Abstract
Introduction:
Prognosis of vasculo-Behçet’s disease (VBD) is not consistent. Patients exhibiting arterial and central large venous involvement have the poorest prognosis. Given the significance of risk stratification in the management of cardiovascular diseases and the lack of specific diagnostic tests for BD, which depends entirely on clinical manifestations, this study aimed to identify clinical features linked to high-risk VBD for early management and enhanced outcomes.
Patients and Methods:
Clinical characteristics of 230 BD patients; 96 VBD and 134 non-VBD; were documented and analysed. For risk stratification, VBD were categorised into two clinical groups. Group (A): 37 patients with isolated arterial and/or central large venous involvement(s) who were identified as having high-risk VBD (Hr-VBD), and Group (B) [low risk VBD (Lr-VBD)] comprised 59 patients with isolated peripheral limb deep venous thrombosis.
Results:
Retinal vasculitis and neuro-BD (NBD) were less frequent in Hr-VBD patients (P = .03, .06, respectively). However, in multivariate analysis, only NBD was found to be less associated with Hr-VBD (P = .08). A comparison of VBD and non-VBD validated the inverse relationship between retinal and peripheral vasculitis in both univariate (P = .01) and multivariate (P = .049) analyses. Moreover, anterior, posterior, and pan-uveitis were all significantly lower in VBD in univariate (P = .01, <.001 and .03) and multivariate (.03, .001 and .045) examination. Furthermore, male gender (P = .01) and disease duration (P = .045) were associated with VBD.
Conclusion:
Ocular and NBD are less associated with VBD, and when they co-occur, VBD tends to be of low risk with consequent better prognosis. BD seems to have two distinct opposing pathologies: central ‘oculo and NBD’ and peripheral vasculitis.
Introduction
Vasculo-Behçet’s disease (VBD) refers to patients with BD who exhibit significant vascular symptoms. 1 The vascular aspects of the illness present a wide range of different manifestations with completely varied clinical outcomes. For example, arterial aneurysms and Budd-Chiari syndrome represent key vascular manifestations that are linked to serious complications and a high risk of mortality. 2 Prior research on VBD has examined the characteristics of vascular involvement as a whole in comparison to non-VBD3–5 without risk stratification and regardless of simultaneous involvement of multiple vascular sites. Recognising that BD diagnosis entirely relies on clinical symptoms and lacks specific diagnostic tools, the present study aimed to perform risk stratification of VBD and analyse the clinical features within each risk group. To our knowledge, this study is the first to address risk stratification of VBD. This approach enables early identification, vigilant monitoring, and management of Hr-VBD, which can lead to a more favourable prognosis.
Patients and Methods
This research involved patients visiting the outpatient clinic of the Rheumatology and Clinical Immunology unit within the Internal Medicine department at Kasr Alainy Medical School, Cairo University. The information was gathered from the available fully completed records of 230 Egyptian BD patients, 96 VBD patients and 134 patients without vascular involvement (non-VBD) from December 2020 to June 2022.
The updated international criteria for Behçet’s disease (ICBD) 2010 6 was selected as the inclusion criteria in this research due to a prior study demonstrating its effective performance among Egyptian BS patients. 7 For risk stratification, VBD patients were divided into two clinical categories based on their vascular involvement: Group (A) and (B). Group (A) consisted of 37 patients with isolated arterial and/or large central venous involvement ‘without peripheral venous limb involvement’ who were categorised as having high-risk VBD (Hr-VBD). Arterial involvement encompassed arterial thrombosis and/or aneurysms at any location. Large central venous involvement included superior and inferior vena cava (SVC, IVC), portal vein (PV), superior and inferior mesenteric veins (SMV, IMV), hepatic veins (HV), and internal jugular vein (IJV) thrombosis as well as pulmonary embolism (PE). Group (B) comprised 59 patients with isolated peripheral lower limb (LL) and/or upper limb (UL) deep venous thrombosis (DVT) without arterial or large central venous involvement, classified as having low-risk VBD (Lr-VBD). The detailed clinical characteristics of both groups, along with non-VBD, were recorded and compared. To specifically study Hr-VBD, patients with combined arterial and peripheral limb venous involvements and those with simultaneous large central and peripheral limb venous involvements were excluded from the study. Only isolated lesions in each group were permitted. The research was conducted following the 1964 Helsinki Declaration and its subsequent amendments. 8
Statistical Analysis
All data were entered and analysed using the Statistical Package of Social Sciences (SPSS) version 17.0, GraphPad Prism and Microsoft Excel XP. Normality was tested by the Kolmogorov-Smirnov test. Data were expressed as mean ± standard deviation or median for normally and non-normally distributed data, respectively. Comparison between groups was performed by the unpaired Student’s t-test ‘for quantitative data’ and chi square test ‘for categorical data’. Multiple logistic regression analysis was performed to study clinical manifestations associated independently with Hr-VBD. For all, the results were considered significant with P < .05.
Results
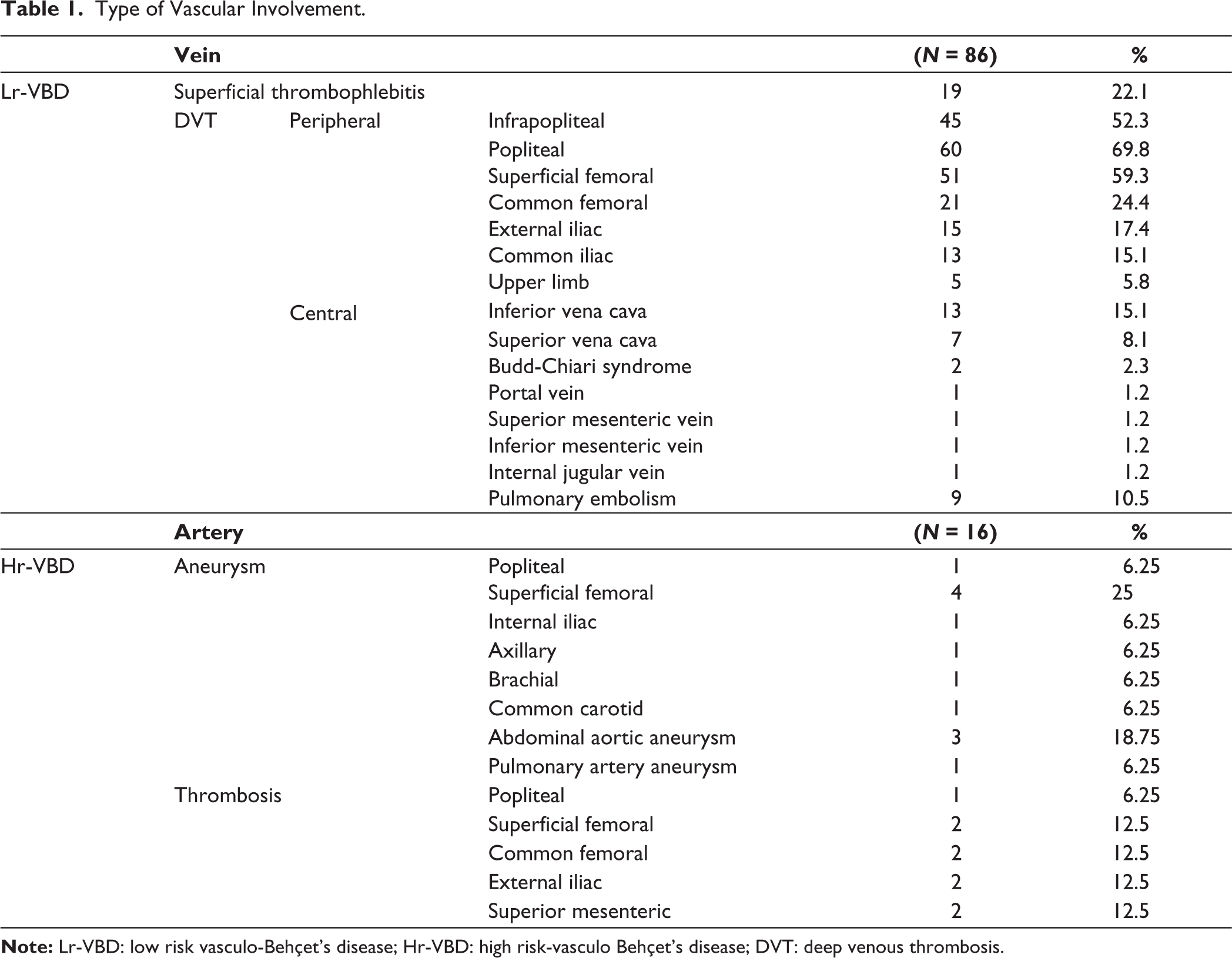
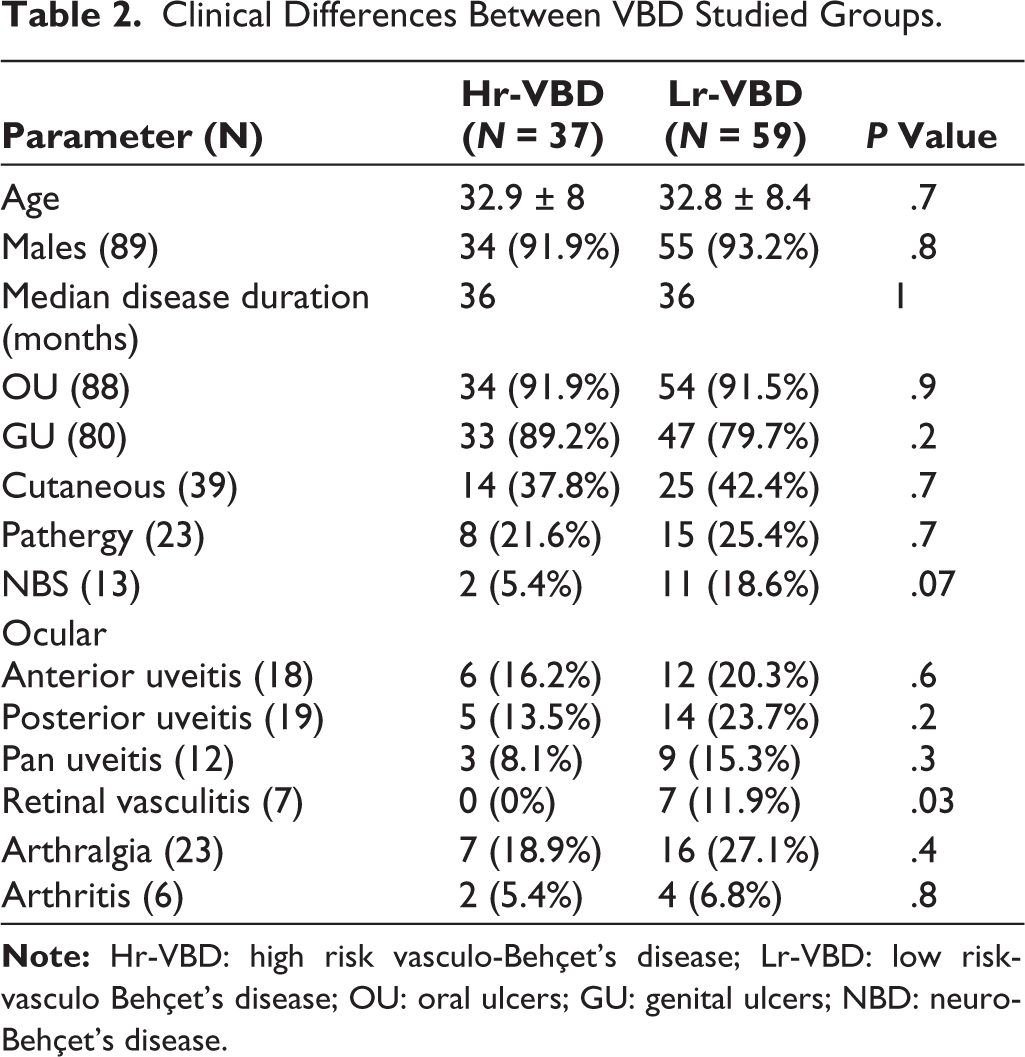
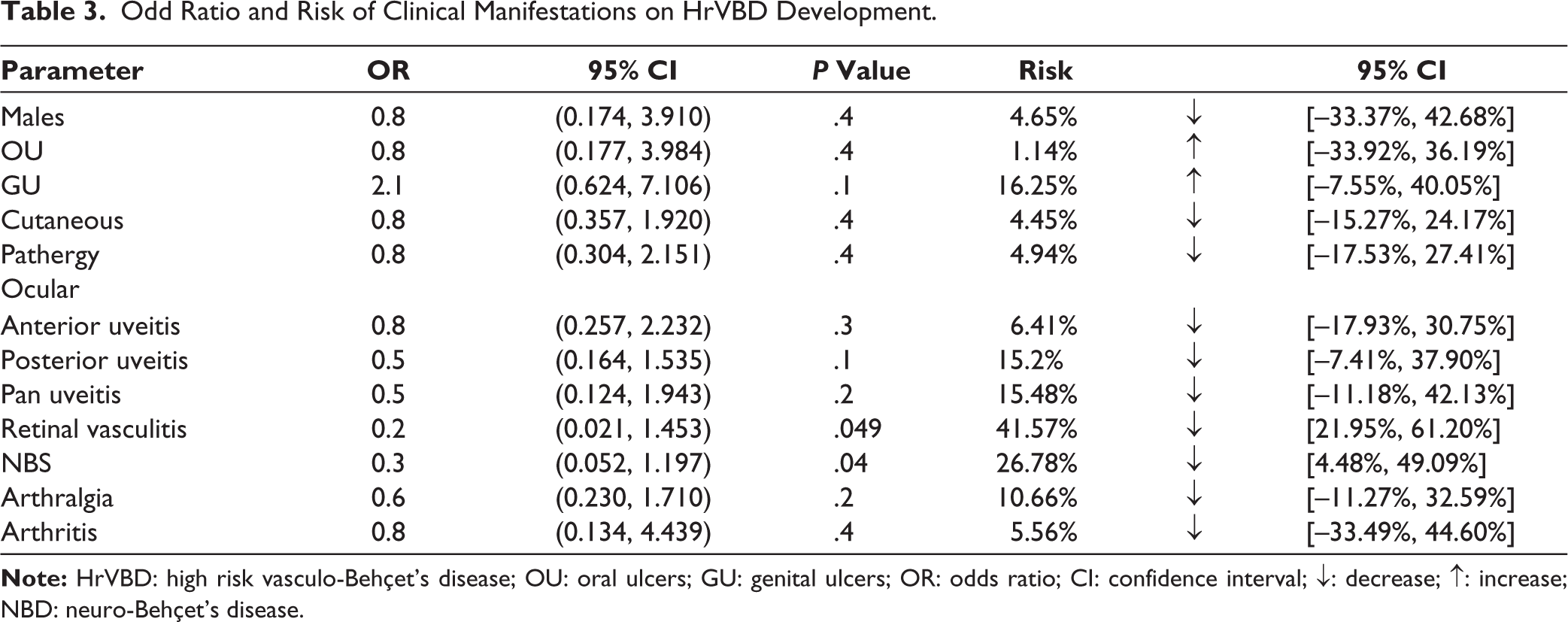
VBD group comprised 96 patients; 80 patients (83.3%) presented with venous lesions and 16 patients (16.7%) had arterial lesions. Documented venous pathologies included superficial thrombophlebitis [19 patients; (19.8%)], 59 patients (61.5%) with peripheral limb DVT; LL DVT was noted in 57 (59.4%) and UL DVT in 5 (5.2%); and large central venous DVT was found in 21 patients (21.9%). The large central venous DVT consisted of thrombosis of SVC ‘7 patients’, IVC ‘13 patients’, HV ‘2 patients’, with each of PV, SMV, IMV, and IJV ‘1 patient’, while PE was present in 5 patients. Among the 16 patients with arterial involvement, 13 patients (13.5%) exhibited arterial aneurysms, while arterial occlusion was seen in 4 patients (4.2%) (Table 1). The Hr-VBD group comprised 37 patients (38.5%) with isolated arterial involvement (16 patients) and 21 patients with isolated large central venous DVT and/or pulmonary embolism (PE), absent of any concomitant peripheral limb venous DVT. Of the Hr-VBD individuals, 34 patients (91.9%) were males with a mean age of (32.9 ± 8) years and a median disease duration of 36 months. The Lr-VBD cohort included 59 patients (61.5%) with isolated peripheral LL and/or UL DVT devoid of large central venous obstruction, PE, or arterial engagement. This group consisted of 55 males (93.2%) and 4 females (6.8%) with a mean age of (32.8 ± 8.4) years and a median disease duration of 36 months (Tables 1 and 2). Univariate analysis indicated that retinal vasculitis and NBD were both less linked with Hr-VBD 0/37 (0%) and 2/37 (5.4%) as opposed to 7/59 (11.9%) and 11/59 (18.6%) in Lr-VBD (P = .03, .07), respectively. Other clinical distinctions between the two groups studied are outlined in Table 2. The odds ratio for having Hr-VBD is significantly diminished in patients with retinal vasculitis and NBD [OR: 0.2 (95% CI: 0.021, 1.453), 0.3 (95% CI: 0.052, 1.197), P = .05, .04), respectively, with a relative risk reduction of 41.6% (95% CI: 21.95%, 61.20%) and 26.8% (95% CI: 4.48%, 49.09%) (Table 3). Following the elimination of non-significant independent variables, multiple logistic regression analysis revealed a negative relationship of Hr-VBD with NBD, [coefficient: -1.39, (95% CI: 0.05–1.2), P = .08] although this was not significant.
Type of Vascular Involvement.
Clinical Differences Between VBD Studied Groups.
Odd Ratio and Risk of Clinical Manifestations on HrVBD Development.
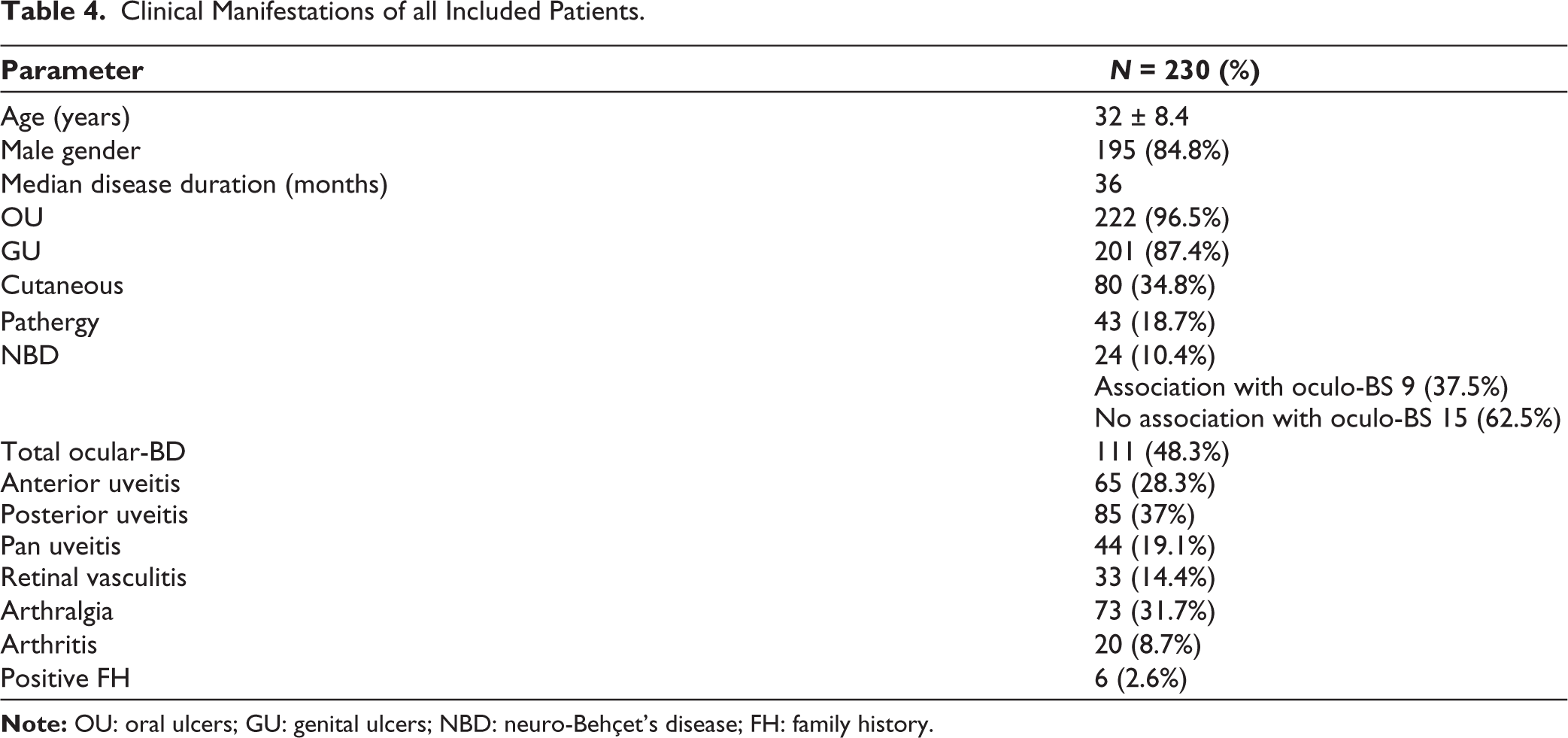
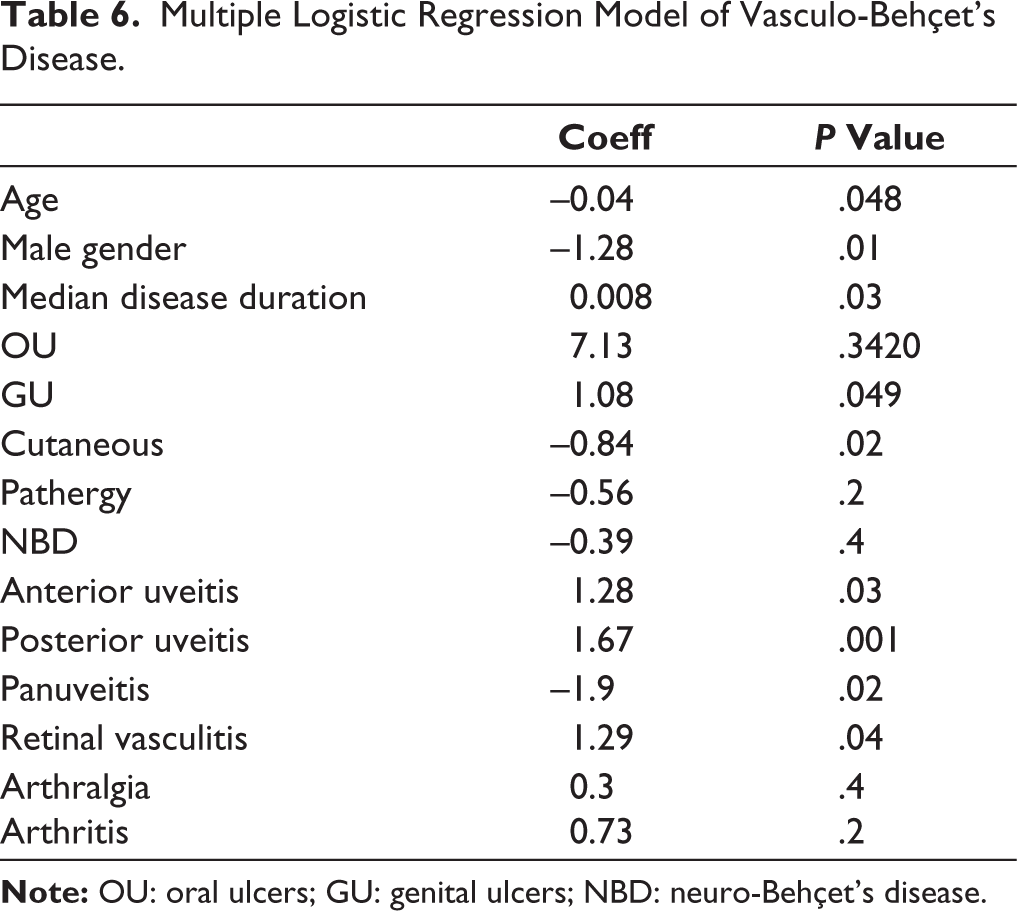
We incorporated non-VBD in this research to validate our results, ensure greater emphasis on vascular involvement in BD, and mitigate biased and random outcomes stemming from the retrospective nature of the study and the relatively small sample size. Considering all patients included (230 BD patients), VBD was present in 96/230 (41.7%), and the clinical manifestations of all included patients (N = 230) are displayed in Table 4. The mean age of the non-VBD group was (31.5 ± 8.4) years and consisted of 106 males (79.1%) and 28 females (20.9%) with a median disease duration of 26 months. Table 5 outlines the clinical spectrum variations between non-VBD and VBD patients. Multivariate analysis indicated that not only retinal vasculitis (P = .049) but also anterior (P = .03), posterior (P = .001), and pan-uveitis (P = .045) were linked to non-VBD. Male gender, disease duration, and cutaneous manifestations were all linked to VBD (Table 6).
Clinical Manifestations of all Included Patients.
Clinical Spectrum Differences Between Non-VBD and VBD.

Multiple Logistic Regression Model of Vasculo-Behçet’s Disease.
Discussion
As a vascular condition, in the present study, we introduced what we consider essential in the management of VBD, which is risk stratification. Risk scores and stratification are standard clinical practices for addressing cardiovascular diseases. These examples include but are not limited to coronary artery disease,9,10 stroke 11 and peripheral vascular disease. 12 Within the domain of vasculitides, risk stratification and scores were established for giant cell arteritis, 13 anti-glomerular basement membrane disease 14 and ANCA-associated vasculitis. 15
Ninety-six (41.7%) of the BD patients in the present study exhibited vascular involvement; this percentage is situated at the higher range of documented cases in previous studies ‘7.7%–43%’ influenced by ethnic variations.16–19 The male predominance was significantly evident in our research, with a male-to-female ratio of 3.8 to 1 in the non-VBD group, which surged to 12.7 to 1 in VBD patients. This observation serves as a notable instance of male predominance in VBD acknowledged by numerous authors.1,3,20,21 Our findings indicated that the duration of the disease was linked to VBD, possibly corresponding with the higher incidence of VBD in Egypt; in other words, it may suggest that through patient follow-up, VBD could develop during the disease progression. It is possible that BD with other clinical manifestations, such as OGU, gets diagnosed earlier and treated, thus preventing further vascular disease advancement. In the current study, venous and arterial involvement were observed in 83.3% and 16.7% of patients correspondingly. This aligns with several previous reports showcasing a clear dominance of venous involvement over arterial involvement in BD.22–24 Conversely, a limited number of studies reported nearly equal frequencies of venous and arterial involvement (24% and 18%) 25 and (36% and 33%), 26 with the absence of male predominance in the former study as well. This variation might be attributed to geographic and ethnic differences and/or sample size discrepancies. Consistent with our findings, numerous prior researchers indicated that superficial venous thrombosis and DVT, particularly in the lower limbs rather than the upper limbs, were the most common vascular manifestations of BD.27–29 Aligning with our results, one study found that the most frequently affected sites in the lower limbs were the popliteal and femoral veins, followed by calf tributary veins and iliac veins. 26 Lin and colleagues documented that the vena cava were the most common locations of large central venous thrombosis, primarily involving IVC thrombosis more than SVC, while hepatic venous obstruction (Budd Chiari syndrome) was only observed in a single patient. 21
According to our findings, retinal vasculitis is inversely related to Hr-VBD. When we included the non-VBD group, additional ocular manifestations were displayed as being negatively correlated with VBD, including anterior, posterior, and panuveitis. These results collectively affirm the inverse association between oculo-BD and VBD noted in previous studies.3,4 However, what is novel in our research is that this inverse relationship is also associated with severity, such that patients with Hr-VBD, who have the most severe cases among VBD and a poorer prognosis, are inversely related to retinal vasculitis, the most serious ocular manifestation in BD, with a significant reduction of Hr-VBD risk by 42% in patients suffering from retinal vasculitis. Furthermore, a similar observation was found in cases with NBD, another severe clinical manifestation of BD, but with significantly less risk reduction of Hr-VBD (15%). In one investigation, Lin and colleagues noted that patients with VBD along with uveitis often achieved remission and stable disease more frequently. 21 However, they attributed their results to the biological treatment these patients received, which was shown to be effective in managing refractory VBD. 30 Based on our results, we proposed another potential explanation that the improvement of VBD may be related to presence of uveitis itself, which is likely associated with less severe VBD with more favorable prognosis regardless of received medications.
In line with our study, ocular manifestations were more evident in BD patients without compared to those with arterial aneurysms with the well-known risk of the latter group. 31
In a previous study, we postulated that the eye as an embryologic outgrowth from the brain may be immunologically protected, so that in the setting of a florid form of peripheral vasculitis, the eye may still be relatively protected. 32
On the other hand, in one cohort of BD, vascular involvement was higher in patients with ocular-BD compared to controls ‘BD patients without ocular disease’. Of note, most of oculo-BD patients in this study had a severe disease (71%). 33 First of all, the mentioned result was statistically insignificant as documented by the authors themselves. Another important remark that in our study we confirmed the inverse relation of oculo and VBD in the multivariate analysis. Finally, we documented the inverse relation in severity between both disease spectrums.
In another study, in contrast to our results, central nervous system (CNS) lesions were more frequent in patients with VBD compared to those without vascular lesions. 21
This discrepancy may be explained by the small sample size of included VBD patients in the mentioned study ‘(5.6%) compared to non-VBD (94.4%)’ versus 41.7% and 58.3%, respectively, in our study, which may augment the results of NBD in the former study. Another important explanation is that included NBD in our study were patients suffering from parenchymatous CNS lesions only which enable us to accurately studying relation of NBD to vascular involvement without the confounding intracerebral vascular affection which carries the same characteristics of extracerebral corresponders according to our postulation. The latter explanation was confirmed by a finding from a previous study where there was a significant positive association between peripheral vascular disease and extra-parenchymal CNS involvement, while a poor association was found between parenchymal neurological and peripheral vascular involvements. 34 In another report, Bettiol and colleagues categorized BD into four distinct entities and classified VBD and intracerebral vascular lesions in a category and both ocular and parenchymal NBD in another different category. 35
All these observations strengthen the concept that vascular involvement in BD is a unique autonomous category in BD whether inside or outside the brain. In this context, immunosuppression represents a key strategy for the therapeutic management of central and peripheral vascular involvements.27,36
Notably, the positive association of ocular and NBD ‘central BD’ had been longely discussed in many previous reports.37–39 Even in patients with oculo-BD without overt CNS manifestations, they were found to have silent neurologic manifestations including neuropsychological deficits, subcortical magnetic resonance imaging (MRI) lesions and nonstructural headache. 40
The embryogenic process was postulated as an explanation for this positive association where neural tube and neural crest were involved in the organogenesis of the eye. 41 Also retinal pericytes are derived from the same embryologic origin as CNS pericytes. 42 However, it is worth noting to mention that despite the longely discussed positive association between ocular BD and NBD, there is still incomprehensible remaining questions like why the use of cyclosporine A was associated with flare of CNS manifestations in BD and should be avoided in such group of patients,43–44 while it is effective and recommended in ocular-BD. 45
All the above mentioned studies refer to two distinct and inverse pathologies in BD. One with peripheral vascular involvement and the other with ocular/neurological involvement(s). There may be a slight difference in the predisposition and pathogenesis of each type.
Plasma angiopoietin-1 (Ang-1) levels ‘an endothelial survival factor and vasculoprotective agent’ was significantly lower in VBD patients than in those without vascular involvement. 46 In contrast, plasma endothelin- 1 and 2, von Willebrand factor, antiendothelial cell antibodies with impairment of prostacyclin production and ischaemia-modified albumin ‘a biomarker of ischaemia, oxidative stress and endothelial dysfunction’ were significantly higher in patients with vascular involvement. 47 Other suggested markers of endothelial dysfunction in VBD include mean platelet volume, neutrophil–lymphocyte ratio, red cell distribution width, g-glutamyl transferase activity and uric acid levels.48–49.
Hamzaoui et al. showed an increase in serum levels of soluble CD28 in BD patients and vasculitis. 50 Other suggested mechanism of VBD includes lymphocyte activation with cytokines production, which stimulates neutrophils with resultant fibrinogen structure modification making it less susceptible to plasmin-induced lysis. This leads to endothelial dysfunction, platelet activation and over-expression of tissue factor leading to inflammatory thrombi, which are usually attached to the wall. 51
We think that presence of one or more of the above mentioned mechanisms may predispose BD patients to vascular involvement.
Though both CD4+ and CD8+ T cells have a potential cytolytic damage in BD via perforin- and Fas–Fas ligand-mediated pathways 52 , there was evidence in the literature that CD4+ T cell-mediated (namely Th1) mechanisms play an important role in the induction of vascular inflammation in a murine model of vasculitis with limited activation of CD8+ T cells in this model. CD4+ T cells specific for vascular antigens could accumulate, activated, clonally expand and induce vasculitis. 53
Auto-reactive CD4+ T cells were specifically activated in co-culture with vascular smooth muscle cells; another suggestion of CD4+ T cells pathogenic role in vasculitis. 54
On the other hand, the predominant intraocular infiltrating cells in BD uveitis were CD8+ T cells versus CD4+ T cells in other uveitides. Natural killer T (NKT) cells were much higher in aqueous humor than in other uveitides, and CD8+CD56+ cells were the predominant subtype among these NKT cells. 55
This predominant CD8+ T and NKT cells intraocular infiltration in active BD uveitis is in line with the unique increase in aqueous IL-15 levels in these patients compared to other uveitides.56–57 Of note, IL-15 is well known to be involved in the development and survival of immune effector cells, such as NK cells, NKT cells and CD8+ T cells.58–59
In another study, CD8bright CD56+ T cells population with cytolytic potential have been reported in patients with active Behçet’s uveitis with obvious harmful effects through both Fas ligand-dependent and perforin-dependent pathways. 60
Conclusions
VBD should not be managed as one bulk; risk stratification is warranted and recommended. BD seems to have two separate and opposing pathologies, which are also severity related; a central form including ‘oculo-BD and parenchymatous NBD’ and a peripheral vascular form with possible different pathogenesis.
Study limitations include its retrospective design and reliance solely on clinical manifestations, which are central to the diagnosis of Behçet’s disease. However, the absence of laboratory and radiological markers makes it challenging to differentiate between high-risk VBD, low-risk VBD and the proposed central and peripheral forms of the disease. Future research is warranted to address these gaps. We also recommend scoring system for risk stratification of VBD in further studies.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Patient Consent
All included patients in the study signed a written informed consent according to the protocol approved by the Research Ethics Committee for clinical studies at the Faculty of Medicine (Cairo University, Egypt) and in accordance with the 1964 Helsinki Declaration and its later amendments.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
