Abstract
Objective
To study the effectiveness of infliximab for the treatment of refractory central neuro-Behçet’s disease.
Methods
In this systematic review and meta-analysis, the research question was designed using the ‘Population, Intervention, Comparator, and Outcomes’ (PICO) model and the search methodology was developed according to the PRISMA statement. The study was registered on PROSPERO. Web of Science, PubMed, and Cochrane Library databases were searched for articles published in English between January 2000 and January 2020. Data were analysed using Meta-Essentials software, version 10.12. Treatment effect size was determined by a random effects model. Interstudy heterogeneity was explored using I2 statistics. Cumulative meta-analysis was conducted to assess the temporal trend for accumulating evidence.
Results
Twenty-one studies, comprising 64 patients (mean age, 38 .21 years and mean disease duration, 84.76 months) were included. Effect-size analysis showed that 93.7% of the treated patients in the analysis were responders to infliximab therapy (95% confidence interval 0.88, 0.993). There was no significant inter-study heterogeneity (I2 = 0%). Cumulative analysis showed accumulating evidence favoring increasing effectiveness over the last 20 years.
Conclusion
Infliximab showed considerable therapeutic effectiveness in the treatment of refractory neuro-Behçet’s disease.
Keywords
Introduction
Behçet’s disease, or Adamantiades, is an idiopathic inflammatory chronic relapsing multisystem immune-mediated disorder, characterized by recurrent oral and genital ulcers and uveitis. The disease displays geographic prevalence along the ancient Silk Road countries, extending from the Mediterranean region to Japan. 1 The major histocompatibility complex, class I, B (HLA-B)51 allele is currently identified as one of the genetic predisposing factors for Behçet’s disease, as it is observed in 50–80% of patients with Behçet’s disease and shows a prevalence of 20–25% in the general populations of the Silk Road countries. 2 Considering the relative deficiency of specific laboratory, radiographic and histopathological criteria, the accurate diagnosis of Behçet’s disease predominantly relies on clinical features. 3 Patients with Behçet’s disease may additionally present with a variety of neurological manifestations that may be related either directly or indirectly to the disease. The prevalence of neurologic involvement in Behçet’s disease varies between 5.3% and 59%, with lesions typically described as primary parenchymal (focal or multifocal) mostly presenting as subacute brainstem or corticospinal tract affection, and/or vascular disease (venous sinus or arterial thrombosis [ventral or spinal]). Despite extensive clinical and radiographic reporting of the neurological manifestations of Behçet’s disease, data regarding the long-term outcome and treatment of neuro-Behçet’s disease are scarce, and up to 25% of patients progress to moderate-to-severe disability. The clinical spectrum for the primary (direct or disease-related) neurological involvement in Behçet’s disease might include headache (migraine-like, non-structural); cerebral venous sinus thrombosis or extra-axial neuro-Behçet disease; central nervous system (CNS) involvement, described as intra-axial neuro-Behçet’s disease; arterial neuro-Behçet’s disease; neuro-psycho-Behçet’s disease; peripheral nervous system (PNS) involvement, and subclinical neuro-Behçet’s disease. 1 Other indirect forms of neurological involvement that might evolve as secondary comorbidities, include cerebral emboli from cardiac complications of Behçet’s disease, and increased intracranial pressure secondary to superior vena cava syndrome. 4 The management and secondary prevention of neuro-Behçet’s disease is challenging. Treatment of neuro-Behçet’s disease largely depends on general treatment guidelines for systemic involvement in Behçet’s disease.5–7 Colchicine, azathioprine, cyclosporine, cyclophosphamide, methotrexate, chlorambucil, thalidomide, interferon alpha, and anti-tumor necrosis factor (TNF) agents are among the list of approved drugs used for preventive treatment of the systemic features of Behçet’s disease and have also been tried for CNS involvement.5,8,9
The aim of the present systematic review and meta-analysis was to examine the effect of anti-TNF therapy on refractory neuro-Behçet’s disease, by performing a systematic review and meta-analysis of previously published studies assessing the effectiveness of anti-TNF therapy in patients with Behçet’s disease having severe, refractory, or recurrent neurological manifestations affecting the CNS.
Materials and methods
The research question for this systematic review and meta-analysis was designed using the ‘population, intervention, comparator, and outcomes’ (PICO) model, as follows:
Study population: adult patients 18 years and older with established diagnosis of Behçet’s disease according to the criteria set forth by the international study group,
10
with clinical and or radiographic features of acute central or spinal involvement as either an isolated feature or as part of systemic organ affection who failed conventional therapy. Intervention: infliximab anti-TNF monoclonal antibody therapy. Comparator: none. Outcome: resolution of clinical and/or radiographic features of central or spinal of neuro-Behçet’s disease.11,12
Study registration
This study was registered in PROSPERO, an international prospective register of systematic reviews: PROSPERO registration No. 132440.
Search strategy and selection criteria
The systematic review and meta-analysis protocol was developed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 13 The Web of Science, PubMed (National Library of Medicine, Bethesda, MD, USA), and Cochrane Library (The Cochrane Collaboration, London, UK) databases were searched for articles published in English language only from January 2000 to May 2020.
The two reviewers of the study (RHM and YWW) used the following search terms [infliximab therapy or chimeric monoclonal antibody therapy to tumor necrosis factor alpha or anti-tumor necrosis factor therapy] and [behcet’s disease or Adamantiades] and [neurological disease] for searching the literature. Duplicate publications were removed by the first reviewer (RHM), using the free text and medical subject heading terms by the two reviewers (RHM and YWW). Search results were independently refined by the two reviewers according to inclusion criteria, and conflicts were resolved via discussions by the same two reviewers. All fully published articles in English language on the date at which the search was performed were selected, screened, and checked for eligibility.
Eligibility criteria
The following inclusion criteria were applied:
Studies including adult patients with the diagnosis of Behçet’s disease according to criteria set forth by the international study group,
10
with features of severe or refractory acute/chronic progressive central or spinal involvement, as either an isolated feature or as part of systemic organ affection, and were started on infliximab either as their first line biologic for severe uncontrolled/refractory disease or after failure of at least one conventional and/or non infliximab biologic therapy who received the anti-TNF chimeric monoclonal antibody infliximab and were properly followed up for neurological disease outcome by clinical activity measures and/or radioimaging studies. Studies published in English language only. Studies published between January 2000 and May 2020 (a preliminary search of databases retrieved no articles matching the search criteria prior to 2000). Studies with available full-text publication.
Due to the significant lack of well-designed randomized controlled trials discussing the effectiveness of infliximab in neuro-Behçet’s disease, and considering this as an unmet need in clinical practice, despite long-term experience with the drug relative to other conventional therapies, all forms of eligible publications that investigated the research question (considering language, search period, and inclusion criteria), were gathered and included.
Exclusion criteria
Anti-TNF therapies other than infliximab, patients with neuro-Behçet’s disease without appropriate follow-up for outcomes, and other CNS diseases or systemic diseases other than Behçet’s disease that might involve the CNS.
Data extraction
Data extracted from studies included: article title, authors, year of publication, patients' age, sex, date of Behçet’s disease/or neuro-Behçet’s disease diagnosis, clinical manifestations of Behçet’s diseases (mucocutaneous lesions, eyes, joint, and vascular involvement) and neurological symptoms, disease score (if available), and brain magnetic resonance imaging (MRI) at diagnosis and follow-up. Data regarding therapeutic modalities (drug, dosage, and duration) were also extracted.
The current authors (RHM and YWW) proposed a simple response score for the study population with neuro-Behçet’s disease included in the meta-analysis (Hamdy and Woldeamanuel neuro-Behcet’s simple response score, 2020). This score is proposed by the reviewers to homogenize the assessment of the response by creating a simple item checklist including clinical, radiographic and or cerebrospinal fluid (CSF) responses with corresponding grading to provide a provisional quantification of the response. The reviewers derived the items of the score from the response assessment measures that were used rather uniformly in the studies and are commonly used in practice for measuring the activity and the response to a specific therapeutic intervention in patients with neurological disease. The proposed score includes the sum of clinical, radiographic points and cerebrospinal fluid analysis (or evoked potentials), resulting in a total score of 0–5, with a score of 5 defined as an excellent response and a score of ≥ 3 defined as a good response. Response items comprise the following:
Clinical response: worsened/no response = 0, stabilization = 1, no further relapses/improvement = 2. Radiographic-MRI response: worsened = 0, no progression = 1, regressed = 2. CSF changes (measured by improvement of neutrophilia and/or decrease in interleukin [IL]-6 and/or proteins): no = 0, yes = 1. Evoked potentials may be used as a substitute to CSF with a score of 1 denoting improvement.
In terms of treatment effect measures, outcome data, assessed as early as 4–12 weeks, were extracted regarding changes in neurological disease and MRI lesions with/without other possibly significant disease-related markers and/or CSF analysis.
Assessment of the risk of bias in the included studies
The Cochrane Risk of Bias in Interventional Studies (ROBINS-1) tool for nonrandomized interventional studies was used for addressing the risk of bias, 14 since all the eligible studies investigating the effectiveness of infliximab for patients with refractory neuro-Behçet’s disease were non-controlled, non-randomized studies, and the majority were case reports.
Statistical analyses
Data were statistically analyzed using Meta-Essentials software, version 10.12. 15 Aggregate data were included in meta-analysis using quantitative synthesis. Random-effects weighted meta-analysis on the proportion of remission rate pre- to post-infliximab treatment was analyzed. A random-effects model was chosen considering between-study differences in study designs. Interstudy heterogeneity was evaluated using I2 statistics. Cumulative meta-analysis was performed to assess for any temporal trend in infliximab effectiveness. Meta-regression was conducted to examine the confounding potential of age, sex, and disease duration. Results are presented as effects estimates with 95% confidence intervals (CIs), and a P value <0.05 was considered statistically significant.
Results
Results of the Web of Science, PubMed and Cochrane Library database search revealed a total of 1481 publications. Following removal of duplicates, 1470 publications were identified and screened for title, abstract, criteria and completeness. A total of 21 studies that directly addressed the effectiveness of therapy for neuro-Behçet’s disease met the study eligibility criteria and were included in the qualitative and quantitative analysis (Figure 1 and Table 1).8,9,16–34 The publications included two studies that investigated the effectiveness of infliximab in patients with multi-organ refractory Behçet’s disease and satisfied the inclusion criteria (Hibi et al., 2016, and Giardina et al., 2011).16,18 These studies were enrolled by the reviewers since they satisfied the inclusion criteria and presented objective measures to elicit the response of the neurological manifestations to infliximab therapy. Additionally, studies addressing different forms of biological anti-TNF monotherapy, including infliximab, in the management of neuro-Behçet’s disease were included using data only for patients receiving infliximab, provided that the inclusion criteria were met.
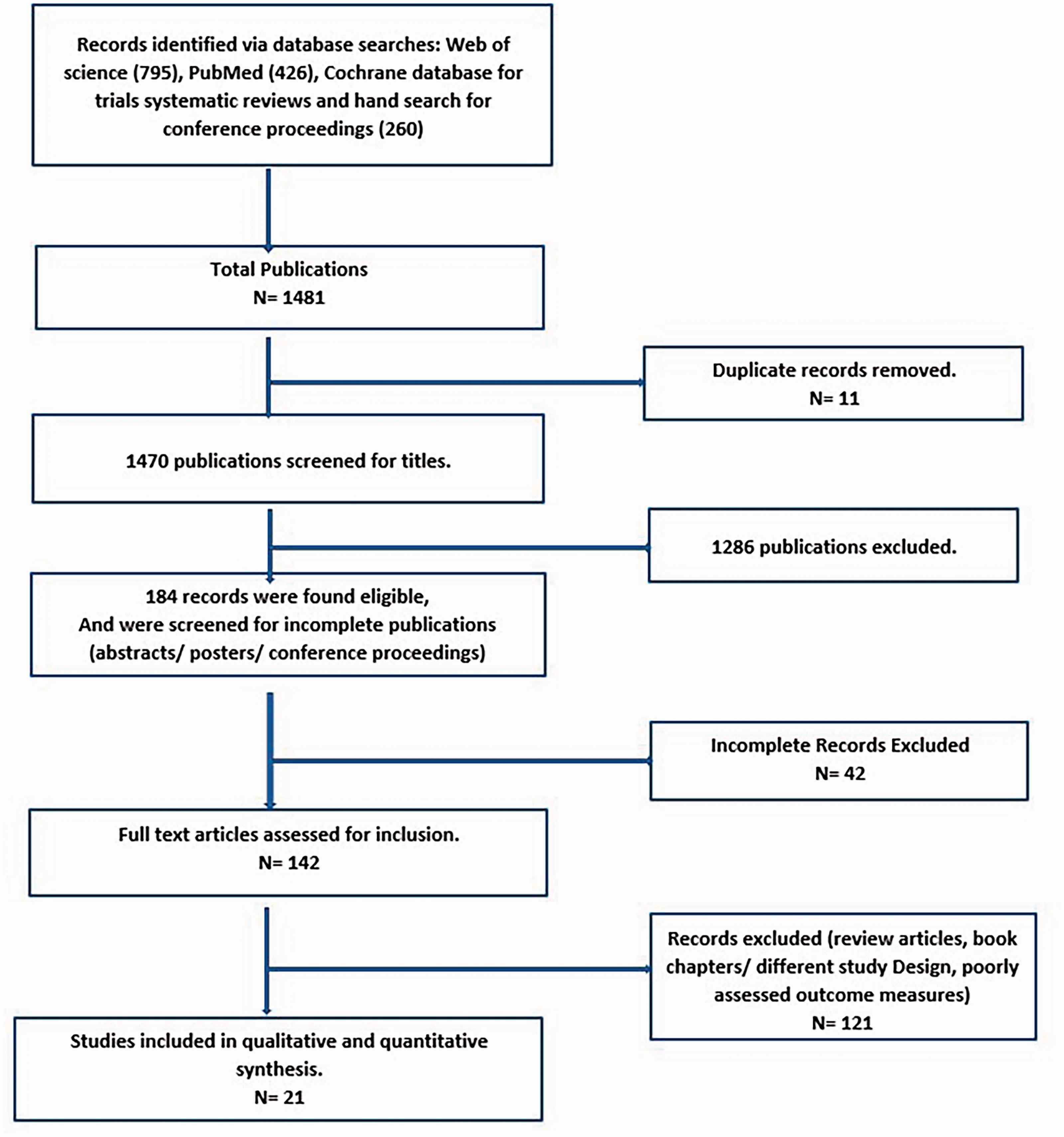
Flow diagram of search results and study selection (PRISMA model).
Details of 21 studies included in the systematic review and meta-analysis of anti-TNF therapy efficacy in patients with neuro-Behçet’s disease.*

BDCAF, Behçet’s Disease Current Activity Form; CNS, central nervous system; CSF, cerebrospinal fluid; IFX, infliximab; MRI, magnetic resonance imaging; NBD, neuro-Behçet’s disease; TNF, tumor necrosis factor; UTI, urinary tract infection.
All studies included adult patients (except Pipitone et al., 2008 that included one juvenile case) with a diagnosis of Behcet’s disease who had severe/ refractory/ relapsing or chronic form of neuro-Behcet’s disease who failed to respond to intravenous or oral corticosteroid therapy and/ or conventional immune suppression.
The presented meta-analysis included four open-label non-randomized single-arm interventional studies (trials) comprising 29 patients, and 17 cohort studies (including one prospective cohort study, two case series and 14 case reports) comprising 37 patients, in the intent-to-treat for drug effectiveness group that were pooled in the current study. Sixty-four patients who completed their assessment per protocol (52 males and 12 females, at a ratio of 4:1; mean age, 38.21 years; and mean disease duration, 84.76 months) were included in the final per-protocol analysis for the effect of the intervention.
The characteristics of the open-label interventional, cohort studies and case series included in this meta-analysis are summarized in Tables 1 and 2. Six studies were assessed for the risk of bias, and are summarised in more detail below.
Risk of bias assessment for non-randomized studies.*
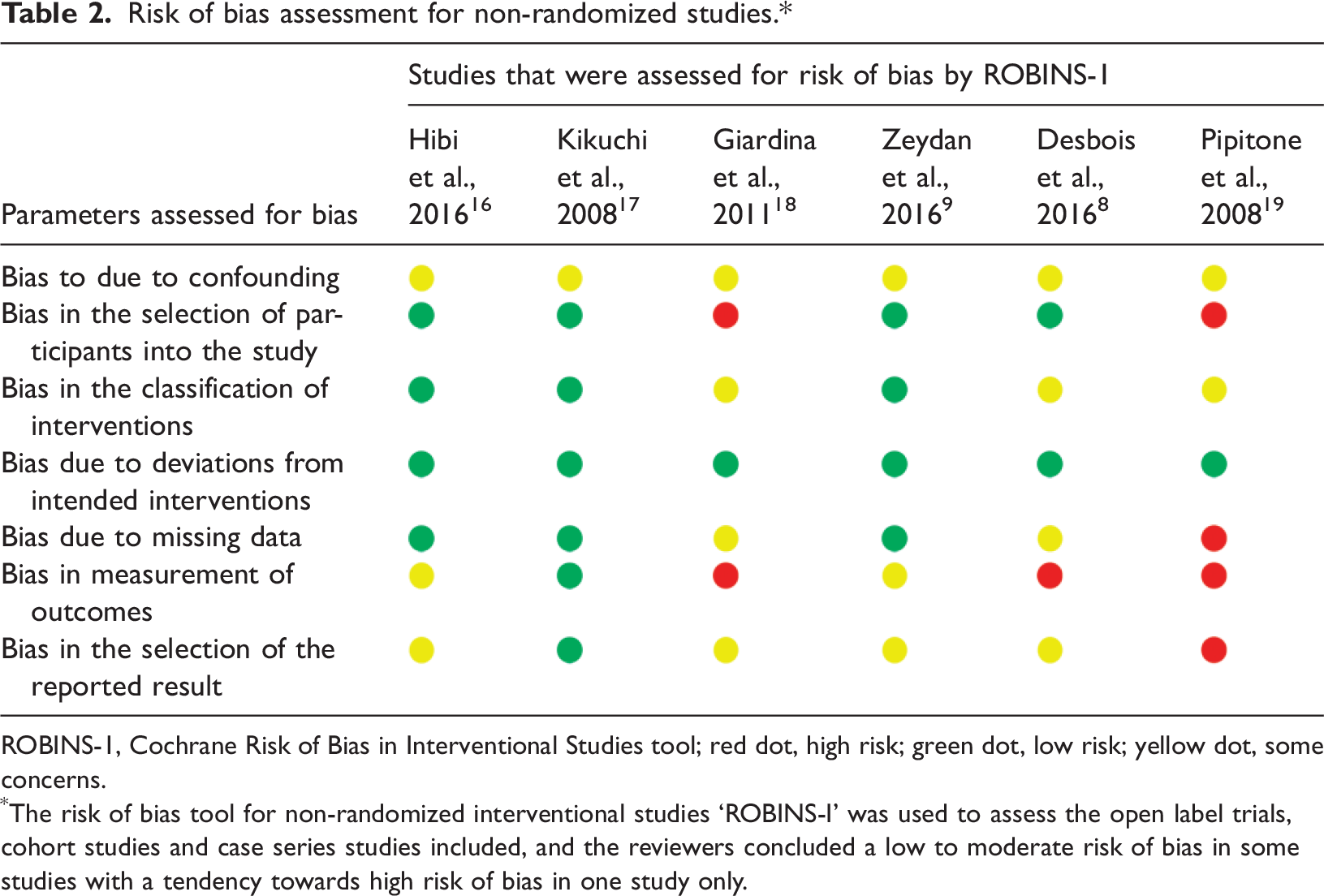
ROBINS-1, Cochrane Risk of Bias in Interventional Studies tool; red dot, high risk; green dot, low risk; yellow dot, some concerns.
The risk of bias tool for non-randomized interventional studies ‘ROBINS-I’ was used to assess the open label trials, cohort studies and case series studies included, and the reviewers concluded a low to moderate risk of bias in some studies with a tendency towards high risk of bias in one study only.
Hibi et al., 2016: 16 This study was a phase 3, prospective, open label, single armed clinical trial (ClinicalTrials.gov, NCT01532570) conducted at 21 medical institutions in Japan between January 2012 and May 2014. The study included patients who had been diagnosed with a complete or incomplete type of Behçet’s disease with intestinal Behçet’s disease, or neuro-Behçet’s disease, or vascular Behçet’s disease, as per the criteria defined by the Ministry of Health, Labour and Welfare (partially revised in 2010) for Japan, who had insufficient response or intolerance to conventional therapy. Acute neuro-Behçet’s disease was diagnosed by the presence of acute or subacute headache, pyrexia, or focal neurological symptoms and a cell count of ≥6.2 cells/mm3 in the CSF, or acute or subacute symptoms at least twice during the year preceding enrollment, with a cell count of ≥6.2 cells/mm3 in the CSF at symptom onset. Chronic neuro-Behçet’s disease was diagnosed by the presence of neuropsychiatric symptoms (dementia-like symptoms, psychiatric symptoms, truncal ataxia, dysarthria, etc.) and a CSF IL-6 concentration of ≥17.0 pg/ml at enrollment and at the most recent measurement within the year preceding enrollment, or a CSF IL-6 concentration of ≥17.0 pg/ml at enrollment and evidence of brainstem atrophy on brain MRI. A total of 25 patients were screened for eligibility, and 16 patients completed the study, including two patients diagnosed with acute neuro-Behçet’s disease and one patient with chronic progressive neuro-Behçet’s disease, who were on oral corticosteroids (mean age, 38.3 ± 8.5 years, all male patients with incomplete type Behçet’s disease). Patients with neuro-Behçet’s disease were assessed for the response by clinical examination, brain MRI (assessed by two Behçet’s disease specialists blinded to patient symptoms and time of imaging), and CSF (cell count and IL-6 concentration) analysis and inflammatory marker (C-reactive protein or erythrocyte sedimentation rate) levels. For patients with neuro-Behçet’s disease, the severity of clinical symptoms was scored at weeks 2 and 6 and every 4 weeks thereafter in terms of changes from the data recorded at week 0, while brain MRI was performed at weeks 0, 14, 30, and 54, with comparison to baseline MRI. Cell count and IL-6 concentrations in the CSF were measured at weeks 0, 14, 30, and 54. The incidence of acute or subacute attacks occurring between weeks 0 and 30 and between weeks 30 and 54 was determined. The first patient with acute neuro-Behçet’s disease experienced resolution of the acute symptoms, apart from mild headache, and the CSF cell count and IL-6 concentration dropped to 7 cells/ml and 1.8 pg/ml, response was sustained until week 54 (at week 54: cell count, 1 cell/ml; IL-6 concentration, 1.5 pg/ml). The second patient with acute neuro-Behçet’s disease experienced dull headache as a chronic symptom until week 22, no acute symptoms, however, the patient withdrew consent and discontinued the study at week 22 (CSF cell count was 2 cells/ml at week 14 and at withdrawal). In the patient with chronic neuro-Behçet’s disease, CSF IL-6 showed an initial rise in concentration that declined to 35.1 pg/ml at week 14 and further decreased to 5.4 pg/ml at week 30, but slightly increased at week 54 (32.1 pg/ml). The use of infliximab was associated with a progressive reduction in the dose of oral steroids in the two patients who completed the study period to a final point of 5 mg/day. The study focused on the effect of infliximab on more than one parameter of Behçet’s disease, although each parameter was studied independently. The small sample size in each group was considered a major limitation, however, the authors declared that sample size was decided after consulting with the Pharmaceuticals and Medical Devices Agency of Japan, with a minimum sample size of three patients per disease type, and a total of 15 patients. Authors declared that they couldn’t include controls because of ethical considerations. The authors reported significant improvement in the visual analogue scale in all patients regardless of the type of presentation. 16 Despite the prospective design, the study possessed additional limitations, as the authors didn’t declare how they excluded other possible causes of neurological deterioration in these patients at study inclusion, with the classification of patients as having incomplete Behçet’s disease, or how they considered the effect of confounders and bias in measuring and reporting their results. Therefore, this study was considered to have a low to moderate risk of bias.
Kikuchi et al., 2008: 17 This was a proof of principle, open label, single center, non-controlled, nonrandomized, unblinded study that aimed to evaluate the efficacy of infliximab in progressive neuro-Behçet's disease refractory to methotrexate (MTX). The study included five male patients aged 35.6 ± 7 years, who showed a variety of clinically identified neurological manifestations commonly observed in chronic progressive neuro-Behçet's disease, including dementia, psychosis, ataxia, myoclonus, and urinary incontinence, in addition to a sustained elevation of CSF IL-6 (> 20 pg/ml) despite administration of MTX and steroid. Intravenous infusion of infliximab was added to MTX (10–17.5 mg/week) and prednisolone (10 mg/day) at the same doses. All five patients were evaluated by a neurologist, with exclusion of features of systemic diseases other than Behçet's disease that cause the progressive neurological manifestation, including hypothyroidism, uremia, liver cirrhosis, atherosclerosis, and HIV infection. The clinical responses were judged by neuropsychiatric findings, revised Wechsler adult intelligence scale (WAIS-R), and IL-6 and TNF-α in the sera, and CSF analysis of patients measured together with brain MRI (MRI) scans at 24 weeks. At the end of the study period, none of the five patients showed exacerbation, and three patients showed significant improvement clinically, with no evidence of significant progression or new onset lesions on brain MRI scans. Interestingly, CSF IL-6 levels were decreased by significantly more than 50% on the next day of the first infusion, attributed by the authors to the direct cytotoxic effects of infliximab on monocytes/macrophages. This drop in CSF IL-6 level was sustained until the last infusion of infliximab at 14 weeks with gradual elevation of CSF IL-6 levels after the last infusion. Adverse events occurred in two patients, including transient headache in one patient and suspected subclinical pneumocystis pneumonia in another patient, with no discontinuation of the drug. The authors reported an observable increase in the CSF IL-6 levels in some patients when the intervals of infliximab infusion reached 8 weeks, recommending adjustment of the infliximab infusion intervals to sustain CSF IL-6 levels below 20 pg/ml for at least 2 years. Although the study by Kikuchi et al. 17 was non-blinded, non-controlled and non-randomized, with a small sample size, the authors used several objective measures to assess the response to the intervention, including clinical assessment, radio-imaging, and CSF analysis, and serum analysis of proinflammatory cytokines involved in the disease process. The study was deemed to carry a low to moderate risk of bias considering the nature of the study design, the small sample size, the unclear representation of how the analysis of potential confounders was handled, and additional concerns about the study being funded by a pharmaceutical company, while details regarding publication, copyright and possible conflicts of interest weren’t clearly declared.
Giardina et al., 2011: 18 This was a non-controlled interventional study that aimed to assess the long-term efficacy and safety of infliximab therapy in the treatment of patients with Behçet’s disease refractory to standard immunosuppressive agents. The enrolled population included 21 adult patients (18 males and three females; mean age, 36 years [range, 18–71 years]) who had ocular and/or CNS involvement refractory to corticosteroids and failed at least one immunosuppressant (cyclosporin, methotrexate, azathioprine, cyclophosphamide). The primary outcome was to assess the clinical efficacy (total or partial recovery) of infliximab, while the secondary outcomes included quality of life and drug safety. Eighteen patients completed the study up to 54 weeks while on fixed doses of prednisone (< 10 mg/day) with discontinuation of immunosuppressants at least 4 weeks prior baseline. The study included five patients with cerebral vasculitis, with three patients having combined cerebral vasculitis and uveitis and one patient with celiac disease. Remission was achieved at a mean time of 4.8 weeks with all patients having the diagnosis of cerebral vasculitis reaching a Behçet’s Disease Current Activity Form (BDCAF) score of 0 post infliximab. Limitations of this study included patients with mixed disease presentations with underrepresentation of patients with neuro-Behçet's disease, as the study included only patients with cerebral vasculitis with other manifestations, and accordingly, results can’t be generalized to all patients diagnosed with neuro-Behçet's disease. The authors used BDCAF as a measure of response to therapy, while comments on radiographic findings weren’t included, which in our opinion might affect the interpretation of data and the measurement of outcomes. The exclusion criteria included screening for infections that might affect the decision of infliximab, however, sufficient data on other comorbid conditions that were encountered in the patients, which might have confounded patient assessment and reporting of outcomes, were not clearly displayed. Accordingly, this study was considered to exhibit a moderate risk of bias.
Zeydan et al., 2016: 9 This was an open-label, prospective, interventional study that evaluated the effectiveness of infliximab in patients with neuro-Behçet’s syndrome, for whom other immunosuppressive medications had failed. The study included 15 patients who were followed prospectively for failure of conventional therapy prior to enrolment, according to the inclusion criteria. The mean age at the initiation of infliximab was 38.5 ± 10.0 years, five patients were treated with infliximab alone, and in the other 10 patients, infliximab was added to other medications: azathioprine, prednisolone, colchicine, cyclophosphamide and cyclosporine. The authors used clinical and radiographic examinations to assess and follow the response to therapy. The Expanded Disability Status Scale modified for neuro-Behçet's syndrome (EDSS-NBS), after exclusion of the section concerning uveitis, was additionally used to assess functional improvement related to neurological improvement with the intervention. Primary and secondary outcomes were defined with the following conclusion: ‘neurologic relapses were completely aborted and there was no further disability accumulation’. Authors implemented measures to overcome the nonstandardized follow-up times of real-world practice, with results displayed in two ways. First, the group effect was addressed using the total of clinical assessments for EDSS-NBS, with the median EDSS-NBS calculated for each month, comparing the preinfliximab period with the postinfliximab period, with censoring between the two time-points of median preinfliximab NBS duration and median infliximab use duration. Secondly, the EDSS-NBS accumulation rate between pre- and postinfliximab periods was calculated for each individual patient. The authors reported no difference at enrollment between patients in whom infliximab was chosen as a single agent, EDSS-NBS scores (median score, 4.5; range, 2.0–7.0) were comparable to those in whom infliximab was used as an adjunct to conventional immunosuppressive drugs (median score, 5.0; range, 3.0–7.0). Authors further clarified that by the end of the study period, both groups had the same half-point drop in median EDSS-NBS scores, with insignificant differences between infliximab monotherapy and infliximab combined with other immune suppressive drugs. Extended follow-up of both the infliximab group and non-infliximab group was included with observations regarding relapse rates rising to folds in the non-infliximab group and sustained efficacy for infliximab for up to 39 months. During infliximab treatment (median duration, 39.0 months, range 16.0–104.9 months), all 15 patients reached the preset primary outcome measure of having no further relapses and the secondary outcome of stabilization of disability. The authors reported common side-effects including rash, headaches, and nausea, however, they noted that such a small number of patients made it difficult to provide an informative report regarding adverse events. There was no mention of whether the assessors were blinded to treatment arms, which might have created some concerns regarding the measurement and selective reporting of outcome, additionally, there was no clear reporting of the effect of other confounders, including age, sex, and other comorbidities, which the present reviewers believe will endure as a shortcoming with the sample size included. The study was considered to have a low risk of bias, being a prospective, open-labelled, interventional study that included an interventional and a comparator arm. Patients were followed-up sufficiently prior to enrollment to identify those who satisfied the inclusion criteria. The strategy for patient recruitment, inclusion and exclusion criteria, outcome measures and follow-up were clearly stated. The sample size and homogeneity supported the reliability of the study. Patients who didn’t complete the study due to complications were excluded from the final analysis.
Desbois et al., 2016: 8 This was a multicenter, observational, retrospective study that included 17 patients with severe/ refractory parenchymatous disease treated with anti-TNF (infliximab or adalimumab; 13 patients were on infliximab). The authors aimed to evaluate the clinical and radiological response, time to obtain remission, occurrence of relapse, side-effects, and the degree of disability using the modified Rankin score. Authors defined complete remission as the disappearance of all neurological symptoms with improvement of radiological abnormalities related to neuro-Behçet's disease at 12 months after anti-TNF initiation, while partial remission was defined as improvement of neurological symptoms and of radiological abnormalities at 12 months after anti-TNF initiation and/or by a decrease > 50% of the corticosteroids dose compared with baseline. All patients had parenchymal neuro-Behçet's disease, associated with meningitis in 10 patients, optic neuritis in one patient, and cerebral thrombophlebitis in one patient. Parenchymal lesions involved severe neurological involvement, as 41.2% had brainstem lesions, 24% had myelitis, and 58.8% had a Rankin score of 2 at anti-TNF initiation. The study concluded that the defined parameters showed an overall improvement following anti-TNF, evidenced in 16/17 (94.1%) patients, complete response was achieved in five patients (29.4%) and partial response in 11 patients (64.7%). The median Rankin score was 2 (range, 1–4) at the initiation of anti-TNF versus 1 (range, 0–4) at the time of remission (P = 0.01). The introduction of infliximab therapy had a steroid-sparing effect with corticosteroids discontinued in four patients (23.5%) and reduced by more than 50% versus baseline in 10 patients (58.8%). The median daily dose of corticosteroids was 6.25 mg by the end of the follow-up period. Adverse events were encountered in 23.5% of patients with treatment discontinuation in 17% of cases. The reported data should be interpreted with caution since the study came up with some limitations that might have affected the interpretation and reporting of results, including the retrospective nature of the study (‘Prospective enrollment and data collection from the time of diagnosis would have been ideal but is more difficult to achieve with rare diseases’), with some patients being seen and assessed on an intermittent basis, as declared by the authors. Furthermore, the study included male patients only, with an insufficient number of patients in the adalimumab and golimumab arms that didn’t promote adequate quantification of their effect versus infliximab. The magnitude of the response to infliximab in each patient with different immunosuppressive regimens and doses couldn’t be extracted from the results, and the effect of potential confounders was unclear, 8 therefore, the study was considered to display a moderate risk of bias.
Pipitone et al., 2008: 19 This study included a series of eight cases, where six patients satisfied the criteria by the International Study Group for the Diagnosis of Behçet's disease. The study was conducted retrospectively; therefore, the assessment of patients was not fully standardized. Furthermore, documentation regarding the patients referred to was not fully available. Patients were started on infliximab due to either refractory or relapsing disease or new onset neuro-Behçet's disease. Some patients were on immunosuppressives, including methotrexate, cyclosporine, cyclophosphamide intravenous therapy, thalidomide, methylprednisolone pulse therapy, in addition to colchicine, and oral corticosteroids prior to and during infliximab therapy, however, in some patients the doses and forms of immunosuppressives were missing in the pre- and postinfliximab study period. Clinical and MRI assessment was available in six out of eight cases, while only clinical evaluation was performed in the other two cases. Infliximab was administered after, as well as with, other medications. The clinical benefit perceived might reflect, in part, the effect of other drugs, for example, the authors mentioned that cyclosporine was used before infliximab in one patient, and withdrawn with the introduction of infliximab, and while considering the possible negative effect of cyclosporine on neurological disease in Behcet’s disease, the drug effect may have been overestimated. Conversely, six patients were still receiving the preinfliximab drug regimen that had failed previously, without any change in dose or frequency, and displayed a favorable clinical response with introduction of infliximab, with no serious adverse events reported in any of the patients. The discontinuation of infliximab was associated with a flare in one of four patients, yet the long-term efficacy of infliximab as maintenance therapy and possible steroid-sparing effect in the enrolled patients wasn’t available due to the limited follow-up periods, with discontinuation of therapy in a few patients. 19 Considering the study’s nature and limitations, this study displayed the highest risk towards bias according to the ROBINS-I tool.
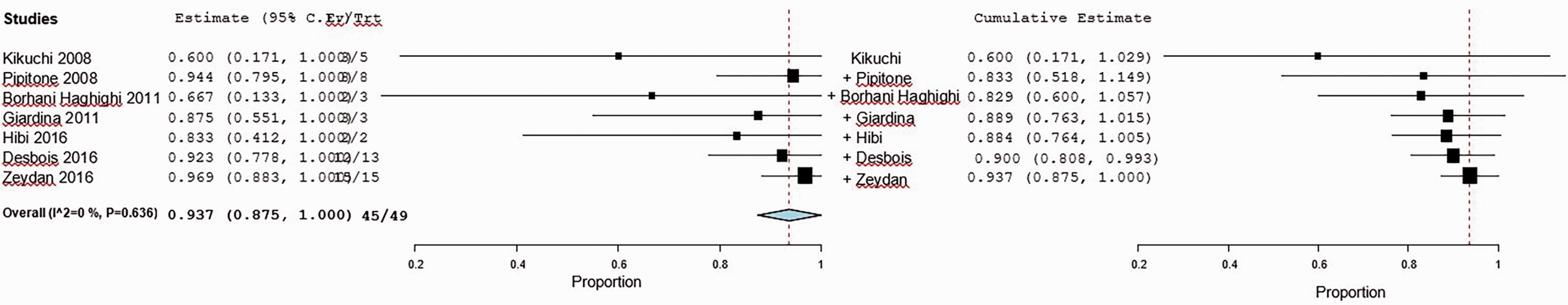
In our presented study overall the random effects meta-analysis showed that 59 out of 64 patients, or 93.7%, exhibited a good response to infliximab therapy (95% CI 0.88, 0.993). Interstudy heterogeneity was not statistically significant (I2 = 0%, P = 0.744; Figure 2). In cumulative analysis, the temporal trend showed accumulating evidence favoring increasing effectiveness over the last 20 years as more studies were accruing (Figure 3). Meta-regression results did not show statistically significant confounding of infliximab effect size by age (P = 0.89), sex ratio (P = 0.29), or disease duration (P = 0.67). Assessment of the risk of publication bias using Egger’s regression test revealed no significant evidence of publication bias (95% CI −30.23, 12.05; Z-value = −0.34; P = 0.366; Figure 4).

Forest plot summarizing meta-analysis results of the effectiveness of infliximab to treat neuro-Behçet’s disease. Overall, 94% of patients responded to infliximab. Dark squares represent study weight, with larger square size representing higher study weight (sample size). Horizontal lines depict 95% confidence intervals. Red broken vertical line represents the summary for the proportion of patients who responded to infliximab. Studies are shown with first author name and year of publication. Ev, responders (patients with response score ≥3 according to Hamdy and Woldeamanuel neuro-Behcet’s simple response score, 2020); Trt, patients who were treated with infliximab.
Forest plot summarizing cumulative meta-analysis of 7 studies showing a temporal trend indicating accumulating evidence for increasing effectiveness of infliximab to treat neuro-Behçet’s disease. Dark squares represent study weight, with larger square size representing higher study weight (sample size). Horizontal lines depict 95% confidence intervals. Red broken vertical line represents the summary for the proportion of patients who responded to infliximab.

Funnel plot and standardized residual histogram for publication bias.
Discussion
Central neuro-Behçet’s disease (Behçet’s disease of the CNS) represents a challenging clinical entity of Behçet’s disease that runs an aggressive course, with significant morbidity and mortality in a considerable population of patients. 35 The lack of targeted effective strategies to manage refractory neuro-Behçet’s disease adds to the disease burden and contributes to a lack of tight control. Considering the importance of this question to evidence-based clinical practice, the authors of this systematic review and meta-analysis aimed to explore the value of anti-TNF-directed biologic therapy with infliximab in refractory/relapsing/progressive central neuro-Behçet’s disease. The study is considered an evidence-based initiative towards better understanding of the effectiveness of infliximab, examining the currently available body of evidence in the field, which might add to current knowledge, highlight unmet needs, and lead the way for additional evidence-based studies that might aid the development of a well-defined treatment algorithm in such cases.
To the best of the authors’ knowledge, the present study is the first published systematic review and meta-analysis reporting the interventional effectiveness of the anti-TNF chimeric monoclonal antibody infliximab in controlling refractory neuro-Behçet’s disease. The research question was designed in accordance with the PICO model, using an explicit search strategy from evidence-based metasearch engines, recruiting full articles published in English language only, between January 2000 and May 2020. The PRISMA model was used to verify the search strategy, and the review was carefully performed by the present authors, with eligibility criteria designed to minimize bias in the selection of studies for inclusion. A total of 21 publications were eligible for inclusion in the quantitative and qualitative analysis; unfortunately, no published double-blind randomized trials addressing the effectiveness of infliximab in neuro-Behçet’s disease were identified. Authors of the current meta-analysis undertook extensive review for the risk of bias, using the risk of bias tool for non-randomized interventional studies ‘ROBINS-I’ in the open label trials, cohort studies and case series studies included. According to evidence-based hierarchy, the included studies were graded as level III–IV evidence.
Examination of the included studies for bias, using the Cochrane ROBINS-1 tool, showed that, although the included studies were non-randomized and cohort studies of evidence level III–IV, the majority had a low to moderate risk of bias, with the exception of the study by Pipitone et al., 2008, 19 which showed a high risk of bias due to the retrospective nature of the study with lack of standardized assessment and as infliximab was administered after, as well as alongside, other medications, which might have affected the interpretation of drug effects. Selection bias was not applicable considering the nature of the studies included being open-label non-randomized, however, almost all of the studies clearly stated that the inclusion criteria for receipt of the intervention was patients who were infliximab-naive with either acute severe, refractory, relapsing or chronic progressive neuro-Behçet’s disease, who were started on infliximab as first line biologic or who failed to respond to at least one of the conventional lines of treatment or previous biologics other than infliximab. Furthermore, there was no tendency towards choosing one drug over the other, as infliximab was the only intervention. Review of performance bias parameters revealed that the methodology and observation period in each study satisfied the minimum period for follow-up of the response to therapy, and all the included studies used objective measures for assessment of neurological response to the intervention. Regarding the response measures used in each study, the assessment scores were found to vary between studies, however, all included studies used at least the clinical response as a measure of improvement, with the majority using clinical assessment, with or without MRI, with or without CSF analysis. Five studies used the combined assessment of clinical, MRI and CSF analysis as a measure of response, 13 studies used clinical and MRI assessment, and one study used combined clinical response and CSF analysis. Only two studies (Giardina et al., 2011 and Zeydan et al., 2016) used clinical response scores as a single measure of improvement on infliximab treatment.9,18 Overall, there was no evidence of significant selective reporting of outcomes, as the majority of studies declared adverse events experienced during infliximab therapy, and whether or not they lead to drug discontinuation, declaring the number of withdrawals, dropouts and reasons, as well as the number of relapses while on treatment, with the exception of the study by Desbois et al., 2016, 8 where the authors declared lack of prospective enrollment and intermittent assessment rather than regular follow up, and the case series by Pipitone et al., 2008, 19 because the data were enrolled retrospectively.
Due to the fact that a small number of included publications defined a specific disease score in their evaluation of infliximab response, and with a lack of score standardization between studies (the majority used a combined assessment of clinical and/or radiographic [MRI] with/without CSF analysis), the reviewers of the present meta-analysis proposed a simple response score that aimed to incorporate standard evaluation parameters for assessment of responses, driven by the measures used in each study. Numerical values were used to grade the pattern of response in each parameter considered, to standardize analysis of the responses and minimize tendency towards overestimation, with a score of ≥ 3 considered to represent a good response, and a score of 5 denoting an excellent response. CSF analysis was used as an additional assessment point, considering that some studies used evoked potential as an assessment measure, and evoked potentials were suggested as a substitute to CSF being used in some studies as a measure of response, with a score of 1 denoting improvement. The lack of detailed reports of the clinical and MRI-related response data was an important shortcoming and a limitation in some studies. The reviewers of the current study aim to validate this score in future research to support the development of objective measures for the response to biologics in patients with neuro-Behçet’s disease.
Interstudy heterogeneity was not found to be significant, and the considerable homogeneity of inclusion criteria, study population, intervention used, and objective measures of response to therapy supported combining case reports into a case series. Results of this meta-analysis showed that 94% of the patients responded to infliximab therapy according to the response measures used in each study. Cumulative analysis showed accumulating evidence favoring increasing effectiveness over the previous 20 years, which may be explained by better acquaintance with the spectrum of CNS disease in Behçet’s disease and improved patient diagnosis, categorization and selection, and earlier diagnosis, in addition to increasing experience with the use of biologics in rheumatology practice in general and in Behçet’s disease. Infliximab effect size was not found to be confounded by age, sex or disease duration.
In the current systematic review and meta-analysis, infliximab was concluded to display a significantly homogenous therapeutic effectiveness in treating refractory neuro-Behçet’s disease. An increasing trend of effectiveness was observed over the previous two decades, which may be due to improved accuracy in diagnosing neuro-Behçet’s disease, increasing confidence in the use of anti-TNF biologics, and the recent adoption of a treat-to-target strategy in the management of autoimmune rheumatic diseases.
The current systematic review and meta-analysis uncovered important limitations in the available published studies, including: (1) A paucity of studies handling the current research question, with an observable deficit in published randomized controlled trials despite the relevance to clinical practice, decision making, and disease outcome; (2) Small sample size and lack of control groups with comparator intervention; (3) Lack of prospective enrollment in some studies; (4) Lack of clarity of exclusion criteria that might have a confounding effect; (5) Lack of a standardized scoring instrument available for outcome assessment of treatment response in neuro-Behçet’s disease; (6) Unclear discussion of the spectrum of comorbidities within some study publications, which might negatively confound the reported results regarding drug effectiveness, and impede proper assessment of response as well as any decision for therapy continuation; and (7) Lack of long-term follow-up in some studies for the perceived initial responses and the frequency of recurrences with discontinuation of infliximab therapy; (8) Underrepresentation of neuro-Behçet’s disease in some studies that included patients with overlapping disease manifestations; and (10) Some studies included patients with incomplete Behçet’s disease in the analysis.
Based on available data, the current authors strongly recommend designing well-structured randomized long-term controlled trials targeting effective therapy of refractory neuro-Behçet’s disease in light of the up-to-date advents in biologic strategies, with consideration of the potential benefit suggested in the current review. The authors additionally intend and recommend validation of the ‘Hamdy and Woldeamanuel simple response score, 2020’ as a possible standard tool that may be used for homogenous assessment of response to therapy in patients with central neuro-Behçet’s disease.
Research Data
sj-pdf-1-imr-10.1177_03000605231169895 - Research Data for The effectiveness of the anti-tumor necrosis factor therapy infliximab in neuro-Behçet’s disease: a systematic review and meta-analysis
Research Data, sj-pdf-1-imr-10.1177_03000605231169895 for The effectiveness of the anti-tumor necrosis factor therapy infliximab in neuro-Behçet’s disease: a systematic review and meta-analysis by Reem Hamdy A Mohammed and Yohannes W Woldeamanuel in Journal of International Medical Research
Footnotes
Data availability statement
All the relevant data, including analysis results, have been supplied within the manuscript, and any further data required will be available upon request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
