Abstract
Objectives:
The spectrum of metabolic disease, including obesity, prediabetes and Type 2 diabetes mellitus (T2DM), is increasing globally at an alarming rate. In addition to the vascular complications classically associated with diabetes, other ‘non-classical’ comorbidities like psychological distress, osteoporosis and sarcopenia are getting increasingly reported with diabetes. Hypogonadism in males has been reported to be associated with diabetes from the early 1990s. Around one-fourth of male subjects with diabetes mellitus were found to have decreased testosterone levels by several studies from both India and other countries. There is apparently a bidirectional link between T2DM and hypogonadism, with each contributing to and adversely affecting the other. This article briefly reviews the literature related to diabetes and hypogonadism, including the burden in an Indian setting, the pathophysiological link between the two, clinical evaluation and the evidence for and against testosterone replacement therapy.
Methodology:
Literature and information regarding this topic were collected from various original articles, review articles, systematic reviews and meta-analyses available on various platforms such as Google Scholar and PubMed.
Results:
The caveat here for clinicians is whether this low testosterone in a diabetic male who is more than 50 years old is due to late-onset hypogonadism (andropause) or is it a consequence per se due to diabetes. Physicians should remind themselves that a diabetic patient may also harbour other conditions directly leading to hypogonadism (like hypothalamo-pituitary lesions or primary testicular conditions). Thus, it is imperative that a thorough history and physical examination are mandatory in all diabetes suspected to have hypogonadism. Judicious use of relevant hormonal tests, including FSH, LH, testosterone and sex hormone binding globulin (SHBG), may be warranted to confirm hypogonadism.
Conclusions:
Appropriate referral to endocrine specialists for further endocrine evaluation of aetiology and management may be required in a subset of patients. In these patients with diabetes with a clear organic aetiology of hypogonadism, the latter is managed with testosterone replacement at age-appropriate doses. However, in the larger group of diabetes patients with functional hypogonadism with or without symptoms of androgen deficiency, testosterone replacement therapy is controversial. There are studies that show improvement in metabolic metrics of body composition, glucose, lipid and inflammatory parameters. However, the main concern of aggravating an underlying androgen-dependent occult malignancy (prostate), rise in haematocrit-related potential thrombotic tendencies, including cardio- and cerebrovascular events, is to be weighed against the potential benefits. Thus, the Endocrine Society guidelines currently recommend against testosterone treatment for improving the metabolic status (glucose levels, dyslipidaemia) alone in a diabetic patient. However, the Indian consensus (Integrated Diabetes and Endocrine Academy [IDEA] group) suggests individualising testosterone replacement after discussing it with the patient.
Introduction
Hypogonadism is defined as a syndrome due to inability of the testicular tissue to produce normal physiological levels of testosterone with or without defective spermatogenesis.[1] Generally, in routine clinical practice, normal testosterone secretion with isolated germ cell dysfunction is not considered equivalent to hypogonadism. From the causal point of view, hypogonadism may be due to defective hypothalamo-pituitary signalling to the testis (secondary hypogonadism) or due to testicular dysfunction (primary hypogonadism).[2] The incidence of diabetes and obesity are rising at epidemic proportions, and India probably has the dubious distinction of being called as ‘diabetes capital’ of the world.[3] Diabetes is directly linked with erectile dysfunction (ED), and the latter may be due to testosterone deficiency in at least in a small but significant proportion of those patients. In addition, after the fifth or sixth decade, testosterone levels may physiologically decline and is usually considered as andropause or late onset hypogonadism. In this background, many studies have demonstrated a significant association between hypogonadism and the spectrum of metabolic syndrome, namely central obesity, prediabetes and Type 2 diabetes mellitus (T2DM). Approximately 30% of diabetic patients are found to have low testosterone concentrations.[4] Testosterone levels correlate inversely with body mass index (BMI) and insulin resistance but usually have no correlation with the degree or duration of hyperglycaemia.[5,6] A potentially addressable and treatable condition like hypogonadism and diabetes has major implications in terms of increased cardiovascular (CV) risk factors and resultant increase in mortality. This review is an attempt to briefly review the current data and literature on hypogonadism and its relationship with diabetes.
Epidemiology
In the early 1990s, low testosterone concentrations were described in patients with T2DM [Table 1].[7] Further studies revealed a structurally normal testis and hypothalamo-pituitary-gonadal (HPG) axis in these patients. This led to the hypotheses of a functional hypogonadotropic hypogonadism in this setting.[8] Studies over the last few years have clearly established that at least one-fourth of men with T2DM have subnormal free testosterone (Free T) concentrations with inappropriately low to normal FSH and LH levels.[9] Another 4% have low testosterone levels with elevated FSH and LH concentrations.[10]
Various Indian studies have assessed the percent prevalence of hypogonadism in subjects with T2DM [Table 1]. In a study by Kumar et al it was found that testosterone levels were low in 70% men with moderate ED and 97% men with severe ED and coexisting diabetes.[11] Similarly, 11% men with diabetes were found to have hypogonadotropic hypogonadism in a study done by Ganesh et al.[12]
Indian data on percentage prevalence of hypogonadism in subjects with Type 2 diabetes mellitus

Diabetes and Gonadal Function: A Possible Bidirectional Link
Ever since the discovery of this high prevalence of hypogonadism in diabetes, studies have tried to establish the mechanistic link. The question of whether hypogonadism is the cause or consequence in a person with diabetes has been an ongoing debate. The following discussion in this chapter focuses on hypogonadism in T2DM, obesity and the insulin resistance spectrum. It is important for a clinician to remember that a male (especially of younger age) with diabetes may have other aetiologies that may have direct or indirect links to hypogonadism. For instance, Type 1 diabetes may be part of autoimmune polyendocrinopathy syndrome (APS 1 or 2) affecting gonads also. Similarly, hemochromatosis may cause diabetes and hypogonadism due to common iron overload related glandular dysfunction (pituitary or testis). Alcohol may cause both testicular dysfunction and chronic pancreatitis-related secondary diabetes. Congenital conditions like Klinefelter syndrome and cystic fibrosis are also associated with primary hypogonadism and infertility. A detailed discussion of these conditions is not dealt with here.
Effect of Diabetes and Obesity on Gonadal Function
Inflammatory mediators (e.g., tumour necrosis factor-alpha [TNF-a] and interleukin-1beta [IL-1b]) can suppress hypothalamic and pituitary hormones (gonadotropin-releasing hormone [GnRH] and LH) in vitro [Figure 1].[18,19] Insulin resistance reduces serum total testosterone by lowering the levels of sex hormone-binding globulin (SHBG).[20] Leptin is known to stimulate GnRH neurons, further inducing LH release under normal circumstances. It has been observed that elevating the adipokine leptin in diabetic patients directly suppresses the effect of gonadotropins on the Leydig cells (testes) to decrease testosterone production. In obese diabetic patients, the resistance to leptin may further diminish androgen function.[21,22] Aromatase activity in the adipose tissue of obese individuals leads to increased oestrogen secretion, which suppresses GnRH.[23] Minor mechanisms like increased glucocorticoid turnover and the dysregulation of the hypothalamic-pituitary-gonadal axis, leading to hypoandrogenism and altered function of leukocytes due to microvascular disruption in the testes, might also contribute.[24,25]
Mechanisms of hypogonadism in males with Type 2 diabetes mellitus
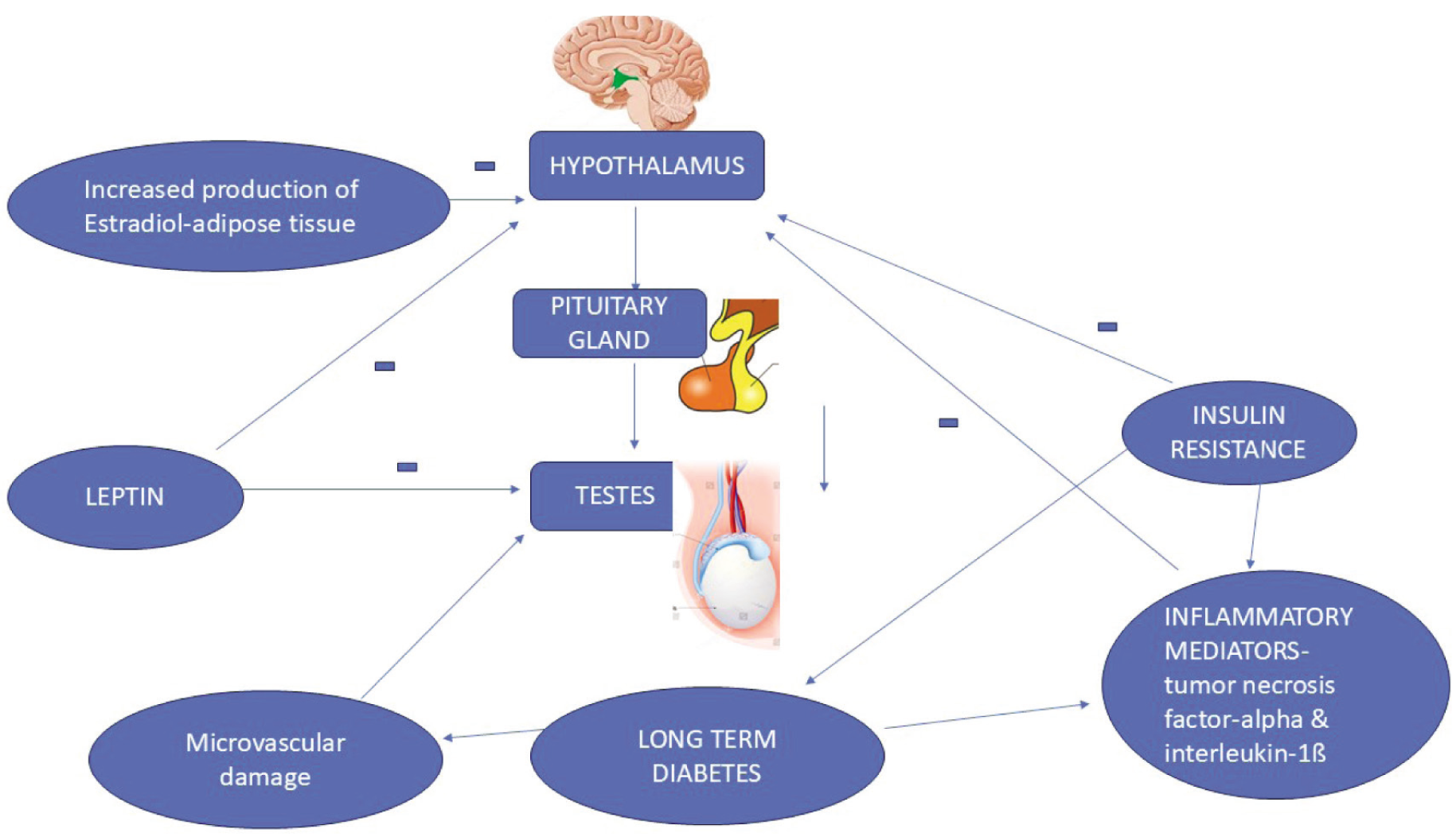
Effect of Hypogonadism and Diabetes
Meta-analysis has shown that men with decreased testosterone levels have an increased tendency to develop T2DM.[26] The main hypothesis proposed is that of central obesity and a higher degree of insulin resistance in individuals with low testosterone.[27] The other mechanism is the low level of GLUT4 in myocytes and adipocytes as a result of low testosterone. This leads to reduced transport of glucose and reduced insulin sensitivity, leading to T2DM.[28,29] Finally, abnormalities in the metabolism of lipids and altered body composition with increased central adiposity lead to diabetes.[30]
Why Should Clinicians Be Concerned About Hypogonadism in Diabetes?
In a patient with co-existing hypogonadism and diabetes mellitus, metabolic consequences extend beyond sexual health. Low testosterone levels in men are associated with increased CV and metabolic risk factor burden, which translates to increased prevalence of dyslipidaemia and atherosclerosis.[31,32] Various studies have shown that subnormal testosterone is associated with an increased incidence of CV events. Laughlin et al. showed that the hazard ratio for all-cause mortality was 1.44 and 1.36 for CV mortality in men with hypogonadism.[33] Another study—Osteoporotic Fracture in Men (MrOS)—a Swedish cohort of 3014 men (mean age, 75 years; mean follow-up, 4.5 years) showed a 65% mortality risk in men with low Free T levels (6.1 ng/dl).[34] Low levels of Free T are associated with a 69% risk of stroke or transient ischemic attack.[35] Hypogonadal men with diabetes have a reduced haematocrit than those with normal testosterone levels.[36] Hypogonadism is clear risk factor for osteoporosis (decreased bone mineral density), altered trabecular bone structure and increased fracture rate among men with diabetes.[37-39]
Evaluation of Hypogonadism in a Patient with Diabetes (Box 1)
Clinical features of hypogonadism are often vague in men with T2DM. Poor glycaemic control and underlying comorbidities might also contribute to fatigue, lethargy and reduced exercise capacity. ED is the most commonly reported symptom. It is reported in 30% men with T2DM and functional hypogonadism.[6] More often than not, ED in men with T2DM is not due to testosterone deficiency. It is often secondary to poor glycaemic control, peripheral vascular and autonomic nervous system involvement.[40] Underlying hypogonadism is indicated by decrease in libido and decrease in early morning erections.
Evaluation of hypogonadism in a patient with diabetes

A careful history is important to confirm the diagnosis of hypogonadism to determine whether they have other medical comorbidities, history of head injury, cerebrovascular accident, intra cranial neoplasms or pituitary surgery, chronic illnesses (CKD/CLD), toxic exposure, substance abuse, concomitant medications that can cause hypogonadism, or co-existing subfertility. Also, history starting from delay in puberty, early morning erection, shaving frequency, sexual history, symptoms of loss of smell, any prior testicular disease, excessive exercise should also be elicited. A history of recent fracture after trivial trauma might give a clue to chronic testosterone deficiency-related severe osteoporosis.
Physical examination includes examination of testes (size/consistency), the distribution and amount of body hair and changes in fat distribution. Post-pubertal testosterone deficiency does not affect penile length. Normal penile size, normal or small soft testes, regression of pubic and axillary hair regression, normal body proportions and decreased shaving frequency indicate post-pubertal hypogonadism, and organic causes other than diabetes should be ruled out. Any history of liver disease and skin pigmentation can give a clue to underlying hemochromatosis. Dysmorphism and cardiac malformation can point towards RASopathies. An assessment of the prostate either clinically or by sonogram and measuring prostate-specific antigen (PSA) are vital for obtaining clues for hypogonadism and monitoring testosterone therapy if planned. Intellectual changes, mood and cognition should also be assessed.[41]
Should We Measure Testosterone Levels in All Males with T2DM?
International societies like the American Association of Clinical Endocrinology (AACE) and American Diabetes Association (ADA) recommend checking levels of testosterone in diabetic patients with BMI > 30 kg/m2 because of the mechanistic link between obesity, diabetes and hypogonadism. But this is not possible in day-to-day practice as men are usually not willing to disclose sexual symptoms. IDEA consensus guideline (2021) recommends measuring serum testosterone levels in men with T2DM with features (signs/symptoms) suggestive of hypogonadism (reduced libido, reduced early morning penile tumescence and ED).[42]
Total Versus Free Testosterone: Which Is Better for Clinical Assessment?
Total testosterone assay is easily available, accepted and cost-effective to perform. Despite the variation in ranges and methods, physicians can check with their local laboratories for the acceptable values in their clinical practice. But in a person with diabetes, this may be confounded, and the value might be low due to reduced SHBG levels. Especially in an aging male with diabetes, elevated SHBG can normalise the testosterone value falsely. In an elderly male presenting with signs of hypogonadism and a normal total testosterone level, the clinician can check levels of Free T or SHBG and calculate bioavailable testosterone. Hence, guidelines recommend the measurement of Free T in patients with T2DM.[1,42]
It is difficult to check Free T by methods like equilibrium dialysis or ultrafiltration. These methods are largely unavailable but highly dependable. A radioimmunoassay for Free T is widely available but less reliable. As total testosterone and SHBG assays are readily available and cost-effective, calculating bioavailable testosterone might be a good alternative. Guidelines recommend using an equation based on SHBG, total testosterone and serum albumin. This can be alternatively used to calculate Free T where the estimation via equilibrium dialysis is not feasible.[1]
Whichever method is chosen, early morning measurements (8 am) after withholding the confounding medications is the norm. Lower levels should prompt repeat measurements to confirm the result.
In patients with repeated low testosterone, it is advised to measure FSH, LH and prolactin. Elevated FSH and LH levels suggest a primary testicular cause, and low or normal levels indicate a hypothalamic or pituitary cause. It is mandatory to rule out organic causes of hypogonadism in men with T2DM, low testosterone and low/low normal FSH/LH. In such situations, it is mandatory to do a pituitary hormone profile along with serum Free T4 and an MRI of the pituitary (as untreated hypothyroidism often mimics hypogonadism). Diagnostic cut-offs include 2.3 ng/ml for the lower limit for total testosterone (with levels <1.5 ng/ml necessitating referral to endocrinologist) and Free T <46 pg/ml.[43,44]
The androgen deficiency in aging male (ADAM) score is a widely used screening tool to identify men at risk for hypogonadism [Box 2]; it has a high sensitivity and a low specificity.[45]
ADAM questionnaire

Treatment
Androgen replacement therapy at age-appropriate, gradually escalating doses according to standard protocols is warranted for testosterone deficiency either due to hypo- or hypergonadotropic hypogonadism, especially in younger males. Monitoring is both clinical and biochemical, and the usual target is to maintain testosterone levels in the normal adult ranges. However, administering testosterone in a male with diabetes for primarily improving metabolic parameters like insulin resistance and dysglycaemia or lipid parameters alone is as yet controversial. Few studies have shown improvement in testosterone levels with a significant reduction in weight (more than 10%) by the patient (reversal of functional hypogonadism) following strict lifestyle measures or bariatric procedures.[46] With potent incretin-based therapies (semaglutide and tirzepatide) showing nearly bariatric intervention like weight loss, obese males with significant comorbidities like hypogonadism may be ameliorated by the use of the latter agents.[47]
Testosterone Therapy in Patients with Diabetes
With regards to testosterone replacement treatment (TRT) in hypogonadal patients with T2DM, the Endocrine Society (USA) recommends that testosterone therapy should not be used as a means of improving glycaemic control in men with T2DM with low testosterone concentrations.[1] An Indian consensus group suggested the use of testosterone replacement in men with T2DM and functional hypogonadism (serum total testosterone <2.3 ng/ml; <8 nmol/l) with sexual dysfunction after discussing the risks versus benefits. A therapeutic trial of testosterone replacement can be considered in the same group in borderline cases (serum total testosterone: 2.3–3.5 ng/ml) with symptoms.[42]
Testosterone is available in multiple forms: intramuscular injections, gel, patch, tablets or subcutaneous implantable pellets. If initiated, choice of testosterone formulation can be individualised (cost/ease of use, availability), etc. Transdermal testosterone gel was the most effective treatment for hypogonadal men with T2DM.[42] However, in an Indian setting, due to tropical weather-related sweating and other skin issues and cost, the latter mode of administration of testosterone is seldom popular.
However, most of the guidelines uniformly recommend monitoring of testosterone administration related adverse effects. Breast and prostate cancer are absolute contraindications for testosterone replacement therapy. The Endocrine Society recommends that testosterone therapy should be avoided in men with PSA >4 ng/ml, unevaluated prostate nodule, severe lower urinary tract symptoms with benign enlargement of prostate hypertrophy, untreated heart failure, haematocrit >48% and chronic liver disease.[1]
In spite of the above limitations and words of caution, there are potential benefits demonstrated in different clinical parameters related to testosterone treatment in patients with diabetes/metabolic syndrome including: glycaemic status, body composition (meta-analysis by Corona and colleagues),[48] QOL (quality of life), sexual symptoms, cognition, mood etc. One significant concern among clinicians regarding testosterone treatment is its potential adverse effect on haematocrit with attendant thrombotic consequences and CV health.
Effect on Glycaemic Control
Trials on the benefits of testosterone therapy on glycaemic control in T2DM patients with functional hypogonadism have yielded mixed results. The discrepancy between various studies mostly reflects the small sample size, differences in age, BMI, testosterone measurements/cut-offs, baseline HbA1c levels, degrees/measures of insulin resistance, and use of oral hypoglycaemic agents.
In the TIMES-2 study (testosterone [transdermal] replacement in hypogonadal men with either metabolic syndrome or T2DM), the investigators found improvement in CV risk factors such as insulin resistance, cholesterol, lipoprotein (a), body fat composition, and sexual function only in patients with metabolic syndrome but not diabetes.[49] Interventional studies showed a positive effect of TRT on insulin resistance and glycaemic status.[49]
Four landmark studies of testosterone replacement therapy in men with late onset hypogonadism have been published recently: (a) TEAAM, (b) TRAVERSE, (c) T4DM, (d) T-TRIALS and the salient points related to the trials are tabulated in Table 2.
Landmark trials on testosterone replacement therapy[50–54]
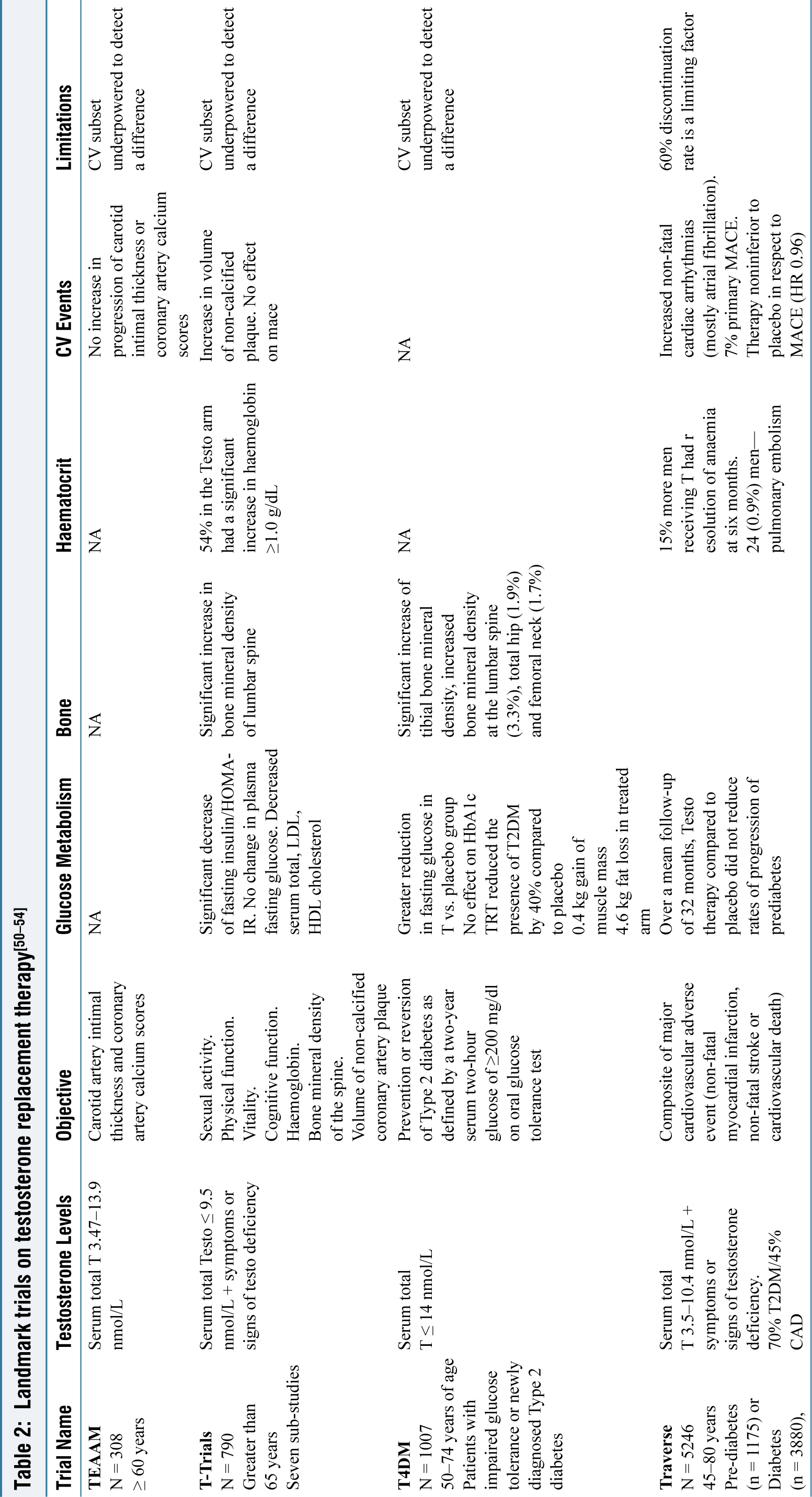
T-Trials and TRAVERSE showed a negligible effect on markers of insulin resistance or glycaemic control. Whereas the most recent T4DM offers a whopping 40% risk reduction in the development of T2DM. It offers not only proof of the bidirectional link between DM and hypogonadism but also a new therapeutic option in combination with lifestyle modification for diabetes remission.
Effect on CV Health
TEAAM, T-Trials and T4DM trials showed very low rates of major adverse cardiovascular events (MACE), but these studies were underpowered in that they failed to detect a difference. The TRAVERSE trial was a primary CVOT trial. However, the major limitation was 60% discontinuation rate. There was higher incidence of pulmonary embolism, arrhythmias (non fatal/atrial fibrillation) requiring intervention and acute kidney injury among patients who received testosterone.
Hence, treatment with testosterone should be individualised after assessing and explaining the risks versus benefits.
Summary and Take Home Points
Testosterone deficiency is recognised to be increasingly prevalent in men with the spectrum of metabolic syndrome (central obesity to prediabetes to diabetes). Among Indian men with T2DM, studies have shown a prevalence of confirmed testosterone deficiency in up to 10%-15%. From a clinician’s point of view, whenever erectile/sexual dysfunction is considered in a male with diabetes, it is prudent to thoroughly examine the patient for clinical clues of hypoandrogenemia as well as its aetiology (hypothalamo-pituitary vs. primary testicular causes). For patients with diabetes and clearly documented hypogonadism, both clinically, biochemically and etiologically, there is little or no controversy in replacement therapy with age-appropriate doses of testosterone. However, in diabetes patients with low normal testosterone values or subjects with symptoms suggestive of androgen deficiency but normal testosterone, option of testosterone replacement is still controversial. Multiple studies have shown the benefits of testosterone replacement by different parameters. However, concerns still remain regarding safety of long-term testosterone therapy with respect to occult prostate malignancy, rise in haematocrit and its thrombotic consequences including CV events. The risks versus benefits of testosterone replacement therapy in diabetes patients needs to be clarified by long-term follow-up studies. Thus, currently, clinicians have to be aware of the strong association between low testosterone and diabetes and the possible benefits; however, they exercise their clinical judgement and caution to individualise whom to screen for hypogonadism and replace with testosterone.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional Ethical Committee Approval Number
Not applicable..
Informed consent
No patients are involved in this review article.
Credit author statement
Both authors have contributed equally to reviewing the literature, discussion points, writing, corrections and re-reviewing manuscript.
Data availability
Not applicable.
Use of artificial intelligence
Nil.
