Abstract
Objective:
The aim of this study is the comparative evaluation of minimal and metabolic flow anaesthesia with regard to hemodynamic stability, trends of anaesthesia gases, problems during maintenance, time to recovery and the consumption of anaesthetic agent.
Methods:
A randomised controlled prospective study was conducted on 60 patients of ASA grades I and II between 20 and 60 years of age undergoing routine surgeries under general anaesthesia using the Dräger Primus workstation. Patients were randomly allocated to Group 1 (minimal flow) and Group 2 (metabolic flow). Anaesthesia consumption was calculated manually using Dion’s method and the Dräger Primus machine also using the Dräger algorithm.
Results:
The two groups were found comparable with regard to the above-mentioned objectives. No statistically significant difference was observed between the two groups.
Conclusion:
Metabolic flow anaesthesia is safe and feasible on a day-to-day basis with the judicious use of advanced anaesthesia workstations and newer lower solubility halogenated agents.
Introduction
Minimal and metabolic flow anaesthesia techniques have been known since long. Many studies are available on low- and minimal-flow techniques in literature, and these techniques are widely used, while the studies utilising metabolic flow anaesthesia are relatively less.[1-3] The introduction of new lower solubility volatile agents and advanced anaesthesia machines with highly reliable monitoring systems has made even metabolic flow anaesthesia safe and feasible on a daily basis.[4,5] Further clinical, pharmacological, environmental and economic benefits have reaffirmed the role of minimal and metabolic flow anaesthesia in modern anaesthesia practice.[6,7]
The aim of our study is to compare minimal and metabolic flow anaesthesia using desflurane by comparing the consumption of the anaesthetic agent and looking for overall safety using the advanced workstation.
For safety, we are looking for hemodynamic stability, trends of inspired and expired anaesthetic gases, problems during maintenance and time to recovery.
Materials and Methods
After obtaining approval from the hospital’s ethics committee, a randomised controlled prospective study was conducted. Written informed consent was taken from 60 healthy patients enrolled for routine elective surgeries to be performed under general anaesthesia.
All ASA grades I and II, body mass index (BMI) less than 30 kg m–2, haemoglobin greater than 12 g dL–1, age between 20 and 60 years, undergoing routine surgeries were included in the study. Patients were randomly allocated into two groups using a table of random numbers. Group 1 (n = 30) patients receiving minimal flow anaesthesia 7 mL kg–1 min–1 with desflurane. Group 2 (n = 30) patients receiving metabolic flow anaesthesia 3.5 mL kg–1 min–1 with desflurane. Patients with cardiac disorders, lung disorders, pregnancy, haemoglobinopathies, haemoglobin less than 10 g dL–1 (anaemia), intoxicated patients and severe metabolic disorders, for example, diabetic-ketoacidosis, increased oxygen consumption states–drugs, endocrine disorders, hyperthermia, end-organ damaged states, undergoing major surgeries and emergency surgeries were excluded. In our study, the Dräger Primus (Lubeck, Germany) anaesthesia machine with a circle absorber with a Dräger D 2000 vaporiser outside the breathing circuit was used. It is suitable for minimal and metabolic flow anaesthesia techniques.
A routine pre-operative check-up of patients was done one day before surgery. A thorough history was taken and physical examination was done. Height, weight, heart rate (HR), blood pressure (BP), temperature and systemic findings were recorded and patients were optimised accordingly. The anaesthesia procedure was explained to patients and informed consent was taken. No pre-medication was given. On arrival at the operation theatre, an intravenous (i.v.) cannula was inserted and intravenous fluid started. Application of standard monitors included electrocardiogram (ECG), non-invasive blood pressure (NIBP), SPO2, end-tidal CO2 (etCO2). baseline HR, systolic BP (SBP), mean BP (MBP), diastolic BP (DBP) and SPO2 (oxygen saturation); the vital parameters were noted. Gas monitoring also included continuous FiO2 (fraction of inspired oxygen), (FeAN and FiAN) expired and inspired concentration of the agent, CO2. Ventilatory parameters monitored were minute volume, tidal volume, peak pressure, plateau pressure, mean pressure, respiratory rate, positive and end-expiratory pressure, and patient’s compliance. Each patient was pre-oxygenated with 100% oxygen for 3 minutes. Patients were induced with propofol 2 mg kg–1, and the analgesic used was opioid fentanyl 2 μg kg–1 i.v. This was followed by non-depolarising muscle relaxant atracurium 0.5 mg kg–1. Patients were ventilated with 6 l of oxygen for 3 minutes, and intermittent boluses of propofol 20 mg i.v. were given at 1 minute interval. After adequate relaxation, a cuffed endotracheal tube made up of polyvinyl chloride was inserted. Patients were connected to the anaesthesia circuit and the circle absorber and ventilated with the tidal volume of 8–10 mL kg–1 and a respiratory rate of 12 per min, and the I:E ratio was 1:2. The initial high-flow phase of oxygen was 6 l min–1 with desflurane at 8 vol%. The high-fresh gas flows of oxygen were reduced to minimal (Group 1) or metabolic (Group 2) on achieving a ratio of 0.8 or 80% between expired and inspired desflurane concentration (FeAN/FiAN = 0.8). This is known as the ‘equilibration point’.[1,8] Minimum alveolar concentration (MAC) was maintained 1 or above. FiO2 was maintained above 80% throughout. Later, analgesic 0.1–0.2 mg kg–1 morphine and 10 mg kg–1 paracetamol were given i.v. in the surgeries of longer duration. Relaxant boluses were given on a regular basis depending on the duration of surgery. Side sampling port drew gases into the respiratory gas monitor and allowed the measurement of FiO2, etCO2, expired and inspired concentration of the agent. Patients’ hemodynamic characters and ventilator parameters were noted. Trends of ventilatory gases were noted, and problems during maintenance, time to recovery and consumption were also noted for every case. Consumption was calculated manually as per Dion’s method.[9,10] The Dräger Primus anaesthesia machine gave the value of consumption as per the Dräger algorithm.[11]
Hemodynamic parameters including HR, SBP, MBP, DBP and trends of expired and inspired oxygen and desflurane were noted and compared between the two groups. Critical events (those which can be harmful to the patient or lead to the abandonment of minimal/metabolic flow), if occurred, were also noted. Such events included hypoxia (oxygen saturation less than 90%), hypoventilation (etCO2 > 55 mmHg), accidental increase in airway pressure (peak pressure > 30 cm of water), episodes of aborted ventilation (leak in the circuit leading to the abandonment of minimal/metabolic flow), alterations in haemodynamics (hypotension and hypertension).
For sample size calculation, we defined a relevant difference of 5% for ease of maintenance between the minimal and metabolic flow groups using desflurane, and about 30 patients per group were enrolled. Statistical analysis was done, and all values were expressed as mean ± standard deviation (SD). Independent continuous data (example HR, blood pressure and mean end-tidal agent concentration, ratio between inspired and expired agent concentration, recovery time and recovery score) were analysed using the analysis of variance (ANOVA) or unpaired t-test, and the P value of less than 0.05 was considered statistically significant. Mann–Whitney’s U-test was used for non-parametric (mean consumption of volatile agent concentration), skewed (non-normal) distribution for test of significance.
Results and Observations
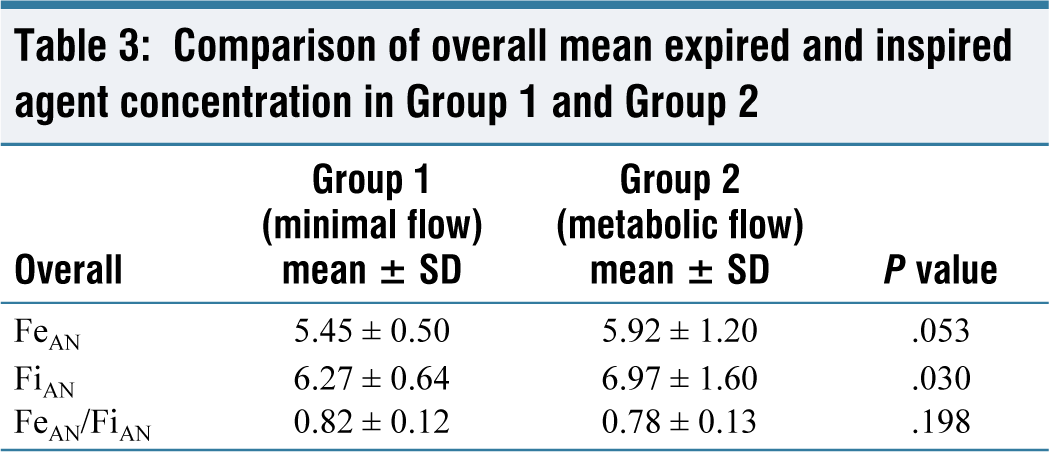
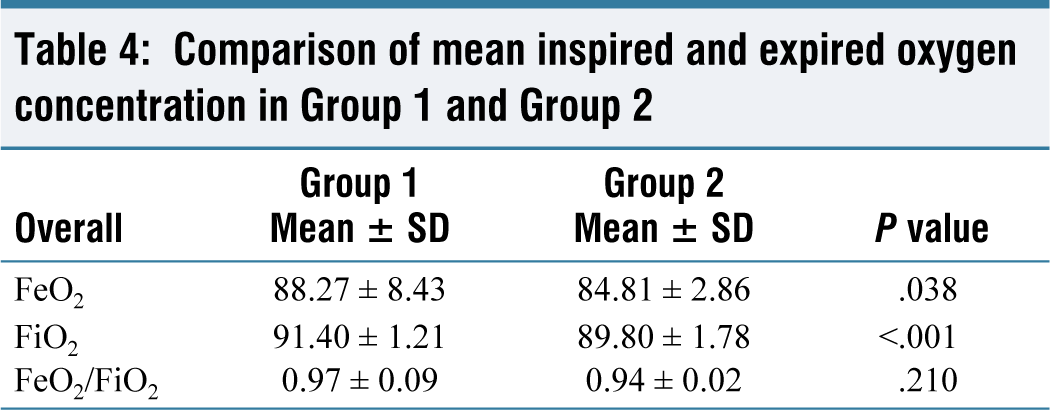
The two groups were statistically comparable with respect to age, weight, height, and BMI as shown in Table 1. There was no statistically significant difference in the mean HR, mean SBP, mean DBP and MBP between the two groups [Figures 1 and 2]. The mean equilibration time was studied between the two groups. The mean equilibration time of desflurane in the two groups did not show a statistically significant difference [Table 2]. Overall, the mean expired agent concentration did not show statistically significant difference in the groups, while the mean inspired agent concentration showed a statistically significant difference between the two groups, but this difference was clinically insignificant. The ratio of the mean expired and mean inspired of agent concentration did not show a statistically significant difference [Table 3]. Overall, the mean expired and inspired oxygen concentration showed statistically significant difference but clinically insignificant difference between the two groups [Table 4]. The recovery time was defined as the time from the discontinuation of the inhalational anaesthetic agent to the time patients opened their eyes. The time taken for recovery was noted in minutes. The values obtained for two groups did not show a statistically significant difference [Table 5].
Demographic profile of Group 1 and Group 2
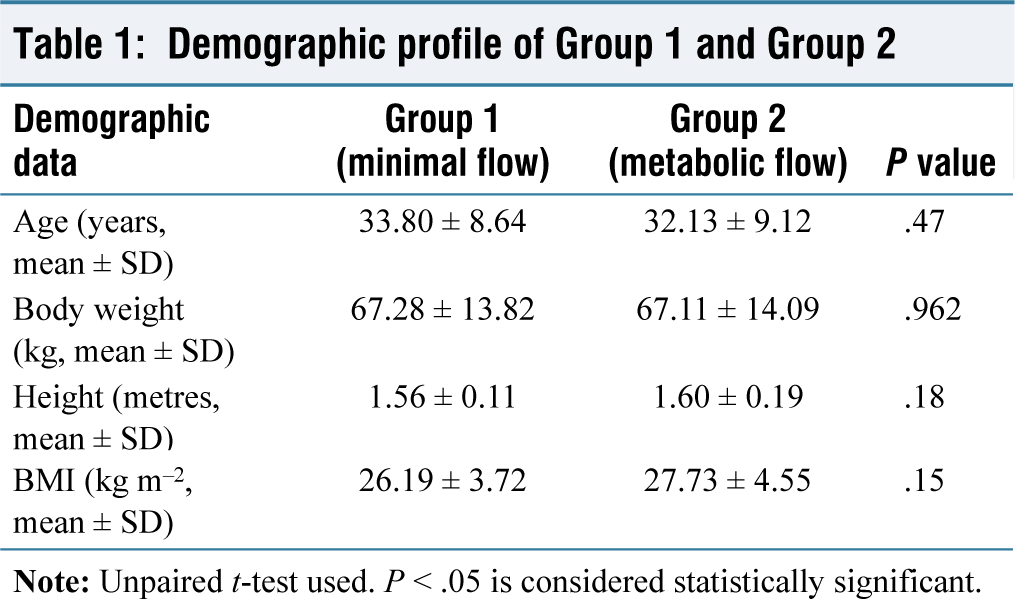
Comparison of heart rate in Group 1 and Group 2
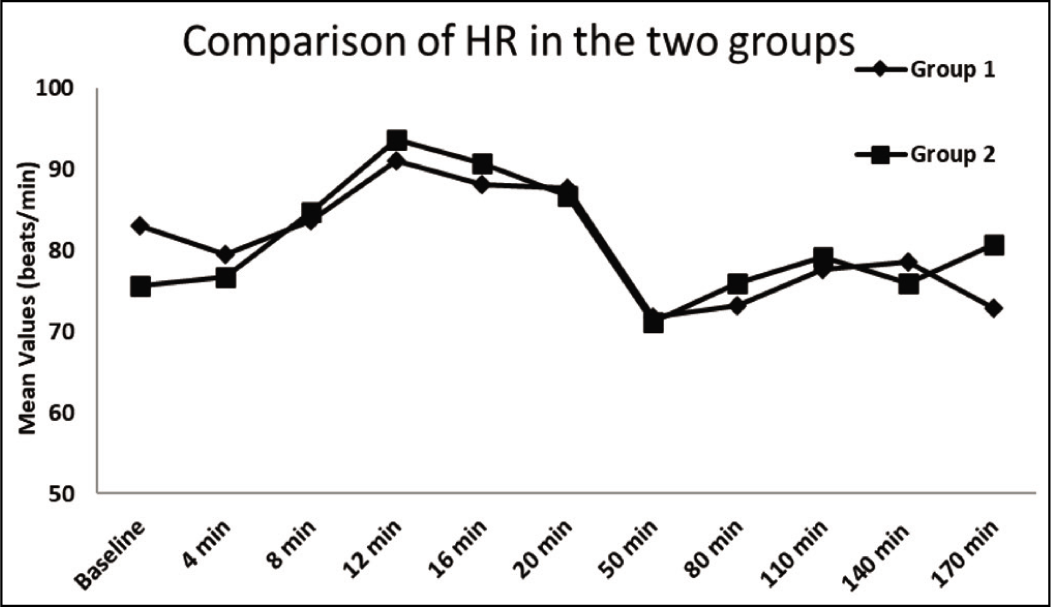
Comparison of mean blood pressure in Group 1 and Group 2
Comparison of equilibration time between Group 1 and Group 2

Comparison of overall mean expired and inspired agent concentration in Group 1 and Group 2
Comparison of mean inspired and expired oxygen concentration in Group 1 and Group 2
Comparison of time to recovery in Group 1 and Group 2

No critical event was recorded in our study. In 10 cases in Group 2, we had an alarm of low fresh gas flows during which the collapse of the reservoir bag was seen. Subsequently, all external connections were checked for leaks and the cuff pressure of the tube was checked. There was no significant leak or disconnection. Flushing was done to overcome the low fresh gas flow alarm. Oxygen flush was used to overcome this.
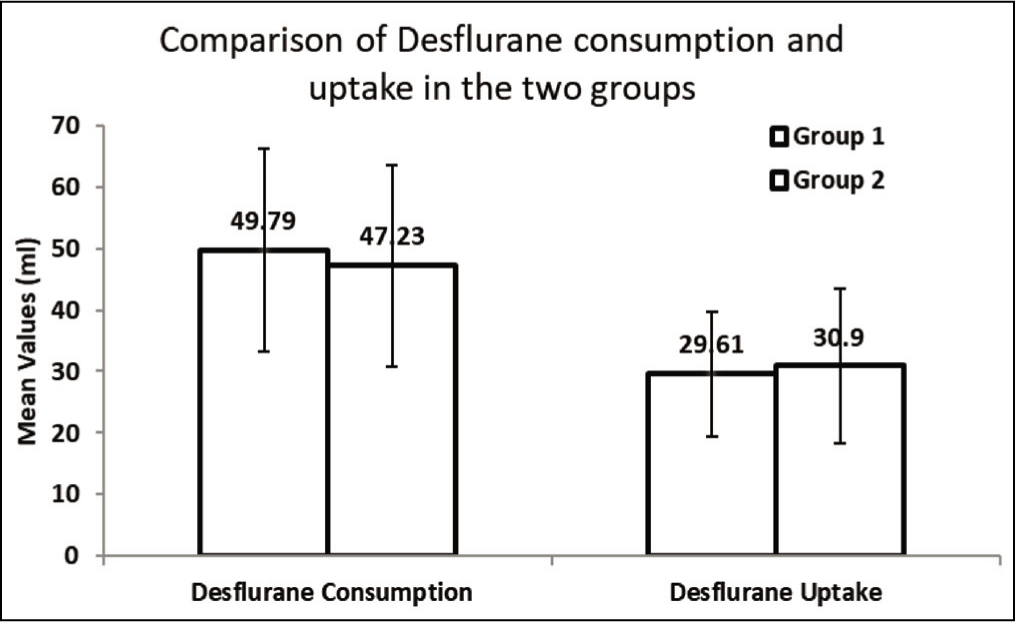
When using the Dräger method, desflurane consumption in the two groups did not show statistically significant difference, whereas Dion’s method showed statistically significant difference in desflurane consumption in the two groups. The reason for the difference could be the oxygen flush that was used in 10 cases of Group 2 whenever there was an alarm of low fresh gas flows. Flushing was not taken into account while calculating consumption manually applying Dion’s formula [Table 6, Figure 3].
Comparison of desflurane consumption in Group 1 and Group 2 by Dräger and Dion’s method

Comparison of desflurane consumption in Group 1 and Group 2
Discussion
We conducted our study in ASA grade I and II individuals of 20–60 years of age of either sex. Other studies using low flows had a similar demographic profile.[1,8] The selection of patients was done keeping in mind the safety from the point of view of balance between oxygen consumption and delivery, by observing contraindications from vulnerability to hyperoxia and individuals at increased risk from trace gases accumulation.[12] Thus, selection was based on patient-specific risks and requirements. Our study results cannot be extrapolated to other groups of patients. Surgeries included were laparoscopic/open-donor nephrectomy, breast surgeries, gynaecological surgeries and plastic surgeries. Our study results cannot be extrapolated to other groups of surgeries such as major emergency surgeries, surgeries of prolonged duration greater than four hours or short procedures with face masks or with imperfectly gas-tight airways.
The earlier definitions of metabolic and minimal flows are based purely on flow, and the recommended flow was a flow of 250 mL min–1 and 500 mL min–1, respectively, for metabolic and minimal flows.[13] This does not take into consideration the requirements according to the patient’s body weight. Total flow was a sole and important criterion as the delivery systems were not fully designed for very low flows. This can be considered fallacious as far as metabolic flow is concerned because it is always related to the metabolic requirement of oxygen, and this is usually based on weight in normal healthy patients and cannot be a fixed value of 250 mL min–1. We have tried to qualify the relation between flows and body weight of the patient by further adding a limiting factor, that is, BMI of more than 30 kg m–2 has been excluded. Hence, our decision to deliver 3.5 mL kg–1 min–1 in the metabolic flow group.[14-16]
Similarly, the definition of the minimal flow—whether based on the limitation of the delivering anaesthesia machine or focusing on the minimum oxygen in the gas mixture supplied—could have been made more rational by making the flow proportional to the body weight of the patient. This would allow us to titrate a flow according to the weight of the patient and not a fixed flow of 500 mL min–1. Hence, we made a decision to administer 7 mL kg–1 min–1 in our minimal flow group. Virtue’s minimal flow (300 mL oxygen and 200 mL nitrous oxide) delivered a metabolic flow of the oxygen component. The reduction in flow was limited by the minimum amount of oxygen of 300 mL, which was enough for a patient below 80 kg as recommended by Virtue et al.[17]
After pre-oxygenating the patient for 3 minutes, we induced anaesthesia with opioid and a hypnotic agent. Thereafter, the patient was mask-ventilated on high flows of oxygen without any inhalational agent. To prevent inhalational agent loss, boluses of propofol (20 mg) were given at 1-minute intervals after initial induction. The similar method was used by other studies as well, where it was found to be an effective technique for saving anaesthesia agent usage in a high-flow period.
Using the technique of ‘equilibration point’, the duration of high flows and thus the usage of the inhalational agent were drastically reduced, thus providing economic and ecological benefits. The mean equilibration time for minimal- and metabolic flows in our study were 4.71 ± 1.98 minutes and 4.97 ± 1.95 minutes, respectively. There was no statistically significant difference between the two groups. This is in contrast to a fixed period of high-flow states as recommended by earlier studies.
An attempt to keep the inhalational agent level constant by the manual adjustment of dial settings was done in our study by maintaining MAC of over 1 to prevent awareness and to meet certain surgical needs. Fresh gas flows were kept constant. This technique has been used in other studies as well, and this was found to be effective even with very low flows being delivered.[1,16] Techniques involving injection of liquid anaesthetics directly into the circuit have been described and used for metabolic flow anaesthesia.[18,19]
Hemodynamic stability was maintained throughout the study in both groups. There were no clinically significant changes from the baseline values in HR, SBP, DBP and MBP in both the groups. Similar results were seen in other studies.[20,21]
The expired/inspired concentration, the agent and oxygen, and their ratios were studied in both groups. The mean expired agent concentration did not show statistically significant difference in both the groups. The mean inspired agent concentration did show statistically significant difference between the groups, but the difference was not clinically significant. The mean inspired oxygen concentration showed statistically significant but clinically insignificant differences in Group 1 and Group 2. It never fell below 82% throughout the whole study. Similar ratios were also studied in other studies.[8,22]
No problem occurred during maintenance in Group 1. In 10 cases in Group 2, we had an alarm of low fresh gas flows during which the collapse of the reservoir bag was seen. Subsequently, all external connections were checked for leak, and the cuff pressure of the tube was checked. There was no significant leak or disconnection. Flushing was done to overcome the low fresh gas flow alarm. Dilution of circuit gases caused by possible room air entrainment was not observed in our study. Dilution of agent concentration was noted after flushing. The dial setting was adjusted to maintain MAC above 1.
We have used the Dräger Primus workstation for the purpose of our study. It is a modern care station with all features required for minimal and metabolic flow.[11] The appropriate use of the workstation results in enhanced safety and ergonomics of anaesthetic delivery and monitoring. The machine is equipped with an electronically driven piston ventilator which displays the alarm of ‘gas volume deficiency’ in case it happens. For any short-term imbalances between the fresh gas volume and the consumption/uptake, as well as leaks, our machine is equipped with a gas reservoir, the manual bag.
Consumption of desflurane was calculated by using Dion’s method. Our machine also gave the consumption as per the in-built Dräger algorithm for each case. Our study showed a statistically significant difference in desflurane consumption by Dion’s method between the two groups. Consumption was lower in Group 2 than Group 1. The Dräger algorithm failed to show statistically significant difference in desflurane consumption between the two groups. A possible reason for this could be intermittent bursts of high flow which were used in a few cases of Group 2 whenever there was an alarm of low fresh gas flows by the machine. This intermittent flushing was not taken into account while calculating consumption manually using Dion’s formula. The two methods of measurement of consumption showed a statistically significant correlation of 88% in Group 1 and 90% in Group 2.
The recovery and reversal were uneventful with our technique. The vaporiser was switched off with last stitch given by the surgeon. The mean time of recovery showed no statistically significant difference between the two groups.
Thus, we found out that the minimal- and metabolic flow anaesthesia were comparable with regard to hemodynamic stability, trends of ventilator gases, time to recovery and the consumption of the anaesthesia agent. No major problem occurred during the maintenance, except for leak in 10 cases of Group 2 for which oxygen flush was used. With newer agents and advanced machines with vigilant monitoring, it is possible to deliver metabolic flow anaesthesia on a day-to-day basis.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional ethical committee approval number
Institute ethical no: 101-20120-132-108951.
Patient consent
Consents from all the patients were taken for enrolling them in the study.
Credit author statement
The author had the following roles in the development of the paper:
Conception or design of the work: Muralidhar V, Gulbul Khurana.
Data collection: Gulbul Khurana, Jaylalitha MV.
Data analysis and interpretation: Gulbul Khurana, Sachin Kumar, Vikram Mahajan.
Drafting the article: Gulbul Khurana, Sachin Kumar, Muralidhar V.
Critical revision of the article: Sachin Kumar, Muralidhar V, Jaylalitha MV, Vikram Mahajan.
Other (study supervision, fundings, materials, etc): Jaylalitha MV, Muralidhar V.
All authors reviewed the results and approved the final version of the manuscript.
Data availability
Not available.
Use of artificial intelligence
There was no use of Artificial Intelligence.
