Abstract
Objectives:
Behcet’s disease is a multisystemic disorder. Most of its clinical manifestations are due to thrombosis or vasculitis. There is a predominance of mucocutaneous and ocular lesions; neurological manifestations are minority. However, this could be the first problem that leads a patient to seek medical help.
Methods:
We report a 30-year-old female with rapidly progressive headaches and confusion. Brain magnetic resonance imaging showed bilateral thalamic T2 hyperintense lesions and hemorrhage. Directed inquiry revealed recurrent oral ulcers and three miscarriages. During hospitalization, the patient developed a deep venous thrombosis.
Results:
After exclusion of other differential diagnoses, the diagnosis of Behcet’s disease was established.
Conclusion:
Behcet’s disease is a protean disease. Hence it should be in mind in patients with otherwise unexplained signs and symptoms, especially in endemic countries, and treatment decision should not be witheld in critical cases if no alternative diagnosis is plausible.
Introduction
Behcet’s disease was first described by Hippocrates, but highlighted to modern medical community attention by Hulusi Behcet in 1937. The hallmark of Behcet’s disease is recurrent oral ulcers beside any of the following systemic manifestations: genital ulcers, ocular disease, skin lesions, gastrointestinal involvement, neurologic disease, vascular disease, or arthritis. The majority of vasculitic processes are venular rather than arterial vasculitis, and “arterial” vasculitis could be crossed.1–3
Ancient Silk Road, which extends from eastern Asia to the Mediterranean, represents the area where Behcet’s disease is more common and has more severe course. The prevalence is similar in men and women in this area. 4
Less than one-fifth of patients with Behcet’s disease suffer from neurologic disease at a certain point during the course5,6 and is observed in men more frequently than women. The most common neurologic abnormalities are parenchymal lesions and complications of vascular thrombosis. Other manifestations include aseptic meningitis or encephalitis, and arterial vasculitis. Progressive personality change, psychiatric disorders, and dementia may also occur.
Although diagnostic criteria were not fulfilled on this case presentation, the symptoms and signs seemed to confluent during the following period. As a result, Behcet’s disease diagnosis and treatment should not be delayed if other differentials are excluded.
Case presentation
A 30-year-old married female with no known medical history presented to the emergency room (ER) with acute delirium and a fever since the day before presentation. Five days earlier she had a severe progressive generalized headache resistant to painkillers, profuse vomiting, fever, and delirium.
Past medical history was unremarkable except for three spontaneous miscarriages at the first trimester and recent irregular menses for which she started oral contraceptive (OCP) 20 days before presentation.
The patient described a history of recurrent painful oral ulcers for an undetermined period. On examination, there were a fever of 38°C and delirium, regular pulse 70 beat per minute (BPM), no signs of meningesmus, and no focal signs. Skin, cardiovascular, and genitourinary systems were within normal limits except for papulo-pustular rash on chest inspection. Fundoscopy was normal.
Brain computed tomography (CT) on admission was unremarkable. Blood tests were also normal except for white blood cells (WBCs) count of 20.91 × 109 per liter (neutrophils: 89.9%), erythrocyte sedimentation rate (ESR) of 75, and C-reactive protein (CRP) of 148.2 mg/L. Widal test for typhoid and Wright test for Brucella were both negative. Blood film, urinalysis, and chest X-ray (CXR) were normal. Cerebrospinal fluid (CSF) analysis showed WBCs of 30 cells/µL (predominantly neutrophils), red blood cells (RBCs) 100–150 cells/µL, glucose 69 mg/dL, protein 75 mg/dL, and negative gram stain.
Intravenous (IV) vancomycin and ceftazidime for suspected bacterial meningitis were initiated with no improvement for 48 h. The brain CT was then repeated and revealed bilateral pulvinar hypodensity and subacute hemorrhage within (Figure 1).

Brain CT shows bilateral pulvinar hypodensity and subacute hemorrhage within.
Brain magnetic resonance imaging (MRI) showed bilateral thalamic T2 hyperintense lesions with restricted diffusion. The lesions did not enhance with contrast. Intrathalamic hemorrhage was also noted (Figure 2). Brain magnetic resonance venography (MRV) showed right transverse, and sigmoid sinuses reduced calibers with no corresponding cerebral lesions (Figure 3).

(a) Sagittal T1 MRI without contrast shows hyperintense vein of Galen and straight sinus; (b, c, d) T2 and fluid attenuation inversion recovery and flair; (e, f) MRI shows bilateral thalamic T2 hypersignal and edema with subacute hemorrhage; (g, h) diffusion-weighted MRI shows restricted diffusion; (i, j, k) susceptibility weighted imaging (SWI) MRI shows bilateral intrathalamic hemorrhage; and (l) time-of-flight (TOF) MRI shows patent basilar artery and posterior cerebral artery (PCA) bilaterally.
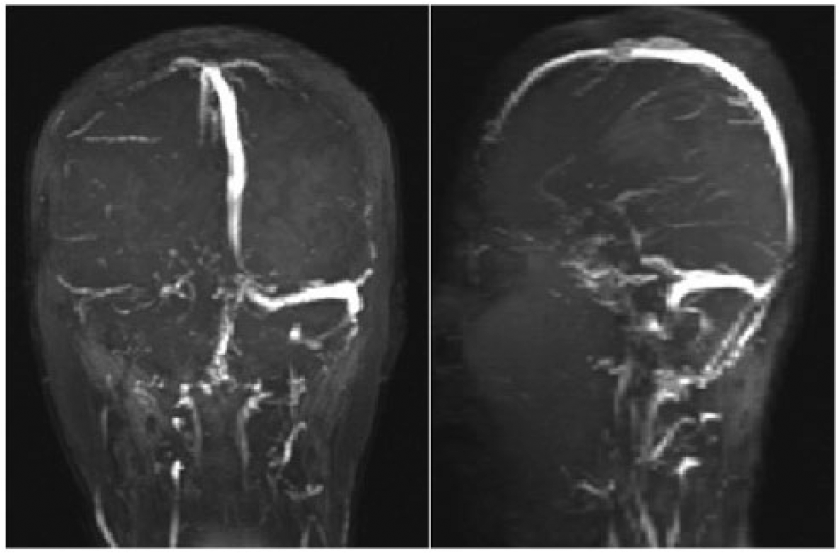
MRV shows patent dural venous sinuses with right transverse and sigmoid sinuses reduced calibers.
CSF analysis for the second time revealed no cells, 70 mg/dL protein, and 65 mg/dL glucose. CSF cytology was negative. Pathergy test was negative. Anticardiolipin antibodies and antinuclear nuclear antibodies (ANA) were both negative.
Behcet’s disease with either vein of Galen thrombosis or mesodiencephalic junction parenchymal lesions was suggested. Other differential diagnoses of bilateral thalamic lesions and/or thrombophilia were also considered.
However, in the light of the rapidly deteriorating clinical picture, the decision of treatment with steroids was made (dexamethasone 10 mg I.V followed by 4 mg/6 h). During the next few days, a dramatic clinical, laboratory, and radiological improvement ensued but the patient did not fully recover.
A week later, the patient developed a painful swollen leg, and echodoppler detected deep venous thrombosis (DVT) of the left lower leg despite the regularly aided ambulation. Pathergy test was repeated again and showed a stark positivity (Figure 4).

Positive pathergy test: postural rash on the right arm.
Anticoagulants (enoxaparin 1 mg/kg q12 h subcutaneously (SC), followed by warfarin 5 mg daily) were added to azathioprine 50 mg as a starting dose plus oral prednisolone 40 mg daily after fulfilling the diagnostic criteria of Behcet’s disease. Follow-up MRI and MRV were done and revealed no active lesions (Figure 5).

(a,b) Flair, (c,d) T2, (e) SWI, (f) T1 MRI show no active lesions and chronic hemorrhage; (g, h) T1 MRI shows disappearance of hyperintense vein of Galen; and (i) follow-up MRV shows reduced calibers of right transverse and sigmoid sinuses along with patent remaining dural sinuses.
Discussion
Diagnosis of Behcet’s disease is made upon clinical criteria published in 1990. These require the presence of oral ulceration (x3 in one 12 month period), plus any two of genital ulceration (aphthuos or scarring), typically defined eye lesions (anterior/posterior uveitis cells in vitreous (slit lamp), retinal vasculitis), typically defined skin lesions (erythema nodosum, pseudofolliculitis, or acneiform nodules), or positive pathergy test (24–48h, oblique insertion of 20-gaige needle. 7
Neurologic manifestation occurs in less than 20% of patients with Behcet’s disease, and none of them is considered a diagnostic criterion. Central nervous system (CNS) involvement stems from either vascular or parenchymal origin. In the former, arterial or venous thrombosis is implicated including dural sinus thrombosis. 8 Each of ischemic stroke, dissection, aneurysmal dilatation, and subarachnoid hemorrhage is a possible consequence of arteritis in Behcet’s disease patients. While in the latter, lesions may affect the corticospinal tract, brainstem, periventricular white matter, spinal cord, and basal ganglia. MRI can detect these lesions of CNS. 9
An association has been observed between dural sinus thrombosis and peripheral DVT. 5 Differential diagnoses of bilateral thalamic lesions were considered excluded by the history or investigations (Table 1).
Differential diagnoses of bilateral thalamic lesions.

The previous use of OCP, the acute presentation of progressive headache with nausea and vomiting, lack of enhancement, presence of thalamic hemorrhage, and the dramatic clinical and radiological response beside the subsequent DVT make vein of Galen thrombosis more likely. Preliminary data advocated the possibility of spontaneous recanalization of cerebral venous thrombosis (CVT), especially in the acute phase. 10
Whether the lesions were of vascular or parenchymal origin has not affected the treatment in this case, but it does affect the prognosis. A more favorable prognosis is expected in patients with dural venous thrombosis and other non-parenchymal processes, where recurrence rate, disability, and premature death are less likely compared to patients with parenchymal disease.5,11
As the diagnostic criteria are created to hasten both sensitivity and specificity, Behcet’s disease should not be excluded if the criteria are not fulfilled unless alternative diagnosis is available.
Conclusion
Behcet’s disease should always be suspected in otherwise unexplained thrombophilia, aseptic meningitis, parenchymal CNS lesions, and systemic inflammatory response, especially in endemic countries. A meticulously taken history is essential to unveil symptoms that patients usually forget to mention.
Footnotes
Acknowledgements
Yasmin Aboras, Khadija Almohamad Alkhalifa, and Husam Razouk revised the manuscript critically for important intellectual content. Rana Alnasser Alsukhni and Mohamed Sourat Ghoubari: Substantially contributed to the concept of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor of this journal.
