 18.94; P <.01) [Table 4].
Pie chart showing systemic autoimmune disease (a), renal involvement (b) and renal biopsy findings (c) of patients under study. AKI: Acute Kidney Injury; APS: Antiphospholipid Syndrome; CGN: Crescentic Glomerulonephritis; FSGS: Focal Segmental Glomerulosclerosis; GN: Glomerulonephritis; LCDD: Light Chain Deposition Disease; LN: Lupus Nephritis; MCTD: Mixed Connective Tissue Disorder; MN: Membranous Nephropathy; RA: Rheumatoid Arthritis; RAS: Renal Artery Stenosis; RTA: Renal Tubular Stenosis; SLE: Systemic Lupus Erythematosus; SpA: Spondyloarthropathy; SS: Systemic Sclerosis; TIN: Tubulointerstitial Nephritis; TMA: Thrombotic Microangiopathy
Association of parameters with Coronavirus Disease 2019 (COVID-19)
aBased on Patients Urine Microscopic Examination; bCalculated using an online calculator based on the 2021 CKD-EPI creatinine equation [
Biochemical parameters of patients under study
Association of parameters with mortality
Binary logistic regression model for analysis of mortality in patients who tested positive for COVID-19 RT-PCR (n = 18)
Discussion
The present study was one of the few studies that assessed outcomes in patients with systemic autoimmune diseases and COVID-19 who developed AKI. There is a limited number of large-scale research available and the existing literature largely focuses on either renal outcomes or rheumatologic diseases and their association with COVID-19. The age of onset for various systemic autoimmune diseases varies significantly.[10] The mean age of patients included in the present study was 38.7 ± 14.7 years; older patients were present in the COVID-19 negative group and no significant association between age and mortality was observed. This was in contrast to a single-centre observational study conducted by Fredi M et al., where COVID-19 association with rheumatologic diseases in general population was done and the median age observed in their study was 69 (59.5–78.2) years.[11] Chang R et al., reported in their retrospective cohort study a mean age of 45.2 ± 17.6 years in patients diagnosed with COVID-19.[12] While Gayathri et al., reported in their cohort of 16 patients with lupus nephritis and COVID-19, a mean age of 20.8 years (n = 8) in patients who had expired.[13] The preponderance of autoimmune diseases in females has been widely documented, and the present study showed similar results.[14] Analysis between gender, COVID-19 and outcome revealed no significant association. The median length of hospital stay observed in the present study was 8 days and patients in the COVID-19-positive group had a longer duration hospital stay [11 (8–14) days]. Fredi M. et al. reported COVID-19 patients with rheumatological involvement had a median length of hospital stay of 15 (11.5–27.5).[11] The most common systemic autoimmune disease in the present study was SLE followed by RA and SpA, while Fredi M. et al. reported RA as the most common disease observed, followed by SpA and SLE.[11] Data on renal histopathology in systemic autoimmune diseases and COVID-19 is still lacking. Data from a recent study conducted by Prasad P. et al. on histopathologic lesions in patients with non-lupus rheumatologic diseases reported ANCA Associated Vasculitis (AAV) as the most common disease observed, followed by Henoch-Schönlein purpura (HSP)/IgA nephropathy and RA. The most common renal histopathologic lesion observed in their study was pauci-immune crescentic glomerulonephritis followed by IgA nephropathy, chronic tubulointerstitial nephritis and hypertensive vasculopathy.[15] Literature on systemic autoimmune diseases, COVID-19 and outcomes is also lacking and the present study did not observe any significant association. Thrombocytopenia has been reported in patients with COVID-19, and a significant association was observed between thrombocytopenia and mortality.[16,17] Biochemical parameters revealed that patients who developed acute deterioration of kidney function were associated with mortality. It was also observed that the majority of the patients in the study developed acute deterioration in renal function once multiorgan dysfunction and acute cytokine storm set in. There are several mechanisms of AKI in COVID-19 which have been described in the existing literature, including tubular, vascular, glomerular, and interstitial injury, all of which significantly contribute to morbidity and mortality. Systemic autoimmune diseases also contribute to impairment of renal function; data regarding AKI in systemic autoimmune disease and COVID-19 is not available in the present literature. The association between AKI, COVID-19 and outcome was assessed in the present study. The study observed a significant association between AKI Stage 3 and mortality, but no significant association between AKI stages and COVID-19. The logistic regression analysis in the COVID-19-positive group revealed a significant association between AKI Stage 3 and mortality. Similarly, patients who required haemodialysis were also significantly associated with mortality. Yüksel E and team conducted a single-centre retrospective cohort study in patients with COVID-19, analysing parameters associated with mortality. According to their study, patients who were older, had higher levels of C-reactive protein, procalcitonin and creatinine kinase and those patients who developed AKI Stages 2 and AKI Stage 3 or needed renal replacement therapy were significantly associated with poor outcomes. Their study also reported mortality was higher in patients who developed in-hospital AKI as compared to AKI on admission.[18] Tan B. W. L. et al., conducted an international multi-centre observational cohort study, which revealed a significant association between patient age, AKI stage, and mortality. Their study reported a higher mortality rate for AKI Stage 3 compared to Stage 2 and Stage 1. Their study also reported that severe COVID-19-associated AKI was associated with poor long-term post-AKI kidney function recovery.[19] In the present study, a significant association between mortality was observed between patients who required inotropic support, ventilatory support, ICU admission and patients who had SPO2 <94% at presentation. The need for ventilatory support emerged as an independent predictor of mortality in the COVID-19-positive group. The association between the need for invasive ventilation and COVID-19 and mortality has been well-documented in multiple studies.[20,21] Wafy S. M. et al., reported older age, severe lung infiltrate and ICU admission were independent predictors of mortality in their cohort study.[22] Assal H. H. et al., and team reported elevated total leucocyte count, urea, the use of invasive mechanical ventilation, and the presence of respiratory bacterial co-infection were found to be independently associated with mortality in their cohort study.[23] The development of ventilator-associated pneumonia after intubation was a significant factor leading to sepsis shock and mortality in the present study. Rouyer M. and colleagues observed similar results in their observational study in ventilator-associated pneumonia patients with and without COVID-19, reporting higher mortality and the development of sepsis in patients who developed ventilator-associated pneumonia with COVID-19.[21] In a retrospective cohort study conducted by Faye A. S. et al., patients with autoimmune disease and COVID-19, reported no significant differences in ICU admission, intubation and death. Their study reported patients with autoimmune disease were not at an increased risk of ICU admission, intubation, or death.[24]
While the study provides valuable insights into the impact of COVID-19 on patients with systemic autoimmune diseases and renal involvement, several limitations must be acknowledged. First, one of the primary limitations of this study is the relatively small sample size and the study’s observations may not be fully generalisable to larger populations. Second, the confounding variables, such as the severity of the underlying autoimmune disease or other comorbid conditions, might have influenced the results. Third, the study included patients who were admitted to the hospital, which may not represent all patients with systemic autoimmune diseases and renal involvement (selection bias). Patients with milder forms of the disease or those managed in outpatient settings were not included, potentially skewing the findings towards more severe cases.
In summary, while this study offers valuable insights into the impact of COVID-19 on patients with systemic autoimmune diseases and renal involvement, it is essential to consider these limitations when interpreting the findings. Further research, with larger sample sizes, longer follow-up periods and more controlled study designs, is needed to fully understand the complex interactions between systemic autoimmune diseases, renal involvement and COVID-19.
Conclusion
The present study highlights renal parameters and outcomes associated with various systemic autoimmune diseases and COVID-19. Patients who presented with low oxygen saturation (<94%) and who required inotropic support, ventilatory support or ICU admission; patients who developed AKI Stage 3 and required haemodialysis were significantly associated with poor outcomes. The presence of COVID-19 was also associated with poor outcomes in the present study. The need for ventilatory support was an independent risk factor for mortality. Further research is required to assess the outcomes in patients with systemic autoimmune disease, AKI and COVID-19.
Supplemental material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
We would like to acknowledge the support and facilities provided by Jawaharlal Nehru Medical College and Hospital (JNMCH) in conducting this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
The research procedure was approved by the Institutional Ethics Committee of Jawaharlal Nehru Medical College (IECJNMC/929) and the study was conducted following the standards of Good Clinical Practice and the Helsinki Declaration.
Patient consent
The authors confirm that no part of this publication contains information that discloses the identity or personal details of any patient. Written informed consent has been obtained from all patients under study and is retained by the authors for reference.
Credit author statement
Ariba Nasar: Conceptualization, methodology design, data collection, and analysis.
Mohd. Aslam: Contributed significantly to the literature review, data interpretation, and manuscript revision.
Ahmad Ghayas Ansari: Involved in data collection, analysis, and contributed to the discussion section.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration
The authors have declared that observations and results from this article have been presented at World Conference of Nephrology, held in New Delhi on 7th February 2025 as poster presentation.
Use of artificial intelligence
The authors confirm that no artificial intelligence tools or software were used in the preparation, analysis, or writing of this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
