Abstract
Immune checkpoint inhibitors, particularly anti–PD-1 agents, are increasingly associated with bullous pemphigoid, a rare yet significant immune-related adverse event. We report a case of immune checkpoint inhibitor-induced bullous pemphigoid in a 74-year-old renal transplant recipient undergoing cemiplimab and later pembrolizumab for recurrent cutaneous squamous cell carcinomas. Bullous pemphigoid developed 23 months after initial immune checkpoint inhibitor exposure and 4 weeks after pembrolizumab reinitiation, presenting with generalized pruritus and tense bullae. Diagnosis was confirmed by histology and direct immunofluorescence. Disease was managed with oral prednisone and topical steroids, with relapses occurring upon tapering. This case highlights the complex interplay between immune checkpoint inhibitors, transplant immunosuppression, and autoimmune toxicity, underscoring the need for personalized, multidisciplinary management strategies in transplant patients receiving immune checkpoint inhibitors.
Keywords
Introduction
Immune checkpoint inhibitors (ICIs) have revolutionized cancer therapy, but are associated with a growing spectrum of immune-related adverse events (irAEs). 1 While a rare irAE, bullous pemphigoid (BP) is the most frequently reported autoimmune blistering disease associated with ICI therapy, especially with anti–PD-1/PD-L1 agents.1,2 Pathophysiologically, PD-1/PD-L1 blockade disrupts peripheral tolerance, enabling autoreactive T cell activation and B cell–mediated production of autoantibodies against hemidesmosomal proteins anti-BP180 and anti-BP230 (Figure 1). 2 Circulating anti-BP180 antibodies may precede overt blistering, and persistent pruritus should prompt early dermatologic assessment. 2

Proposed mechanism and timeline of ICI-induced BP. Illustration created by Ghassan Barnawi, MD using Biorender. (a) Schematic representation of ICI-induced BP. ICIs block PD-1/PD-L1 and CTLA-4 pathways, restoring T cell activation. Activated T cells stimulate B cell activation and proliferation, resulting in autoantibody production. These autoantibodies target auto antigens at the dermoepidermal junction, causing BP. (b) Timeline of ICI administration, BP onset, and treatment course. The graph illustrates the progression of BP severity over time, along with the timing of treatment interventions using oral prednisone and topical clobetasol.
ICI-induced BP poses unique therapeutic challenges, particularly in transplant patients, who are at elevated risk of cutaneous malignancies and risk of graft rejection due to autoimmunity. The management of such cases becomes more complex when balancing ICI-related toxicity with effective cancer control and prevention of graft rejection. We present a challenging case of anti–PD1-induced BP in a renal transplant recipient with multiple cutaneous squamous cell carcinomas (SCCs), treated with cemiplimab and pembrolizumab. To our knowledge, this is the first case described in the literature of ICI-induced BP in a renal transplant recipient.
Case presentation
A 74-year-old male with a history of chronic kidney disease and three renal transplants (1981, 1983, and 1995) was on a long-term immunosuppressive therapy with mycophenolate mofetil (MMF) and tacrolimus. His medical history was complicated by multiple cutaneous SCCs due to chronic immunosuppression.
In September 2022, he developed two high-risk invasive SCCs on the left eyebrow and left ear, with the latter having incomplete histological clearance after Mohs surgery. The patient received adjuvant radiation therapy, followed by initiation of cemiplimab in April 2023. He received 20 infusions every 3–4 weeks until July 2024, resulting in complete clinical response. He was subsequently switched to pembrolizumab every 6 weeks, beginning on August 30, 2024. However, therapy was discontinued after the first cycle due to progressive acute renal failure requiring dialysis.
In January 2025, his cutaneous SCCs relapsed, and he received a second cycle of pembrolizumab on February 18, 2025. On March 19, 2025, he presented with a 2-week history of generalized pruritus and multiple erythematous tense blisters and excoriated plaques on the trunk, scalp, and extremities (Figure 2(a) and (b)). Skin biopsies confirmed BP, showing dermo-epidermal separation and linear direct immunofluorescence staining.
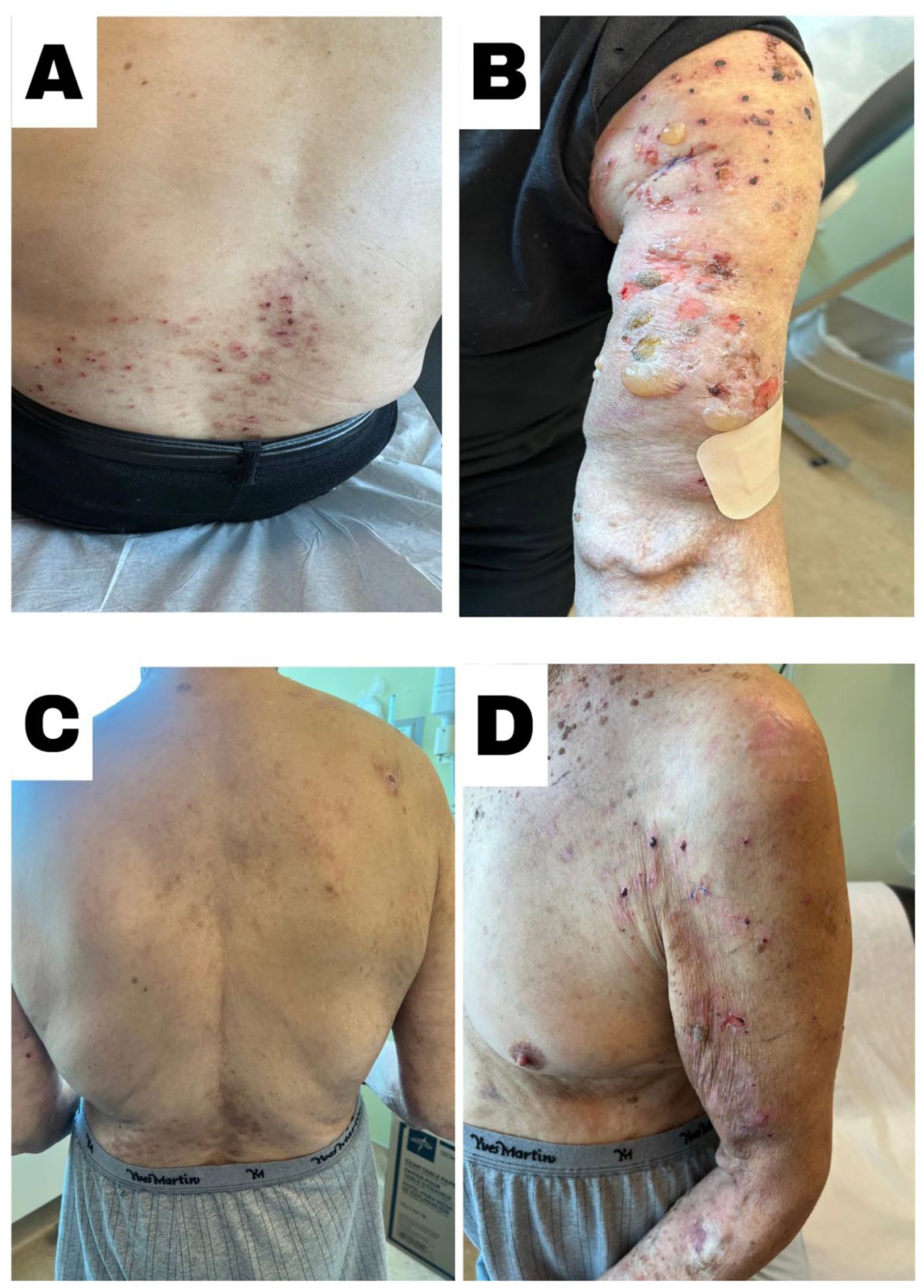
Clinical presentation and treatment response of immune checkpoint inhibitor-induced BP. (a and b) Early presentation with pruritic, erythematous, and excoriated papules plaques and tense bullous lesions on the lower back, trunk, and arm. (c and d) Two weeks following prednisone treatment showing interval improvement of skin lesions and postinflammatory hyperpigmentation posttreatment resolution on the back, with residual postinflammatory hyperpigmentation.
Following the diagnosis, his oncologist held pembrolizumab, and he was started on oral prednisone 50 mg daily for 2 weeks, along with wound care and topical fucidin cream, which led to significant clinical improvement (Figure 2(c) and (d)). The prednisone dose was tapered to 25 mg daily in April 2025 and then discontinued.
Despite cessation of immunotherapy, he experienced a relapse on April 16, 2025, manifesting as tense blisters on the left arm and new erythematous, pruritic plaques and papules on the chest. He was restarted on prednisone 25 mg for 3 weeks. A second localized relapse occurred on May 14th, while the patient was off prednisone for 10 days, and was managed with topical clobetasol. The patient did not experience any occurrence of new lesions while on topical clobetasol for 30 days (Figure 1).
Discussion
ICI-induced BP may manifest weeks to months after therapy initiation or following discontinuation. 2 In our case, BP developed 23 months after initial exposure to cemiplimab and 4 weeks after pembrolizumab reinitiation, consistent with the variable latency in the literature. 2 The temporal and dose-dependent association supports a pharmacodynamic mechanism underlying ICI-induced BP. 1
Solid-organ transplant recipients face a unique vulnerability due to baseline immunosuppression, which skews immune responses toward a type 2-dominant phenotype with certain medications, such as cyclosporine, as seen in our patient. 3 A select number of cases reported the incidence of BP in transplant patients. 4 Although the mechanism underlying this remains elusive, a growing number of these reports have linked BP to the onset of either acute or chronic graft rejection, as seen in our patient. 4
Therapeutic strategies for ICI-induced BP are guided by disease severity. 5 In mild cases, high-potency topical corticosteroids may suffice. 5 Moderate-to-severe disease typically warrants initiating systemic therapy including corticosteroids, doxycycline, and/or niacinamide. 4 For refractory cases, MMF, azathioprine, or methotrexate is recommended, with rituximab or intravenous immunoglobulin reserved for more severe presentations. 4 Biologics targeting type 2 inflammation, such as dupilumab, may also be used safely in such cases. 4 Holding ICI therapy requires a multidisciplinary approach to balance the risk and benefits of ongoing cancer treatment. 4
In our case, disease control was achieved with short-term systemic and topical steroids, which achieved an excellent clinical response. However, relapses occurred following corticosteroid taper or discontinuation, underscoring the importance of gradual tapering, co-treatment with topical steroids and close surveillance.
ICI interruption in our patient led to development of three new low-risk SCCs that were managed with shave and electrodessication and curettage. Rechallenge decisions should be individualized, factoring in cancer activity and risk, BP severity, corticosteroid responsiveness, and the availability of targeted therapies.
Footnotes
Consent for publication
Informed consent was obtained from the patient for the publication of this case and accompanying images.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
