Abstract
Background:
Acute kidney injury (AKI) is a potentially fatal complication of Coronavirus Disease-2019 (COVID-19). Binding of the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), the virus responsible for COVID-19, to its viral receptor, angiotensin converting enzyme 2 (ACE2), results in viral entry and may cause AKI.
Objectives:
We performed a systematic review and meta-analysis of the frequencies of AKI and renal replacement therapy (RRT) in critically ill COVID-19 patients and compared those frequencies with patients who were infected by respiratory viruses that bind or downregulate ACE2 (ACE2-associated viruses) and viruses that do not bind nor downregulate ACE2 (non-ACE2-associated viruses).
Design:
Systematic review and meta-analysis.
Setting:
Observational studies on COVID-19 and other respiratory viral infections reporting AKI and RRT were included. The exclusion criteria were non-English articles, non-peer-reviewed articles, review articles, studies that included patients under the age of 18, studies including fewer than 10 patients, and studies not reporting AKI and RRT rates.
Patients:
Adult COVID-19, Severe Acute Respiratory Syndrome (SARS), Middle East Respiratory Syndrome (MERS), and influenza patients.
Measurements:
We extracted the following data from the included studies: author, year, study location, age, sex, race, diabetes mellitus, hypertension, chronic kidney disease, shock, vasopressor use, mortality, intensive care unit (ICU) admission, ICU mortality, AKI, and RRT.
Methods:
We systematically searched PubMed and EMBASE for articles reporting AKI or RRT. AKI was defined by authors of included studies. Critical illness was defined by ICU admission. We performed a random effects meta-analysis to calculate pooled estimates for the AKI and RRT rate within each virus group using a random intercept logistic regression model.
Results:
Of 23 655 hospitalized, critically ill COVID-19 patients, AKI frequencies were not significantly different between COVID-19 patients (51%, 95% confidence interval [CI]: 44%-57%) and critically ill patients infected with ACE2-associated (56%, 95% CI: 37%-74%, P = .610) or non-ACE2-associated viruses (63%, 95% CI: 43%-79%, P = .255). Pooled RRT rates were also not significantly different between critically ill, hospitalized patients with COVID-19 (20%, 95% CI: 16%-24%) and ACE2-associated viruses (18%, 95% CI: 8%-33%, P = .747). RRT rates for both COVID-19 and ACE2-associated viruses were significantly different (P < .001 for both) from non-ACE2-associated viruses (49%, 95% CI: 44%-54%). After adjusting for shock or vasopressor use, AKI and RRT rates were not significantly different between groups.
Limitations:
Limitations of this study include the heterogeneity of definitions of AKI that were used across different virus studies. We could not match severity of infection or do propensity matching across studies. Most of the included studies were conducted in retrospective fashion. Last, we did not include non-English publications.
Conclusions:
Our findings suggest that viral ACE2 association does not significantly alter the rates of AKI and RRT among critically ill patients admitted to the ICU. However, the rate of RRT is lower in patients with COVID-19 or ACE2-associated viruses when compared with patients infected with non-ACE2-binding viruses, which might partly be due to the lower frequencies of shock and use of vasopressors in these two virus groups. Prospective studies are necessary to demonstrate whether modulation of the ACE2 axis with Renin-Angiotensin System inhibitors impacts the rates of AKI and whether they are beneficial or harmful in COVID-19 patients.
What was known before
While a small proportion of patients with Coronavirus Disease-2019 (COVID-19) were known to develop acute kidney injury or require renal replacement therapy, the true rates of acute kidney injury and renal replacement therapy in hospitalized, critically ill COVID-19 patients were unknown. Furthermore, the rates of acute kidney injury and renal replacement therapy in hospitalized patients infected with other respiratory viruses that do or do not bind or downregulate angiotensin converting enzyme 2 (ACE2) have been described in isolation but, it remains unclear whether these rates are different than those in COVID-19. The ongoing debate of whether Renin-Angiotensin System (RAS) inhibitors confer protection against or increases susceptibility to COVID-19 infection underscores the dire need to understand whether viral association with ACE2, through binding or downregulation, is correlated with the development of end-organ injury such as AKI.
What this adds
Acute kidney injury occurs to approximately the same degree in critically ill patients, that is, patients admitted to the ICU, regardless of viral association with ACE2. The minimal impact of ACE2 association with rates of acute kidney injury restates the need for and provides a rationale for trials of RAS inhibitors for the therapeutic management of COVID-19.
Introduction
Coronavirus-Disease 2019 (COVID-19) has resulted in over 140 000 000 cases and over 3 000 000 deaths since it was first identified in December 2019.1-3
Angiotensin converting enzyme 2 (ACE2) is the viral receptor for Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2).4-6 ACE2 cleaves angiotensin II (Ang II) into the vasodilatory, anti-inflammatory peptide angiotensin 1-7 (Ang [1-7]).7,8
ACE2 is also the viral receptor for SARS-CoV-1. 9 The cellular distribution of ACE2, on the vascular endothelium, kidney, heart, and lungs, may explain, in part, the frequent occurrence of acute kidney injury (AKI), acute cardiac injury (ACI), and acute respiratory distress syndrome (ARDS) in COVID-19 patients.10-13 SARS-CoV-2 binding to ACE2 leads to SARS-CoV-2 entry and downregulation of ACE2 at the cell membrane, similar to SARS-CoV-1. 14 Other respiratory viruses also downregulate ACE2 expression (eg, influenza H1N1, H5N1, H7N9).15-17
The main proposed mechanisms of AKI in COVID-19 include (1) renal hypoperfusion due to fever, gastrointestinal losses, cardiomyopathy, shock, and use of vasopressors, (2) direct viral injury, (3) sepsis and septic shock-associated acute tubular necrosis, (4) increased inflammation due to increased pro-inflammatory cytokines and cytokine storm, (5) rhabdomyolysis, (6) micro- and macro-vascular thrombosis, and (7) micro-embolism.18-20
To our knowledge, no studies compare AKI and RRT in COVID-19 with other respiratory viruses that bind to or downregulate ACE2 and those viruses that are not associated with ACE2. This comparison enhances understanding of the role of ACE2 in the pathophysiology of AKI and could inform therapeutic strategies such as targeting of ACE2 with angiotensin converting enzyme inhibitors (ACEi) or angiotensin receptor blockers (ARBs) to mitigate AKI in COVID-19.
Accordingly, our objectives were to review and perform a meta-analysis of the frequencies of AKI and RRT in critically ill COVID-19 patients and compare those frequencies with critically ill patients who were infected by respiratory viral infections due to viruses that bind or downregulate ACE2 (ACE2-associated viruses) and viruses that do not involve ACE2 (non-ACE2-associated viruses).
Methods
Eligibility Criteria
We searched PubMed/MEDLINE and EMBASE from database inception up until March 25, 2021, for cohort studies, case control studies, and other systematic reviews and meta-analyses that reported the frequency of AKI, acute renal failure (ARF), or RRT in adults (18 years old or greater) infected with COVID-19. Full-text peer-reviewed articles and peer-reviewed articles published ahead of print were included. Letters and commentaries were also included if they provided patient characteristics. Only articles in English were included in the final analysis. Studies with less than 10 patients were excluded as were studies on cohorts of deceased patients and studies that reported only patients on dialysis or kidney transplant recipients. Non-peer-reviewed articles were excluded as were article abstracts, incomplete articles, research posters, conference abstracts, books, theses, and retracted articles.
Search Strategy and Study Selection
We used a broad search strategy to find relevant articles using the following search terms: COVID, COVID-19, 2019-nCov, and SARS-CoV-2. These terms were combined using “AND” with the terms clinical features, clinical characteristics, kidney injury, renal, kidney, creatinine, renal replacement, and dialysis. For ACE2 and non-ACE2 associated viruses, we used similar search terms for kidney injury as above and replaced COVID-19 with these search terms: influenza, H1N1, H5N1, H3N2, H7N9, SARS, SARS-CoV-1, and MERS (see Supplemental Appendix for full search strategy details).
One reviewer (A.C.) screened titles and abstracts to identify reports of hospitalized COVID-19 patients with AKI or RRT. After reading the full texts of publications, studies were included if they reported the frequency of AKI, acute renal failure (ARF), or RRT in adults (18 years old or greater) infected with COVID-19. We similarly reviewed the literature on patients (18 years old or greater) infected with respiratory viruses that are associated with ACE2 through ACE2 binding or downregulation (SARS-CoV-1, influenza H1N1, influenza H7N9) and respiratory viruses that are not associated with ACE2 (e.g., Middle East Respiratory Syndrome Coronavirus [MERS-CoV] and other influenza strains besides H1N1, H7N9).9,14-17,21 Other studies were identified from the references in review articles and meta-analyses and were included if they met the inclusion criteria.
Data Extraction
One reviewer (A.C.) extracted the studies from the literature in duplicate using a systematic template. We extracted the following data from each included study: author, journal of publication, study type, study location, age, sex, race, diabetes mellitus, hypertension, chronic kidney disease, shock, vasopressor use, intensive care unit (ICU) admission, ICU mortality, overall mortality, AKI, and RRT.AKI in the original publications was defined using a variety of definitions including the Kidney Disease Improving Global Outcomes (KDIGO) criteria, the Acute Kidney Injury Network (AKIN) criteria, the risk of renal failure, injury to the kidney, failure of kidney function, loss of kidney function, and end-stage renal failure (RIFLE) criteria or the presence of elevated serum creatinine.22-24 Critical illness was defined as admission to the intensive care unit (ICU). Results were not reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist, and a risk of bias assessment was performed on individual studies (Supplemental Table 4).
Statistical Analysis
Results of all included studies were summarized quantitatively. Pooled estimates for the AKI and RRT rate within each virus group and comparison of rates between groups were obtained using random effects (RE) meta-analysis based on a random intercept logistic regression model with logit link and covariate being virus group.25,26 Pairwise comparisons between virus groups were done by testing the appropriate regression coefficient using Wald-type test from the estimated model. Mixed effects meta-regression was further conducted to adjust for the prevalence of shock and vasopressors use in each study when comparing the virus groups. Among the included studies, only overall prevalence of shock and vasopressors use was reported, but not the ones within critically ill. Since shock is a critical illness and vasopressors are only administered in critically ill patients, we will assume all patients who had shock or used vasopressors are critically ill patients for the purpose of computing these prevalence rates within critically ill. Given that few studies have both shock and vasopressors use data available, we examined these variables as two separate adjusted analyses. Log odds of AKI or RRT was considered as the outcome variable in the regression. Results from the meta-regression were thus expressed as odds ratios. Random effects meta-analysis and mixed effects meta-regression were performed using the meta package in R 3.6.2 (R Foundation for Statistical Computing, Vienna, Austria). Study heterogeneity was assessed visually by funnel plot with a minimum of 10 studies and asymmetry was assessed by the Begg adjusted rank correlation test. Statistical heterogeneity was assessed by I2.
Results
Study Selection and Characteristics
After screening 6560 COVID-19 titles and abstracts, 5622 studies were excluded. We assessed 938 full-text articles for their eligibility. Following assessment for eligibility, 867 COVID-19 articles were excluded because they (1) were review articles or meta-analyses, (2) did not report rates of AKI or RRT in critically ill patients, (3) were not written in English, (4) included only patients on dialysis or kidney transplant recipients, or (5) included patients under 18 years of age or did not specify the minimum age of study participants. After reviewing the full texts of 938 articles, 71 COVID-19 studies were included in the meta-analysis (Figure 1A).1-3,13,27-94 Similarly, 7742 articles on ACE2-associated (SARS-CoV-1, influenza H1N1, H7N9) and non-ACE2-associated viruses (MERS-CoV, other influenza strains) were screened based on their titles and abstracts and 6841 articles were excluded. The full-texts of 901 articles were reviewed and 20 studies on ACE2-associated viruses and 3 studies on non-ACE2-associated viruses were included in the meta-analysis (Figure 1B).95-117 The majority of the included COVID-19 studies were from the United States (n = 28) and China (n = 17) but other included studies were from England (n = 5), France (n = 4), Brazil (n = 3), South Korea (n = 2), Mexico (n = 2), and Canada, Sweden, Netherlands, Italy, Switzerland, Bahrain, India, United Arab Emirates, and Saudi Arabia (n = 1 each). The last study was conducted jointly in France and Spain. 38 Included studies on ACE2-associated and non-ACE2-associated viruses were not predominantly associated with any country and were variable. The characteristics of the included studies are provided in Supplemental Table 1.

Flow diagrams of the search strategies showing included and excluded studies in the meta-analysis of acute kidney injury (AKI) and renal replacement therapy (RRT) in a) COVID-19 and b) ACE2- and non-ACE2-associated viral infections.
Characteristics of Included Study Populations
Females made up 38% (n = 20 615) of the studied hospitalized COVID-19 population and represented 66% (n = 1082) and 32% (n = 120) of the populations infected with ACE2-associated viruses and non-ACE2-associated viruses, respectively (Supplemental Tables 1a-c). Of the 30 COVID-19 studies that reported race, 24% (n = 12 157) of the included population was Black or African American (Supplemental Table 1a). Similarly, 32% (n = 15 167), 27% (n = 8535), and 5% (n = 1163) of the included population were white, Hispanic or Latino, and Asian, respectively (data not shown). The frequency of hospitalized COVID-19 patients that had diabetes mellitus, hypertension, and chronic kidney disease (CKD) were 35% (n = 18 959), 58% (n = 27 072), and 13% (n = 6054), respectively (Supplemental Table 1a). In hospitalized patients infected with ACE2-associated viruses, 20% (n = 261), 31% (n = 159), and 6% (n = 69) had underlying diabetes mellitus, hypertension, and CKD, respectively (Supplemental Table 1b). Similarly, rates of these underlying comorbidities in hospitalized patients infected with non-ACE2-associated viruses were 50% (n = 170), 50% (n = 6), and 29% (n = 107), respectively (Supplemental Table 1c).
Acute Kidney Injury in COVID-19 and in ACE2-Associated and Non-ACE2-Associated Viruses
Among hospitalized COVID-19 patients (n = 54 173), 44% were critically ill (n = 23 655) (Table 1). In patients hospitalized with ACE2-associated viruses (n = 1575), 94% (n = 1485) were critically ill whereas in hospitalized patients with non-ACE2-associated viruses (n = 370), 100% (n = 370) were critically ill. AKI frequencies did not differ between virus groups in critically ill patients: COVID-19 51% (95% confidence interval [CI]: 44%-57%), ACE2-associated respiratory viruses 56% (95% CI: 37%-74%, P = .610), and non-ACE2-associated viruses 63% (95% CI: 43%-79%, P = .255) or between ACE2-associated respiratory viruses and non-ACE2-associated viruses (P = .624) (Figure 2).
AKI in Cohorts of Hospitalized Adult (≥18 Years Old) COVID-19 Patients.
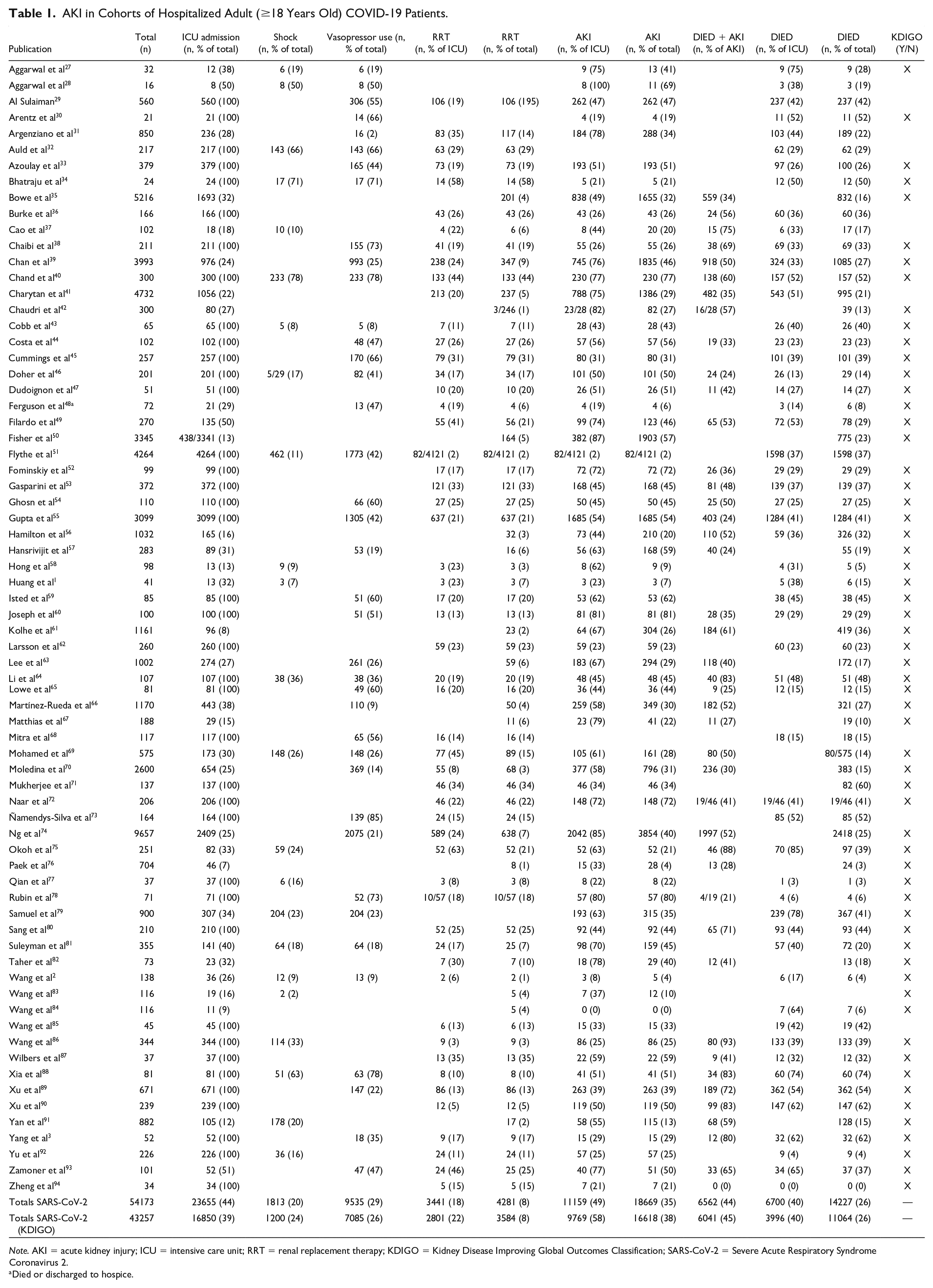
Note. AKI = acute kidney injury; ICU = intensive care unit; RRT = renal replacement therapy; KDIGO = Kidney Disease Improving Global Outcomes Classification; SARS-CoV-2 = Severe Acute Respiratory Syndrome Coronavirus 2.
Died or discharged to hospice.

Forest plots of acute kidney injury (AKI) in critically ill patients with COVID-19 and ACE2- and non-ACE2-associated respiratory viral infections.
Definitions of AKI varied between studies (Supplemental Table 2). After only including studies that defined AKI according to KDIGO criteria (61 studies, n = 43 257), the RE meta-analysis pooled AKI rate among critically ill COVID-19 patients was 52% (95% CI: 46%-58%).
Substantial statistical heterogeneity between studies was observed within each virus type (I2 = 98.1%, 83.9%, and 79.9% in critically ill patients for COVID-19, ACE2-associated, and non-ACE2-associated, respectively). The funnel plots (Figure 4) revealed no significant asymmetry (Begg test P > .05), but there seemed to exist some heterogeneity between studies. Four studies reported extreme AKI rate of either 100% or less than 1% (2 COVID-19 studies and 2 studies which examined ACE2-associated viruses).28,84,110,112 Sensitivity analysis which excluded these studies did not alter our conclusion for the comparisons between the 3 viruses.
Renal Replacement Therapy in COVID-19 and in ACE2-Associated and Non-ACE2-Associated Viruses
In critically ill patients, there were no significant differences in the pooled RRT rates between COVID-19 (20%, 95% CI: 16%-24%) and ACE2-associated viruses (18%, 95% CI 8%-33%; P = .747). However, both COVID-19 and ACE2-associated viruses were significantly different (P < .001 for both) from non-ACE2-binding viruses (49%, 95% CI: 44%-54%; Figure 3). Substantial statistical heterogeneity between studies was observed within each virus type, except for non-ACE2-associated viruses (I2 = 95.4%, 89.6%, 0% in critically ill for COVID-19, ACE2-associated, and non-ACE2-associated, respectively). The funnel plots (Figure 4) revealed one outlying study on ACE2-associated viruses. 112 Removal of this study did not alter our conclusion for the comparisons between the 3 groups of viruses.
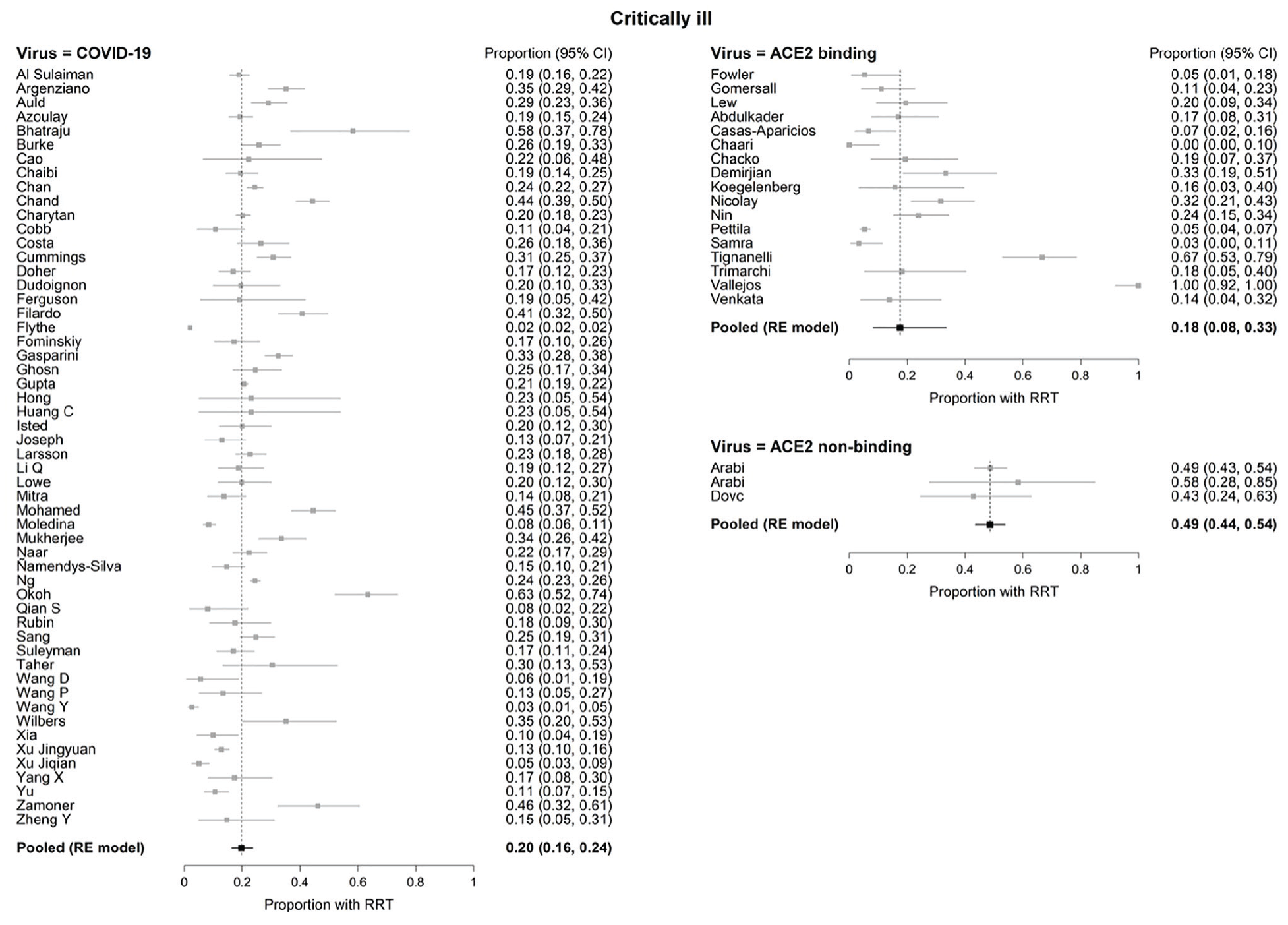
Forest plots of renal replacement therapy (RRT) in critically ill patients with COVID-19 and ACE2- and non-ACE2-associated respiratory viral infections.
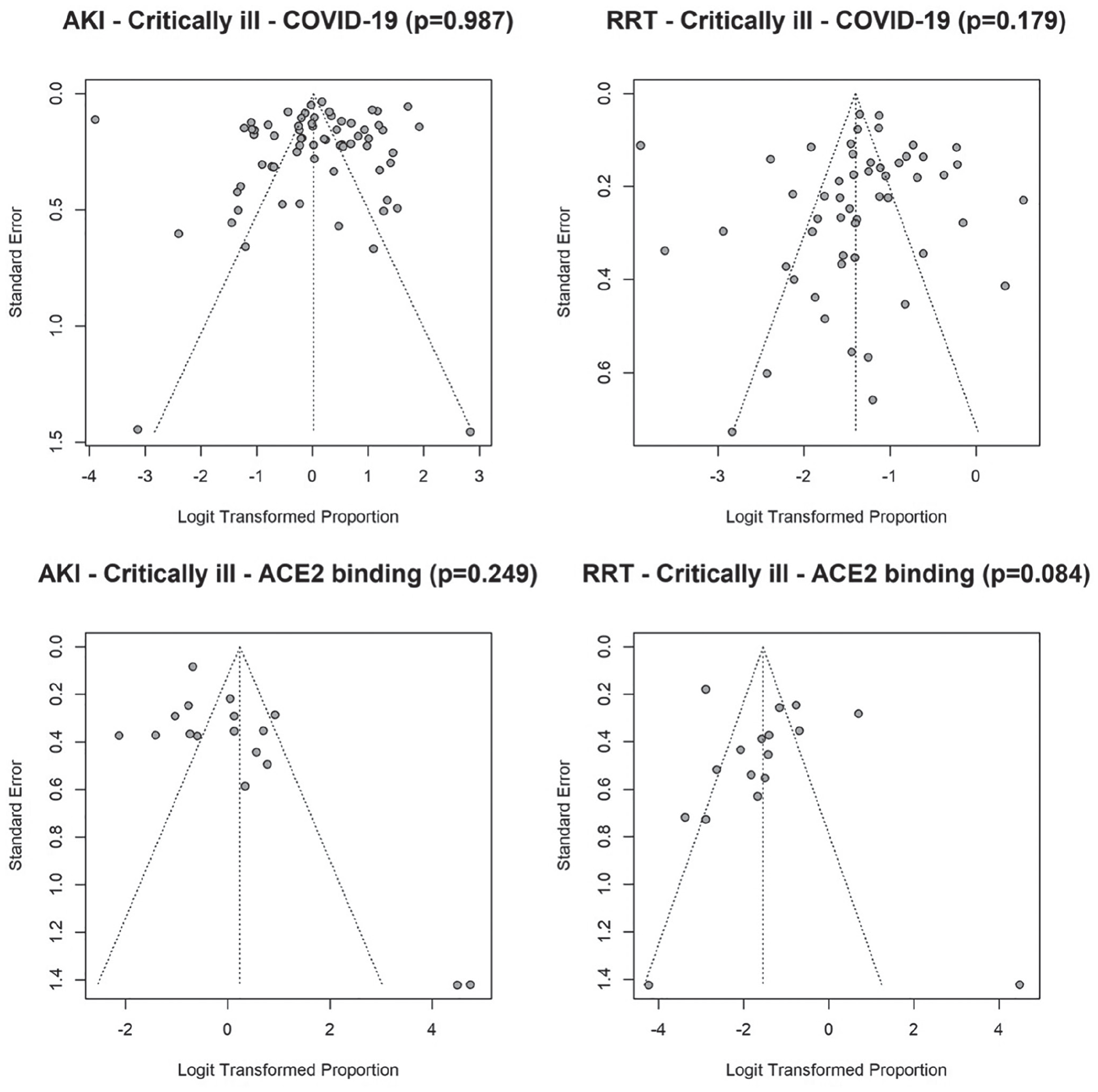
Funnel plots of acute kidney injury (AKI) and renal replacement therapy (RRT) in critically ill patients with COVID-19 and ACE2- and non-ACE2-associated respiratory viral infections.
After only including studies that defined AKI according to KDIGO criteria (61 studies, n = 43 257), RRT frequency among critically ill COVID-19 patients was 22% (n = 2801) (Table 1; RE meta-analysis pooled rate: 21% (95% CI: 17%-25%).
Shock and Vasopressors in COVID-19 and Viruses That Are or Are not Associated With ACE2
In critically ill patients, shock occurred in 24% (n = 1813/6621) of COVID-19 patients, whereas vasopressors were used in 26% (n = 9535/17 170) of patients (Table 1). In cohorts of patients infected with ACE2-associated viruses, shock was seen in 25% (n = 76/299), whereas in patients infected with non-ACE2-associated viruses, 78% (n = 267/342) of patients experienced shock (Tables 2D and 3C). Similarly, in cohorts of patients infected with ACE2-associated viruses, vasopressors were initiated in 44% (n = 390/884) whereas 78% (n = 289/370) of patients infected with non-ACE2 associated viruses received vasopressor treatment (Tables 2D and 3C). After adjusting for differences in prevalence of shock or vasopressor use between studies, the AKI and RRT rates were not significantly different between the 3 viruses (Supplemental Table 3). Shock and vasopressor use were, however, both positively associated with AKI (odds ratio [OR]: 1.31, 95% CI: 1.14-1.49 and OR: 1.26, 95% CI: 1.07-1.48, respectively) and RRT use (OR: 1.36, 95% CI: 1.18-1.56 and OR: 1.26, 95% CI: 1.11-1.42, respectively) (Supplemental Table 3).
AKI in Cohorts of Hospitalized Adult (≥18 Years Old) Patients Infected With Respiratory Viruses That Bind and/or Downregulate ACE2.

Note. AKI = acute kidney injury; ACE2 = angiotensin converting enzyme 2; ICU = intensive care unit; RRT = renal replacement therapy; SARS-CoV-1 = Severe Acute Respiratory Syndrome Coronavirus 1.
Vasopressor and inotrope use reported together.
AKI in Cohorts of Hospitalized Adult (≥18 Years) Patients Infected With Respiratory Viral Infections That Do Not Bind or Downregulate ACE2.

Note. AKI = acute kidney injury; ACE2 = angiotensin converting enzyme 2; ICU = intensive care unit; RRT = renal replacement therapy; MERS-CoV = Middle East Respiratory Syndrome Coronavirus.
Discussion
Among critically ill patients, there were no differences in frequencies of AKI between COVID-19 patients and patients infected with other respiratory viruses. However, RRT rate was significantly lower in patients with COVID-19 or ACE2-associated viruses when compared with patients infected with non-ACE2 associated viruses. Lower frequencies of shock and use of vasopressors may partially account for lower RRT frequencies in COVID-19 and ACE2 associated viruses, because adjustments for shock and vasopressor use revealed no significant differences in rates of RRT between virus classes. CKD prevalence was similar in COVID-19 (13%) and in patients who had infection with ACE2-associated viruses (6%) but lower than in patients with non-ACE2-associated viruses (29%).
The similar rates of AKI in critically ill patients across viral groups suggests, but does not prove, that downregulation or binding of ACE2 may not be associated with increased rates of AKI. The AKI rates in COVID-19 in critically ill patients may not differ from other viruses because there may be common biological mechanisms by which COVID 19, other ACE2-associated and non-ACE2-associated viruses lead to AKI. Studies of renal structure and function in COVID-19 suggest several mechanisms of AKI. Post-mortem renal tissue of COVID-19 patients with AKI shows viral particles in the kidney and SARS-CoV-2 nucleoprotein antigen accumulation in renal tubules and in one study (n = 3), in all renal compartments.118-120 However, there is debate on whether these particles are indeed viral particles or other intracellular structures. 121 Indications of diffuse proximal tubule injury, severe acute tubular necrosis, and lymphocyte infiltration have also been found.118-120 Evidence of renal tropism has also been found in cohorts of SARS, MERS, and influenza H1N1 patients.122-124 In one large study of patients with COVID-19-associated AKI, the most common cause of AKI was acute tubular necrosis, likely from hemodynamic instability possibly due to volume depletion, sepsis, or nephrotoxic drugs. 69 Similarly, in another study, acute tubular injury (ATI) was the main post-mortem renal finding in 33 COVID-19 patients who had AKI. 125 Post-mortem analysis of renal tissues of SARS and influenza H1N1 patients also revealed evidence of ATN.124,126
It is known that AKI and use of RRT are associated with an increased mortality rate. 127 Here, we found that the AKI-associated mortality rate was 42% for COVID-19 patients, including those who were not critically ill, which is much less than what was seen in MERS patients (81%). Despite potential common pathophysiological mechanisms, this finding lends support to distinct pathophysiological processes of AKI, even among coronaviruses. The high frequencies of AKI and use of RRT in Middle East Respiratory Syndrome (MERS) may be due to the high renal expression of dipeptidyl peptidase-4, the viral receptor for MERS-CoV. 21
A few other meta-analyses to date have also directly compared AKI and/or RRT rates among the different coronaviruses.128,129 In a meta-analysis comparing hospitalized patients infected with different coronaviruses and included studies until June 2020, the rates of AKI were found to be 9.0%, 9.6%, and 42.0% in COVID-19 patients, SARS patients, and MERS patients, respectively. 128 In addition, urgent start kidney replacement therapy rates were reported to be 3.4% and 35.0% for COVID-19 and MERS patients, respectively. However, the study did not report rates of AKI or RRT in critically ill, or ICU, patients as we have done here.
With respect to mortality, it was found that among patients infected with different coronaviruses including SARS-CoV-2, the COVID-19 AKI mortality rate (78.0%) was lower than in SARS (86.6%) patients but higher than in MERS (68.5%) patients. 129 Furthermore, another meta-analysis found that 72.3%, 98.9%, and 100% hospitalized patients with AKI who died had COVID-19, SARS, and MERS. 128 The latter but not the former analysis is consistent with our findings that COVID-19-associated AKI mortality rates are lower than those in SARS or MERS-associated AKI (COVID-19 46%, SARS 90%, MERS 81%). Discrepancies in mortality rates across studies may be due in part to emerging evidence that suggests that AKI in COVID-19 has two distinct clinical phenotypes based on whether AKI presents early or late in patients during the course of illness. 130 It has been suggested that different mechanisms underlie these phenotypes, each associated with an independent risk of mortality, with AKI-early possibly inciting multiple organ dysfunction whereas AKI-late may be a consequence of multiple organ dysfunction. Furthermore, AKI rates and AKI mortality appear to be much higher in the United States than in China, resulting in heterogeneity in AKI mortality rates among different studies. 131
Variable definitions of AKI over these eras of COVID-19 versus ACE2-associated and non-ACE2-associated viruses may have masked differing rates of AKI across viruses, which may have emerged had one standard definition of AKI been used across all included studies. Earlier and better recognition and resuscitation may also have decreased the risks of AKI in the COVID-19 era compared with the earlier eras of infection by ACE2-associated and non-ACE2 associated viruses. Given the difference in mortality rates between groups (COVID-19 25%, patients infected with ACE2-associated viruses 30%, patients infected with non-ACE2-associated viruses 63%), it is possible that more patients in the non-ACE2 associated groups died before developing AKI, thereby underestimating true AKI rates among these patients.
Shock, such as septic shock due to bacterial sepsis, and use of vasopressors are common precedents and causes of AKI and need for RRT because of shock- and vasopressor-associated induced changes in total renal blood flow and distribution of renal blood flow as well as increased levels of pro-inflammatory cytokines and other mediators of AKI.132-136 In cohorts of critically ill patients with COVID-19, 84% and 70% of non-survivors had septic shock while 60% and 50% had AKI, respectively, suggesting that sepsis and septic shock contribute to AKI among critically ill COVID-19 patients.12,86 Septic AKI may also be due to pro-inflammatory cytokines which worsen AKI as well as angiotensin II because it is pro-inflammatory.132-135 Here, adjusting for shock and vasopressors using meta-regression analysis removed the significant differences in rate of RRT between virus classes suggesting—but not proving—that the lower rates of shock and use of vasopressors explains in part the lower rate of RRT in COVID-19 and ACE2-associated viruses compared with the non-ACE2-associated viruses.
Our study complements and extends a meta-analysis by Silver and colleagues that found similar rates of AKI (46%, 95% CI: 35%-57%) in critically ill COVID-19 patients but included fewer studies (because they only searched up until October 14, 2020, and excluded studies not using KDIGO criteria to define AKI), did not adjust for presence of shock or use of vasopressors, and did not compare rates of AKI and RRT in COVID-19 with those rates in patients infected with viruses that do or do not downregulate ACE2. 137 Similarly, they found that kidney replacement therapy rates among critically ill COVID-19 patients was 19% (95% CI: 15%-22%). 137 After including studies that defined AKI using KDIGO criteria, we found that AKI and RRT rates among hospitalized, critically ill COVID-19 patients were 52% and 22%, respectively.
The low prevalence (6%) of pre-existing CKD among cohorts of COVID-19 patients suggests that having CKD may not increase patient susceptibility to SARS-CoV-2 infection (Table 1). Use of ACEi or ARBs are cornerstones of CKD treatment and may have conferred a protective effect on these patients. However, the role of ACEi and ARBs in protecting from COVID-19 is controversial and unproven thus far. It is unclear whether developing AKI associated with COVID-19 alters the risk of developing CKD later in life. 138
Our result that ACE2 association has a minimal impact on rates of AKI in hospitalized patients provides a rationale for trials of renin-angiotensin system inhibitors for the therapeutic management of COVID-19. Two recent randomized controlled trials of continuing or discontinuing ARBs in hospitalized patients already on ARBs found no difference in mortality between groups. However, we note that the mortality rates in the Brazil trial were very low (2.8% vs 2.7% continuing vs discontinuing ARBs, respectively) and therefore the trial was underpowered for such low rates.139,140
Our study has limitations which include the variable definitions for AKI used in different eras and in different cohorts. AKIN and RIFLE criteria were used to diagnose AKI in earlier SARS, influenza, and MERS studies whereas KDIGO was used in the majority of COVID-19 studies.22-24 While there is much overlap in the criteria, they are not identical, so some of the difference in AKI rates may be attributable to the different criteria used. We acknowledge that there is a possibility of overlap in reported cases as has been reported in other COVID-19 studies. 141 Another limitation is that we could not match severity of infection or do propensity matching across studies. Other limitations include the high I2 value associated with COVID-19 studies (98.1%) and that only one reviewer screened the literature and extracted the data. Last, we did not include non-English publications. The inclusion of many studies from China aided our accurate reflection of AKI frequency in COVID-19.
Conclusions
In conclusion, AKI frequencies were not significantly different between critically ill patients, that is, patients admitted to the ICU, with COVID-19 and other respiratory viral infections. However, the rate of RRT is lower in patients with COVID-19 or ACE2-associated viruses when compared with patients infected with non-ACE2-binding viruses, which might partly be due to the lower frequencies of shock and use of vasopressors in these two virus groups. However, despite being statistically significant, only 3 studies on non-ACE2-associated viruses were included in the meta-analysis and thus, more studies on non-ACE2-associated viruses are necessary to support this finding. Shock and vasopressor use may contribute to AKI and RRT in COVID-19. Lastly, underlying CKD rates were similar in COVID-19 and in patients who had infection with ACE2-associated and non-ACE2-associated viruses. Data from comprehensive prospective registries may elucidate true rates of AKI and RRT in COVID-19.
Supplemental Material
sj-pdf-1-cjk-10.1177_20543581211052185 – Supplemental material for Acute Kidney Injury and Renal Replacement Therapy in COVID-19 Versus Other Respiratory Viruses: A Systematic Review and Meta-Analysis
Supplemental material, sj-pdf-1-cjk-10.1177_20543581211052185 for Acute Kidney Injury and Renal Replacement Therapy in COVID-19 Versus Other Respiratory Viruses: A Systematic Review and Meta-Analysis by A. Cau, M. P. Cheng, Terry Lee, A. Levin, T. C. Lee, D. C. Vinh, F. Lamontagne, J. Singer, K. R. Walley, S. Murthy, D. Patrick, O. Rewa, B. Winston, J. Marshall, J. Boyd and Russell JA in Canadian Journal of Kidney Health and Disease
Footnotes
Authors Note
D. C. Vinh is now affiliated with McGill University Health Centre, Montreal, QC, Canada.
Ethics Approval and Consent to Participate
This work did not involve human subjects.
Consent for Publication
All authors provided consent for publication.
Availability of Data and Materials
Not applicable as original data was not collected for this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Russell reports patents owned by the University of British Columbia (UBC) that are related to the use of PCSK9 inhibitor(s) in sepsis and related to the use of vasopressin in septic shock. Dr Russell is an inventor on these patents. Dr Russell was a founder, Director, and shareholder in Cyon Therapeutics Inc and is a shareholder in Molecular You Corp. Dr Russell reports receiving consulting fees in the last 3 years from (1) Asahi Kesai Pharmaceuticals of America (AKPA) (was developing recombinant thrombomodulin in sepsis), (2) SIB Therapeutics LLC (developing a sepsis drug), and (3) Ferring Pharmaceuticals (manufactures vasopressin and developing selepressin). Dr Russell is no longer actively consulting for the following: (4) La Jolla Pharmaceuticals (developing angiotensin II; Dr Russell chaired the DSMB of a trial of angiotensin II from 2015 to 2017) and (5) PAR Pharma (sells prepared bags of vasopressin). Dr Russell reports having received an investigator-initiated grant from Grifols (entitled “Is HBP a mechanism of albumin’s efficacy in human septic shock?”) that was provided to and administered by UBC.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by grants from the Canadian Institutes of Health Research (CIHR).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
