Abstract
Cancer immunotherapy including immune checkpoint inhibitors (ICI) has revolutionised the field of oncology and has been in use in India since 2015. ICI includes monoclonal antibodies directed against programmed cell death-1(PD-1), programmed cell death ligand 1(PD-L1) and cytotoxic T-lymphocyte antigen 4 (CTLA-4). They are currently approved in various stages of cancer immunotherapy. By inhibiting the negative co-stimulatory signals, unchecked T-cell stimulation can occur which helps enhance anti-tumour responses but at the same time cause immune-related adverse events (irAE) due to excessive/off-target inflammation. The most common irAE is cutaneous (30%) followed by hypothyroidism (20%). Other irAEs like rash and colitis present early in the treatment course, while inflammatory arthritis (ICI-IA) and pneumonitis have variable times of onset. Musculoskeletal AEs are rarely life-threatening and include arthralgia, inflammatory arthritis, polymyalgia rheumatica (PMR) and myositis. Most patients of ICI-IA have undifferentiated phenotypes at onset and do not satisfy any diagnostic or classification criteria for primary rheumatic diseases. Caucasian origin, female sex, combination therapy (anti-CTLA-4 and anti-PD-1/PD-L1) and body mass index (BMI) >25 kg/m2 are independently associated risk factors. Phenotypic subtypes include rheumatoid arthritis (RA)-like, reactive arthritis (ReA)-like, spondyloarthritis (SpA)-like, PMR-like, undifferentiated arthritis, isolated tenosynovitis and RS3PE-like syndromes. Pre-treatment autoantibodies like RF, anti-cyclic citrullinated peptide (anti-CCP) and anti-nuclear antibody (ANA) have been detected in some patients receiving ICI who went on to develop ICI-IA later. Management includes early rheumatology referral, continuing ICI therapy in grade 1 irAE, holding ICI, non-steroidal anti-inflammatory drug (NSAID)/intra-articular steroids for grade 2 irAE, while grades 3 and 4 require high-dose glucocorticoids prednisolone 0.5–1 mg/kg and conventional synthetic Disease Modifying Anti-Rheumatic Drug (DMARD)/biological DMARDs if not responding within two weeks.
Introduction
Cancer immunotherapy including immune checkpoint inhibitors (ICIs) has revolutionised the field of oncology in the last decade and has been in use in India since 2015. ICIs are monoclonal antibodies directed against immune checkpoints like programmed cell death-1(PD-1), programmed cell death ligand 1(PD-L1) and cytotoxic T- lymphocyte antigen 4 (CTLA-4). They are currently approved for the treatment of various cancers.1,2 By inhibiting the negative co-stimulatory signals unchecked T-cell stimulation can occur that helps enhance anti-tumour responses but at the same time can lead to immune-related adverse events (irAE) due to excessive/off-target inflammation.3,4 These adverse events have a wide spectrum with ICI-induced arthritis (ICI-IA) reported scarcely and variably across studies. 5 ICI-induced arthritis can mimic more common rheumatic disorders and hence needs to differentiate early to facilitate adequate treatment.
Pathogenesis
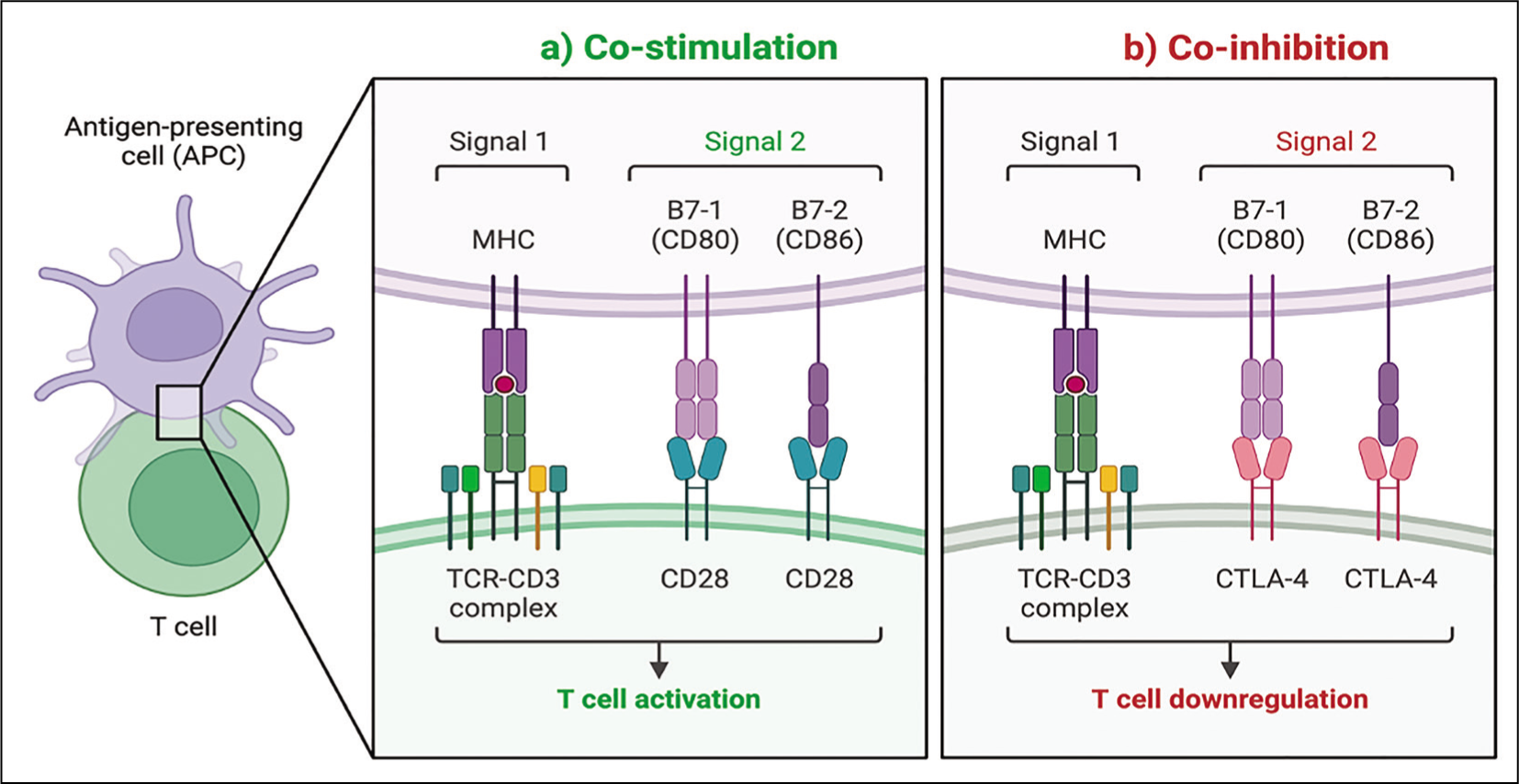
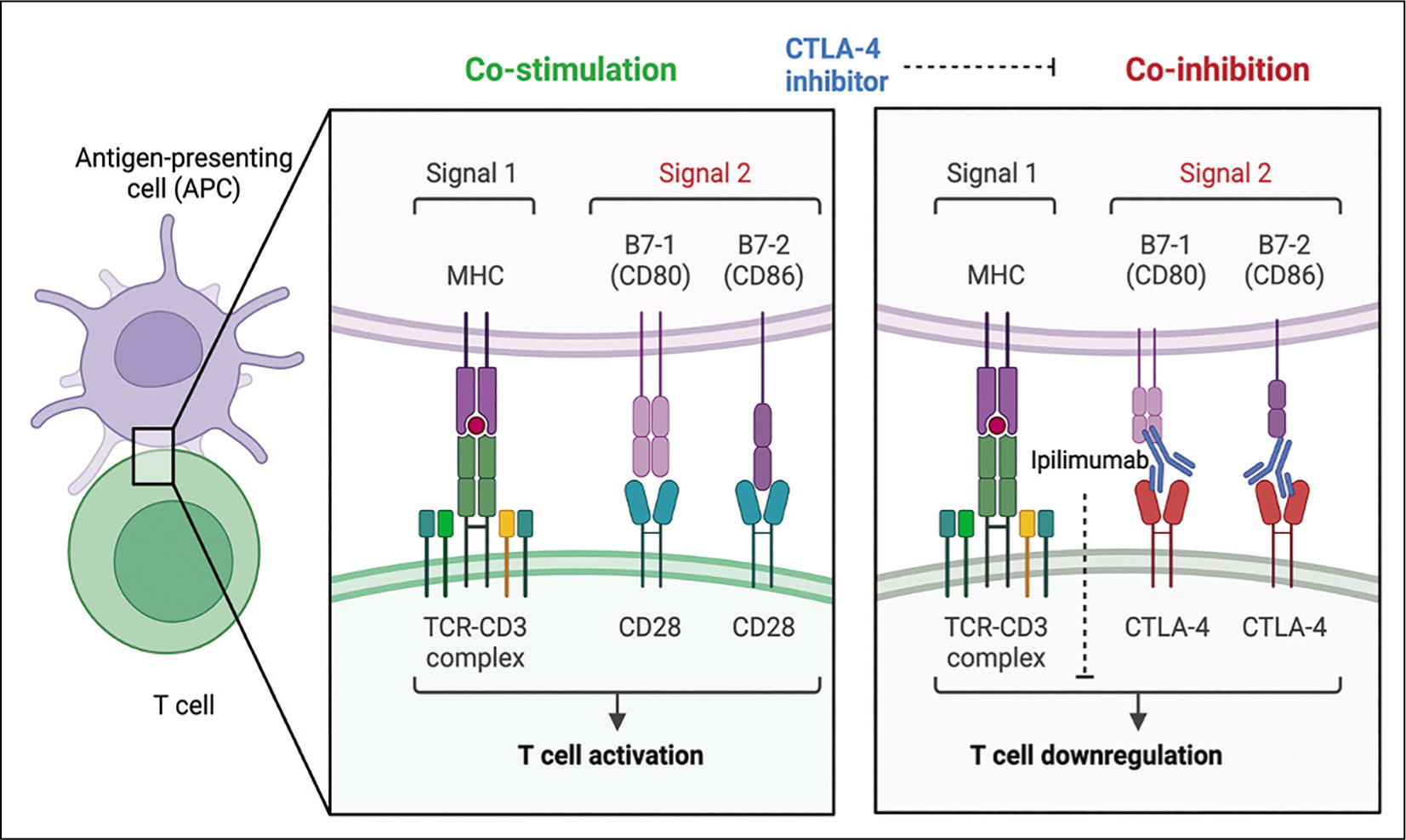
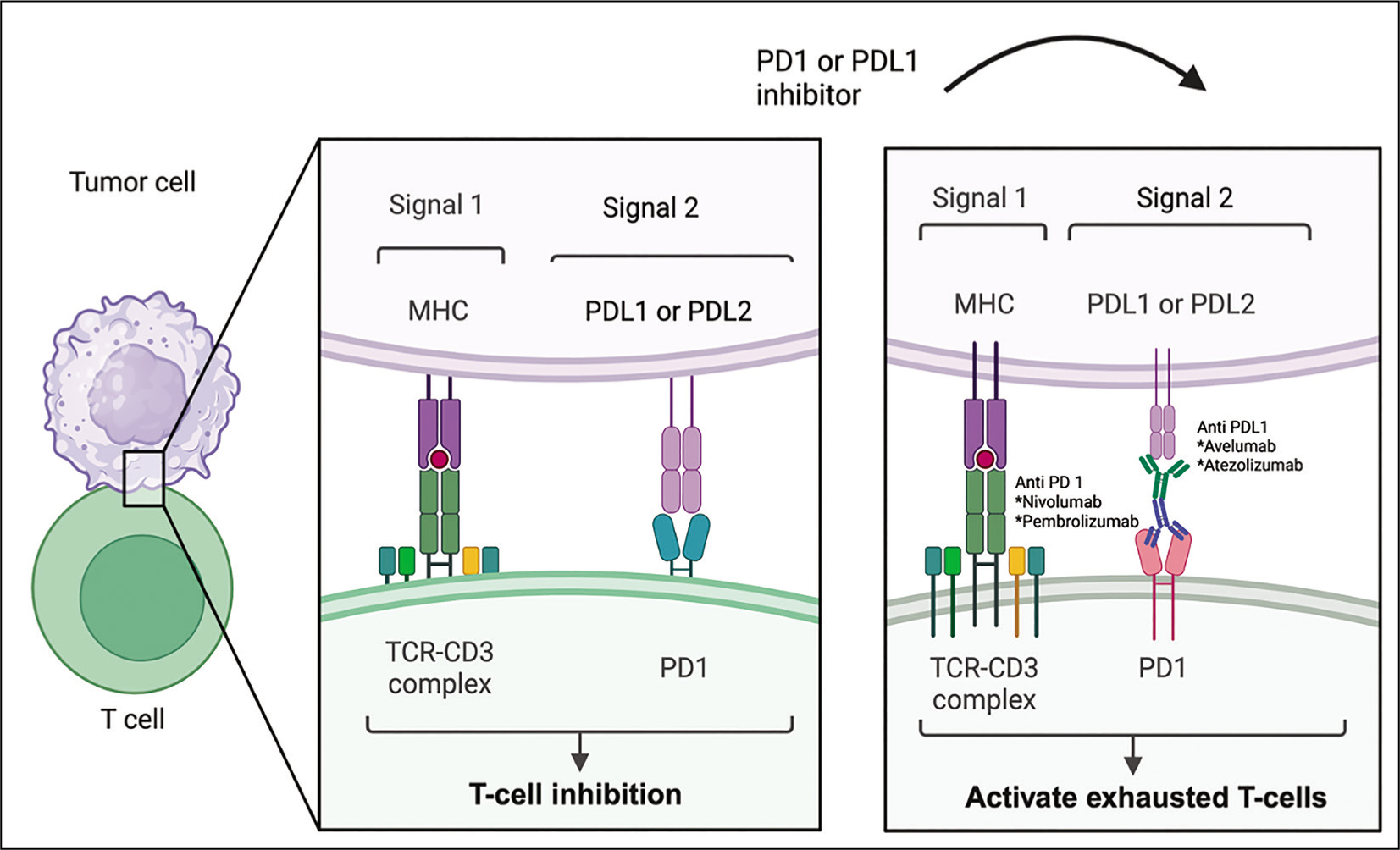
Effective antigen binding and complete T-cell activation requires two signals. Signal 1 involves the binding of T-cell Receptor on T-cells with major histocompatibility complex on antigen presenting cell, while signal 2 involves the binding of CD28 on T-cells with co-stimulatory molecules B7.1(CD80) and B7.2 (CD86) (Figure 1A).To negate excessive T-cell activation caused by chronic antigenic stimulation, certain inhibitory immune responses are activated which include the upregulation of inhibitory molecules such as CTLA-4, PD-1 and PDL-1, also known as immune checkpoints. ICI therapies for the treatment of cancer aim to block such inhibitory signals on T-cells or tumour cells through the use of antibodies. 6 CTLA-4 and PD-1/PDL-1 inhibitors are associated with different mechanisms and effects. While CTLA-4 inhibition lowers the threshold for proliferation of activated T-cells (Figure 1B), PD-1 or PD-L1 blockade can activate exhausted T-cells but may also work through blocking the inhibitory effects on dendritic cells (DC’s) and natural killer (NK) cells. 7 (Figure 1C). Since these inhibitory signals play a major role in preventing autoimmunity, their inhibition leads to numerous irAEs during cancer therapy such as arthritis, colitis, pneumonitis and thyroiditis.
a) Effective Antigen Binding and Complete T-Cell Activation. Signal 1 Involves the Binding of T-cell Receptor on T-Cells with Major Histocompatibility Complex on Antigen Presenting Cell, While Signal 2 Involves the Binding of CD28 on T-Cells with Co-stimulatory Molecules B7.1(CD80) and B7.2 (CD86); b) Chronic Antigen Stimulation Leads to Upregulation of Inhibitory Molecules Such As CTLA-4, PD1 and PD-L1. CTLA-4 Prevents Effective Co-stimulation by Binding to CD80 and CD 86 Thus Causing Co-inhibition.
CTLA-4 Inhibitors, e.g. Ipilimumab Lowers the Threshold for the Proliferation of Activated T-cells.
PD-1 (e.g. Nivolumab, Pembrolizumab) or PD-L1 (Avelumab, Atezolizumab) Blockade Can Activate Exhausted T-Cells.
Mechanistic Clues for ICI-IA from Animal Models
Proposed Animal Model for ICI-IA
Humanised BALB/c-hPD1/hCTLA4 mice were injected with vehicle or collagen-specific antibodies (CA) and ICIs (ipilimumab and nivolumab) to establish a mouse model for ICI-IA and ICI-mediated lung injury. Anti-tumour necrosis factor-alpha (TNF-α) antibodies were also checked for therapeutic strategy. Joint and lung symptoms were longitudinally assessed along with histological analysis. Flow cytometry was used to identify T-cell subsets in peripheral blood, lymphoid tissues and lung. Multiplex immunofluorescence and flow cytometric analysis of CAIA+ICI mice revealed a higher frequency of splenic TNF-α +CD4+ and TNF-α +CD8+ T-cells compared to wild type and unaltered other T-cell subset (interferon-γ (IFN-ɣ), interleukin-4 (IL-4), IL-17 secreting T-cells and Tregs). Treatment with TNF-α inhibitor mitigated the severity of arthritis, possibly by decreasing the frequency of TNF-α + CD4, TNF-α + CD8 cells and effector memory cells in peripheral lymphatic organs and lungs, making it a primary therapeutic target in ICI-IA. 14
Insights into Synovial Fluid Analysis in ICI-IA
A recent study retrieved distinct cell clusters in peripheral blood and synovial fluid in newly diagnosed ICI-IA patients. Ki67+ T-cells and CD25hi CD127lo Tregs were significantly higher in synovial fluid, while CD45RAhi CCR7hi CD3+ naive T-cells, CD16hi NK and NK T-cells and CD19+ B cells were higher in peripheral blood. IFN-γ producing Th1/Tc1 cells might play a critical role in the pathogenesis of arthritis-irAE. Synovial fluid Tregs had CTLA-4, CD25 and FOXP3 positivity and showed higher suppressor function than peripheral blood Tregs. Clonotypes and trafficking analysis of T-cell suggested migration of clonally expanded CX3CR1hi effector CD8+ T-cells into joints by CXCR3 and CXCR6. 12 Circulating CX3CR1hi CD8+ T-cells are increased after anti PD-1 agent in patients with certain malignancies like renal cell carcinoma. 15 CX3CR1hi CD8+ T-cells play a critical role in tumor eradication in vivo. Therefore, it is possible that CX3CR1hi effector CD8+ cells from tumours cross-react with issues (e.g. joint), causing inflammation.
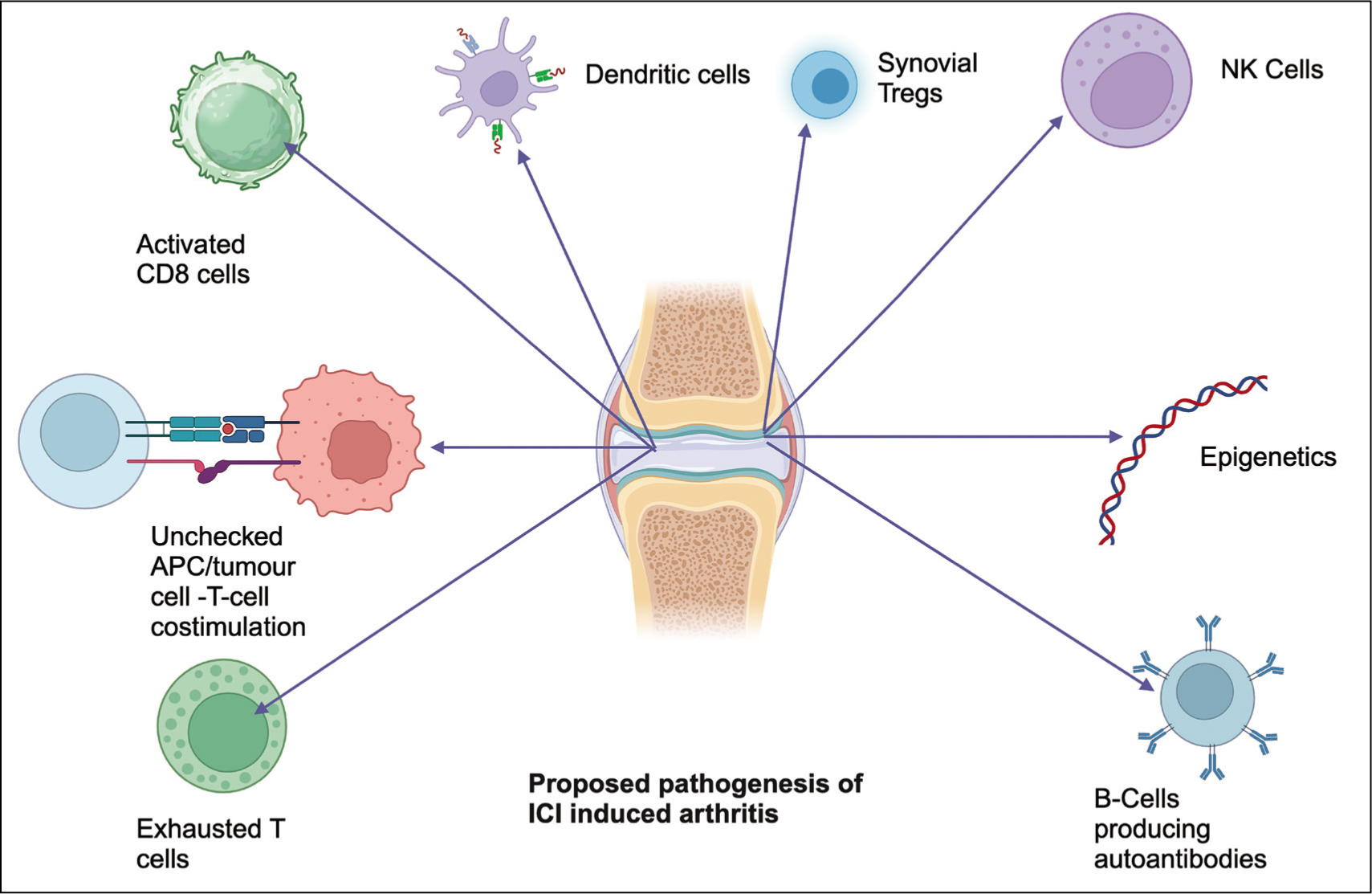
The possible mechanisms in ICI-associated arthritis have been illustrated in Figure 2.
Mechanistic Understanding of Pathogenesis of Immune Checkpoint Inhibitor-Induced Arthritis.
Clinical Spectrum
irAEs are increasingly recognised off-target effects of ICI therapy. The most common irAE is cutaneous (30%) followed by hypothyroidism (20%). Other irAEs like rash and colitis present early in the treatment course, while inflammatory arthritis and pneumonitis have variable onset.16,17 Rheumatic irAEs are rarely severe or life-threatening and less likely reported by patients as well as oncologists. Musculoskeletal AEs include inflammatory arthritis, polymyalgia rheumatica (PMR) and myositis.
In a single-centre prospective study, inflammatory arthritis was the most common rheumatic irAE, occurring in 3.8% of cases. 18 Data from a retrospective analysis showed arthralgia (13.3%) as the predominant manifestation in metastatic melanoma treated with anti-PD1 blocker. 19 ICI-IA can manifest early during the initiation phase (esp. after 1st dose) of ICI therapy or after years. Though the arthritis usually resolves with cessation of therapy, it might persist after discontinuation in some. Most patients have the undifferentiated phenotype at onset.
Caucasian origin, female sex, combination therapy (anti-CTLA-4 & anti-PD-1/PD-L1) and body mass index (BMI) >25kg/m2 are independently associated with risk of ICI -IA.20,21 Although no striking racial disparity was identified in arthritis-related irAEs, type of cancer (melanoma and genitourinary Ca), choice of ICI regimen, glucocorticoid use within one year before ICI initiation and history of pre-existing autoimmune disease are associated with irAE development. 20
Arthralgia is well documented in clinical trials with an incidence of 1 to 43%, but overt synovitis is uncommon (1–7%). 22 A VigiBase (WHO database of case reports) analysis by Arnaud et al. on autoimmune arthritis with ICI treatment mentions 86 cases. 23 Rheumatoid arthritis (RA)-like arthritis was seen in 68.6% of patients, followed by unclassifiable autoimmune arthritis in 16.3%. The disease was frequent in the elderly population (median age: 63 years) with a mean duration of ICI exposure of 2.7 months before arthritis onset. Lung carcinomas (39.7%) and malignant melanomas (36.8%) were frequently associated with irAEs, who had received anti-PD1 monotherapy (86%) or combination therapy (10.5%). 23
Phenotypic patterns and differences from primary rheumatologic phenotype (Table 1):
Phenotypic Patterns of ICI-IA and Associated Clinical, Laboratory and Radiological Features.
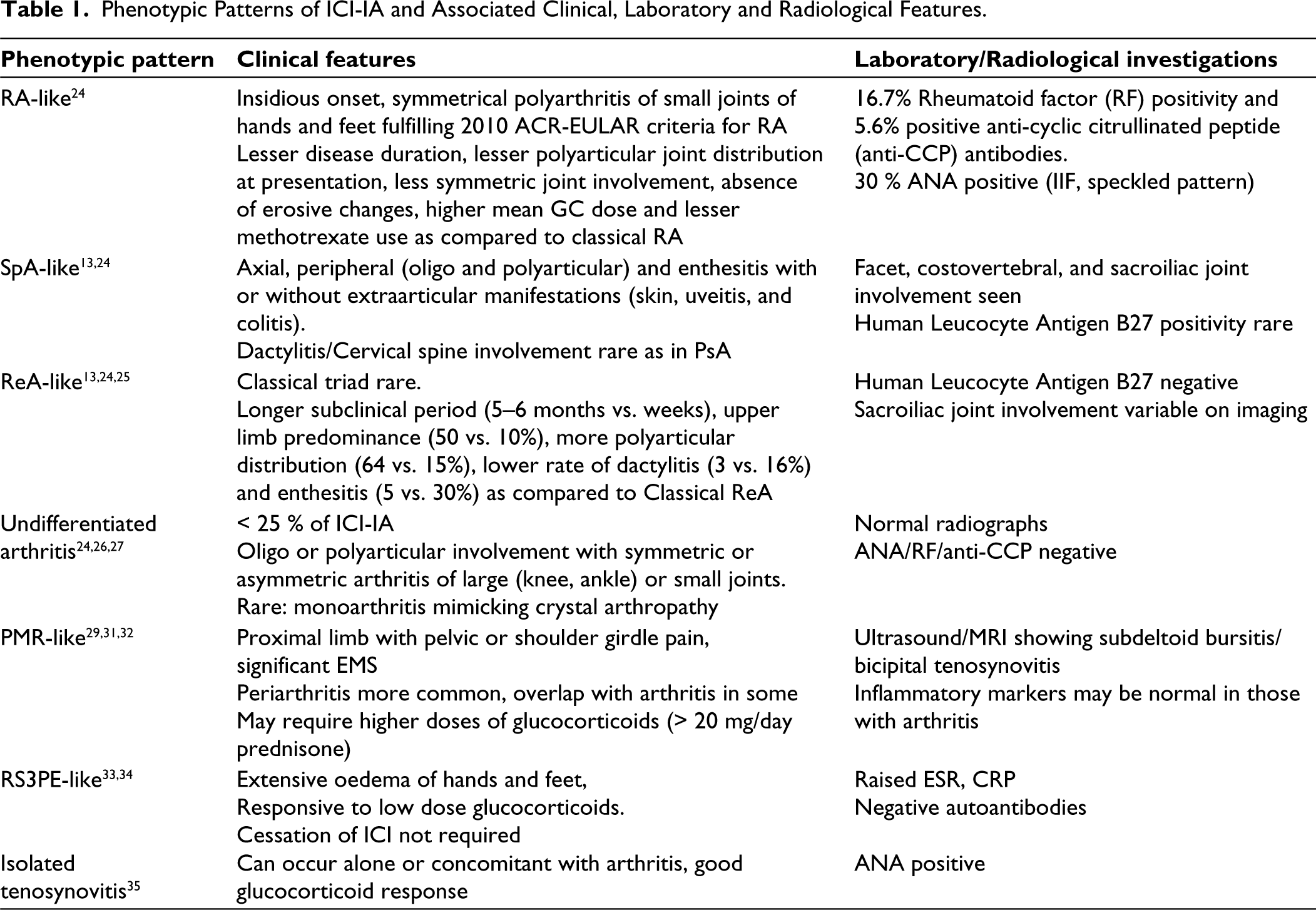
Patients may have a predisposing genotype like HLA-DRB1 shared epitope or non-HLA associations, or a pre-existing subclinical disease. 36 Those with pre-existing autoimmunity (RA, PsA or SpA) can experience arthritis flares upon ICI initiation, specifically those with uncontrolled disease and who have discontinued immunosuppressant therapy at ICI initiation. Though new autoantibodies weren’t identifiable in de-novo IA-irAEs, in some cases in whom autoantibodies (RF, anti- CCP, anti-TPO and ANA) were detected before ICI therapy, patients later developed irAEs. 37 Finally, a newer entity termed ‘activated osteoarthritis’ explains the exacerbation of previously identified OA joints after ICI therapy without any inflammatory features. 38 The exact cause of joint flare is unknown and could be due to crystal deposition or an epiphenomenon rather than a true irAE. Further clinical studies are necessary to reveal basic pathogenetic differences and explain the phenotypic variability.
Approach to Diagnosis and Assessment
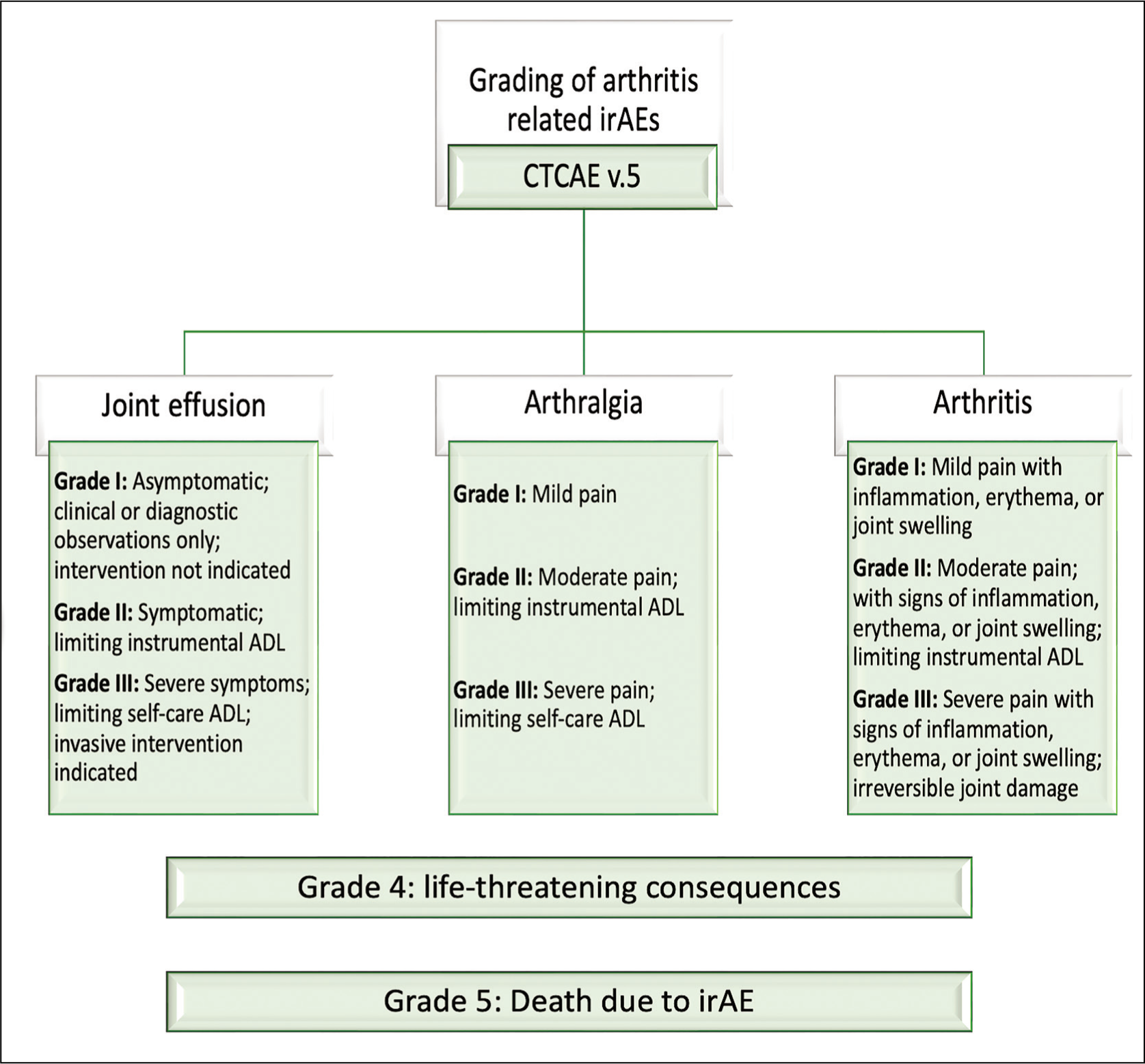
IrAEs may present after stopping ICI therapy, so a prior history of ICI exposure in the last one to two years is helpful. Patient characterisation based on the Common Terminology Criteria for Adverse Events (CTCAE) version 5 grading system (Figure 3) can grade the severity of symptoms according to the difficulty in daily living. Clinical Disease activity score (CDAI) measurement adopted from RA can help monitor treatment response but hasn’t been validated yet. Patient-reported outcome measures (PROMIS-PF10a, RAPID3) and physical function scores (HAQII, HAQ-DI) have been used to denote functional status and its improvement with therapy. Elevated inflammatory markers and imaging evidence of synovitis can guide the clinical outcome.
Flow Diagram of Common Terminology Criteria for Adverse Events (CTCAE) Grading for Arthritis-Related Adverse Events; Grade 4 and Grade 5 Are Not Mentioned in Arthritis but in Common Rheumatological irAE (Data from National Cancer Institute). 58
Patients of ICI-IA should be evaluated in a structured manner (Figure 4) to confirm the clinical diagnosis and to decide about further treatment plans. A careful review of oncological notes and temporal correlation of symptom onset is necessary to establish a causal effect of ICI therapy. Patient assessment should be done to characterise the clinical pattern and full differential diagnosis to be considered even if the symptoms started after ICI.
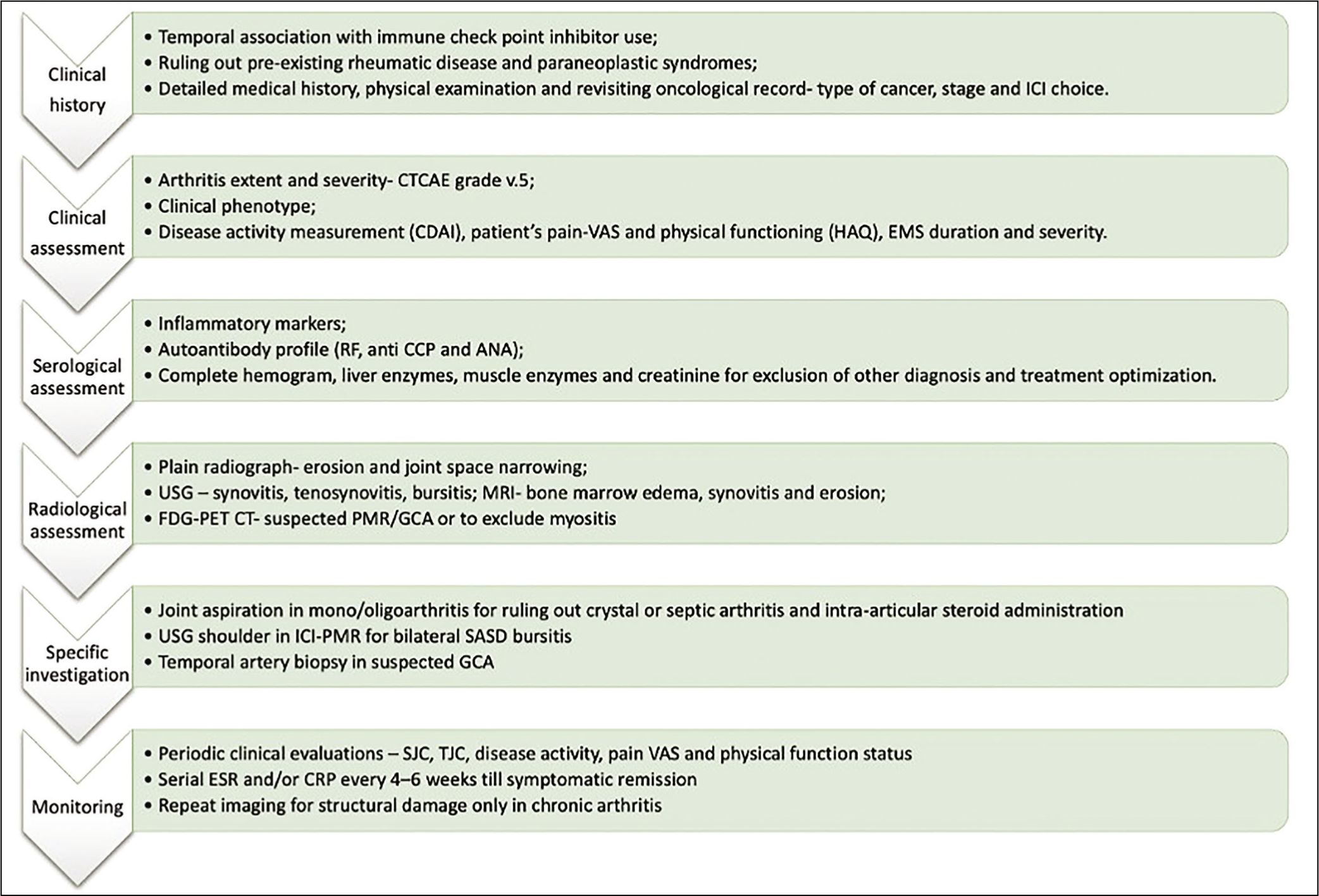
Differentials
Clinical history gives clues about the temporal association of symptom onset and ICI use. Other diagnoses like pre-existing autoimmune disease, paraneoplastic arthritis, crystal arthritis and OA should be excluded, as all of these can present with a joint flare after ICI therapy. Metastasis to adjacent joint and periarticular structures and septic arthritis are ruled out in mono-articular involvement. Acute leukaemia and lymphomas may present with bony pain which needs to be differentiated from true arthritis in younger individuals. Paraneoplastic arthritis should be considered especially if symptom onset correlates with cancer diagnosis.
Treatment
Management of arthritis-related irAEs has been published by several oncology and rheumatology societies. Oncological societies like the American Society of Clinical Oncology (ASCO) and the European Society for Medical Oncology (ESMO) have also given guidelines regarding patient assessment based on their symptom severity as per CTCAE grade. EULAR has recommended a multi-disciplinary approach and shared decision-making as a primary step for management. 26 Patients with suspected rheumatic irAE should be referred to a rheumatologist for detailed evaluation. Rheumatologists must consult the treating oncologist and converse with the patient, to get an overview of the condition and decide the achievable therapy targets. The following points are necessary and should be considered. 26
Arthritis-related irAE can have a wide spectrum and may not fit into one classification criteria.
Differential diagnosis like metastatic disease, paraneoplastic syndromes and unrelated RMDs should be excluded.
No need to check for pre-existing autoimmunity routinely in cancer patients before initiation of ICI therapy.
Withdrawal of ICI therapy must be a shared decision between patient, oncologist and treating rheumatologist.
Baseline glucocorticoid dosage should be below 10mg/day before starting ICI therapy, which negates the negative impact of GC on tumour response and long-term survival.
Symptomatic management is essential for patients with mild disease. Local or systemic glucocorticoids may be necessary for adequate symptom control but should be advised to taper to the lowest effective dose when improvement is achieved.
For those who have failed symptomatic management or require a higher dose of corticosteroids, conventional synthetic Disease Modifying Anti-Rheumatic Drug (DMARDs) (CsDMARDs) are recommended.
In case of inadequate response to CsDMARDs, biological DMARDs like TNF-α inhibitors or IL-6R inhibitors are preferred.
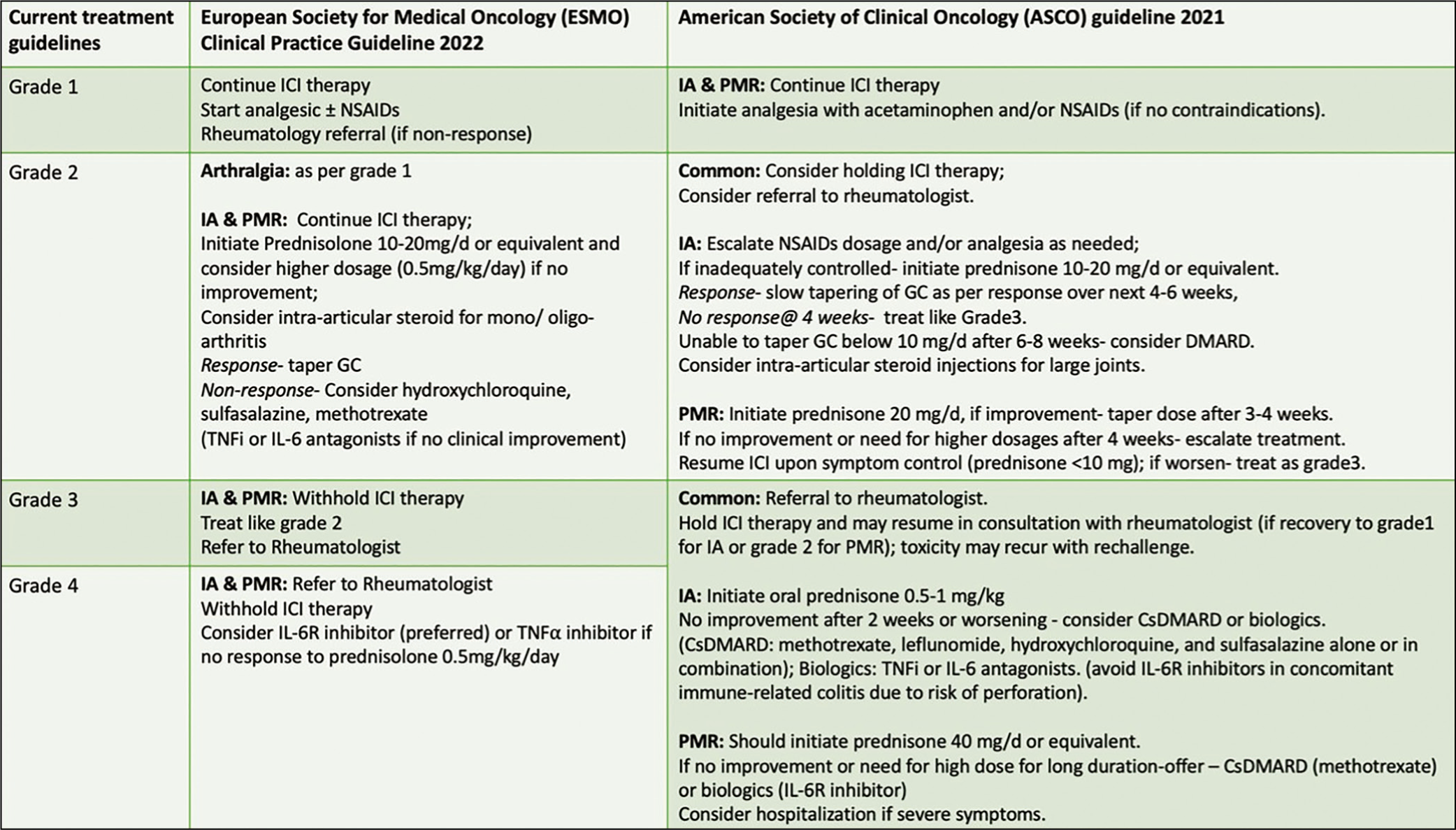
ASCO has recommended early referral to a rheumatologist in patients with definite joint swelling (synovitis) or arthralgia persistent for more than four weeks. Early recognition is the key to avoiding permanent joint damage. Though corticosteroids are essential in the initial therapy regime, prolonged use is better voided and early consideration of DMARDs should be considered. In oligo articular variant, intra-articular steroids are the mainstay of therapy along with analgesics. 39 Current treatment guidelines for ICI-induced arthritis and PMR are summarised in Figure 5.
Current Treatment Guidelines for ICI-Induced Arthritis.49,59
Evidence of Treatment and Safety
Animal Models of Humanised BALB/c-hPD-1/hCTLA-4 mice treated with ICI therapy can induce rheumatic irAE and anti-TNF-α successfully mitigated the disease phenotype. 14 In vitro models have supported the use of combination therapy including antirheumatic drugs and ICIs to mitigate the development of rheumatic irAEs. Higher expression of monocyte chemoattractant protein 1 (MCP- 1) is documented in pembrolizumab-treated patients and co-culture with anti-TNF-α ameliorates the disease, as opposed to anti-IL-6 receptor therapy (40). Patients with irAE have a distinct immune effector or metabolic profile than irAE-free individuals and in vitro tofacitinib or infliximab treatment was unable to impact the immune-metabolic profile and cytokine release capacity of CD8 T-cells. 41
The safety and efficacy of concurrent ICI and antirheumatic therapy use are well documented. 42 In a cohort of 69 patients, the use of non-steroidal anti-inflammatory drugs (NSAIDs), oral corticosteroids, CsDMARDs and anti-TNF-α therapy showed no negative impact on cancer outcomes. 43 However, Faje et al reported a potential negative impact of high-dose steroid use in melanoma patients. 44 Grade 3 ICI-induced arthritis treated with methotrexate was able to maintain symptomatic improvement with long-term cancer remission. 45 Concomitant use of anti-TNF-α with ICI therapy was found safe in infliximab-treated patients but at the cost of higher toxicity in the certolizumab study field. 46 Multiple studies have established the safety and efficacy of anti-IL-6 receptor (tocilizumab) in ICI-induced arthritis.47,48 Anti-IL-17A was able to demonstrate clinical remission in melanoma patients treated with ICI therapy. 49
As irAEs are an off-target effect of cancer immunotherapy, they act as a surrogate for effective anti-tumour response. 50 In non-melanoma cancer, an investigation into anti-PD1 blockers has shown improved objective response rate, disease control rate and overall survival with irAE, as well as the highest regression (80%) with grade 3 or more adverse events. 51 Patients with irAEs esp. musculoskeletal irAEs have better tumour response. 52 ICI-treated patients’ prednisone use >10mg/day proved detrimental in terms of cancer outcomes and overall survival esp. when used within the first two months of ICI therapy.44,53,54 On the other hand, steroid-sparing agents like conventional and biological DMARDs did not impact the tumour response to ICI. 43
In a systematic review, pre-existing autoimmunity had experienced exacerbation in 75% of cases, 25% had denovo irAE after ICI therapy but ICI discontinuation rate was only 17.1%. 55 Generally, the rate of exacerbation is higher with anti-PD-1/PDL1 therapy, but denovo disease is frequent with CTLA-4 blockade.
The CANRIO registry aimed to study the clinical patterns of iRAE’s and their rheumatological and oncological outcomes. Amongst 73 patients with a mean follow-up period of 14 ±10.8 months, 80% 59 with ICI- induced arthritis became chronic requiring continuation of ICI, predominantly those with synovitis at onset. About 15.1% 11 could discontinue treatment, and 4.1 % 3 combined therapy was switched to monotherapy. Chronic irAEs were associated with improved overall and progression- free survival compared to those with acute episodes. 56
Limitation in Current Research
Though current research suggests that combination therapy predisposes to the persistence of inflammatory arthritis, the potential reason behind differential occurrence rates in different ICI therapies is still unknown. Whether mechanistic difference or the genetic susceptibility of the host plays a major role in the occurrence of ICI-induced arthritis and its outcome is unclear. There is no universal consensus regarding optimal treatment, appropriate dose, tapering schedule and duration of therapy. Some studies have reported variable persistence of IA after ICI cessation. Duration of IA therapy ranges from 9 to > 24 months after ICI cessation, depending upon the clinical scenario and initial response.43,57 The heterogeneity of presentation and joint distribution separate ICI-induced arthritis from other primary rheumatological diseases and that’s why it requires a separate tool for measurement of disease activity and response than traditional one. Also, the universal recommendation for remission and low disease activity state is yet to be defined.
Understanding the biology of specific irAE is crucial to developing targeted therapy. Janus Kinase (JAK) inhibitors are the popular alternatives to biological DMARDs in csDMARD refractory state for rheumatic diseases but role in ICI-induced arthritis is unclear. They have been successfully tried in refractory irAEs like colitis in post-ICI patients but use in refractory arthritis is still limited. Post hoc analysis of the oral surveillance study showed an increased risk of malignancy in patients treated with JAK inhibitors as compared to TNF-α inhibitors. The real question arises, whether JAK inhibitors can be employed as a potential therapeutic agent in refractory ICI- induced arthritis or will it be linked with cancer relapse.
Future Directions
Ongoing clinical and translational research on bigger cohorts will improve our understanding of pathophysiology and help us select the optimum course of therapy in patients with ICI-induced IA. Clinical trial on early induction with adalimumab is ongoing (NCT06037811).
Future research areas are as follows:
Precision medicine approach to develop an ex-vivo personalised predictive assay to guide the optimal selection of antirheumatic drugs to reduce rheumatological irAEs. Development of an ex-vivo model to assist the selection of ICI and anti-rheumatic drug combinations in irAEs. Development of validated diagnostic assays or response tools to aid clinicians. Role of small molecule like JAK inhibitors in ICI-induced arthritis. Long-term follow-up of ICI-induced arthritis patients to study clinical outcomes. Larger cohort studies or registries which would provide data regarding the safety of ICI concerning the progression of underlying malignancy and recurrence of arthritis post- cessation of drug.
The current therapeutic approach is based on case reports and expert opinions. Newer therapeutic inclusion in oncology necessitates more accurate methods for the optimal management of ICI-treated arthritis in a targeted manner.
Footnotes
Acknowledgements
Figures 1 and ![]() were made using Biorender.Com with a valid publication license.
were made using Biorender.Com with a valid publication license.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
Not applicable.
